Inhaled budesonide for COVID-19 in people at high risk of complications in the community in the UK (PRINCIPLE): a randomised, controlled, open-label, adaptive platform trial
et al., The Lancet, doi:10.1016/S0140-6736(21)01744-X, PRINCIPLE, ISRCTN86534580, Apr 2021 (preprint)
Budesonide for COVID-19
27th treatment shown to reduce risk in
September 2021, now with p = 0.000003 from 15 studies, recognized in 10 countries.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
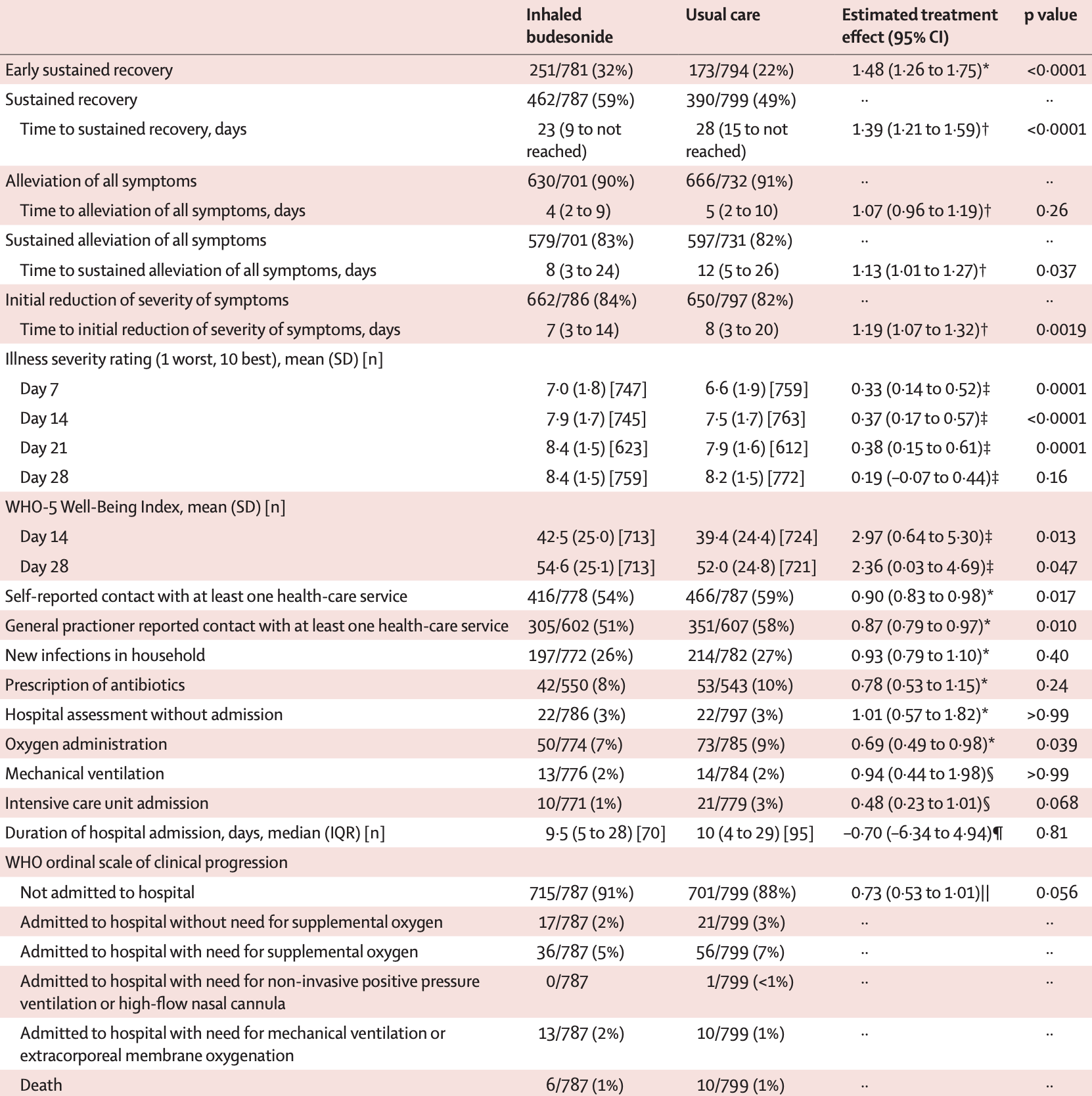
Results from the PRINCIPLE trial, 1,073 treated with budesonide starting a median of 6 days after symptom onset, showing lower hospitalization/death, and faster recovery with treatment.
