Oral Fluvoxamine With Inhaled Budesonide for Treatment of Early-Onset COVID-19
et al., Annals of Internal Medicine, doi:10.7326/M22-3305, TOGETHER, NCT04727424, Apr 2023
Budesonide for COVID-19
27th treatment shown to reduce risk in
September 2021, now with p = 0.000003 from 15 studies, recognized in 10 countries.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
The TOGETHER trial has extreme COI, impossible data, blinding failure, randomization failure, uncorrected errors, and many protocol violations. Authors do not respond to these issues and they have refused to release the data as promised. Some issues may apply only to specific arms.
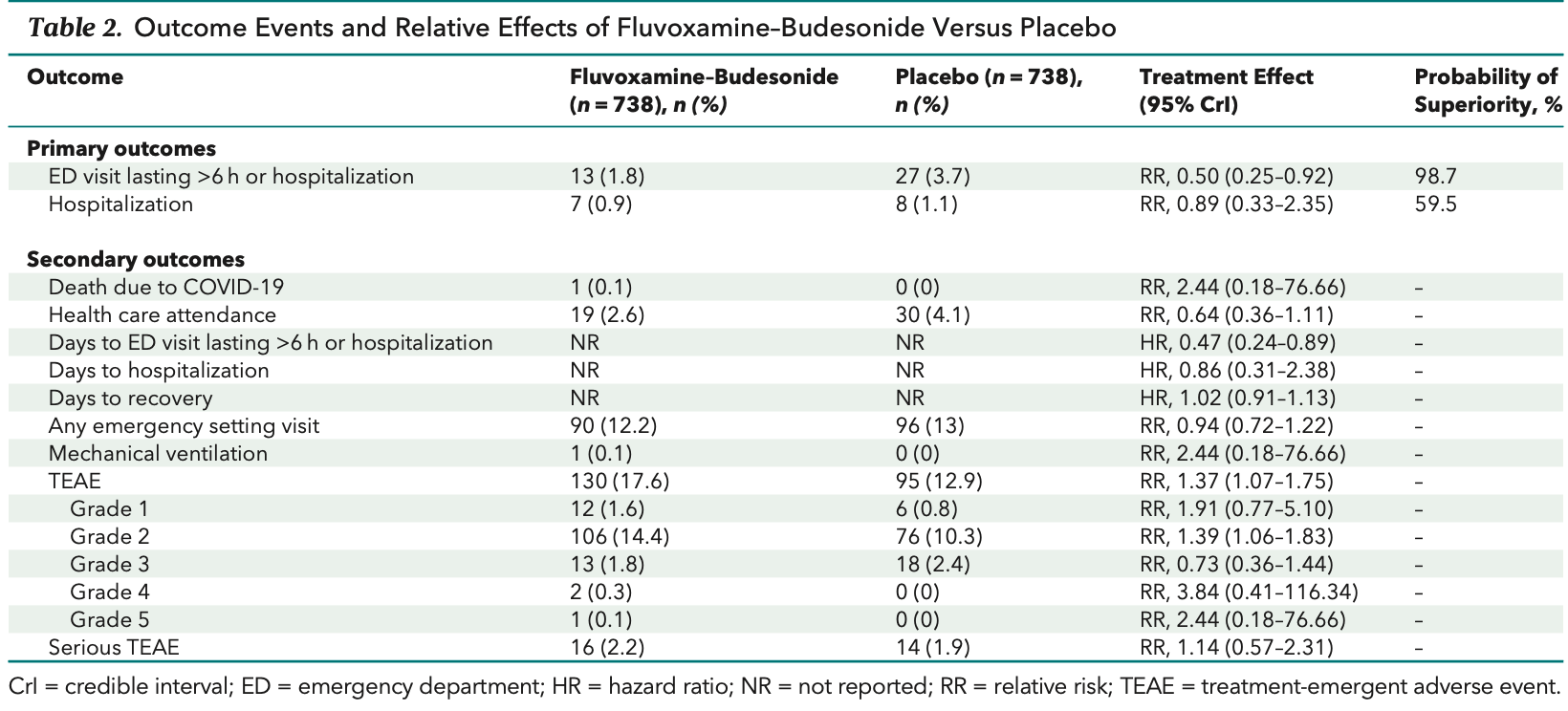
Low-risk (1% hospitalization) outpatient RCT with 738 fluvoxamine + budesonide patients and 738 placebo patients, showing significantly lower hospitalization/ER visits with treatment.
The TOGETHER trial has extreme COI, impossible data,
blinding failure, randomization failure, uncorrected errors, and many
protocol violations. Authors do not respond to these issues and they
have refused to release the data as promised. Some issues may apply only
to specific arms. For more details see1-5.
Targeted administration to the respiratory tract provides treatment directly
to the typical source of initial SARS-CoV-2 infection and replication, and
allows for rapid onset of action, higher local drug concentration, and reduced systemic side effects.
Study covers budesonide and fluvoxamine.
|
risk of death, 200.0% higher, RR 3.00, p = 1.00, treatment 1 of 738 (0.1%), control 0 of 738 (0.0%), continuity correction due to zero event (with reciprocal of the contrasting arm).
|
|
risk of hospitalization, 12.5% lower, RR 0.88, p = 1.00, treatment 7 of 738 (0.9%), control 8 of 738 (1.1%), NNT 738.
|
|
hospitalization or ER >6hrs, 50.0% lower, RR 0.50, p = 0.04, treatment 13 of 738 (1.8%), control 27 of 738 (3.7%), NNT 53, adjusted per study, day 28, primary outcome.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
1.
Reis et al., Effect of Early Treatment with Ivermectin among Patients with Covid-19, New England Journal of Medicine, doi:10.1056/NEJMoa2115869.
2.
Reis (B) et al., RETRACTED: Effect of early treatment with metformin on risk of emergency care and hospitalization among patients with COVID-19: The TOGETHER randomized platform clinical trial, The Lancet Regional Health - Americas, doi:10.1016/j.lana.2021.100142.
3.
Reis (C) et al., Effect of early treatment with fluvoxamine on risk of emergency care and hospitalisation among patients with COVID-19: the TOGETHER randomised, platform clinical trial, The Lancet Global Health, doi:10.1016/S2214-109X(21)00448-4.
