Unveiling the potential pleiotropic effects of metformin in treating COVID-19: a comprehensive review
et al., Frontiers in Molecular Biosciences, doi:10.3389/fmolb.2023.1260633, Oct 2023
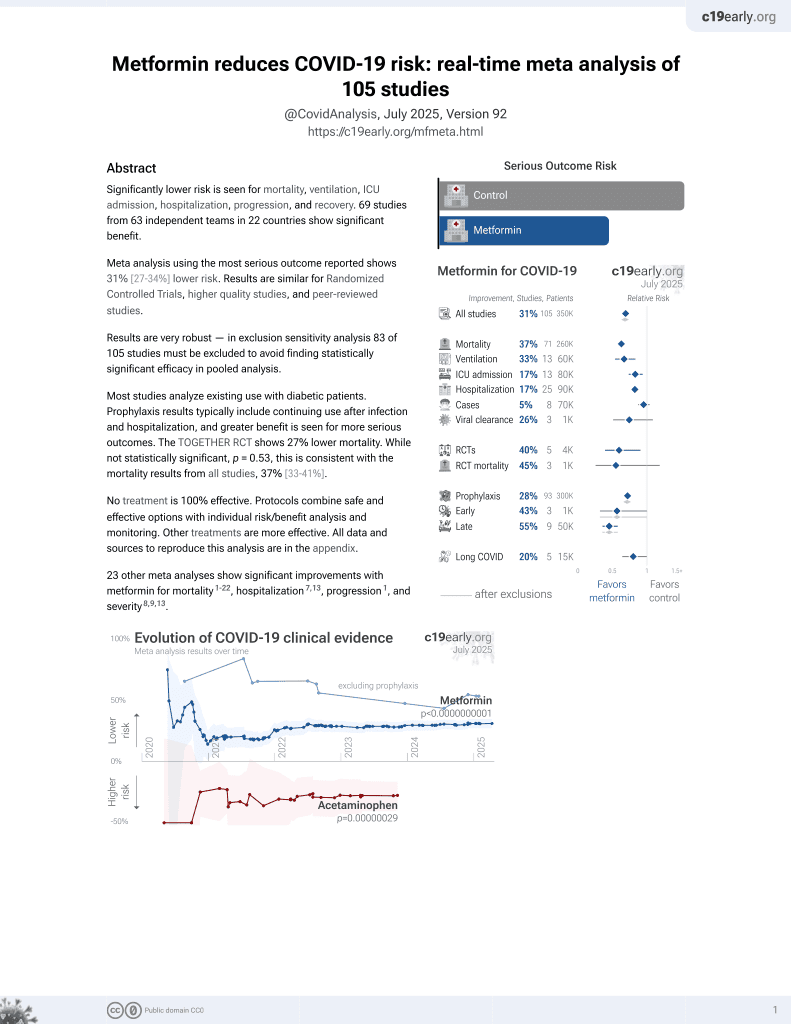
Metformin for COVID-19
3rd treatment shown to reduce risk in
July 2020, now with p < 0.00000000001 from 111 studies.
Lower risk for mortality, ventilation, ICU, hospitalization, progression, recovery, and viral clearance.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
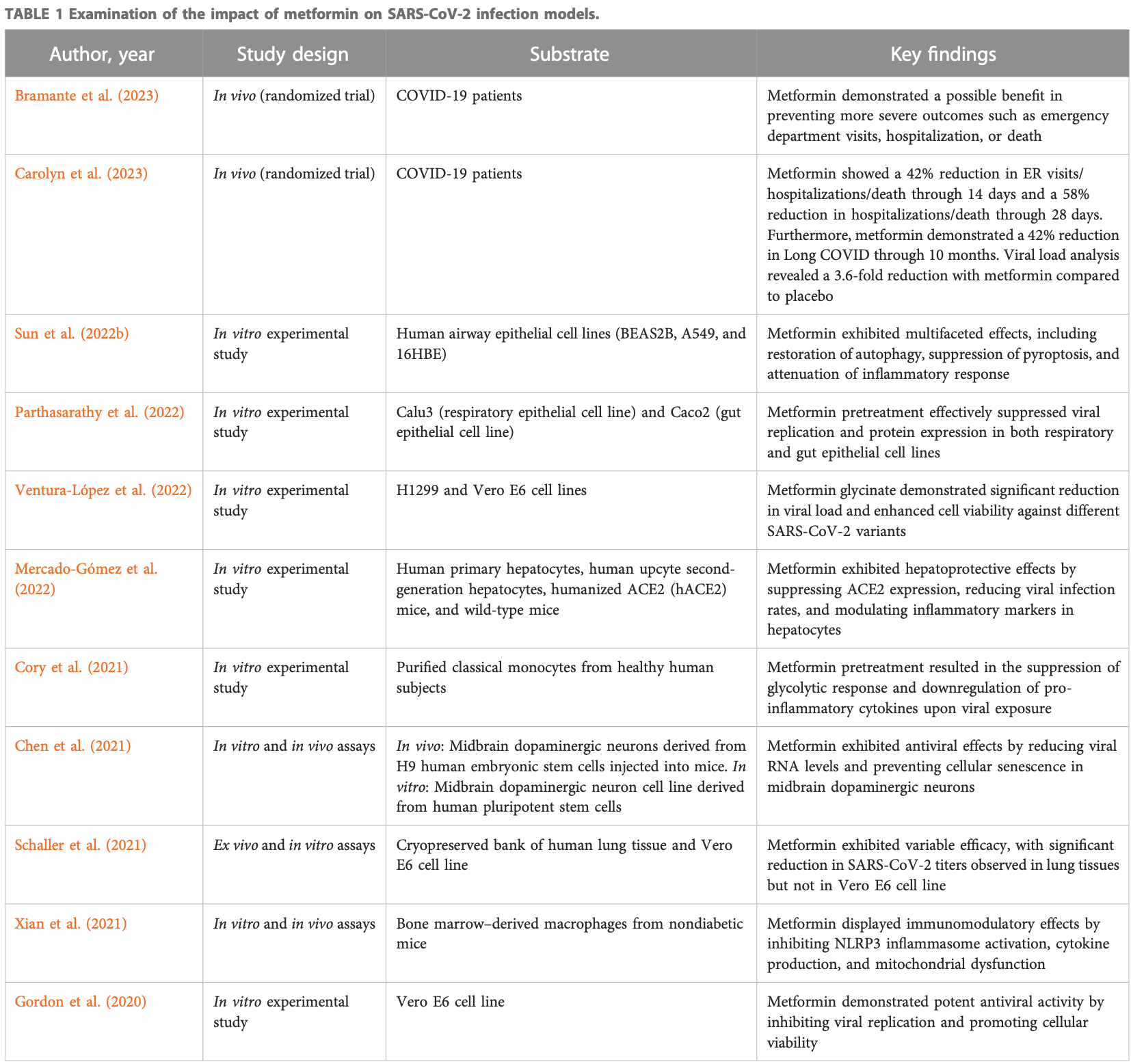
Review of the potential pleiotropic effects of metformin in treating COVID-19. Authors explore the potential of metformin, a medication commonly used for type 2 diabetes, as an antiviral and anti-inflammatory agent in COVID-19. Metformin has demonstrated inhibitory effects on SARS-CoV-2 growth in cell culture models and promising results in reducing viral load and achieving undetectable viral levels in clinical trials. Additionally, metformin exhibits anti-inflammatory properties by reducing pro-inflammatory cytokines and modulating immune cell function, which may help prevent cytokine storm. The drug's ability to regulate Th17/Treg balance suggests its potential in mitigating inflammation and restoring T cell functionality. Furthermore, metformin's modulation of the gut microbiota and production of short-chain fatty acids may contribute to its therapeutic effects.
1.