Targeted administration to the respiratory tract provides treatment directly
to the typical source of initial SARS-CoV-2 infection and replication, and
allows for rapid onset of action, higher local drug concentration, and reduced systemic side effects (early treatment may be more beneficial).
Standard of Care (SOC) for COVID-19 in the study country,
the United Kingdom, is very poor with very low average efficacy for approved treatments1.
The United Kingdom focused on expensive high-profit treatments, approving only one low-cost early treatment, which required a prescription and had limited adoption. The high-cost prescription treatment strategy reduces the probability of early treatment due to access and cost barriers, and eliminates complementary and synergistic benefits seen with many low-cost treatments.
|
risk of death, 39.1% lower, RR 0.61, p = 0.45, treatment 6 of 787 (0.8%), control 10 of 799 (1.3%), NNT 204.
|
|
risk of mechanical ventilation, 6.0% lower, RR 0.94, p = 1.00, treatment 13 of 776 (1.7%), control 14 of 784 (1.8%), NNT 905.
|
|
risk of ICU admission, 52.0% lower, RR 0.48, p = 0.07, treatment 10 of 771 (1.3%), control 21 of 779 (2.7%), NNT 71.
|
|
risk of death/hospitalization, 25.0% lower, RR 0.75, p = 0.96, treatment 72 of 787 (9.1%), control 116 of 1,069 (10.9%), NNT 59, adjusted per study, day 28.
|
|
recovery time, 17.4% lower, relative time 0.83, p = 0.001, treatment 787, control 1,069, adjusted per study, inverted to make RR<1 favor treatment.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Yu et al., 12 Apr 2021, Randomized Controlled Trial, United Kingdom, peer-reviewed, 26 authors, study period 27 November, 2020 - 31 March, 2021, average treatment delay 6.0 days, trial ISRCTN86534580 (PRINCIPLE).
Inhaled budesonide for COVID-19 in people at high risk of complications in the community in the UK (PRINCIPLE): a randomised, controlled, open-label, adaptive platform trial
The Lancet, doi:10.1016/s0140-6736(21)01744-x
Background A previous efficacy trial found benefit from inhaled budesonide for COVID-19 in patients not admitted to hospital, but effectiveness in high-risk individuals is unknown. We aimed to establish whether inhaled budesonide reduces time to recovery and COVID-19-related hospital admissions or deaths among people at high risk of complications in the community.