Reis et al., 17 Apr 2023, Double Blind Randomized Controlled Trial, placebo-controlled, Brazil, peer-reviewed, 35 authors, study period 15 January, 2022 - 6 July, 2022, average treatment delay 3.0 days, this trial uses multiple treatments in the treatment arm (combined with fluvoxamine) - results of individual treatments may vary, trial NCT04727424 (history) (TOGETHER).
Oral Fluvoxamine With Inhaled Budesonide for Treatment of Early-Onset COVID-19 A Randomized Platform Trial
doi:10.7326/M22-3305
Background: Previous trials have demonstrated the effects of fluvoxamine alone and inhaled budesonide alone for prevention of disease progression among outpatients with COVID-19. Objective: To determine whether the combination of fluvoxamine and inhaled budesonide would increase treatment effects in a highly vaccinated population.
Author contributions are available at Annals.org.
References
Bramante, Huling, Tignanelli, Randomized trial of metformin, ivermectin, and fluvoxamine for covid-19, N Engl J Med, doi:10.1056/NEJMoa2201662
Grambsch, Therneau, Proportional hazards tests and diagnostics based on weighted residuals, Biometrika, doi:10.1093/biomet/81.3.515
Guo, Harari, Chernecki, Fluvoxamine for the early treatment of COVID-19: A meta-analysis of randomized clinical trials, Am J Trop Med Hyg, doi:10.4269/ajtmh.21-1310
Guo, Harari, Chernecki, Fluvoxamine for the early treatment of COVID-19: A meta-analysis of randomized clinical trials, Am J Trop Med Hyg, doi:10.4269/ajtmh.21-1310
Lee, Vigod, Bortolussi-Courval, Fluvoxamine for outpatient management of COVID-19 to prevent hospitalization: A systematic review and meta-analysis, JAMA Netw Open, doi:10.1001/jamanetworkopen.2022.6269
Lenze, Mattar, Zorumski, Fluvoxamine vs placebo and clinical deterioration in outpatients with symptomatic COVID-19: A randomized clinical trial, JAMA, doi:10.1001/jama.2020.22760
Mills, Thorlund, Ioannidis, Calculating additive treatment effects from multiple randomized trials provides useful estimates of combination therapies, J Clin Epidemiol, doi:10.1016/j.jclinepi.2012.07.012
Omi, Tanimukai, Kanayama, Fluvoxamine alleviates ER stress via induction of Sigma-1 receptor, Cell Death Dis, doi:10.1038/cddis.2014.301
Park, Siden, Zoratti, Systematic review of basket trials, umbrella trials, and platform trials: a landscape analysis of ORIGINAL RESEARCH Oral Fluvoxamine With Inhaled Budesonide for Early-Onset COVID-19 master protocols, Trials, doi:10.1186/s13063-019-3664-1
Reis, Santos Moreira, Silva, Medeiros Silva, Thabane et al., Oral Fluvoxamine With Inhaled Budesonide for Early-Onset COVID-19 ORIGINAL RESEARCH Annals.org Author Contributions: Conception and design
Reis, Santos Moreira-Silva, Silva, Effect of early treatment with fluvoxamine on risk of emergency care and hospitalisation among patients with COVID-19: the TOGETHER randomised, platform clinical trial, Lancet Glob Health, doi:10.1016/S2214-109X(21)00448-4
Reis, Santos, Silva, Silva, Effect of early treatment with metformin on risk of emergency care and hospitalization among patients with COVID-19: The TOGETHER randomized platform clinical trial, Lancet Reg Health Am, doi:10.1016/j.lana.2021.100142
Reis, Silva, Silva, A multi-center, adaptive, randomized, platform trial to evaluate the effect of repurposed medicines in outpatients with early coronavirus disease 2019 (COVID-19) and high-risk for complications: the TOGETHER master trial protocol, Gates Open Research
Reis, Silva, Silva, A multi-center, adaptive, randomized, platform trial to evaluate the effect of repurposed medicines in outpatients with early coronavirus disease 2019 (COVID-19) and high-risk for complications: the TOGETHER master trial protocol, Gates Open Research
Reis, Silva, Silva, Early treatment with pegylated interferon lambda for covid-19, N Engl J Med, doi:10.1056/NEJMoa2209760
Reis, Silva, Silva, Effect of early treatment with hydroxychloroquine or lopinavir and ritonavir on risk of hospitalization among patients with COVID-19: the TOGETHER randomized clinical trial, JAMA Netw Open, doi:10.1001/jamanetworkopen.2021.6468
Reis, Silva, Silva, Effect of early treatment with ivermectin among patients with covid-19
Schandelmaier, Briel, Varadhan, Development of the Instrument to assess the Credibility of Effect Modification Analyses (ICEMAN) in randomized controlled trials and metaanalyses, CMAJ, doi:10.1503/cmaj.200077
Stanaland, Once-daily budesonide aqueous nasal spray for allergic rhinitis: a review, Clin Ther, doi:10.1016/s0149-2918(04)90050-1
Sukhatme, Reiersen, Vayttaden, Fluvoxamine: A review of its mechanism of action and its role in COVID-19, Front Pharmacol, doi:10.3389/fphar.2021.652688
Woodcock, Lavange, Master protocols to study multiple therapies, multiple diseases, or both, N Engl J Med, doi:10.1056/NEJMra1510062
Yu, Bafadhel, Dorward, Inhaled budesonide for COVID-19 in people at high risk of complications in the community in the UK (PRINCIPLE): a randomised, controlled, open-label, adaptive platform trial, Lancet, doi:10.1016/S0140-6736(21)01744-X
DOI record:
{
"DOI": "10.7326/m22-3305",
"ISSN": [
"0003-4819",
"1539-3704"
],
"URL": "http://dx.doi.org/10.7326/M22-3305",
"alternative-id": [
"10.7326/M22-3305"
],
"author": [
{
"ORCID": "http://orcid.org/0000-0002-4847-1034",
"affiliation": [
{
"name": "ViRx@Stanford, Stanford Biosecurity and Pandemic Preparedness Initiative, Stanford, California; Research Division, Cardresearch - Cardiologia Assistencial e de Pesquisa, Belo Horizonte, Brazil; Department of Medicine, Pontifícia Universidade Católica de Minas Gerais, Minas Gerais, Brazil; and Department of Health Research Methods, Evidence, and Impact, McMaster University, Hamilton, Ontario, Canada (G.R.)"