Monsalve et al., NETosis: A key player in autoimmunity, COVID-19, and long COVID, Journal of Translational Autoimmunity, doi:10.1016/j.jtauto.2025.100280.
2.
Halabitska et al., Metformin in Antiviral Therapy: Evidence and Perspectives, Viruses, doi:10.3390/v16121938.
3.
Plowman et al., Anti-Inflammatory Potential of the Anti-Diabetic Drug Metformin in the Prevention of Inflammatory Complications and Infectious Diseases Including COVID-19: A Narrative Review, International Journal of Molecular Sciences, doi:10.3390/ijms25105190.
4.
De Jesús-González et al., A Dual Pharmacological Strategy against COVID-19: The Therapeutic Potential of Metformin and Atorvastatin, Microorganisms, doi:10.3390/microorganisms12020383.
5.
Halma et al., Exploring autophagy in treating SARS-CoV-2 spike protein-related pathology, Endocrine and Metabolic Science, doi:10.1016/j.endmts.2024.100163.
6.
Zhang et al., SARS-CoV-2 ORF3a Protein as a Therapeutic Target against COVID-19 and Long-Term Post-Infection Effects, Pathogens, doi:10.3390/pathogens13010075.
7.
Gomaa et al., Pharmacological evaluation of vitamin D in COVID-19 and long COVID-19: recent studies confirm clinical validation and highlight metformin to improve VDR sensitivity and efficacy, Inflammopharmacology, doi:10.1007/s10787-023-01383-x.
Petakh et al., 10 Oct 2023, placebo-controlled, multiple countries, peer-reviewed, 3 authors.
Contact: pavlo.petakh@uzhnu.edu.ua, kamyshnyi_om@tdmu.edu.ua.
