Metformin and risk of mortality in patients hospitalised with COVID-19: a retrospective cohort analysis
et al., The Lancet Healthy Longevity, doi:10.1016/S2666-7568(20)30033-7, Dec 2020
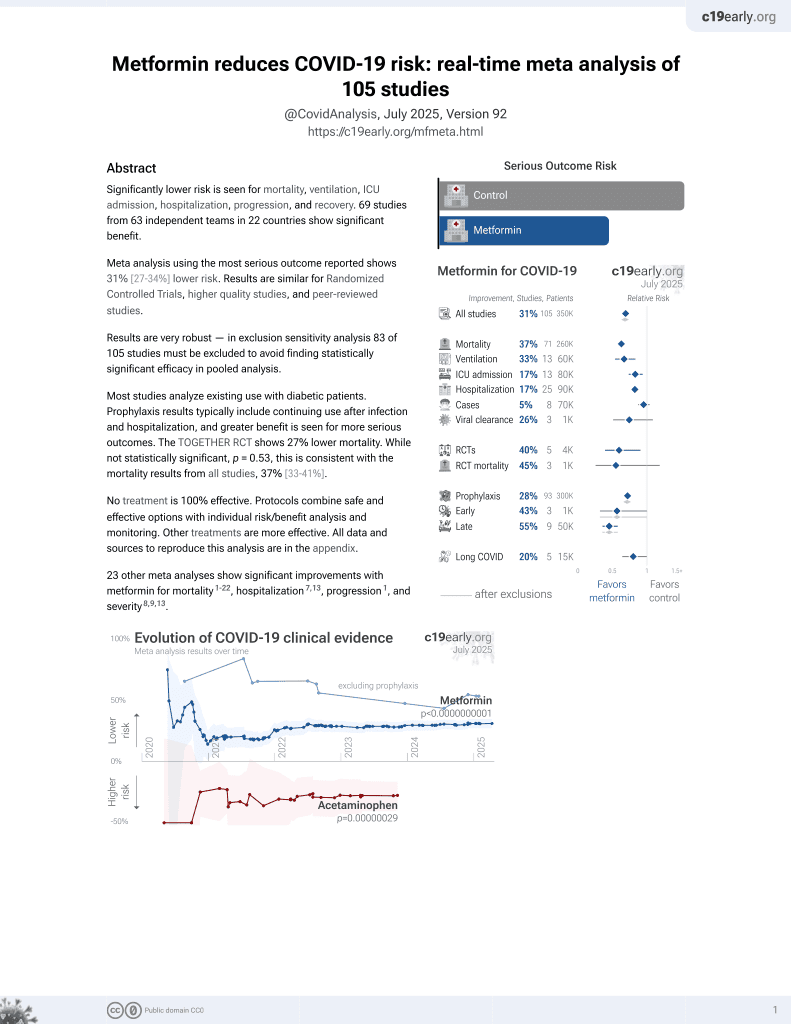
Metformin for COVID-19
3rd treatment shown to reduce risk in
July 2020, now with p < 0.00000000001 from 111 studies.
Lower risk for mortality, ventilation, ICU, hospitalization, progression, recovery, and viral clearance.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
Retrospective 6,256 COVID-19+ diabetes patients in the USA, showing lower mortality with existing metformin treatment, statistically significant only for women.
Standard of Care (SOC) for COVID-19 in the study country,
the USA, is very poor with very low average efficacy for approved treatments1.
Only expensive, high-profit treatments were approved for early treatment. Low-cost treatments were excluded, reducing the probability of early treatment due to access and cost barriers, and eliminating complementary and synergistic benefits seen with many low-cost treatments.
This may explain in part the very high mortality seen in this study.
Results may differ in countries with improved SOC.
