RETRACTED: Effect of early treatment with metformin on risk of emergency care and hospitalization among patients with COVID-19: The TOGETHER randomized platform clinical trial
et al., The Lancet Regional Health - Americas, doi:10.1016/j.lana.2021.100142, TOGETHER, NCT04727424, Aug 2021 (preprint)
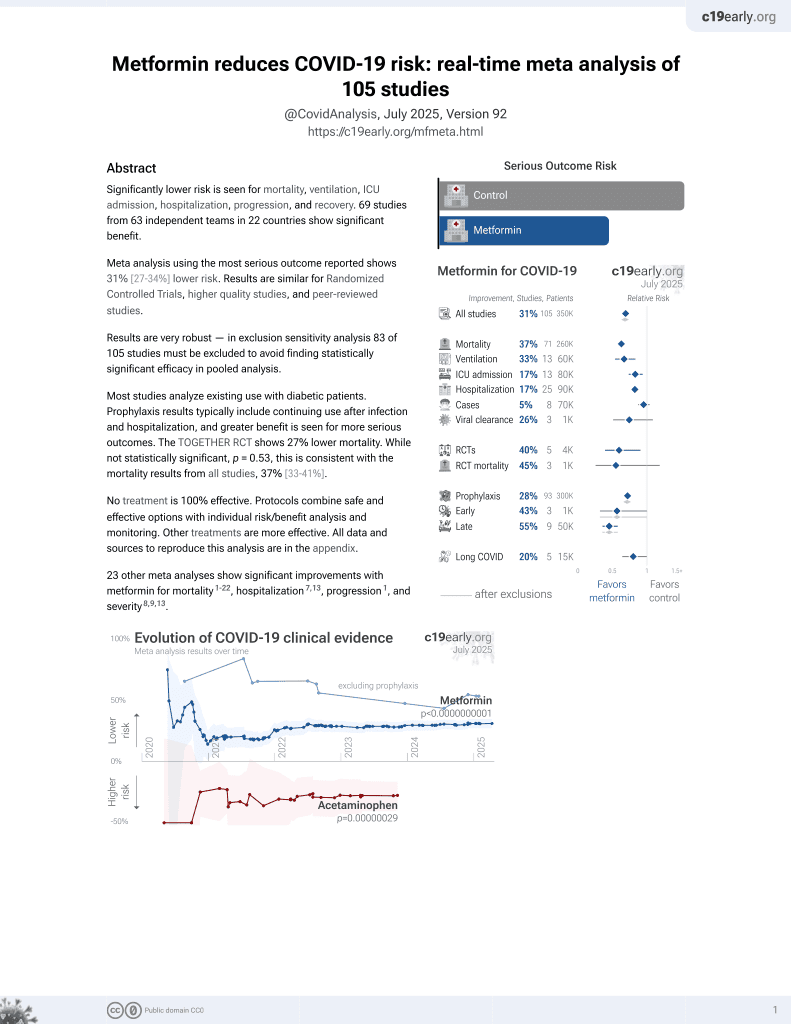
Metformin for COVID-19
3rd treatment shown to reduce risk in
July 2020, now with p < 0.00000000001 from 111 studies.
Lower risk for mortality, ventilation, ICU, hospitalization, progression, recovery, and viral clearance.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
The TOGETHER trial has extreme COI, impossible data, blinding failure, randomization failure, uncorrected errors, and many protocol violations. Authors do not respond to these issues and they have refused to release the data as promised. Some issues may apply only to specific arms.
See also
This study was retracted due to incorrect data and compromised integrity1.
Data for the primary outcome in this trial appears to be impossible2. For example, considering the metformin arm and the ITT population: 24 were hospitalized and 8 had an ER visit (tables S2/S3), therefore the number for combined ER or hospitalization must be between 24 and 32. However, authors report 34 events for ER/hospitalization.