Unveiling the potential pleiotropic effects of metformin in treating COVID-19: a comprehensive review
Frontiers in Molecular Biosciences, doi:10.3389/fmolb.2023.1260633
This review article explores the potential of metformin, a medication commonly used for type 2 diabetes, as an antiviral and anti-inflammatory agent in the context of coronavirus disease 2019 . Metformin has demonstrated inhibitory effects on the growth of SARS-CoV-2 in cell culture models and has shown promising results in reducing viral load and achieving undetectable viral levels in clinical trials. Additionally, metformin exhibits anti-inflammatory properties by reducing the production of pro-inflammatory cytokines and modulating immune cell function, which may help prevent cytokine storms associated with severe COVID-19. The drug's ability to regulate the balance between pro-inflammatory Th17 cells and anti-inflammatory Treg cells suggests its potential in mitigating inflammation and restoring T cell functionality. Furthermore, metformin's modulation of the gut microbiota, particularly changes in bacterial taxa and the production of short-chain fatty acids, may contribute to its therapeutic effects. The interplay between metformin, bile acids, the gut microbiome, glucagon-like peptide-1 secretion, and glycemic control has implications for the management of diabetes and potential interventions in COVID-19. By refreshing the current evidence, this review highlights the potential of metformin as a therapeutic option in the management of COVID-19, while also exploring its effects on the gut microbiome and immunometabolism.
Author contributions PP: Visualization, Writing-original draft. IK: Writing-review and editing. AK: Conceptualization, Formal Analysis, Writing-review and editing.
Conflict of interest The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher's note All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
Adrian, Gariballa, Parekh, Thomas, Saadi et al., Rectal taurocholate increases L cell and insulin secretion, and decreases blood glucose and food intake in obese type 2 diabetic volunteers, Diabetologia, doi:10.1007/s00125-012-2593-2
Al-Kuraishy, Al-Gareeb, Alblihed, Guerreiro, Cruz-Martins et al., COVID-19 in relation to hyperglycemia and diabetes mellitus, J Front. Cardiovasc. Med, doi:10.3389/fcvm.2021.644095
Alexander, Mullish, Danckert, Liu, Olbei et al., The gut microbiota and metabolome are associated with diminished COVID-19 vaccine-induced antibody responses in immunosuppressed inflammatory bowel disease patients, EBioMedicine, doi:10.1016/j.ebiom.2022.104430
Allin, Tremaroli, Caesar, Jensen, Damgaard et al., Aberrant intestinal microbiota in individuals with prediabetes, J. Diabetol, doi:10.1007/s00125-018-4550-1
Ancona, Alagna, Alteri, Palomba, Tonizzo et al., Gut and airway microbiota dysbiosis and their role in COVID-19 and long-COVID, Front. Immunol, doi:10.3389/fimmu.2023.1080043
Atzrodt, Maknojia, Mccarthy, Oldfield, Po et al., A Guide to COVID-19: a global pandemic caused by the novel coronavirus SARS-CoV-2, FEBS J, doi:10.1111/febs.15375
Bailey, Gwilt, Diabetes, Metformin and the Clinical Course of Covid-19: outcomes, Mechanisms and Suggestions on the Therapeutic Use of Metformin, Front. Pharmacol, doi:10.3389/fphar.2022.784459
Bailey, Wilcock, Day, Effect of metformin on glucose metabolism in the splanchnic bed, Br. J. Pharmacol, doi:10.1111/j.1476-5381.1992.tb09093.x
Balakumar, Prabhu, Sathishkumar, Prabu, Rokana et al., Improvement in glucose tolerance and insulin sensitivity by probiotic strains of Indian gut origin in high-fat diet-fed C57BL/6J mice, Eur. J. Nutr, doi:10.1007/s00394-016-1317-7
Barengolts, Green, Eisenberg, Akbar, Reddivari et al., Gut microbiota varies by opioid use, circulating leptin and oxytocin in African American men with diabetes and high burden of chronic disease, PLoS One, doi:10.1371/journal.pone.0194171
Bauer, Duca, Waise, Rasmussen, Abraham et al., Metformin Alters Upper Small Intestinal Microbiota that Impact a Glucose-SGLT1-Sensing Glucoregulatory Pathway, Cell Metab, doi:10.1016/j.cmet.2017.09.019