}
],
"authenticated-orcid": false,
"family": "Reis",
"given": "Gilmar",
"sequence": "first"
},
{
"ORCID": "http://orcid.org/0000-0001-5111-9528",
"affiliation": [
{
"name": "Research Division, Cardresearch - Cardiologia Assistencial e de Pesquisa, Belo Horizonte, Brazil, and Department of Medicine, Pontifícia Universidade Católica de Minas Gerais, Minas Gerais, Brazil (E.A.d.S.M.S., D.C.M.S., V.H.d.S.C., C.V.Q.d.S.)"
}
],
"authenticated-orcid": false,
"family": "dos Santos Moreira Silva",
"given": "Eduardo Augusto",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0003-2275-1190",
"affiliation": [
{
"name": "Research Division, Cardresearch - Cardiologia Assistencial e de Pesquisa, Belo Horizonte, Brazil, and Department of Medicine, Pontifícia Universidade Católica de Minas Gerais, Minas Gerais, Brazil (E.A.d.S.M.S., D.C.M.S., V.H.d.S.C., C.V.Q.d.S.)"
}
],
"authenticated-orcid": false,
"family": "Medeiros Silva",
"given": "Daniela Carla",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0003-0355-9734",
"affiliation": [
{
"name": "Department of Health Research Methods, Evidence, and Impact, McMaster University, Hamilton, Ontario, Canada (L.T., S.S., P.M., G.H.G.)"
}
],
"authenticated-orcid": false,
"family": "Thabane",
"given": "Lehana",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-3842-8016",
"affiliation": [
{
"name": "Research Division, Cardresearch - Cardiologia Assistencial e de Pesquisa, Belo Horizonte, Brazil, and Department of Medicine, Pontifícia Universidade Católica de Minas Gerais, Minas Gerais, Brazil (E.A.d.S.M.S., D.C.M.S., V.H.d.S.C., C.V.Q.d.S.)"
}
],
"authenticated-orcid": false,
"family": "de Souza Campos",
"given": "Vitoria Helena",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Research Division, Cardresearch - Cardiologia Assistencial e de Pesquisa, Belo Horizonte, Brazil (T.S.F., M.I.C.S., L.B.R., R.O.)"
}
],
"family": "Ferreira",
"given": "Thiago Santiago",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Research Division, Cardresearch - Cardiologia Assistencial e de Pesquisa, Belo Horizonte, Brazil, and Department of Medicine, Pontifícia Universidade Católica de Minas Gerais, Minas Gerais, Brazil (E.A.d.S.M.S., D.C.M.S., V.H.d.S.C., C.V.Q.d.S.)"
}
],
"family": "Quirino dos Santos",
"given": "Castilho Vitor",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Public Health Care Division, City of Ibirité, Brazil (A.M.R.N.)"
}
],
"family": "Ribeiro Nogueira",
"given": "Ana Maria",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Public Health at UNIFIPMoc and Family Medicine Fellowship Program, City of Montes Claros, Brazil (A.P.F.G.A.)"
}
],
"family": "Figueiredo Guimaraes Almeida",
"given": "Ana Paula",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0001-6780-0377",
"affiliation": [
{
"name": "Public Health, Mental and Family Medicine Department, Ouro Preto Federal University, Minas Gerais, Brazil (L.C.M.S.)"
}
],
"authenticated-orcid": false,
"family": "Cançado Monteiro Savassi",
"given": "Leonardo",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Public Health Fellowship Program, Governador Valadares Public Health Authority, Minas Gerais, Brazil (A.D.d.F.N.)"
}
],
"family": "de Figueiredo Neto",
"given": "Adhemar Dias",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-2267-2233",
"affiliation": [
{
"name": "Public Health, Mental and Family Medicine Department, Ouro Preto Federal University, Minas Gerais, Brazil, and Public Health Care Division, City of Ibirité, Brazil (C.B., A.C.M.)"
}
],
"authenticated-orcid": false,
"family": "Bitarães",
"given": "Carina",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Public Health, Mental and Family Medicine Department, Ouro Preto Federal University, Minas Gerais, Brazil, and Public Health Care Division, City of Ibirité, Brazil (C.B., A.C.M.)"
}
],
"family": "Cruz Milagres",
"given": "Aline",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Public Health at UNIFIPMoc and Family Medicine Fellowship Program, City of Montes Claros, Brazil, and Public Health Care Division, City of Brumadinho, Brazil (E.D.C.)"
}
],
"family": "Diniz Callegari",
"given": "Eduardo",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Research Division, Cardresearch - Cardiologia Assistencial e de Pesquisa, Belo Horizonte, Brazil (T.S.F., M.I.C.S., L.B.R., R.O.)"
}
],
"family": "Campos Simplicio",
"given": "Maria Izabel",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Research Division, Cardresearch - Cardiologia Assistencial e de Pesquisa, Belo Horizonte, Brazil (T.S.F., M.I.C.S., L.B.R., R.O.)"
}
],
"family": "Barra Ribeiro",
"given": "Luciene",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Research Division, Cardresearch - Cardiologia Assistencial e de Pesquisa, Belo Horizonte, Brazil (T.S.F., M.I.C.S., L.B.R., R.O.)"
}
],
"family": "Oliveira",
"given": "Rosemary",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Cytel, Vancouver, British Columbia, Canada (O.H., H.R.)"
}
],
"family": "Harari",
"given": "Ofir",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Platform Life Sciences, Vancouver, British Columbia, Canada (L.A.W., J.I.F., C.M.G., J.R.S.)"
}
],
"family": "Wilson",
"given": "Lindsay A.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Platform Life Sciences, Vancouver, British Columbia, Canada (L.A.W., J.I.F., C.M.G., J.R.S.)"
}
],
"family": "Forrest",
"given": "Jamie I.",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0003-0538-275X",
"affiliation": [
{
"name": "Cytel, Vancouver, British Columbia, Canada (O.H., H.R.)"