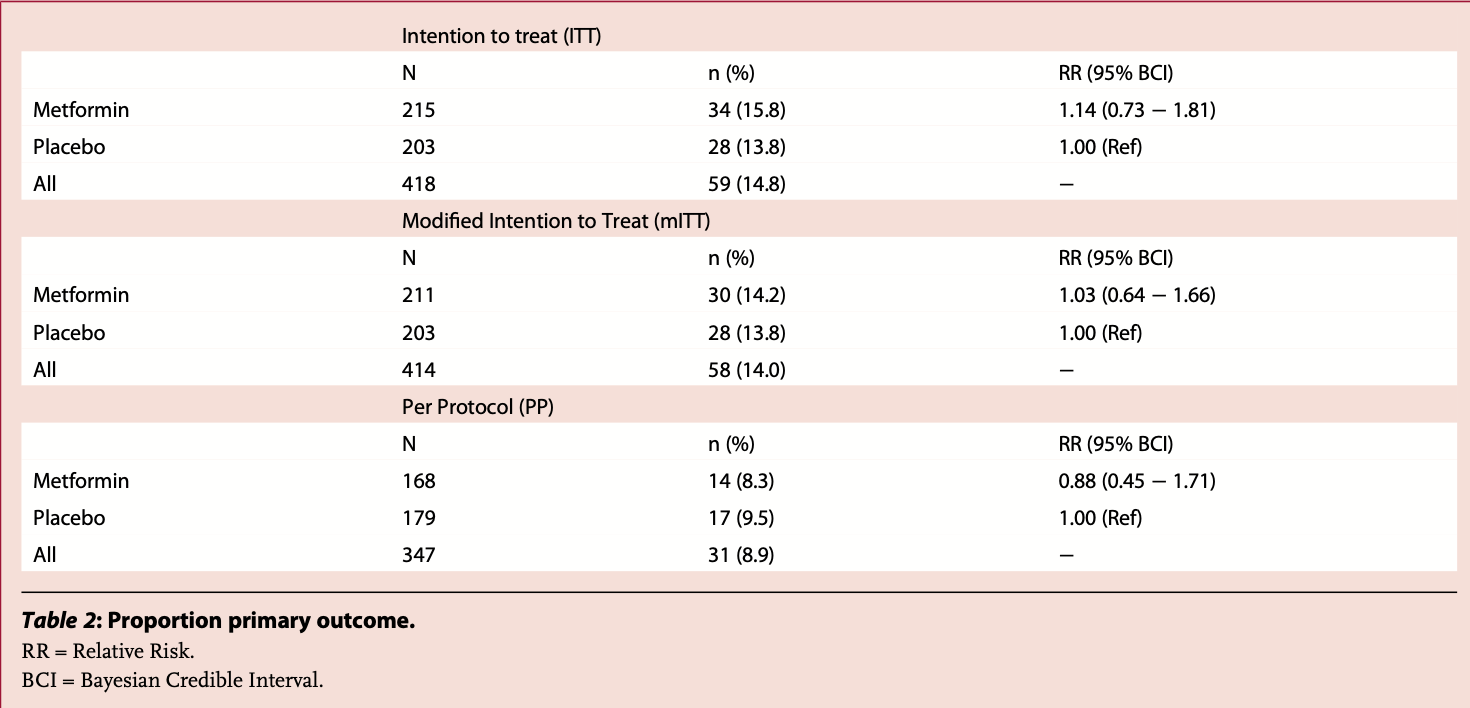
RCT with 215 patients treated with metformin and 203 controls, showing no significant difference with treatment.
The hospitalization risk for off-protocol patients was several times higher in both arms, resulting in Simpson's paradox when combining per-protocol and off-protocol patients5.
750mg twice daily for 10 days.
The TOGETHER trial has extreme COI, impossible data,
blinding failure, randomization failure, uncorrected errors, and many
protocol violations. Authors do not respond to these issues and they
have refused to release the data as promised. Some issues may apply only
to specific arms. For more details see6-10.
Although the 27% lower mortality is not statistically significant, it is consistent with the significant 36% lower mortality [32‑40%] from meta-analysis of the 74 mortality results to date.