Bell, Saad, Tillett, Mcguire, Bordbar et al., Metabolite-based dietary supplementation in human type 1 diabetes is associated with microbiota and immune modulation, Microbiome, doi:10.1186/s40168-021-01193-9
Bramante, Beckman, Mehta, Karger, Odde et al., Metformin reduces SARS-CoV-2 in a Phase 3 Randomized Placebo Controlled Clinical Trial, medRxiv, doi:10.1101/2023.06.06.23290989
Bryrup, Thomsen, Kern, Allin, Brandslund et al., Metformin-induced changes of the gut microbiota in healthy young men: results of a non-blinded, one-armed intervention study, Diabetologia, doi:10.1007/s00125-019-4848-7
Brønden, Albér, Rohde, Gasbjerg, Rehfeld et al., The bile acid-sequestering resin sevelamer eliminates the acute GLP-1 stimulatory effect of endogenously released bile acids in patients with type 2 diabetes, Diabetes, Obes. metabolism, doi:10.1111/dom.13080
Brønden, Albér, Rohde, Rehfeld, Holst et al., Single-Dose Metformin Enhances Bile Acid-Induced Glucagon-Like Peptide-1 Secretion in Patients With Type 2 Diabetes, J. Clin. Endocrinol. metabolism, doi:10.1210/jc.2017-01091
Cameron, Morrison, Levin, Mohan, Forteath et al., Anti-Inflammatory Effects of Metformin Irrespective of Diabetes Status, Circ. Res, doi:10.1161/CIRCRESAHA.116.308445
Candela, Biagi, Soverini, Consolandi, Quercia et al., Modulation of gut microbiota dysbioses in type 2 diabetic patients by macrobiotic Ma-Pi 2 diet, Br. J. Nutr, doi:10.1017/S0007114516001045
Carolyn, Kenneth, Tanvi, Amy, David et al., Metformin reduces SARS-CoV-2 in a Phase 3 Randomized Placebo Controlled Clinical Trial, medRxiv, doi:10.1101/2023.06.06.23290989
Carreca, Gaetani, Busà, Francipane, Gulotta et al., Galectin-9 and Interferon-Gamma Are Released by Natural Killer Cells upon Activation with Interferon-Alpha and Orchestrate the Suppression of Hepatitis C Virus Infection, Viruses, doi:10.3390/v14071538
Chambers, Byrne, Morrison, Murphy, Preston et al., Dietary supplementation with inulin-propionate ester or inulin improves insulin sensitivity in adults with overweight and obesity with distinct effects on the gut microbiota, plasma metabolome and systemic inflammatory responses: a randomised cross-over trial, Gut, doi:10.1136/gutjnl-2019-318424
Chambers, Viardot, Psichas, Morrison, Murphy et al., Effects of targeted delivery of propionate to the human colon on appetite regulation, body weight maintenance and adiposity in overweight adults, Gut, doi:10.1136/gutjnl-2014-307913
Chen, Chien, Yang, The alteration of gut microbiota in newly diagnosed type 2 diabetic patients, Nutrition, doi:10.1016/j.nut.2018.11.019
Chen, Han, Yang, Kim, Nair et al., SARS-CoV-2 Infection Causes Dopaminergic Neuron Senescence, Res. square, doi:10.21203/rs.3.rs-513461/v1
Cheung, Lam, Zhang, Ooi, Tan et al., Association between Recent Usage of Antibiotics and Immunogenicity within Six Months after COVID-19 Vaccination, Vaccines, doi:10.3390/vaccines10071122
Chávez-Carbajal, Pizano-Zárate, Hernández-Quiroz, Ortiz-Luna, Morales-Hernández et al., Characterization of the gut microbiota of individuals at different T2D stages reveals a complex relationship with the host, Microorganisms, doi:10.3390/microorganisms8010094
Cory, Emmons, Yarbro, Davis, Pence, Metformin Suppresses Monocyte Immunometabolic Activation by SARS-CoV-2 Spike Protein Subunit 1, Front. Immunol, doi:10.3389/fimmu.2021.733921
Dang, Marsland, Microbes, metabolites, and the gut-lung axis, Mucosal Immunol, doi:10.1038/s41385-019-0160-6
De Oliveira, Oliveira, Pinzan, De Salis, Cardoso, Microbiota Modulation of the Gut-Lung Axis in COVID-19, Front. Immunol, doi:10.3389/fimmu.2021.635471
De, Cuesta-Zuluaga, Mueller, Corrales-Agudelo, Velásquez-Mejía et al., Metformin is associated with higher relative abundance of mucin-degrading Akkermansia muciniphila and several short-chain fatty acid-producing microbiota in the gut, Diabetes care, doi:10.2337/dc16-1324