|
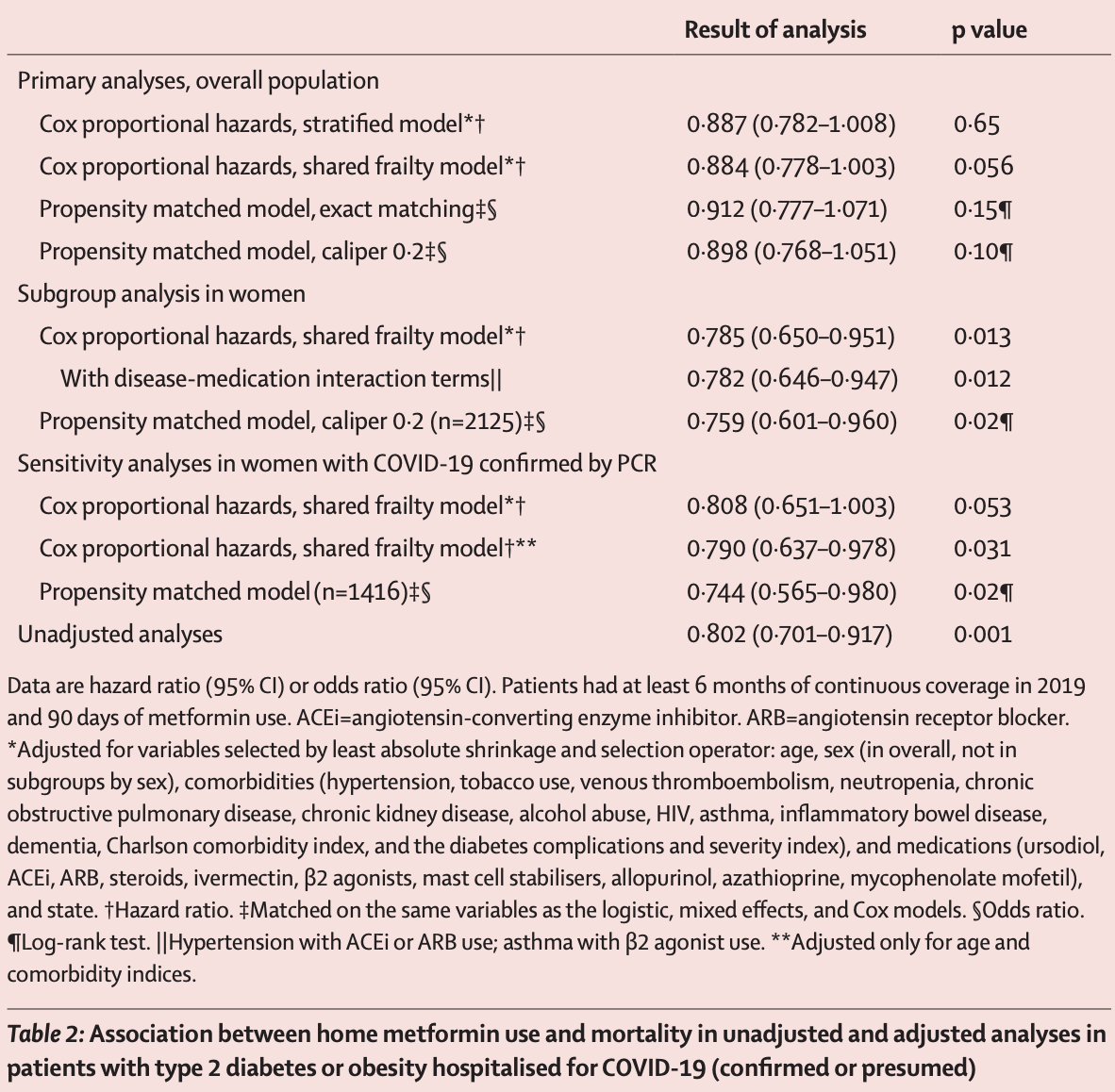
risk of death, 11.6% lower, HR 0.88, p = 0.65, treatment 394 of 2,333 (16.9%), control 791 of 3,923 (20.2%), NNT 31, adjusted per study, multivariable, Cox proportional hazards.
|
|
risk of death, 21.5% lower, HR 0.79, p = 0.01, treatment 1,129, control 2,173, adjusted per study, women, multivariable, Cox proportional hazards.
|
|
risk of death, 4.3% lower, HR 0.96, p = 0.69, treatment 1,204, control 1,750, adjusted per study, men, multivariable, Cox proportional hazards.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Bramante et al., 3 Dec 2020, retrospective, database analysis, USA, peer-reviewed, 17 authors.
Metformin and risk of mortality in patients hospitalised with COVID-19: a retrospective cohort analysis
The Lancet Healthy Longevity, doi:10.1016/s2666-7568(20)30033-7
Background Type 2 diabetes and obesity, as states of chronic inflammation, are risk factors for severe COVID-19. Metformin has cytokine-reducing and sex-specific immunomodulatory effects. Our aim was to identify whether metformin reduced COVID-19-related mortality and whether sex-specific interactions exist.