Methods PRINCIPLE is a multicentre, open-label, multi-arm, randomised, controlled, adaptive platform trial done remotely from a central trial site and at primary care centres in the UK. Eligible participants were aged 65 years or older or 50 years or older with comorbidities, and unwell for up to 14 days with suspected COVID-19 but not admitted to hospital. Participants were randomly assigned to usual care, usual care plus inhaled budesonide (800 µg twice daily for 14 days), or usual care plus other interventions, and followed up for 28 days. Participants were aware of group assignment. The coprimary endpoints are time to first self-reported recovery and hospital admission or death related to COVID-19, within 28 days, analysed using Bayesian models. The primary analysis population included all eligible SARS-CoV-2-positive participants randomly assigned to budesonide, usual care, and other interventions, from the start of the platform trial until the budesonide group was closed. This trial is registered at the ISRCTN registry (ISRCTN86534580) and is ongoing. Findings The trial began enrolment on April 2, 2020, with randomisation to budesonide from Nov 27, 2020, until March 31, 2021, when the prespecified time to recovery superiority criterion was met. 4700 participants were randomly assigned to budesonide (n=1073), usual care alone (n=1988), or other treatments (n=1639). The primary analysis model includes 2530 SARS-CoV-2-positive participants, with 787 in the budesonide group, 1069 in the usual care group, and 974 receiving other treatments. There was a benefit in time to first self-reported recovery of an estimated 2•94 days (95% Bayesian credible interval [BCI] 1•19 to 5•12) in the budesonide group versus the usual care group (11•8 days [95% BCI 10•0 to 14•1] vs 14•7 days [12•3 to 18•0]; hazard ratio 1•21 [95% BCI 1•08 to 1•36]), with a probability of superiority greater than 0•999, meeting the prespecified superiority threshold of 0•99. For the hospital admission or death outcome, the estimated rate was 6•8% (95% BCI 4•1 to 10•2) in the budesonide group versus 8•8% (5•5 to 12•7) in the usual care group (estimated absolute difference 2•0% [95% BCI -0•2 to 4•5]; odds ratio 0•75 [95% BCI 0•55 to 1•03]), with a probability of superiority 0•963, below the prespecified superiority threshold of 0•975. Two participants in the budesonide group and four in the usual care group had serious adverse events (hospital admissions unrelated to COVID-19). Interpretation Inhaled budesonide improves time to recovery, with a chance of also reducing hospital admissions or deaths (although our results..
References
Aveyard, Gao, Lindson, Association between pre-existing respiratory disease and its treatment, and severe COVID-19: a population cohort study, Lancet Respir Med, doi:10.1016/S2213-2600(21)00095-3
Beigel, Tomashek, Dodd, Remdesivir for the treatment of Covid-19-final report, N Engl J Med
Butler, Dorward, Yu, Azithromycin for community treatment of suspected COVID-19 in people at increased risk of an adverse clinical course in the UK (PRINCIPLE): a randomised, controlled, open-label, adaptive platform trial, Lancet
Butler, Yu, Dorward, Doxycycline for community treatment of suspected COVID-19 in people at high risk of adverse outcomes in the UK (PRINCIPLE): a randomised, controlled, open-label, adaptive platform trial, Lancet Respir Med, doi:10.1016/S2213-2600(21)00310-6
Gordon, Mouncey, Al-Beidh, Interleukin-6 receptor antagonists in critically ill patients with Covid-19, N Engl J Med
Patel, Dorward, Yu, Hobbs, Butler, Inclusion and diversity in the PRINCIPLE trial, Lancet
Ramakrishnan, Nicolau, Jr, Langford, Inhaled budesonide in the treatment of early COVID-19 (STOIC): a phase 2, open-label, randomised controlled trial, Lancet Respir Med
Schultze, Walker, Mackenna, Risk of COVID-19-related death among patients with chronic obstructive pulmonary disease or asthma prescribed inhaled corticosteroids: an observational cohort study using the OpenSAFELY platform, Lancet Respir Med
Skipper, Pastick, Engen, Hydroxychloroquine in nonhospitalized adults with early COVID-19 : a randomized trial, Ann Intern Med
Tardif, Bouabdallaoui, Allier, Efficacy of colchicine in non-hospitalized patients with COVID-19, medRxiv, doi:10.1101/2021.01.26.21250494
Topp, Østergaard, Søndergaard, Bech, The WHO-5 Well-Being Index: a systematic review of the literature, Psychother Psychosom
Verity, Okell, Dorigatti, Estimates of the severity of coronavirus disease 2019: a model-based analysis, Lancet Infect Dis
Welte, SARS-CoV-2-triggered immune reaction: for COVID-19, nothing is as old as yesterday's knowledge, Am J Respir Crit Care Med
Who, WHO model list of essential medicines, 21st list
Woloshin, Patel, Kesselheim, False negative tests for SARS-CoV-2 infection -challenges and implications, N Engl J Med
Woodcock, Lavange, Master protocols to study multiple therapies, multiple diseases, or both, N Engl J Med
DOI record:
{
"DOI": "10.1016/s0140-6736(21)01744-x",
"ISSN": [
"0140-6736"
],
"URL": "http://dx.doi.org/10.1016/S0140-6736(21)01744-X",
"alternative-id": [
"S014067362101744X"
],
"assertion": [
{
"label": "This article is maintained by",
"name": "publisher",
"value": "Elsevier"
},
{
"label": "Article Title",
"name": "articletitle",
"value": "Inhaled budesonide for COVID-19 in people at high risk of complications in the community in the UK (PRINCIPLE): a randomised, controlled, open-label, adaptive platform trial"
},
{
"label": "Journal Title",
"name": "journaltitle",
"value": "The Lancet"
},
{
"label": "CrossRef DOI link to publisher maintained version",
"name": "articlelink",
"value": "https://doi.org/10.1016/S0140-6736(21)01744-X"
},
{
"label": "CrossRef DOI link to the associated document",
"name": "associatedlink",
"value": "https://doi.org/10.1016/S0140-6736(21)01809-2"
},
{
"label": "Content Type",
"name": "content_type",
"value": "article"
},
{
"label": "Copyright",
"name": "copyright",
"value": "© 2021 The Author(s). Published by Elsevier Ltd."