Den Besten, Van Eunen, Groen, Venema, Reijngoud et al., The role of short-chain fatty acids in the interplay between diet, gut microbiota, and host energy metabolism, J. lipid Res, doi:10.1194/jlr.R036012
Ding, Yang, Wang, Huang, Bile acid nuclear receptor FXR and digestive system diseases, Acta Pharm. Sin. B, doi:10.1016/j.apsb.2015.01.004
Dotson, Zhang, Xu, Shin, Vigues et al., Bitter taste receptors influence glucose homeostasis, PLoS One, doi:10.1371/journal.pone.0003974
Duan, Ding, Yu, Ma, Yang et al., Metformin mitigates autoimmune insulitis by inhibiting Th1 and Th17 responses while promoting Treg production, Am. J. Transl. Res
Duca, Côté, Rasmussen, Zadeh-Tahmasebi, Rutter et al., Metformin activates a duodenal Ampk-dependent pathway to lower hepatic glucose production in rats, Nat. Med, doi:10.1038/nm.3787
Eaton, Merkulova, Brown, The H(+)-ATPase (V-ATPase): from proton pump to signaling complex in health and disease, Am. J. physiology Cell physiology, doi:10.1152/ajpcell.00442.2020
Ejtahed, Mohtadi-Nia, Homayouni-Rad, Niafar, Asghari-Jafarabadi et al., Probiotic yogurt improves antioxidant status in type 2 diabetic patients, Nutrition, doi:10.1016/j.nut.2011.08.013
El Aidy, Merrifield, Derrien, Van Baarlen, Hooiveld et al., The gut microbiota elicits a profound metabolic reorientation in the mouse jejunal mucosa during conventionalisation, Gut, doi:10.1136/gutjnl-2011-301955
Erickson, Fenno, Barzilai, Kuchel, Bartley et al., Metformin for Treatment of Acute COVID-19: systematic Review of Clinical Trial Data Against SARS-CoV-2, Diabetes Care, doi:10.2337/dc22-2539
Esam, A proposed mechanism for the possible therapeutic potential of Metformin in COVID-19, Diabetes Res. Clin. Pract, doi:10.1016/j.diabres.2020.108282
Farfan-Morales, Cordero-Rivera, Osuna-Ramos, Monroy-Muñoz, De Jesús-González et al., The antiviral effect of metformin on zika and dengue virus infection, Sci. Rep, doi:10.1038/s41598-021-87707-9
Forslund, Hildebrand, Nielsen, Falony, Le Chatelier et al., Disentangling type 2 diabetes and metformin treatment signatures in the human gut microbiota, Nature, doi:10.1038/nature15766
Giron, Dweep, Yin, Wang, Damra et al., Plasma Markers of Disrupted Gut Permeability in Severe COVID-19 Patients, Front. Immunol, doi:10.3389/fimmu.2021.686240
Gomaa, Human gut microbiota/microbiome in health and diseases: a review, Antonie Leeuwenhoek, doi:10.1007/s10482-020-01474-7
Gorboulev, Schürmann, Vallon, Kipp, Jaschke et al., Na(+)-D-glucose cotransporter SGLT1 is pivotal for intestinal glucose absorption and glucose-dependent incretin secretion, Diabetes, doi:10.2337/db11-1029
Gorboulev, Schürmann, Vallon, Kipp, Jaschke et al., Na+-d-glucose Cotransporter SGLT1 is Pivotal for Intestinal Glucose Absorption and Glucose-Dependent Incretin Secretion, Diabetes, doi:10.2337/db11-1029
Gordon, Jang, Bouhaddou, Xu, Obernier et al., A SARS-CoV-2 protein interaction map reveals targets for drug repurposing, Nature, doi:10.1038/s41586-020-2286-9
Govers, Calder, Savelkoul, Albers, Van Neerven, Ingestion, Immunity, and Infection: nutrition and Viral Respiratory Tract Infections, Front. Immunol, doi:10.3389/fimmu.2022.841532
Gradisteanu Pircalabioru, Grigore, Czobor Barbu, Chifiriuc, Savu, Impact of COVID-19 on the Microbiome and Inflammatory Status of Type 2 Diabetes Patients, Biomedicines, doi:10.3390/biomedicines11010179
Hameed, Masoodi, Mir, Nabi, Ghazanfar et al., Type 2 diabetes mellitus: from a metabolic disorder to an inflammatory condition, World J. diabetes, doi:10.4239/wjd.v6.i4.598
Han, Zheng, Deng, Nan, Xiao et al., SARS-CoV-2 ORF10 antagonizes STING-dependent interferon activation and autophagy, J. Med. Virol, doi:10.1002/jmv.27965
Hansen, Scheltema, Sonne, Hansen, Sperling et al., Effect of chenodeoxycholic acid and the bile acid sequestrant colesevelam on glucagon-like peptide-1 secretion, Diabetes, Obes. metabolism, doi:10.1111/dom.12648
Hansen, Sonne, Mikkelsen, Gluud, Vilsbøll et al., Bile acid sequestrants for glycemic control in patients with type 2 diabetes: A systematic review with meta-analysis of randomized controlled trials, J. diabetes its Complicat, doi:10.1016/j.jdiacomp.2017.01.011