Methods In this retrospective cohort analysis, we assessed de-identified claims data from UnitedHealth Group (UHG)'s Clinical Discovery Claims Database. Patient data were eligible for inclusion if they were aged 18 years or older; had type 2 diabetes or obesity (defined based on claims); at least 6 months of continuous enrolment in 2019; and admission to hospital for COVID-19 confirmed by PCR, manual chart review by UHG, or reported from the hospital to UHG. The primary outcome was in-hospital mortality from COVID-19. The independent variable of interest was home metformin use, defined as more than 90 days of claims during the year before admission to hospital. Covariates were comorbidities, medications, demographics, and state. Heterogeneity of effect was assessed by sex. For the Cox proportional hazards, censoring was done on the basis of claims made after admission to hospital up to June 7, 2020, with a best outcome approach. Propensity-matched mixed-effects logistic regression was done, stratified by metformin use. Findings 6256 of the 15 380 individuals with pharmacy claims data from Jan 1 to June 7, 2020 were eligible for inclusion. 3302 (52•8%) of 6256 were women. Metformin use was not associated with significantly decreased mortality in the overall sample of men and women by either Cox proportional hazards stratified model (hazard ratio [HR] 0•887 [95% CI 0•782-1•008]) or propensity matching (odds ratio [OR] 0•912 [95% CI 0•777-1•071], p=0•15). Metformin was associated with decreased mortality in women by Cox proportional hazards (HR 0•785, 95% CI 0•650-0•951) and propensity matching (OR 0•759, 95% CI 0•601-0•960, p=0•021). There was no significant reduction in mortality among men (HR 0•957, 95% CI 0•82-1•14; p=0•689 by Cox proportional hazards). Interpretation Metformin was significantly associated with reduced mortality in women with obesity or type 2 diabetes who were admitted to hospital for COVID-19. Prospective studies are needed to understand mechanism and causality. If findings are reproducible, metformin could be widely distributed for prevention of COVID-19 mortality, because it is safe and inexpensive.
References
Beigel, Tomashek, Dodd, Remdesivir for the treatment of COVID-19-preliminary report. Reply, N Engl J Med
Blüher, Fasshauer, Tönjes, Kratzsch, Schön et al., Association of interleukin-6, C-reactive protein, interleukin-10 and adiponectin plasma concentrations with measures of obesity, insulin sensitivity and glucose metabolism, Exp Clin Endocrinol Diabetes
Bosch, Steinberger, Sinaiko, Identification of sex-specific thresholds for accumulation of visceral adipose tissue in adults, Obesity
Bray, Medical consequences of obesity, J Clin Endocrinol Metab
Brookhart, Schneeweiss, Rothman, Glynn, Avorn et al., Variable selection for propensity score models, Am J Epidemiol
Cameron, Morrison, Levin, Anti-inflammatory effects of metformin irrespective of diabetes status, Circ Res
Cariou, Hadjadj, Wargny, Phenotypic characteristics and prognosis of inpatients with COVID-19 and diabetes: the CORONADO study, Diabetologia
Chen, Sang, Jiang, Longitudinal hematologic and immunologic variations associated with the progression of COVID-19 patients in China, J Allergy Clin Immunol
Izeboud, Vermeulen, Zwart, Voss, Van Miert et al., Stereoselectivity at the beta2-adrenoceptor on macrophages is a major determinant of the anti-inflammatory effects of beta2-agonists, Naunyn Schmiedebergs Arch Pharmacol
Jadhav, Ferrell, Greer, Petrie, Cobbe et al., Effects of metformin on microvascular function and exercise tolerance in women with angina and normal coronary arteries: a randomized, double-blind, placebo-controlled study, J Am Coll Cardiol
Kelly, Tannahill, Murphy, Neill, Metformin inhibits the production of reactive oxygen species from NADH:ubiquinone oxidoreductase to limit induction of interleukin-1β (IL-1β) and boosts interleukin-10 (IL-10) in lipopolysaccharide (LPS)-activated macrophages, J Biol Chem
Klein, Flanagan, Sex differences in immune responses, Nat Rev Immunol