This study is excluded in meta-analysis:
retracted due to incorrect data.
|
risk of death, 26.6% lower, RR 0.73, p = 0.53, treatment 7 of 215 (3.3%), control 9 of 203 (4.4%), NNT 85, day 28.
|
|
risk of hospitalization, 5.6% lower, RR 0.94, p = 0.88, treatment 24 of 215 (11.2%), control 24 of 203 (11.8%), NNT 152, ITT.
|
|
risk of hospitalization, 39.1% lower, RR 0.61, p = 0.28, treatment 8 of 168 (4.8%), control 14 of 179 (7.8%), NNT 33, PP.
|
|
risk of extended ER observation or hospitalization, 14.0% higher, RR 1.14, p = 0.58, treatment 34 of 215 (15.8%), control 28 of 203 (13.8%), ITT, primary outcome.
|
|
risk of extended ER observation or hospitalization, 12.0% lower, RR 0.88, p = 0.72, treatment 14 of 168 (8.3%), control 17 of 179 (9.5%), NNT 86, PP.
|
|
risk of ER visit, 31.0% lower, RR 0.69, p = 0.48, treatment 8 of 216 (3.7%), control 11 of 205 (5.4%), NNT 60, ITT.
|
|
risk of ER visit, 25.9% lower, RR 0.74, p = 0.62, treatment 7 of 171 (4.1%), control 10 of 181 (5.5%), NNT 70, PP.
|
|
risk of no viral clearance, 1.0% lower, RR 0.99, p = 0.85, treatment 215, control 203, adjusted per study.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
2.
doyourownresearch.substack.com, doyourownresearch.substack.com/p/together-trial-and-the-negative-number.
3.
doyourownresearch.substack.com (B), doyourownresearch.substack.com/p/together-trial-false-interim-analyses.
5.
web.archive.org, web.archive.org/web/*/https://twitter.com/Covid19Crusher/status/1470733348079288333.
6.
Reis et al., Effect of Early Treatment with Ivermectin among Patients with Covid-19, New England Journal of Medicine, doi:10.1056/NEJMoa2115869.
7.
Reis (B) et al., Effect of early treatment with fluvoxamine on risk of emergency care and hospitalisation among patients with COVID-19: the TOGETHER randomised, platform clinical trial, The Lancet Global Health, doi:10.1016/S2214-109X(21)00448-4.
8.
Reis (C) et al., Effect of Early Treatment With Hydroxychloroquine or Lopinavir and Ritonavir on Risk of Hospitalization Among Patients With COVID-19 The TOGETHER Randomized Clinical Trial, JAMA Network Open, doi:10.1001/jamanetworkopen.2021.6468.
Reis et al., 31 Aug 2021, Double Blind Randomized Controlled Trial, Brazil, peer-reviewed, 23 authors, study period 15 January, 2021 - 3 April, 2021, impossible data, see notes, trial NCT04727424 (history) (TOGETHER).
Effect of early treatment with metformin on risk of emergency care and hospitalization among patients with COVID-19: The TOGETHER randomized platform clinical trial
doi:10.17605/OSF.IO/EG37X)
Background Observational studies have postulated a therapeutic role of metformin in treating COVID-19. We conducted an adaptive platform clinical trial to determine whether metformin is an effective treatment for high-risk patients with early COVID-19 in an outpatient setting. Methods The TOGETHER Trial is a placebo-controled, randomized, platform clinical trial conducted in Brazil. Eligible participants were symptomatic adults with a positive antigen test for SARS-CoV-2. We enroled eligible patients over the age of 50 years or with a known risk factor for disease severity. Patients were randomly assigned to receive either placebo or metformin (750 mg twice daily for 10 days or placebo, twice daily for 10 days). The primary outcome was hospitalization defined as either retention in a COVID-19 emergency setting for > 6 h or transfer to tertiary hospital due to COVID-19 at 28 days post randomization. Secondary outcomes included viral clearance at day 7, time to hospitalization, mortality, and adverse drug reactions. We used a Bayesian framework to determine probability of success of the intervention compared to placebo.
Supplementary materials Supplementary material associated with this article can be found, in the online version, at doi:10.1016/j. lana.2021.100142.