Holz, Kühtreiber, Habener, Pancreatic beta-cells are rendered glucose-competent by the insulinotropic hormone glucagon-like peptide-1(7-37), Nature, doi:10.1038/361362a0
Huang, Wang, Ma, Ji, Chen et al., Dysbiosis and Implication of the Gut Microbiota in Diabetic Retinopathy
Ibrahim, Lowe, Bramante, Shah, Klatt et al., Metformin and Covid-19: focused Review of Mechanisms and Current Literature Suggesting Benefit, Front. Endocrinol, doi:10.3389/fendo.2021.587801
Justice, Gubbi, Kulkarni, Bartley, Kuchel et al., A geroscience perspective on immune resilience and infectious diseases: a potential case for metformin, GeroScience, doi:10.1007/s11357-020-00261-6
Kamyshnyi, Matskevych, Lenchuk, Strilbytska, Storey et al., Metformin to decrease COVID-19 severity and mortality: molecular mechanisms and therapeutic potential, Biomed. Pharmacother, doi:10.1016/j.biopha.2021.112230
Karlsson, Tremaroli, Nookaew, Bergström, Behre et al., Gut metagenome in European women with normal, impaired and diabetic glucose control, Nature, doi:10.1038/nature12198
Ke, Li, Deng, Li, Wang et al., Metformin Exerts Antiinflammatory and Mucus Barrier Protective Effects by Enriching Akkermansia muciniphila in Mice With Ulcerative Colitis, Front. Pharmacol, doi:10.3389/fphar.2021.726707
Koffert, Mikkola, Virtanen, Andersson, Faxius et al., Metformin treatment significantly enhances intestinal glucose uptake in patients with type 2 diabetes: results from a randomized clinical trial, Diabetes Res. Clin. Pract, doi:10.1016/j.diabres.2017.07.015
Kuhre, Frost, Svendsen, Holst, Molecular mechanisms of glucose-stimulated GLP-1 secretion from perfused rat small intestine, Diabetes, doi:10.2337/db14-0807
Kuhre, Wewer Albrechtsen, Larsen, Jepsen, Balk-Møller et al., Bile acids are important direct and indirect regulators of the secretion of appetite-and metabolism-regulating hormones from the gut and pancreas, Mol. Metab, doi:10.1016/j.molmet.2018.03.007
Kulkarni, Gubbi, Barzilai, Benefits of Metformin in Attenuating the Hallmarks of Aging, Cell Metab, doi:10.1016/j.cmet.2020.04.001
Larraufie, Roberts, Mcgavigan, Kay, Li et al., Important Role of the GLP-1 Axis for Glucose Homeostasis after Bariatric Surgery, Cell Rep, doi:10.1016/j.celrep.2019.01.047
Larsen, Vogensen, Van Den, Berg, Nielsen et al., Gut microbiota in human adults with type 2 diabetes differs from non-diabetic adults, PloS one, doi:10.1371/journal.pone.0009085
Lee, Chae, Jo, Jerng, Bae, The Relationship between the Gut Microbiome and Metformin as a Key for Treating Type 2 Diabetes Mellitus, Int. J. Mol. Sci, doi:10.3390/ijms22073566
Lenzen, Lortz, Tiedge, Effect of metformin on SGLT1, GLUT2, and GLUT5 hexose transporter gene expression in small intestine from rats, Biochem. Pharmacol, doi:10.1016/0006-2952(95)02243-0
Li, Jiang, Krausz, Li, Albert et al., Microbiome remodelling leads to inhibition of intestinal farnesoid X receptor signalling and decreased obesity, Nat. Commun, doi:10.1038/ncomms3384
Li, Zhou, Yan, An Update on the Mutual Impact between SARS-CoV-2 Infection and Gut Microbiota, Viruses, doi:10.3390/v14081774
Ma, Patel, Vemparala, Krishnamurthy, Metformin is associated with favorable outcomes in patients with COVID-19 and Type 2 diabetes mellitus, J. Sci. Rep, doi:10.1038/s41598-022-09639-2
Madsen, Baggesen, Richelsen, Thomsen, Effect of Roux-en-Y gastric bypass surgery on diabetes remission and complications in individuals with type 2 diabetes: a Danish population-based matched cohort study, Diabetologia, doi:10.1007/s00125-019-4816-2
Makishima, Okamoto, Repa, Tu, Learned et al., Identification of a nuclear receptor for bile acids, Science, doi:10.1126/science.284.5418.1362
Mannucci, Tesi, Bardini, Ognibene, Petracca et al., Effects of metformin on glucagon-like peptide-1 levels in obese patients with and without Type 2 diabetes, Diabetes, Nutr. metabolism
Mercado-Gómez, Prieto-Fernández, Goikoetxea-Usandizaga, Vila-Vecilla, Azkargorta et al., The spike of SARS-CoV-2 promotes metabolic rewiring in hepatocytes, Commun. Biol, doi:10.1038/s42003-022-03789-9