Kritas, Ronconi, Caraffa, Gallenga, Ross et al., Mast cells contribute to coronavirus-induced inflammation: new anti-inflammatory strategy, J Biol Regul Homeost Agents
Li, Li, Zhang, Wang, Expression of the SARS-CoV-2 cell receptor gene ACE2 in a wide variety of human tissues, Infect Dis Poverty
Liu, Li, Lu, AMPK: a balancer of the renin-angiotensin system, Biosci Rep
Lloyd, Blackwell, Wei, Howell, Shrank, Validity of a claims-based diagnosis of obesity among medicare beneficiaries, Eval Health Prof
Luo, Qiu, Liu, Metformin treatment was associated with decreased mortality in COVID-19 patients with diabetes in a retrospective analysis, Am J Trop Med Hyg
Mackey, Ayyadurai, Pohl, Costa, Li et al., Sexual dimorphism in the mast cell transcriptome and the pathophysiological responses to immunological and psychological stress, Biol Sex Differ
Matsiukevich, Piraino, Lahni, Metformin ameliorates gender-and age-dependent hemodynamic instability and myocardial injury in murine hemorrhagic shock, Biochim Biophys Acta Mol Basis Dis
Mehta, Mcauley, Brown, Sanchez, Tattersall et al., COVID-19: consider cytokine storm syndromes and immunosuppression, Lancet
Nascimento, Sales, Dienstmann, Souza, Fleig et al., Metformin for prevention of cesarean delivery and large-for-gestational-age newborns in non-diabetic obese pregnant women: a randomized clinical trial, Arch Endocrinol Metab
O'brien, Dropping highly collinear variables from a model: why it typically is not a good idea, Soc Sci Q
Park, Lee, Park, Sex-dependent difference in the effect of metformin on colorectal cancer-specific mortality of diabetic colorectal cancer patients, World J Gastroenterol
Quan, Zhang, Wei, Fang, Gender-related different effects of a combined therapy of exenatide and metformin on overweight or obesity patients with type 2 diabetes mellitus, J Diabetes Complications
Rena, Hardie, Pearson, The mechanisms of action of metformin, Diabetologia
Sharma, Ray, Sadasivam, Metformin in COVID-19: a possible role beyond diabetes, Diabetes Res Clin Pract
Simonnet, Chetboun, Poissy, High prevalence of obesity in severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) requiring invasive mechanical ventilation, Obesity
Tibshirani, Regression shrinkage and selection via the LASSO, J R Stat Soc B
Tufan, Güler, Matucci-Cerinic, COVID-19, immune system response, hyperinflammation and repurposing antirheumatic drugs, Turk J Med Sci
Vanderweele, Ding, Sensitivity analysis in observational research: introducing the e-value, Ann Intern Med
Vj, Illescas-Montes, Puerta-Puerta, Ruiz, Melguizo-Rodríguez, SARS-CoV-2 infection: the role of cytokines in COVID-19 disease, Cytokine Growth Factor Rev
Wang, Huang, Metformin inhibits IgE-and aryl hydrocarbon receptor-mediated mast cell activation in vitro and in vivo, Eur J Immunol
Zeng, Li, Zeng, Can we predict the severity of COVID-19 with a routine blood test?, Pol Arch Intern Med
Zhou, Myers, Li, Role of AMP-activated protein kinase in mechanism of metformin action, J Clin Invest
DOI record:
{
"DOI": "10.1016/s2666-7568(20)30033-7",
"ISSN": [
"2666-7568"
],
"URL": "http://dx.doi.org/10.1016/S2666-7568(20)30033-7",
"alternative-id": [
"S2666756820300337"
],
"assertion": [
{
"label": "This article is maintained by",
"name": "publisher",
"value": "Elsevier"
},
{
"label": "Article Title",
"name": "articletitle",
"value": "Metformin and risk of mortality in patients hospitalised with COVID-19: a retrospective cohort analysis"
},
{
"label": "Journal Title",
"name": "journaltitle",
"value": "The Lancet Healthy Longevity"
},
{
"label": "CrossRef DOI link to publisher maintained version",
"name": "articlelink",
"value": "https://doi.org/10.1016/S2666-7568(20)30033-7"
},
{
"label": "CrossRef DOI link to the associated document",
"name": "associatedlink",
"value": "https://doi.org/10.1016/S2666-7568(20)30047-7"
},
{
"label": "Content Type",
"name": "content_type",
"value": "article"
},
{
"label": "Copyright",
"name": "copyright",
"value": "© 2020 The Author(s). Published by Elsevier Ltd."