}
],
"author": [
{
"affiliation": [],
"family": "Yu",
"given": "Ly-Mee",
"sequence": "first"
},
{
"affiliation": [],
"family": "Bafadhel",
"given": "Mona",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Dorward",
"given": "Jienchi",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Hayward",
"given": "Gail",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Saville",
"given": "Benjamin R",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Gbinigie",
"given": "Oghenekome",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Van Hecke",
"given": "Oliver",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ogburn",
"given": "Emma",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Evans",
"given": "Philip H",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Thomas",
"given": "Nicholas P B",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Patel",
"given": "Mahendra G",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Richards",
"given": "Duncan",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Berry",
"given": "Nicholas",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Detry",
"given": "Michelle A",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Saunders",
"given": "Christina",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Fitzgerald",
"given": "Mark",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Harris",
"given": "Victoria",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Shanyinde",
"given": "Milensu",
"sequence": "additional"
},
{
"affiliation": [],
"family": "de Lusignan",
"given": "Simon",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Andersson",
"given": "Monique I",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Barnes",
"given": "Peter J",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Russell",
"given": "Richard E K",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Nicolau",
"given": "Dan V",
"sequence": "additional",
"suffix": "Jr"
},
{
"affiliation": [],
"family": "Ramakrishnan",
"given": "Sanjay",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Hobbs",
"given": "F D Richard",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Butler",
"given": "Christopher C",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Yu",
"given": "Ly-Mee",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Bafadhel",
"given": "Mona",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Dorward",
"given": "Jienchi",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Hayward",
"given": "Gail",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Saville",
"given": "Benjamin R",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Gbinigie",
"given": "Oghenekome",
"sequence": "additional"
},
{
"affiliation": [],
"family": "van Hecke",
"given": "Oliver",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ogburn",
"given": "Emma",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Evans",
"given": "Philip H",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Thomas",
"given": "Nicholas PB",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Patel",
"given": "Mahendra G",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Richards",
"given": "Duncan",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Berry",
"given": "Nicholas",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Detry",
"given": "Michelle A",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Saunders",
"given": "Christina T",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Fitzgerald",
"given": "Mark",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Harris",
"given": "Victoria",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Shanyinde",
"given": "Milensu",
"sequence": "additional"
},
{
"affiliation": [],