}
],
"authenticated-orcid": false,
"family": "Ruton",
"given": "Hinda",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Health Research Methods, Evidence, and Impact, McMaster University, Hamilton, Ontario, Canada (L.T., S.S., P.M., G.H.G.)"
}
],
"family": "Sprague",
"given": "Sheila",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Health Research Methods, Evidence, and Impact, McMaster University, Hamilton, Ontario, Canada (L.T., S.S., P.M., G.H.G.)"
}
],
"family": "McKay",
"given": "Paula",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Platform Life Sciences, Vancouver, British Columbia, Canada (L.A.W., J.I.F., C.M.G., J.R.S.)"
}
],
"family": "Guo",
"given": "Christina M.",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0003-2352-5718",
"affiliation": [
{
"name": "Department of Health Research Methods, Evidence, and Impact, McMaster University, Hamilton, Ontario, Canada (L.T., S.S., P.M., G.H.G.)"
}
],
"authenticated-orcid": false,
"family": "Guyatt",
"given": "Gordon H.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Certara, Princeton, New Jersey, and Monash Institute of Pharmaceutical Sciences, Monash University, Melbourne, Victoria, Australia (C.R.R.)"
}
],
"family": "Rayner",
"given": "Craig R.",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-4715-0060",
"affiliation": [
{
"name": "Division of Infectious Diseases and International Medicine, Department of Medicine, University of Minnesota, Minneapolis, Minnesota (D.R.B.)"
}
],
"authenticated-orcid": false,
"family": "Boulware",
"given": "David R.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Medicine, Division of Respiratory Medicine, McGill University Health Centre, Montréal, Québec, Canada (N.E.)"
}
],
"family": "Ezer",
"given": "Nicole",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-2267-4239",
"affiliation": [
{
"name": "Division of Infectious Diseases, Department of Medicine, McGill University Health Centre, Montréal, Québec, Canada (T.C.L.)"
}
],
"authenticated-orcid": false,
"family": "Lee",
"given": "Todd C.",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0003-0783-0624",
"affiliation": [
{
"name": "Division of General Internal Medicine, Department of Medicine, McGill University Health Centre, Montréal, Québec, Canada (E.G.M.)"
}
],
"authenticated-orcid": false,
"family": "McDonald",
"given": "Emily Gibson",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-9993-2478",
"affiliation": [
{
"name": "School of Immunology and Microbial Sciences, Faculty of Life Sciences and Medicine, King’s College London, London, United Kingdom (M.B.)"
}
],
"authenticated-orcid": false,
"family": "Bafadhel",
"given": "Mona",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-0102-3453",
"affiliation": [
{
"name": "Nuffield Department of Primary Care Health Sciences, University of Oxford, Oxford, United Kingdom (C.B.)"
}
],
"authenticated-orcid": false,
"family": "Butler",
"given": "Christopher",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0001-7814-2349",
"affiliation": [
{
"name": "Platform Life Sciences, Vancouver, British Columbia, Canada (L.A.W., J.I.F., C.M.G., J.R.S.)"
}
],
"authenticated-orcid": false,
"family": "Rodrigues Silva",
"given": "Josue",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-2108-8246",
"affiliation": [
{
"name": "Global Health Institute, Georgetown University, Washington, DC (M.D.)"
}
],
"authenticated-orcid": false,
"family": "Dybul",
"given": "Mark",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "ViRx@Stanford, Stanford Biosecurity and Pandemic Preparedness Initiative, Stanford, California; Department of Health Research Methods, Evidence, and Impact, McMaster University, Hamilton, Ontario, Canada; Cytel, Vancouver, British Columbia, Canada; and Platform Life Sciences, Vancouver, British Columbia, Canada (E.J.M.)."
}
],
"family": "Mills",
"given": "Edward J.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Reis",
"given": "Gilmar",
"sequence": "additional"
},
{
"affiliation": [],
"family": "dos Santos Moreira Silva",
"given": "Eduardo Augusto",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Medeiros Silva",
"given": "Daniela Carla",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Quirino dos Santos",
"given": "Castilho Vitor",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ribeiro Nogueira",
"given": "Ana Maria",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Figueiredo Guimaraes Almeida",
"given": "Ana Paula",
"sequence": "additional"
},
{
"affiliation": [],
"family": "de Figueiredo Neto",
"given": "Adhemar Dias",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Cançado Monteiro Savassi",
"given": "Leonardo",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Barra Ribeiro",
"given": "Luciene",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Campos Simplicio",
"given": "Maria Izabel",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ferreira",
"given": "Thiago Santiago",
"sequence": "additional"
},
{
"affiliation": [],
"family": "de Souza Campos",
"given": "Vitoria Helena",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Thabane",
"given": "Lehana",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Forrest",
"given": "Jamie",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Mills",
"given": "Edward J.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Cameron",
"given": "William",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Orbinski",
"given": "James",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Singh",
"given": "Sonal",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Thorlund",
"given": "Kristian",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Haggstrom",
"given": "Jonas",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Costa",
"given": "Heloísa A.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Drumont",
"given": "Nayara S.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Bessa",
"given": "Stephanny K.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Vieira",
"given": "João V.B.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Campos",
"given": "Vania R",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Oliveira",
"given": "Rosemary M",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Suterio",
"given": "Lineria HM",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Rodrigues Silva",
"given": "Josue",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Junior",
"given": "Adelino de melo Freire",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Rocha",
"given": "Ana Carolina Fialho Dias",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Gonçalves",
"given": "Ana Paula de Faria",
"sequence": "additional"
},
{
"affiliation": [],
"family": "França",
"given": "Andressa",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Igídio",
"given": "Carlos Eduardo Dias",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Sousa",
"given": "Igor César de Oliveira",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Carvalho",
"given": "Iracema Luisa Quintino de",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Benavides",
"given": "Larissa Maria Manini",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Pereira",
"given": "Patrícia de Oliveira",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Sousa",
"given": "Phelipy Marques de",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Lima",
"given": "Sabrina de Almeida",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Pedroso",
"given": "Silvia Helena Sousa Pietra",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Resende",
"given": "Snaydia Viegas",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Rocha",
"given": "Wellington da Silva",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Alves",
"given": "Thamiris Caldeira",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Martins",
"given": "Aline Lúcia de Jesus",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Mesquita",
"given": "Amanda Gouvêa",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Mendes",
"given": "Amanda Larissa Alves",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Mendes",
"given": "Anna Beatriz Oliveira",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Oliveira",
"given": "Anna Clara de Jesus",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Cysne",
"given": "Carolina Couto de Azevedo",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Pereira",
"given": "Clara Bensemann Gontijo",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Siqueira",
"given": "Cecília Campos",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Dias",
"given": "Ednilson Barbosa",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Gonçalves",
"given": "Emanuelle Cristina Soares",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Guimarães",