}
],
"author": [
{
"affiliation": [],
"family": "Bramante",
"given": "Carolyn T",
"sequence": "first"
},
{
"affiliation": [],
"family": "Ingraham",
"given": "Nicholas E",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Murray",
"given": "Thomas A",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Marmor",
"given": "Schelomo",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Hovertsen",
"given": "Shane",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Gronski",
"given": "Jessica",
"sequence": "additional"
},
{
"affiliation": [],
"family": "McNeil",
"given": "Chace",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Feng",
"given": "Ruoying",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Guzman",
"given": "Gabriel",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Abdelwahab",
"given": "Nermine",
"sequence": "additional"
},
{
"affiliation": [],
"family": "King",
"given": "Samantha",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Tamariz",
"given": "Leonardo",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Meehan",
"given": "Thomas",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Pendleton",
"given": "Kathryn M",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Benson",
"given": "Bradley",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Vojta",
"given": "Deneen",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Tignanelli",
"given": "Christopher J",
"sequence": "additional"
}
],
"container-title": "The Lancet Healthy Longevity",
"container-title-short": "The Lancet Healthy Longevity",
"content-domain": {
"crossmark-restriction": true,
"domain": [
"thelancet.com",
"elsevier.com",
"sciencedirect.com"
]
},
"created": {
"date-parts": [
[
2020,
12,
3
]
],
"date-time": "2020-12-03T23:45:19Z",
"timestamp": 1607039119000
},
"deposited": {
"date-parts": [
[
2022,
10,
4
]
],
"date-time": "2022-10-04T10:38:01Z",
"timestamp": 1664879881000
},
"funder": [
{
"DOI": "10.13039/100000050",
"award": [
"T32HL07741"
],
"doi-asserted-by": "publisher",
"name": "National Heart, Lung, and Blood Institute"
},
{
"DOI": "10.13039/100000054",
"award": [
"P30CA077598"
],
"doi-asserted-by": "publisher",
"name": "National Cancer Institute"
},
{
"DOI": "10.13039/100000133",
"doi-asserted-by": "publisher",
"name": "Agency for Healthcare Research and Quality"
},
{
"DOI": "10.13039/100004374",
"doi-asserted-by": "publisher",
"name": "Medtronic"
},
{
"DOI": "10.13039/100006093",
"award": [
"K12HS026379"
],
"doi-asserted-by": "publisher",
"name": "Patient-Centered Outcomes Research Institute"
},
{
"DOI": "10.13039/100006108",
"award": [
"KL2TR002492",
"UL1TR002494"
],
"doi-asserted-by": "publisher",
"name": "National Center for Advancing Translational Sciences"
}
],
"indexed": {
"date-parts": [
[
2024,
4,
9
]
],
"date-time": "2024-04-09T09:08:54Z",
"timestamp": 1712653734258
},
"is-referenced-by-count": 133,
"issue": "1",
"issued": {
"date-parts": [
[
2021,
1
]
]
},
"journal-issue": {
"issue": "1",
"published-print": {
"date-parts": [
[
2021,
1
]
]
}
},
"language": "en",
"license": [
{
"URL": "https://www.elsevier.com/tdm/userlicense/1.0/",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2021,
1,
1
]
],
"date-time": "2021-01-01T00:00:00Z",
"timestamp": 1609459200000
}
},
{
"URL": "http://creativecommons.org/licenses/by-nc-nd/4.0/",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2020,
10,
7
]
],
"date-time": "2020-10-07T00:00:00Z",
"timestamp": 1602028800000
}
}
],
"link": [
{
"URL": "https://api.elsevier.com/content/article/PII:S2666756820300337?httpAccept=text/xml",
"content-type": "text/xml",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://api.elsevier.com/content/article/PII:S2666756820300337?httpAccept=text/plain",
"content-type": "text/plain",
"content-version": "vor",
"intended-application": "text-mining"
}
],
"member": "78",
"original-title": [],
"page": "e34-e41",
"prefix": "10.1016",
"published": {
"date-parts": [
[
2021,
1
]
]
},
"published-print": {
"date-parts": [
[
2021,
1
]
]
},