References
Bramante, Ingraham, Murray, Metformin and risk of mortality in patients hospitalised with COVID-19: a retrospective cohort analysis, Lancet Healthy Longev
Chen, Guo, Qiu, Zhang, Deng et al., Immunomodulatory and antiviral activity of metformin and its potential implications in treating coronavirus disease 2019 and lung injury, Front Immunol
Cheng, Liu, Li, Metformin is associated with higher incidence of acidosis, but not mortality, in individuals with COVID-19 and pre-existing type 2 diabetes, Cell Metab
Dimairo, Pallmann, Wason, The adaptive designs CON-SORT extension (ACE) statement: a checklist with explanation and elaboration guideline for reporting randomised trials that use an adaptive design, BMJ
Forrest, Rayner, Park, Mills, Early treatment of COVID-19 disease: a missed opportunity, Infect Dis Ther
Goodman, Stopping at nothing? Some dilemmas of data monitoring in clinical trials, Ann Intern Med
Matsiukevich, Piraino, Lahni, Metformin ameliorates gender-and age-dependent hemodynamic instability and myocardial injury in murine hemorrhagic shock, Biochim Biophys Acta Mol Basis Dis
Park, Harari, Dron, Lester, Thorlund et al., An overview of platform trials with a checklist for clinical readers, J Clin Epidemiol
Park, Lee, Park, Sex-dependent difference in the effect of metformin on colorectal cancer-specific mortality of diabetic colorectal cancer patients, World J Gastroenterol
Quan, Zhang, Wei, Fang, Gender-related different effects of a combined therapy of exenatide and metformin on overweight or obesity patients with type 2 diabetes mellitus, J Diabetes Complic
Reis, Silva, Silva, A multi-center, adaptive, randomized, platform trial to evaluate the effect of repurposed medicines in outpatients with early coronavirus disease 2019 (COVID-19) and high-risk for complications: the TOGETHER master trial protocol, Gates Open Res
Reis, Silva, Silva, Effect of early treatment with hydroxychloroquine or lopinavir and ritonavir on risk of hospitalization among patients with COVID-19: the TOGETHER randomized clinical trial, JAMA Netw Open
Thorlund, Dron, Park, Hsu, Forrest et al., A real-time dashboard of clinical trials for COVID-19, Lancet Digit Health
DOI record:
{
"DOI": "10.1016/j.lana.2021.100142",
"ISSN": [
"2667-193X"
],
"URL": "http://dx.doi.org/10.1016/j.lana.2021.100142",
"alternative-id": [
"S2667193X21001381"
],
"article-number": "100142",
"assertion": [
{
"label": "This article is maintained by",
"name": "publisher",
"value": "Elsevier"
},
{
"label": "Article Title",
"name": "articletitle",
"value": "Effect of early treatment with metformin on risk of emergency care and hospitalization among patients with COVID-19: The TOGETHER randomized platform clinical trial"
},
{
"label": "Journal Title",
"name": "journaltitle",
"value": "The Lancet Regional Health - Americas"
},
{
"label": "CrossRef DOI link to publisher maintained version",
"name": "articlelink",
"value": "https://doi.org/10.1016/j.lana.2021.100142"
},
{
"label": "Content Type",
"name": "content_type",
"value": "article"
},
{
"label": "Copyright",
"name": "copyright",
"value": "© 2021 The Authors. Published by Elsevier Ltd."
}
],
"author": [
{
"ORCID": "http://orcid.org/0000-0002-4847-1034",
"affiliation": [],
"authenticated-orcid": false,
"family": "Reis",
"given": "Gilmar",
"sequence": "first"
},
{
"affiliation": [],
"family": "dos Santos Moreira Silva",
"given": "Eduardo Augusto",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Medeiros Silva",
"given": "Daniela Carla",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Thabane",
"given": "Lehana",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Cruz Milagres",
"given": "Aline",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ferreira",
"given": "Thiago Santiago",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Quirino dos Santos",
"given": "Castilho Vitor",
"sequence": "additional"
},
{
"affiliation": [],
"family": "de Figueiredo Neto",
"given": "Adhemar Dias",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Diniz Callegari",
"given": "Eduardo",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Monteiro Savassi",