"given": "Fernanda Leal",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Giorgeto",
"given": "Flávio Eduardo",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Rodrigues",
"given": "Gabriel Torga Saade",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Sousa",
"given": "Gislânia Neres de",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Rezende",
"given": "Júlia Campos de",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Moreira",
"given": "Júlia Diniz Assis",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Soares",
"given": "Isabela Canabrava",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Silveira",
"given": "João Victor Barbosa",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Souza",
"given": "Joselaine Gomes de",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ferreira",
"given": "Juliana Marques Santos",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Gonçalves",
"given": "Kênia Sthéfhane Guimarães",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Oliveira",
"given": "Laura Campos",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Almeida",
"given": "Layane Maria de",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Siqueira",
"given": "Letícia Campos",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Costa",
"given": "Letícia de Fátimas",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Maciel",
"given": "Lorena Gabrielle Miranda",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Peixoto",
"given": "Lorena Giovanna Silva",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Alves",
"given": "Lorena Mesquita",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Rodrigues",
"given": "Luana Rosa",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Almeida",
"given": "Luisa Andrade de",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Lara",
"given": "Marcela Luíza Amaral Resende",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Reis",
"given": "Luíza Lanna França",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Horácio",
"given": "Maria Eduarda Gomes",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Soares",
"given": "Maria Eduarda Martins",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Parreiras",
"given": "Maria Luiza Magalhães",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Siqueira",
"given": "Mariana Campos",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Mariano",
"given": "Mariana Cristina Flores",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Bragatto",
"given": "Marina Guimarães",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Silva",
"given": "Matheus Andrade Almeida e",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Santos",
"given": "Mayra Estefane Silva dos",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ribeiro",
"given": "Mirele Augusta Batista",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Drumond",
"given": "Nayara Santos",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Guimarães",
"given": "Pedro Hosken Fernandes",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Diniz",
"given": "Pollyana Maria Silva",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Lopes",
"given": "Roberta Dumont Paiva",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Oliveira",
"given": "Rosemary Muniz de",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Bessa",
"given": "Stephanny Kelvyn",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Anselmo",
"given": "Sheila Soares",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Vieira",
"given": "Tainara Silva",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Silva",
"given": "Tatiana Aparecida Gomes da",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Siqueira",
"given": "Thais Campos",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Carmo",
"given": "Vânia Regina",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Vale",
"given": "Vitor Augusto Lima do",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Oliveira",
"given": "Vitor Vilaça",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Santos",
"given": "Wedsley Sander Lopes dos",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Abreu",
"given": "Aline Daniele de Almeida",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Camargos",
"given": "Bernardo Saraiva Resende de",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Heringer",
"given": "Camila Araújo",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Filho",
"given": "Daniel Jacinto Mendonça",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Abreu",
"given": "Eriane Damasia Faria de Almeida",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Barnabé",
"given": "Felipe augusto Pereira",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Vieira",
"given": "Felipe Alves",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Carvalho",
"given": "Gabriella Fagundes",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Mussi",
"given": "Higor Gomes",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Macedo",
"given": "Hilton Cardoso Arruda",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Caciquinho",
"given": "Igor Bernardes",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Santos",
"given": "João Pedro Ribeiro",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Costa",
"given": "João Ricardo Garcia",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Cardoso",
"given": "José Lauro Guimarães",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Abreu",
"given": "Júlia Berlim de",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Carvalho",
"given": "Lorena Rodrigues de",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Oliveira",
"given": "Lorena Amaral de",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Matos",
"given": "Luana Rocha Oliveira",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Brito",
"given": "Marcela Ferreira",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Silva",
"given": "Mariana Cavaliere Batista e",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Rodrigues",
"given": "Matheus Lucca Ângelo Costa",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ferreira",
"given": "Mateus de Oliveira",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Perez",
"given": "Murilo Passinati",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Zanetti",
"given": "Natalia Batista",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Almeida",
"given": "Rafaela Ribeiro de",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Jamar",
"given": "Sarah Jeniffer da Silva",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Barbosa",
"given": "Sofia Caporalli",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Rios",
"given": "Sofia Vianna Von Bentzeen",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Rosa",
"given": "Stella Silva",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Morimoto",
"given": "Thomaz Takashi Ferreira",
"sequence": "additional"
},
{
"affiliation": [],
"family": "costa",
"given": "Verônica de Araújo Libânio",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Silva",
"given": "Vitor Augusto Alves da",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Batista",
"given": "Amanda Cypestre Alves",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Viana",
"given": "Ana Flávia Gomes",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Santos",
"given": "Ângela Rodrigues Vieira",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Flores",
"given": "Anna Luiza Ribeiro",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Dutra",
"given": "Arthur Guimarães Rodrigues",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Maia",
"given": "Bruna Luiza",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Almeida",
"given": "Clarisse Lourdes Nadú de",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Barbosa",
"given": "Daniel Domingues",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Medeiros",
"given": "Daniela Carla",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Silva",
"given": "Daniele Saionara da",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Rezende",
"given": "Diuly Ane Faria",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Andrade",
"given": "Felipe Menezes",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Batista",
"given": "Fernanda Rúbia",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Reis",
"given": "Gabriela Ferreira",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Paula",
"given": "Giovane Carvalho Constantino Ferreira de",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Júnior",
"given": "Geovani de Araújo",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Pires",
"given": "Henrique Amaral Fonseca",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Silva",