"publisher": "Elsevier BV",
"reference": [
{
"DOI": "10.1002/oby.22831",
"article-title": "High prevalence of obesity in severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) requiring invasive mechanical ventilation",
"author": "Simonnet",
"doi-asserted-by": "crossref",
"first-page": "1195",
"journal-title": "Obesity",
"key": "10.1016/S2666-7568(20)30033-7_bib1",
"volume": "28",
"year": "2020"
},
{
"DOI": "10.1002/oby.20961",
"article-title": "Identification of sex-specific thresholds for accumulation of visceral adipose tissue in adults",
"author": "Bosch",
"doi-asserted-by": "crossref",
"first-page": "375",
"journal-title": "Obesity",
"key": "10.1016/S2666-7568(20)30033-7_bib2",
"volume": "23",
"year": "2015"
},
{
"DOI": "10.1210/jc.2004-0535",
"article-title": "Medical consequences of obesity",
"author": "Bray",
"doi-asserted-by": "crossref",
"first-page": "2583",
"journal-title": "J Clin Endocrinol Metab",
"key": "10.1016/S2666-7568(20)30033-7_bib3",
"volume": "89",
"year": "2004"
},
{
"DOI": "10.3906/sag-2004-168",
"article-title": "COVID-19, immune system response, hyperinflammation and repurposing antirheumatic drugs",
"author": "Tufan",
"doi-asserted-by": "crossref",
"first-page": "620",
"journal-title": "Turk J Med Sci",
"key": "10.1016/S2666-7568(20)30033-7_bib4",
"volume": "50",
"year": "2020"
},
{
"DOI": "10.1055/s-2005-872851",
"article-title": "Association of interleukin-6, C-reactive protein, interleukin-10 and adiponectin plasma concentrations with measures of obesity, insulin sensitivity and glucose metabolism",
"author": "Blüher",
"doi-asserted-by": "crossref",
"first-page": "534",
"journal-title": "Exp Clin Endocrinol Diabetes",
"key": "10.1016/S2666-7568(20)30033-7_bib5",
"volume": "113",
"year": "2005"
},
{
"DOI": "10.1016/j.bbadis.2017.05.027",
"article-title": "Metformin ameliorates gender-and age-dependent hemodynamic instability and myocardial injury in murine hemorrhagic shock",
"author": "Matsiukevich",
"doi-asserted-by": "crossref",
"first-page": "2680",
"journal-title": "Biochim Biophys Acta Mol Basis Dis",
"key": "10.1016/S2666-7568(20)30033-7_bib6",
"volume": "1863",
"year": "2017"
},
{
"DOI": "10.1016/j.jdiacomp.2016.01.013",
"article-title": "Gender-related different effects of a combined therapy of exenatide and metformin on overweight or obesity patients with type 2 diabetes mellitus",
"author": "Quan",
"doi-asserted-by": "crossref",
"first-page": "686",
"journal-title": "J Diabetes Complications",
"key": "10.1016/S2666-7568(20)30033-7_bib7",
"volume": "30",
"year": "2016"
},
{
"DOI": "10.3748/wjg.v23.i28.5196",
"article-title": "Sex-dependent difference in the effect of metformin on colorectal cancer-specific mortality of diabetic colorectal cancer patients",
"author": "Park",
"doi-asserted-by": "crossref",
"first-page": "5196",
"journal-title": "World J Gastroenterol",
"key": "10.1016/S2666-7568(20)30033-7_bib8",
"volume": "23",
"year": "2017"
},
{
"DOI": "10.4269/ajtmh.20-0375",
"article-title": "Metformin treatment was associated with decreased mortality in COVID-19 patients with diabetes in a retrospective analysis",
"author": "Luo",
"doi-asserted-by": "crossref",
"first-page": "69",
"journal-title": "Am J Trop Med Hyg",
"key": "10.1016/S2666-7568(20)30033-7_bib9",
"volume": "103",
"year": "2020"
},
{
"DOI": "10.1007/s00125-020-05180-x",
"article-title": "Phenotypic characteristics and prognosis of inpatients with COVID-19 and diabetes: the CORONADO study",
"author": "Cariou",
"doi-asserted-by": "crossref",
"first-page": "1500",
"journal-title": "Diabetologia",
"key": "10.1016/S2666-7568(20)30033-7_bib10",
"volume": "63",
"year": "2020"
},
{
"article-title": "Regression shrinkage and selection via the LASSO",
"author": "Tibshirani",
"first-page": "267",
"journal-title": "J R Stat Soc B",
"key": "10.1016/S2666-7568(20)30033-7_bib11",
"volume": "58",
"year": "1996"
},
{
"DOI": "10.1056/NEJMoa2007764",
"article-title": "Remdesivir for the treatment of COVID-19—preliminary report. Reply",