"given": "Leonardo Cançado",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Campos Simplicio",
"given": "Maria Izabel",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Barra Ribeiro",
"given": "Luciene",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Oliveira",
"given": "Rosemary",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Harari",
"given": "Ofir",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Bailey",
"given": "Holly",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Forrest",
"given": "Jamie I",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Glushchenko",
"given": "Alla",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Sprague",
"given": "Sheila",
"sequence": "additional"
},
{
"affiliation": [],
"family": "McKay",
"given": "Paula",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Rayner",
"given": "Craig R.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ruton",
"given": "Hinda",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Guyatt",
"given": "Gordon H.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Mills",
"given": "Edward J.",
"sequence": "additional"
}
],
"container-title": "The Lancet Regional Health - Americas",
"container-title-short": "The Lancet Regional Health - Americas",
"content-domain": {
"crossmark-restriction": true,
"domain": [
"elsevier.com",
"sciencedirect.com"
]
},
"created": {
"date-parts": [
[
2021,
12,
14
]
],
"date-time": "2021-12-14T04:48:44Z",
"timestamp": 1639457324000
},
"deposited": {
"date-parts": [
[
2022,
2,
17
]
],
"date-time": "2022-02-17T14:15:26Z",
"timestamp": 1645107326000
},
"indexed": {
"date-parts": [
[
2024,
3,
26
]
],
"date-time": "2024-03-26T11:01:57Z",
"timestamp": 1711450917393
},
"is-referenced-by-count": 9,
"issued": {
"date-parts": [
[
2022,
2
]
]
},
"language": "en",
"license": [
{
"URL": "https://www.elsevier.com/tdm/userlicense/1.0/",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2022,
2,
1
]
],
"date-time": "2022-02-01T00:00:00Z",
"timestamp": 1643673600000
}
},
{
"URL": "http://creativecommons.org/licenses/by-nc-nd/4.0/",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2021,
11,
24
]
],
"date-time": "2021-11-24T00:00:00Z",
"timestamp": 1637712000000
}
}
],
"link": [
{
"URL": "https://api.elsevier.com/content/article/PII:S2667193X21001381?httpAccept=text/xml",
"content-type": "text/xml",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://api.elsevier.com/content/article/PII:S2667193X21001381?httpAccept=text/plain",
"content-type": "text/plain",
"content-version": "vor",
"intended-application": "text-mining"
}
],
"member": "78",
"original-title": [],
"page": "100142",
"prefix": "10.1016",
"published": {
"date-parts": [
[
2022,
2
]
]
},
"published-print": {
"date-parts": [
[
2022,
2
]
]
},
"publisher": "Elsevier BV",
"reference": [
{
"DOI": "10.1016/j.bbadis.2017.05.027",
"article-title": "Metformin ameliorates gender-and age-dependent hemodynamic instability and myocardial injury in murine hemorrhagic shock",
"author": "Matsiukevich",
"doi-asserted-by": "crossref",
"first-page": "2680",
"issue": "10 Pt B",
"journal-title": "Biochim Biophys Acta Mol Basis Dis",
"key": "10.1016/j.lana.2021.100142_bib0001",
"volume": "1863",
"year": "2017"
},
{
"DOI": "10.3748/wjg.v23.i28.5196",
"article-title": "Sex-dependent difference in the effect of metformin on colorectal cancer-specific mortality of diabetic colorectal cancer patients",
"author": "Park",
"doi-asserted-by": "crossref",
"first-page": "5196",
"issue": "28",
"journal-title": "World J Gastroenterol",
"key": "10.1016/j.lana.2021.100142_bib0002",
"volume": "23",
"year": "2017"
},
{
"DOI": "10.1016/j.jdiacomp.2016.01.013",
"article-title": "Gender-related different effects of a combined therapy of exenatide and metformin on overweight or obesity patients with type 2 diabetes mellitus",
"author": "Quan",
"doi-asserted-by": "crossref",
"first-page": "686",
"issue": "4",
"journal-title": "J Diabetes Complic",
"key": "10.1016/j.lana.2021.100142_bib0003",
"volume": "30",
"year": "2016"
},
{
"DOI": "10.3389/fimmu.2020.02056",
"article-title": "Immunomodulatory and antiviral activity of metformin and its potential implications in treating coronavirus disease 2019 and lung injury",