"given": "Jéssica Aguilar",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Rovetta",
"given": "Isabela Lovatti",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Rangel",
"given": "Júlia Silva",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Paula",
"given": "Júlia Lima Soares de",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Costa",
"given": "Juliana Oliveira",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Silva",
"given": "Lucas Júnio da",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Dias",
"given": "Luiz Gustavo Fernandes",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Oliveira",
"given": "Lívia Francino",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Barros",
"given": "Lucas Rodrigues Pereira",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Pessoa",
"given": "Lucas de Souza Gontijo",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Silva",
"given": "Lucas Klier",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Stabile",
"given": "Maria Luisa Lara Lanza",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Pinto",
"given": "Mariana da Rocha Assis",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Cunha",
"given": "Nicholas Vanzin",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Silva",
"given": "Paloma Alister Vilela da",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Soares",
"given": "Rafael Silva Rodrigues",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Oliveira",
"given": "Thais Gomes de",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Araújo",
"given": "Victor Oliveira de",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Arcanjo",
"given": "Wesley Costa",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Vanetti",
"given": "Alice Pinheiro",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Santos",
"given": "Ana Carolina Araújo Lage",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Freitas",
"given": "Ariade Gomes",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Santos",
"given": "Felipe Viana",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Pinheiro",
"given": "Fernanda Freitas",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Rocha",
"given": "Fernanda Perez",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Santos",
"given": "Filipe Viana",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Marcolino",
"given": "Giulia de Jesus",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Filho",
"given": "Humberto Drumond",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Mendonça",
"given": "Isabela Gontijo",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Faria",
"given": "Isadora Gomides",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Milhomem",
"given": "José Victor Mendes",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Mol",
"given": "Larissa Milagres",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Costa",
"given": "Letícia de Fátima",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Júnior",
"given": "Marcelo Bernardes Rocha",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Miranda",
"given": "Maria Constâncio",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Gonçalves",
"given": "Nathália Euclides",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ricco",
"given": "Suzana Cristina",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Nascimento",
"given": "Thiago Araújo do",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Pinheiro",
"given": "Amanda Luiza do Espírito Santo",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ferreira",
"given": "Ana Carolina Matos",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Silva",
"given": "Ana Caroline Santos",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Taveira",
"given": "Ana Luiza Silva",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Lima",
"given": "Bruno Victor de Souza",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Diana",
"given": "Débora Siqueira",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Silveira",
"given": "Emanuela Pontes Pereira",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Gonçalves",
"given": "Francisca Lina Campos",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Costa",
"given": "Gabriel Corrêa",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Souza",
"given": "Geraldo César Barroso de",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Alves",
"given": "Guilherme Aurelio D'Oliveira",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Neto",
"given": "Isadora Luiza Teixeira",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Inácio",
"given": "Jordanna Porto",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Silvério",
"given": "José Paulo Ribeiro",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Marques",
"given": "Lara Silveira",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Nascimento",
"given": "Larissa Martins do",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Moreira",
"given": "Larissa Oliveira",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Oliveira",
"given": "Lorena Luiza Soares de",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Mussi",
"given": "Lucas Eduardo Lessa",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ruas",
"given": "Lucas Medeiros",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Barbosa",
"given": "Luiza Nogueira Assis",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Sousa",
"given": "Luiz Otávio de",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Fernandes",
"given": "Marcelo Rodrigues da Costa",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Souza",
"given": "Maria Eduarda Guimarães de",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Calazans",
"given": "Maria Thereza Figueiredo Bélem",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Bastos",
"given": "Mariana Rodrigues Marinho de",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Alvarenga",
"given": "Pedro Henrique Arcanjo",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Cujuí",
"given": "Quimberli Vassinave",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Diniz",
"given": "Rafaela Valadares",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Souza",
"given": "Rafael Cota Andrade Ferreira de",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Silva",
"given": "Rita de Cássia",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Corrêa",
"given": "Vinícius Alves",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Oliveira",
"given": "Vinícius Carvalho de",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Cunha",
"given": "Vitória Aparecida",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Santos",
"given": "Viviane Costa",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Almeida",
"given": "Ana Paula Figueiredo Guimarães de",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Carvalho",
"given": "Ana Paula Guimarães Alves de",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Jorge",
"given": "Antônio Henrique Batista",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ribeiro",
"given": "Artur Pimenta",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Sena",
"given": "Elisângela Santos",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Veloso",
"given": "Cleide Rocha",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Murta",
"given": "Gonçalvino Eleutério",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Cardoso",
"given": "Inara Maria Gomes",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Alves",
"given": "João Warley",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Veloso",
"given": "Jorge Fernando Rocha",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Figueiredo",
"given": "Joyce Souto Barbosa Pimenta de",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Lopes",
"given": "Karolina Campos Sampaio",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Souza",
"given": "Larissa Vieira",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Costa",
"given": "Loren Montanha",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Souza",
"given": "Ludimila Pereira de",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Lima",
"given": "Maria de Fátima César",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Gonçalves",
"given": "Maria Izabel Silveira",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Mendes",
"given": "Marizete Gislaine",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Silva",
"given": "Mayra Darlliane Loiola",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Fonseca",
"given": "Raissa Rocha e",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Oliveira",
"given": "Samuel de Paiva",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ferro",