Mohammed, Hollenberg, Ding, Triggle, A Critical Review of the Evidence That Metformin Is a Putative Anti-Aging Drug That Enhances Healthspan and Extends Lifespan, Front. Endocrinol, doi:10.3389/fendo.2021.718942
Ng, Leung, Cheung, Association between Gut Microbiota and SARS-CoV-2 Infection and Vaccine Immunogenicity, Microorganisms, doi:10.3390/microorganisms11020452
Omarjee, Janin, Perrot, Laviolle, Meilhac et al., Targeting T-cell senescence and cytokine storm with rapamycin to prevent severe progression in COVID-19, Clin. Immunol. Orl. Fla), doi:10.1016/j.clim.2020.108464
Oo, Zandi, Shepard, Bassit, Musall et al., Elimination of Aicardi-Goutières syndrome protein SAMHD1 activates cellular innate immunity and suppresses SARS-CoV-2 replication, J. Biol. Chem, doi:10.1016/j.jbc.2022.101635
Parker, Adriaenssens, Rogers, Richards, Koepsell et al., Predominant role of active versus facilitative glucose transport for glucagon-like peptide-1 secretion, Diabetologia, doi:10.1007/s00125-012-2585-2
Parthasarathy, Tandel, Siddiqui, Harshan, Metformin suppresses SARS-CoV-2 in cell culture, Virus Res, doi:10.1016/j.virusres.2022.199010
Perry, Peng, Barry, Cline, Zhang et al., Acetate mediates a microbiome-brain-β-cell axis to promote metabolic syndrome, Nature, doi:10.1038/nature18309
Petakh, Griga, Mohammed, Loshak, Poliak et al., Effects of Metformin, Insulin on Hematological Parameters of COVID-19 Patients with Type 2 Diabetes, Med. archives (Sarajevo, Bosnia Herzegovina), doi:10.5455/medarh.2022.76.329-332
Petakh, Isevych, Mohammed, Loshak, Poliak et al., Association between Use of Metformin and Insulin with Hematological Parameters in COVID-19 Patients with Type 2 Diabetes: A Single Center, Cross-Sectional Study, Clin. Diabetol, doi:10.5603/dk.a2022.0055
Petakh, Kamyshna, Kamyshnyi, Effects of metformin on the gut microbiota: A systematic review, Mol. Metab, doi:10.1016/j.molmet.2023.101805
Petakh, Kamyshna, Nykyforuk, Yao, Imbery et al., Immunoregulatory Intestinal Microbiota and COVID-19 in Patients with Type Two Diabetes: A Double-Edged Sword, J. Viruses, doi:10.3390/v14030477
Petakh, Kamyshna, Oksenych, Kainov, Kamyshnyi, Metformin Therapy Changes Gut Microbiota Alpha-Diversity in COVID-19 Patients with Type 2 Diabetes: the Role of SARS-CoV-2 Variants and Antibiotic Treatment, Pharm, doi:10.3390/ph16060904
Petakh, Kobyliak, Kamyshnyi, Gut microbiota in patients with COVID-19 and type 2 diabetes: A culture-based method, Front. Cell. Infect. Microbiol, doi:10.3389/fcimb.2023.1142578
Petakh, Loshak, Kamyshnyi, Hematological features of patients with type 2 diabetes depending on the variant of SARS-COV-2, Fiziolohichnyĭ Zhurnal, doi:10.15407/fz69.01.035
Petakh, Oksenych, Kamyshnyi, The F/B ratio as a biomarker for inflammation in COVID-19 and T2D: impact of metformin, Biomed. Pharmacother, doi:10.1016/j.biopha.2023.114892
Pénicaud, Hitier, Ferré, Girard, Hypoglycaemic effect of metformin in genetically obese (fa/fa) rats results from an increased utilization of blood glucose by intestine, Biochem. J, doi:10.1042/bj2620881
Qin, Li, Cai, Li, Zhu et al., A metagenome-wide association study of gut microbiota in type 2 diabetes, Nature, doi:10.1038/nature11450
Reis, Santos Moreira, Silva, Medeiros Silva, Thabane et al., Effect of early treatment with metformin on risk of emergency care and hospitalization among patients with COVID-19: the TOGETHER randomized platform clinical trial, Lancet reg. health Am, doi:10.1016/j.lana.2021.100142
Rooj, Kimura, Buddington, Metabolites produced by probiotic Lactobacilli rapidly increase glucose uptake by Caco-2 cells, BMC Microbiol, doi:10.1186/1471-2180-10-16
Sakar, Meddah, Faouzi, Cherrah, Bado et al., Metformin-induced regulation of the intestinal D-glucose transporters, J. physiology Pharmacol
Salvatore, Pafundi, Galiero, Gjeloshi, Masini et al., Metformin: A Potential Therapeutic Tool for Rheumatologists, Pharm, doi:10.3390/ph13090234