"author": "Beigel",
"doi-asserted-by": "crossref",
"first-page": "994",
"journal-title": "N Engl J Med",
"key": "10.1016/S2666-7568(20)30033-7_bib12",
"volume": "383",
"year": "2020"
},
{
"DOI": "10.1093/aje/kwj149",
"article-title": "Variable selection for propensity score models",
"author": "Brookhart",
"doi-asserted-by": "crossref",
"first-page": "1149",
"journal-title": "Am J Epidemiol",
"key": "10.1016/S2666-7568(20)30033-7_bib13",
"volume": "163",
"year": "2006"
},
{
"DOI": "10.7326/M16-2607",
"article-title": "Sensitivity analysis in observational research: introducing the e-value",
"author": "VanderWeele",
"doi-asserted-by": "crossref",
"first-page": "268",
"journal-title": "Ann Intern Med",
"key": "10.1016/S2666-7568(20)30033-7_bib14",
"volume": "167",
"year": "2017"
},
{
"DOI": "10.1016/j.cytogfr.2020.06.001",
"article-title": "SARS-CoV-2 infection: the role of cytokines in COVID-19 disease",
"author": "Costela-Ruiz",
"doi-asserted-by": "crossref",
"first-page": "62",
"journal-title": "Cytokine Growth Factor Rev",
"key": "10.1016/S2666-7568(20)30033-7_bib15",
"volume": "54",
"year": "2020"
},
{
"DOI": "10.1172/JCI13505",
"article-title": "Role of AMP-activated protein kinase in mechanism of metformin action",
"author": "Zhou",
"doi-asserted-by": "crossref",
"first-page": "1167",
"journal-title": "J Clin Invest",
"key": "10.1016/S2666-7568(20)30033-7_bib16",
"volume": "108",
"year": "2001"
},
{
"DOI": "10.1007/s00125-017-4342-z",
"article-title": "The mechanisms of action of metformin",
"author": "Rena",
"doi-asserted-by": "crossref",
"first-page": "1577",
"journal-title": "Diabetologia",
"key": "10.1016/S2666-7568(20)30033-7_bib17",
"volume": "60",
"year": "2017"
},
{
"DOI": "10.1016/j.diabres.2020.108183",
"article-title": "Metformin in COVID-19: a possible role beyond diabetes",
"author": "Sharma",
"doi-asserted-by": "crossref",
"journal-title": "Diabetes Res Clin Pract",
"key": "10.1016/S2666-7568(20)30033-7_bib18",
"volume": "164",
"year": "2020"
},
{
"DOI": "10.1186/s40249-020-00662-x",
"article-title": "Expression of the SARS-CoV-2 cell receptor gene ACE2 in a wide variety of human tissues",
"author": "Li",
"doi-asserted-by": "crossref",
"first-page": "45",
"journal-title": "Infect Dis Poverty",
"key": "10.1016/S2666-7568(20)30033-7_bib19",
"volume": "9",
"year": "2020"
},
{
"DOI": "10.1016/j.jacc.2006.04.088",
"article-title": "Effects of metformin on microvascular function and exercise tolerance in women with angina and normal coronary arteries: a randomized, double-blind, placebo-controlled study",
"author": "Jadhav",
"doi-asserted-by": "crossref",
"first-page": "956",
"journal-title": "J Am Coll Cardiol",
"key": "10.1016/S2666-7568(20)30033-7_bib20",
"volume": "48",
"year": "2006"
},
{
"DOI": "10.1016/j.jaci.2020.05.003",
"article-title": "Longitudinal hematologic and immunologic variations associated with the progression of COVID-19 patients in China",
"author": "Chen",
"doi-asserted-by": "crossref",
"first-page": "89",
"journal-title": "J Allergy Clin Immunol",
"key": "10.1016/S2666-7568(20)30033-7_bib21",
"volume": "146",
"year": "2020"
},
{
"article-title": "Can we predict the severity of COVID-19 with a routine blood test?",
"author": "Zeng",
"first-page": "400",
"journal-title": "Pol Arch Intern Med",
"key": "10.1016/S2666-7568(20)30033-7_bib22",
"volume": "130",
"year": "2020"
},
{
"DOI": "10.1161/CIRCRESAHA.116.308445",
"article-title": "Anti-inflammatory effects of metformin irrespective of diabetes status",
"author": "Cameron",
"doi-asserted-by": "crossref",
"first-page": "652",
"journal-title": "Circ Res",
"key": "10.1016/S2666-7568(20)30033-7_bib23",
"volume": "119",
"year": "2016"
},
{
"DOI": "10.1074/jbc.M115.662114",
"article-title": "Metformin inhibits the production of reactive oxygen species from NADH:ubiquinone oxidoreductase to limit induction of interleukin-1β (IL-1β) and boosts interleukin-10 (IL-10) in lipopolysaccharide (LPS)-activated macrophages",
"author": "Kelly",
"doi-asserted-by": "crossref",