"author": "Chen",
"doi-asserted-by": "crossref",
"first-page": "2056",
"journal-title": "Front Immunol",
"key": "10.1016/j.lana.2021.100142_bib0004",
"volume": "11",
"year": "2020"
},
{
"DOI": "10.1016/S2666-7568(20)30033-7",
"article-title": "Metformin and risk of mortality in patients hospitalised with COVID-19: a retrospective cohort analysis",
"author": "Bramante",
"doi-asserted-by": "crossref",
"first-page": "e34",
"issue": "1",
"journal-title": "Lancet Healthy Longev",
"key": "10.1016/j.lana.2021.100142_bib0005",
"volume": "2",
"year": "2021"
},
{
"DOI": "10.1016/j.cmet.2020.08.013",
"article-title": "Metformin is associated with higher incidence of acidosis, but not mortality, in individuals with COVID-19 and pre-existing type 2 diabetes",
"author": "Cheng",
"doi-asserted-by": "crossref",
"first-page": "537",
"issue": "4",
"journal-title": "Cell Metab",
"key": "10.1016/j.lana.2021.100142_bib0006",
"volume": "32",
"year": "2020"
},
{
"DOI": "10.1007/s40121-020-00349-8",
"article-title": "Early treatment of COVID-19 disease: a missed opportunity",
"author": "Forrest",
"doi-asserted-by": "crossref",
"first-page": "715",
"issue": "4",
"journal-title": "Infect Dis Ther",
"key": "10.1016/j.lana.2021.100142_bib0007",
"volume": "9",
"year": "2020"
},
{
"DOI": "10.12688/gatesopenres.13304.2",
"article-title": "A multi-center, adaptive, randomized, platform trial to evaluate the effect of repurposed medicines in outpatients with early coronavirus disease 2019 (COVID-19) and high-risk for complications: the TOGETHER master trial protocol",
"author": "Reis",
"doi-asserted-by": "crossref",
"first-page": "117",
"issue": "117",
"journal-title": "Gates Open Res",
"key": "10.1016/j.lana.2021.100142_bib0008",
"volume": "5",
"year": "2021"
},
{
"DOI": "10.1136/bmj.m115",
"article-title": "The adaptive designs CONSORT extension (ACE) statement: a checklist with explanation and elaboration guideline for reporting randomised trials that use an adaptive design",
"author": "Dimairo",
"doi-asserted-by": "crossref",
"first-page": "m115",
"journal-title": "BMJ",
"key": "10.1016/j.lana.2021.100142_bib0009",
"volume": "369",
"year": "2020"
},
{
"DOI": "10.1001/jamanetworkopen.2021.6468",
"article-title": "Effect of early treatment with hydroxychloroquine or lopinavir and ritonavir on risk of hospitalization among patients with COVID-19: the TOGETHER randomized clinical trial",
"author": "Reis",
"doi-asserted-by": "crossref",
"issue": "4",
"journal-title": "JAMA Netw Open",
"key": "10.1016/j.lana.2021.100142_bib0010",
"volume": "4",
"year": "2021"
},
{
"DOI": "10.1016/j.jclinepi.2020.04.025",
"article-title": "An overview of platform trials with a checklist for clinical readers",
"author": "Park",
"doi-asserted-by": "crossref",
"first-page": "1",
"journal-title": "J Clin Epidemiol",
"key": "10.1016/j.lana.2021.100142_bib0011",
"volume": "125",
"year": "2020"
},
{
"DOI": "10.7326/0003-4819-146-12-200706190-00010",
"article-title": "Stopping at nothing? Some dilemmas of data monitoring in clinical trials",
"author": "Goodman",
"doi-asserted-by": "crossref",
"first-page": "882",
"issue": "12",
"journal-title": "Ann Intern Med",
"key": "10.1016/j.lana.2021.100142_bib0012",
"volume": "146",
"year": "2007"
},
{
"DOI": "10.1016/S2589-7500(20)30086-8",
"article-title": "A real-time dashboard of clinical trials for COVID-19",
"author": "Thorlund",
"doi-asserted-by": "crossref",
"first-page": "e286",
"issue": "6",
"journal-title": "Lancet Digit Health",
"key": "10.1016/j.lana.2021.100142_bib0013",
"volume": "2",
"year": "2020"
}
],
"reference-count": 13,
"references-count": 13,
"relation": {},
"resource": {
"primary": {
"URL": "https://linkinghub.elsevier.com/retrieve/pii/S2667193X21001381"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subtitle": [],
"title": "Effect of early treatment with metformin on risk of emergency care and hospitalization among patients with COVID-19: The TOGETHER randomized platform clinical trial",
"type": "journal-article",
"update-policy": "http://dx.doi.org/10.1016/elsevier_cm_policy",
"volume": "6"
}