"family": "de Lusignan",
"given": "Simon",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Andersson",
"given": "Monique I",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Barnes",
"given": "Peter J",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Russell",
"given": "Richard EK",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Nicolau",
"given": "Dan V",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ramakrishnan",
"given": "Sanjay",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Hobbs",
"given": "FD Richard",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Butler",
"given": "Christopher C",
"sequence": "additional"
}
],
"container-title": "The Lancet",
"container-title-short": "The Lancet",
"content-domain": {
"crossmark-restriction": true,
"domain": [
"clinicalkey.fr",
"clinicalkey.jp",
"clinicalkey.com.au",
"clinicalkey.es",
"clinicalkey.com",
"em-consulte.com",
"thelancet.com",
"elsevier.com",
"sciencedirect.com"
]
},
"created": {
"date-parts": [
[
2021,
8,
10
]
],
"date-time": "2021-08-10T22:31:45Z",
"timestamp": 1628634705000
},
"deposited": {
"date-parts": [
[
2022,
11,
2
]
],
"date-time": "2022-11-02T20:10:08Z",
"timestamp": 1667419808000
},
"funder": [
{
"DOI": "10.13039/501100000272",
"doi-asserted-by": "publisher",
"name": "NIHR"
}
],
"indexed": {
"date-parts": [
[
2024,
4,
9
]
],
"date-time": "2024-04-09T06:37:33Z",
"timestamp": 1712644653121
},
"is-referenced-by-count": 194,
"issue": "10303",
"issued": {
"date-parts": [
[
2021,
9
]
]
},
"journal-issue": {
"issue": "10303",
"published-print": {
"date-parts": [
[
2021,
9
]
]
}
},
"language": "en",
"license": [
{
"URL": "https://www.elsevier.com/tdm/userlicense/1.0/",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2021,
9,
1
]
],
"date-time": "2021-09-01T00:00:00Z",
"timestamp": 1630454400000
}
},
{
"URL": "http://creativecommons.org/licenses/by/4.0/",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2021,
7,
28
]
],
"date-time": "2021-07-28T00:00:00Z",
"timestamp": 1627430400000
}
}
],
"link": [
{
"URL": "https://api.elsevier.com/content/article/PII:S014067362101744X?httpAccept=text/xml",
"content-type": "text/xml",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://api.elsevier.com/content/article/PII:S014067362101744X?httpAccept=text/plain",
"content-type": "text/plain",
"content-version": "vor",
"intended-application": "text-mining"
}
],
"member": "78",
"original-title": [],
"page": "843-855",
"prefix": "10.1016",
"published": {
"date-parts": [
[
2021,
9
]
]
},
"published-print": {
"date-parts": [
[
2021,
9
]
]
},
"publisher": "Elsevier BV",
"reference": [
{
"DOI": "10.1001/jama.2020.22813",
"article-title": "Therapy for early COVID-19: a critical need",
"author": "Kim",
"doi-asserted-by": "crossref",
"first-page": "2149",
"journal-title": "JAMA",
"key": "10.1016/S0140-6736(21)01744-X_bib1",
"volume": "324",
"year": "2020"
},
{
"DOI": "10.1016/S2213-2600(20)30314-3",
"article-title": "Inhaled corticosteroids in virus pandemics: a treatment for COVID-19?",
"author": "Nicolau",
"doi-asserted-by": "crossref",
"first-page": "846",
"journal-title": "Lancet Respir Med",
"key": "10.1016/S0140-6736(21)01744-X_bib2",
"volume": "8",
"year": "2020"
},
{
"DOI": "10.1016/S2213-2600(20)30167-3",
"article-title": "Do chronic respiratory diseases or their treatment affect the risk of SARS-CoV-2 infection?",
"author": "Halpin",
"doi-asserted-by": "crossref",
"first-page": "436",
"journal-title": "Lancet Respir Med",
"key": "10.1016/S0140-6736(21)01744-X_bib3",
"volume": "8",
"year": "2020"
},
{
"DOI": "10.1016/j.jaci.2020.09.034",
"article-title": "Inhaled corticosteroids downregulate the SARS-CoV-2 receptor ACE2 in COPD through suppression of type I interferon",