"given": "Thiago Rodrigues",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Cruz Milagres",
"given": "Aline",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Estevam",
"given": "Ana Carolina Aguiar",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Oliveira",
"given": "Ana Martins de",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Oliveira",
"given": "André Henrique de Sousa",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Bitarães",
"given": "Carina",
"sequence": "additional"
},
{
"affiliation": [],
"family": "França",
"given": "Carla Stefany Oliveira",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Freire",
"given": "Hayka K. Rodrigues",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Gama",
"given": "Laís Aparecida dos Reis",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Pereira",
"given": "Laylla Michelle",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Silva",
"given": "Luciléia Alcântara Gomes e",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Assis",
"given": "Patrícia Santana",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Andrade",
"given": "Regiane Aparecida de",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Silva",
"given": "Thaís de Paula Ferreira da",
"sequence": "additional"
},
{
"affiliation": [],
"family": "César",
"given": "Thiago de Alcântara",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Rabelo",
"given": "Adriana Marcos Mendes",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Rosa",
"given": "Aline do Carmo",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Wherberth",
"given": "Ana Paula Vilas Boas",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Souza",
"given": "André Gustavo Pinto de",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Gomes",
"given": "André Luiz Souza",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Oliveira",
"given": "Arthur Henrique Nunes Leite",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Siqueira",
"given": "Damiana Rogai",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Pinto",
"given": "Emanuel Peixoto",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Oliveira",
"given": "Felipe Coelho Soares de",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Damaceno",
"given": "Felipe Fraga",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Rhis",
"given": "Guilherme",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Almeida",
"given": "Iandra Silva",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Borges",
"given": "Igor Barros de Araújo",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Rocha",
"given": "Igor Brandão",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Silva",
"given": "Jéssica Genoveva Boline Passarelli Capaz Pinto da",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Júnior",
"given": "Jorge Carlos do Amaral",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Marques",
"given": "José Pio",
"sequence": "additional",
"suffix": "II"
},
{
"affiliation": [],
"family": "Vargas",
"given": "Laura Lima",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Batista",
"given": "Lorena Cristina Ferreira",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Fava",
"given": "Luiz Eduardo Coelho",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Giles",
"given": "Luiza de Azevedo Freitas",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Marques",
"given": "Marina Lacerda",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Veríssimo",
"given": "Maura Alina Morais",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Paula",
"given": "Neila Rodrigues Vargas de",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Demuner",
"given": "Pâmela de Sousa Dias",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Pouzas",
"given": "Ramail Santos",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Marco",
"given": "Roberta Coelho de",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Fernandes",
"given": "Rafaella Rosa de Oliveira",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Mourão",
"given": "Ronan Figueiredo",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Souza",
"given": "Sabrina Stefany da Silva",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ferraz",
"given": "Sara Silva",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Amorim",
"given": "Ana Beatriz",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Martins",
"given": "Carolina Francisca",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Diniz Callegari",
"given": "Eduardo",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Cordeiro",
"given": "Geovanna Carla",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Lourenço",
"given": "Huisnei Ferreira",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Santos",
"given": "João Victor Fagundes dos",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Veloso",
"given": "João Pedro Donato",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Santos",
"given": "Júlia Alves",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Nogueira",
"given": "Júlia senra",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Nascimento",
"given": "Jussara Carolina Assis Ribeiro",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Martins",
"given": "Lavínia de Fátima Baldim",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Menezes",
"given": "Lucca Batista Rocha de",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Silva",
"given": "Maria Clara Damaso da",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Santos",
"given": "Mariana das Graças de Aquino",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Leão",
"given": "Rafael Augusto de Carvalho",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Sales",
"given": "Solange Santos Moreira",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Moreira",
"given": "Vitor Franco",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Friedmann",
"given": "Amanda Diniz",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Chaves",
"given": "Barbara Carvalho",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Oliveira",
"given": "Janaína Leyner de Andrade",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Diniz",
"given": "Júlia Silva",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Braga",
"given": "Luiz Felipe de Oliveira",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Duarte",
"given": "Luiza Gabriele Dutra",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Duque",
"given": "Marcela Julia Rufino",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Emiliano",
"given": "Mariana Cristina Flores",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Silveira",
"given": "Rafaela Carolina",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Moreira",
"given": "Virgínia Mara Santos",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Bedetti",
"given": "Aline Dias",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Campos",
"given": "Amanda Faria Simoni",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Almeida",
"given": "Andressa de",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Costa",
"given": "Anna Carolina Motta",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Cunha",
"given": "Clara Tornelli de Almeida",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Mascarenhas",
"given": "Cristiana Silva",
"sequence": "additional"
},
{
"affiliation": [],
"family": "silva",
"given": "Daniel Ananias da",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ribeiro",
"given": "Diana Antoniazzi de Sá",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Brandão",
"given": "Diana Luiza Marinho",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Morais",
"given": "Érico Henrique Araújo de",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Soares",
"given": "Felipe César",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Borba",
"given": "Flávia Marcella Sena Gonçalves",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Gomes",
"given": "Karinna Guimarães",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Estevam",
"given": "Marco Wellington Junio",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Lopes",
"given": "Matheus Ramos",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Santos",
"given": "Marina Eduarda",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Silva",
"given": "Maryane de Oliveira",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Silva",
"given": "Nágella Samara Eleutério da",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Carneir",
"given": "Rafael Rocha",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Silva",
"given": "Rayane Elen Fernandes",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Wondollinger",
"given": "Ruan Carling Schott",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Menezes",