"first-page": "20348",
"journal-title": "J Biol Chem",
"key": "10.1016/S2666-7568(20)30033-7_bib24",
"volume": "290",
"year": "2015"
},
{
"DOI": "10.1016/S0140-6736(20)30628-0",
"article-title": "COVID-19: consider cytokine storm syndromes and immunosuppression",
"author": "Mehta",
"doi-asserted-by": "crossref",
"first-page": "1033",
"journal-title": "Lancet",
"key": "10.1016/S2666-7568(20)30033-7_bib25",
"volume": "395",
"year": "2020"
},
{
"DOI": "10.1038/nri.2016.90",
"article-title": "Sex differences in immune responses",
"author": "Klein",
"doi-asserted-by": "crossref",
"first-page": "626",
"journal-title": "Nat Rev Immunol",
"key": "10.1016/S2666-7568(20)30033-7_bib26",
"volume": "16",
"year": "2016"
},
{
"DOI": "10.1002/eji.201847706",
"article-title": "Metformin inhibits IgE- and aryl hydrocarbon receptor-mediated mast cell activation in vitro and in vivo",
"author": "Wang",
"doi-asserted-by": "crossref",
"first-page": "1989",
"journal-title": "Eur J Immunol",
"key": "10.1016/S2666-7568(20)30033-7_bib27",
"volume": "48",
"year": "2018"
},
{
"article-title": "Mast cells contribute to coronavirus-induced inflammation: new anti-inflammatory strategy",
"author": "Kritas",
"first-page": "9",
"journal-title": "J Biol Regul Homeost Agents",
"key": "10.1016/S2666-7568(20)30033-7_bib28",
"volume": "34",
"year": "2020"
},
{
"DOI": "10.1186/s13293-016-0113-7",
"article-title": "Sexual dimorphism in the mast cell transcriptome and the pathophysiological responses to immunological and psychological stress",
"author": "Mackey",
"doi-asserted-by": "crossref",
"first-page": "60",
"journal-title": "Biol Sex Differ",
"key": "10.1016/S2666-7568(20)30033-7_bib29",
"volume": "7",
"year": "2016"
},
{
"DOI": "10.1042/BSR20181994",
"article-title": "AMPK: a balancer of the renin-angiotensin system",
"author": "Liu",
"doi-asserted-by": "crossref",
"journal-title": "Biosci Rep",
"key": "10.1016/S2666-7568(20)30033-7_bib30",
"volume": "39",
"year": "2019"
},
{
"DOI": "10.1007/s002100000281",
"article-title": "Stereoselectivity at the beta2-adrenoceptor on macrophages is a major determinant of the anti-inflammatory effects of beta2-agonists",
"author": "Izeboud",
"doi-asserted-by": "crossref",
"first-page": "184",
"journal-title": "Naunyn Schmiedebergs Arch Pharmacol",
"key": "10.1016/S2666-7568(20)30033-7_bib31",
"volume": "362",
"year": "2000"
},
{
"DOI": "10.20945/2359-3997000000251",
"article-title": "Metformin for prevention of cesarean delivery and large-for-gestational-age newborns in non-diabetic obese pregnant women: a randomized clinical trial",
"author": "Nascimento",
"doi-asserted-by": "crossref",
"first-page": "290",
"journal-title": "Arch Endocrinol Metab",
"key": "10.1016/S2666-7568(20)30033-7_bib32",
"volume": "64",
"year": "2020"
},
{
"DOI": "10.1177/0163278714553661",
"article-title": "Validity of a claims-based diagnosis of obesity among medicare beneficiaries",
"author": "Lloyd",
"doi-asserted-by": "crossref",
"first-page": "508",
"journal-title": "Eval Health Prof",
"key": "10.1016/S2666-7568(20)30033-7_bib33",
"volume": "38",
"year": "2015"
},
{
"DOI": "10.1111/ssqu.12273",
"article-title": "Dropping highly collinear variables from a model: why it typically is not a good idea",
"author": "O'Brien",
"doi-asserted-by": "crossref",
"first-page": "360",
"journal-title": "Soc Sci Q",
"key": "10.1016/S2666-7568(20)30033-7_bib35",
"volume": "98",
"year": "2017"
}
],
"reference-count": 34,
"references-count": 34,
"relation": {},
"resource": {
"primary": {
"URL": "https://linkinghub.elsevier.com/retrieve/pii/S2666756820300337"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [
"Family Practice",
"Psychiatry and Mental health",
"Geriatrics and Gerontology",
"Health (social science)"
],
"subtitle": [],
"title": "Metformin and risk of mortality in patients hospitalised with COVID-19: a retrospective cohort analysis",
"type": "journal-article",
"update-policy": "http://dx.doi.org/10.1016/elsevier_cm_policy",
"volume": "2"
}