"author": "Finney",
"doi-asserted-by": "crossref",
"first-page": "510",
"journal-title": "J Allergy Clin Immunol",
"key": "10.1016/S0140-6736(21)01744-X_bib4",
"volume": "147",
"year": "2021"
},
{
"DOI": "10.1164/rccm.202003-0821OC",
"article-title": "COVID-19-related genes in sputum cells in asthma. Relationship to demographic features and corticosteroids",
"author": "Peters",
"doi-asserted-by": "crossref",
"first-page": "83",
"journal-title": "Am J Respir Crit Care Med",
"key": "10.1016/S0140-6736(21)01744-X_bib5",
"volume": "202",
"year": "2020"
},
{
"DOI": "10.1016/j.cell.2020.02.052",
"article-title": "SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor",
"author": "Hoffmann",
"doi-asserted-by": "crossref",
"first-page": "271",
"journal-title": "Cell",
"key": "10.1016/S0140-6736(21)01744-X_bib6",
"volume": "181",
"year": "2020"
},
{
"DOI": "10.1128/JVI.01648-20",
"article-title": "The inhaled steroid ciclesonide blocks SARS-CoV-2 RNA replication by targeting the viral replication-transcription complex in cultured cells",
"author": "Matsuyama",
"doi-asserted-by": "crossref",
"first-page": "e01620",
"journal-title": "J Virol",
"key": "10.1016/S0140-6736(21)01744-X_bib7",
"volume": "95",
"year": "2020"
},
{
"DOI": "10.1056/NEJMoa2021436",
"article-title": "Dexamethasone in hospitalized patients with Covid-19",
"author": "Horby",
"doi-asserted-by": "crossref",
"first-page": "693",
"journal-title": "N Engl J Med",
"key": "10.1016/S0140-6736(21)01744-X_bib8",
"volume": "384",
"year": "2021"
},
{
"DOI": "10.1001/jama.2020.17023",
"article-title": "Association between administration of systemic corticosteroids and mortality among critically ill patients with COVID-19: a meta-analysis",
"author": "Sterne",
"doi-asserted-by": "crossref",
"first-page": "1330",
"journal-title": "JAMA",
"key": "10.1016/S0140-6736(21)01744-X_bib9",
"volume": "324",
"year": "2020"
},
{
"DOI": "10.1164/rccm.202011-4194ED",
"article-title": "SARS-CoV-2-triggered immune reaction: for COVID-19, nothing is as old as yesterday's knowledge",
"author": "Welte",
"doi-asserted-by": "crossref",
"first-page": "156",
"journal-title": "Am J Respir Crit Care Med",
"key": "10.1016/S0140-6736(21)01744-X_bib10",
"volume": "203",
"year": "2021"
},
{
"DOI": "10.1016/S2213-2600(20)30415-X",
"article-title": "Risk of COVID-19-related death among patients with chronic obstructive pulmonary disease or asthma prescribed inhaled corticosteroids: an observational cohort study using the OpenSAFELY platform",
"author": "Schultze",
"doi-asserted-by": "crossref",
"first-page": "1106",
"journal-title": "Lancet Respir Med",
"key": "10.1016/S0140-6736(21)01744-X_bib11",
"volume": "8",
"year": "2020"
},
{
"DOI": "10.1016/S2213-2600(21)00095-3",
"article-title": "Association between pre-existing respiratory disease and its treatment, and severe COVID-19: a population cohort study",
"author": "Aveyard",
"doi-asserted-by": "crossref",
"journal-title": "Lancet Respir Med",
"key": "10.1016/S0140-6736(21)01744-X_bib12",
"year": "2021"
},
{
"DOI": "10.1016/S2213-2600(21)00160-0",
"article-title": "Inhaled budesonide in the treatment of early COVID-19 (STOIC): a phase 2, open-label, randomised controlled trial",
"author": "Ramakrishnan",
"doi-asserted-by": "crossref",
"first-page": "763",
"journal-title": "Lancet Respir Med",
"key": "10.1016/S0140-6736(21)01744-X_bib13",
"volume": "9",
"year": "2021"
},
{
"DOI": "10.1056/NEJMra1510062",
"article-title": "Master protocols to study multiple therapies, multiple diseases, or both",
"author": "Woodcock",
"doi-asserted-by": "crossref",
"first-page": "62",
"journal-title": "N Engl J Med",
"key": "10.1016/S0140-6736(21)01744-X_bib14",
"volume": "377",