"given": "Yuri Barbosa de",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Rezende",
"given": "Bárbara Ellen Souza",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Costa",
"given": "Catarina Giovanna Simões",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Costa",
"given": "Gabriel Antônio Simões",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Cardoso",
"given": "Gabriel Marcos theodoro",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Sousa",
"given": "Graziela César de",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Silva",
"given": "Graziele Aparecida",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Campos",
"given": "Isabela Cristina de Oliveira",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Souza",
"given": "Lara Silva",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Rabello",
"given": "Laura Nacife",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Oliveira",
"given": "Roberta Ellen Santos",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Roberti",
"given": "Bárbara Nogueira",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Garcia",
"given": "Bruno Reis",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Gelape",
"given": "Bruno Rocha",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Figueiredo",
"given": "Carina Daniela Lira Moreira",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Cachoeira",
"given": "João Vitor Rocha",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Santos",
"given": "Júlia Romano Ferreira",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Silva",
"given": "Sarah de Resende",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Rios",
"given": "Thalita Oliva",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Quadros",
"given": "Túlio Fonseca e Silva",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Barber",
"given": "Bethany",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Capraru",
"given": "Camelia",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Lemieux",
"given": "Camille",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Li",
"given": "Daniel",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Smookler",
"given": "David",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Booth",
"given": "Joshua",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Kuczynski",
"given": "Magdalena",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Marquez",
"given": "Maria Kristina",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Noureldin",
"given": "Seham",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Jeganathan",
"given": "Shinthuka",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Tharmaratnam",
"given": "Tharmegan",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Aleyadeh",
"given": "Wesam",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Powis",
"given": "Jeff",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Taylor",
"given": "Maureen",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Chan",
"given": "Adrienne",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Salvant",
"given": "Elsa",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Graham",
"given": "Christopher",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Dagher",
"given": "Marc",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Duarte",
"given": "William Parreira",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Castro",
"given": "Duílio de",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Silveira",
"given": "Flávio Pimenta",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ribeiro",
"given": "Alber Alípio",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Medioli",
"given": "Vittorio",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Rocha",
"given": "Augusto Viana da",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Oliveira",
"given": "Hilton Soares de",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Amaral",
"given": "Tânia Maria de Resende",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Pereira",
"given": "Christiano Augusto Xavier",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Tomé",
"given": "Nádia Cristina Dias Duarte",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Pereira",
"given": "João Marcelo Diegues",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ribeiro",
"given": "Diogo Jonata",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Souto",
"given": "Humberto Guimarães",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Gonçalves",
"given": "Dulce Pimenta",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Barcelos",
"given": "Avimar de Melo",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Merlo",
"given": "André Luiz Coelho",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Leite",
"given": "Edna Gomes",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Sangali",
"given": "Caroline Martins",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Santos",
"given": "Angelo Oswaldo de Araujo",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Nascimento",
"given": "Glauciane Resende do",
"sequence": "additional"
},
{
"affiliation": [],
"name": "TOGETHER Investigators",
"sequence": "additional"
}
],
"container-title": "Annals of Internal Medicine",
"container-title-short": "Ann Intern Med",
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2023,
4,
17
]
],
"date-time": "2023-04-17T21:00:33Z",
"timestamp": 1681765233000
},
"deposited": {
"date-parts": [
[
2023,
5,
20
]
],
"date-time": "2023-05-20T21:03:36Z",
"timestamp": 1684616616000
},
"funder": [
{
"name": "Latona Foundation"
},
{
"DOI": "10.13039/100016608",
"doi-asserted-by": "publisher",
"name": "Rainwater Charitable Foundation"
},
{
"name": "FastGrants"
}
],
"indexed": {
"date-parts": [
[
2024,
4,
2
]
],
"date-time": "2024-04-02T05:16:16Z",
"timestamp": 1712034976143
},
"is-referenced-by-count": 15,
"issue": "5",
"issued": {
"date-parts": [
[
2023,
5
]
]
},
"journal-issue": {
"issue": "5",
"published-print": {
"date-parts": [
[
2023,
5
]
]
}
},
"language": "en",
"member": "4285",
"original-title": [],
"page": "667-675",
"prefix": "10.7326",
"published": {
"date-parts": [
[
2023,
5
]
]
},
"published-print": {
"date-parts": [
[
2023,
5
]
]
},
"publisher": "American College of Physicians",
"reference": [
{
"DOI": "10.1038/cddis.2014.301",
"doi-asserted-by": "publisher",
"key": "r1-M223305"
},
{
"DOI": "10.1016/S2214-109X(21)00448-4",
"doi-asserted-by": "publisher",
"key": "r2-M223305"
},
{
"DOI": "10.1001/jama.2020.22760",
"doi-asserted-by": "publisher",
"key": "r3-M223305"
},
{
"DOI": "10.4269/ajtmh.21-1310",
"doi-asserted-by": "publisher",
"key": "r4-M223305"
},
{
"DOI": "10.4269/ajtmh.21-1310",
"doi-asserted-by": "publisher",
"key": "r5-M223305"
},
{
"DOI": "10.1016/S0140-6736(21)01744-X",
"doi-asserted-by": "publisher",
"key": "r6-M223305"
},
{
"DOI": "10.3389/fphar.2021.652688",
"doi-asserted-by": "publisher",
"key": "r7-M223305"
},
{
"DOI": "10.1016/s0149-2918(04)90050-1",
"doi-asserted-by": "publisher",
"key": "r8-M223305"
},
{
"DOI": "10.1056/NEJMoa2201662",
"doi-asserted-by": "publisher",
"key": "r9-M223305"
},
{
"DOI": "10.1186/s13063-019-3664-1",
"doi-asserted-by": "publisher",
"key": "r10-M223305"
},
{
"DOI": "10.1056/NEJMra1510062",
"doi-asserted-by": "publisher",
"key": "r11-M223305"
},
{
"DOI": "10.12688/gatesopenres.13304.2",
"doi-asserted-by": "publisher",
"key": "r12-M223305"
},
{
"DOI": "10.1001/jamanetworkopen.2021.6468",
"doi-asserted-by": "publisher",
"key": "r13-M223305"
},
{
"DOI": "10.1016/j.lana.2021.100142",
"doi-asserted-by": "publisher",
"key": "r14-M223305"
},
{
"DOI": "10.1056/NEJMoa2115869",
"doi-asserted-by": "publisher",
"key": "r15-M223305"
},
{
"DOI": "10.1056/NEJMoa2209760",
"doi-asserted-by": "publisher",
"key": "r16-M223305"
},
{
"DOI": "10.1016/S1473-3099(20)30483-7",
"doi-asserted-by": "publisher",
"key": "r17-M223305"
},
{
"DOI": "10.1503/cmaj.200077",
"doi-asserted-by": "publisher",
"key": "r18-M223305"
},
{
"DOI": "10.12688/gatesopenres.13304.2",
"doi-asserted-by": "publisher",
"key": "r19-M223305"
},
{
"DOI": "10.1093/biomet/81.3.515",
"doi-asserted-by": "publisher",
"key": "r20-M223305"
},
{
"DOI": "10.1001/jamanetworkopen.2022.6269",
"doi-asserted-by": "publisher",
"key": "r21-M223305"
},
{
"DOI": "10.1016/j.jclinepi.2012.07.012",
"doi-asserted-by": "publisher",
"key": "r22-M223305"
}
],
"reference-count": 22,
"references-count": 22,
"relation": {},
"resource": {
"primary": {
"URL": "https://www.acpjournals.org/doi/10.7326/M22-3305"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [
"General Medicine",
"Internal Medicine"
],
"subtitle": [
"A Randomized Platform Trial"
],
"title": "Oral Fluvoxamine With Inhaled Budesonide for Treatment of Early-Onset COVID-19",
"type": "journal-article",
"volume": "176"
}
reis10