"year": "2017"
},
{
"DOI": "10.1016/S0140-6736(21)00461-X",
"article-title": "Azithromycin for community treatment of suspected COVID-19 in people at increased risk of an adverse clinical course in the UK (PRINCIPLE): a randomised, controlled, open-label, adaptive platform trial",
"author": "Butler",
"doi-asserted-by": "crossref",
"first-page": "1063",
"journal-title": "Lancet",
"key": "10.1016/S0140-6736(21)01744-X_bib15",
"volume": "397",
"year": "2021"
},
{
"article-title": "Doxycycline for community treatment of suspected COVID-19 in people at high risk of adverse outcomes in the UK (PRINCIPLE): a randomised, controlled, open-label, adaptive platform trial",
"author": "Butler",
"journal-title": "Lancet Respir Med",
"key": "10.1016/S0140-6736(21)01744-X_bib16",
"year": "2021"
},
{
"DOI": "10.1016/S0140-6736(21)00945-4",
"article-title": "Inclusion and diversity in the PRINCIPLE trial",
"author": "Patel",
"doi-asserted-by": "crossref",
"first-page": "2251",
"journal-title": "Lancet",
"key": "10.1016/S0140-6736(21)01744-X_bib19",
"volume": "397",
"year": "2021"
},
{
"DOI": "10.1016/S1473-3099(20)30243-7",
"article-title": "Estimates of the severity of coronavirus disease 2019: a model-based analysis",
"author": "Verity",
"doi-asserted-by": "crossref",
"first-page": "669",
"journal-title": "Lancet Infect Dis",
"key": "10.1016/S0140-6736(21)01744-X_bib23",
"volume": "20",
"year": "2020"
},
{
"DOI": "10.7326/M20-4207",
"article-title": "Hydroxychloroquine in nonhospitalized adults with early COVID-19 : a randomized trial",
"author": "Skipper",
"doi-asserted-by": "crossref",
"first-page": "623",
"journal-title": "Ann Intern Med",
"key": "10.1016/S0140-6736(21)01744-X_bib24",
"volume": "173",
"year": "2020"
},
{
"DOI": "10.1056/NEJMoa2007764",
"article-title": "Remdesivir for the treatment of Covid-19—final report",
"author": "Beigel",
"doi-asserted-by": "crossref",
"first-page": "1813",
"journal-title": "N Engl J Med",
"key": "10.1016/S0140-6736(21)01744-X_bib25",
"volume": "383",
"year": "2020"
},
{
"DOI": "10.1159/000376585",
"article-title": "The WHO-5 Well-Being Index: a systematic review of the literature",
"author": "Topp",
"doi-asserted-by": "crossref",
"first-page": "167",
"journal-title": "Psychother Psychosom",
"key": "10.1016/S0140-6736(21)01744-X_bib26",
"volume": "84",
"year": "2015"
},
{
"article-title": "Efficacy of colchicine in non-hospitalized patients with COVID-19",
"author": "Tardif",
"journal-title": "medRxiv",
"key": "10.1016/S0140-6736(21)01744-X_bib27",
"year": "2021"
},
{
"DOI": "10.1056/NEJMp2015897",
"article-title": "False negative tests for SARS-CoV-2 infection - challenges and implications",
"author": "Woloshin",
"doi-asserted-by": "crossref",
"first-page": "e38",
"journal-title": "N Engl J Med",
"key": "10.1016/S0140-6736(21)01744-X_bib28",
"volume": "383",
"year": "2020"
},
{
"DOI": "10.1056/NEJMoa2100433",
"article-title": "Interleukin-6 receptor antagonists in critically ill patients with Covid-19",
"author": "Gordon",
"doi-asserted-by": "crossref",
"first-page": "1491",
"journal-title": "N Engl J Med",
"key": "10.1016/S0140-6736(21)01744-X_bib29",
"volume": "384",
"year": "2021"
}
],
"reference-count": 24,
"references-count": 24,
"relation": {},
"resource": {
"primary": {
"URL": "https://linkinghub.elsevier.com/retrieve/pii/S014067362101744X"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [
"General Medicine"
],
"subtitle": [],
"title": "Inhaled budesonide for COVID-19 in people at high risk of complications in the community in the UK (PRINCIPLE): a randomised, controlled, open-label, adaptive platform trial",
"type": "journal-article",
"update-policy": "http://dx.doi.org/10.1016/elsevier_cm_policy",
"volume": "398"
}