Sansome, Xie, Veedfald, Horowitz, Rayner et al., Mechanism of glucose-lowering by metformin in type 2 diabetes: role of bile acids, Diabetes, Obes. metabolism, doi:10.1111/dom.13869
Scarpello, Hodgson, Howlett, Sharma, Dupee et al., Ex vivo SARS-CoV-2 infection of human lung reveals heterogeneous host defense and therapeutic responses, Diabet. Med. a J. Br. Diabet. Assoc, doi:10.1172/jci.insight.148003
Scheen, Metformin and COVID-19: from cellular mechanisms to reduced mortality, J. Diabetes Metab, doi:10.1016/j.diabet.2020.07.006
Sharma, Ray, Sadasivam, Metformin in COVID-19: a possible role beyond diabetes, Diabetes Res. Clin. Pract, doi:10.1016/j.diabres.2020.108183
Shen, Yang, Li, Wang, Lv et al., Metformin promotes cGAS/STING signaling pathway activation by blocking AKT phosphorylation in gastric cancer, Heliyon, doi:10.1016/j.heliyon.2023.e18954
Shih, Yeh, Lin, Yang, Chiang, Akkermansia muciniphila is Negatively Correlated with Hemoglobin A1c in Refractory Diabetes, Microorganisms, doi:10.3390/microorganisms8091360
Smigiel, Srivastava, Stolley, Campbell, Regulatory T-cell homeostasis: steady-state maintenance and modulation during inflammation, Immunol. Rev, doi:10.1111/imr.12170
Su, Shen, Hu, Chen, Cheng et al., SARS-CoV-2 ORF3a inhibits cGAS-STING-mediated autophagy flux and antiviral function, J. Med. Virol, doi:10.1002/jmv.28175
Sun, Liu, Huang, Xu, Hu et al., SARS-CoV-2 nonstructural protein 6 triggers NLRP3-dependent pyroptosis by targeting ATP6AP1, Cell death Differ, doi:10.1038/s41418-021-00916-7
Sun, Song, Liu, Tan, Lin et al., Gut microbiome alterations and gut barrier dysfunction are associated with host immune homeostasis in COVID-19 patients, BMC Med, doi:10.1186/s12916-021-02212-0
Sun, Xie, Wang, Wu, Wu et al., Gut microbiota and intestinal FXR mediate the clinical benefits of metformin, Nat. Med, doi:10.1038/s41591-018-0222-4
Synodinou, Nikolaki, Triantafyllou, Kasti, Immunomodulatory Effects of Probiotics on COVID-19 Infection by Targeting the Gut-Lung Axis Microbial Cross-Talk, Microorganisms, doi:10.3390/microorganisms10091764
Tan, Heng, Mak, The Potential Use of Metformin, Dipyridamole, N-Acetylcysteine and Statins as Adjunctive Therapy for Systemic Lupus Erythematosus, Syst. Lupus Erythematosus. Cells, doi:10.3390/cells8040323
Thomas, Gioiello, Noriega, Strehle, Oury et al., TGR5-mediated bile acid sensing controls glucose homeostasis, Cell Metab, doi:10.1016/j.cmet.2009.08.001
Titov, Baker, Brusko, Sobel, Morel, Metformin Inhibits the Type 1 IFN Response in Human CD4(+) T Cells, J. Immunol, doi:10.4049/jimmunol.1801651
Trabelsi, Daoudi, Prawitt, Ducastel, Touche et al., Farnesoid X receptor inhibits glucagon-like peptide-1 production by enteroendocrine L cells, Nat. Commun, doi:10.1038/ncomms8629
Triggle, Mohammed, Bshesh, Marei, Ye et al., Metformin: is it a drug for all reasons and diseases?, Metabolism Clin. Exp, doi:10.1016/j.metabol.2022.155223
Tsai, Chang, Sun, Chan, Wu et al., Metformin activates type I interferon signaling against HCV via activation of adenosine monophosphate-activated protein kinase, Oncotarget, doi:10.18632/oncotarget.20248
Ventura-López, Cervantes-Luevano, Aguirre-Sánchez, Flores-Caballero, Alvarez-Delgado et al., Treatment with metformin glycinate reduces SARS-CoV-2 viral load: an in vitro model and randomized, double-blind, Phase IIb clinical trial, Biomed. Pharmacother. = Biomedecine Pharmacother, doi:10.1016/j.biopha.2022.113223
Wang, Zhang, Chen, Zhang, Lv et al., A comparative study of microbial community and functions of type 2 diabetes mellitus patients with obesity and healthy people, Appl. Microbiol. Biotechnol, doi:10.1007/s00253-020-10689-7
Wang, Zhang, Wang, Dai, Qin et al., Alterations in microbiota of patients with COVID-19: potential mechanisms and therapeutic interventions, Signal Transduct. Target. Ther, doi:10.1038/s41392-022-00986-0
Watson, Xie, Wang, Li, Phillips et al., Gastric Emptying in Patients With Well-Controlled Type 2 Diabetes Compared With Young and Older Control Subjects Without Diabetes, J. Clin. Endocrinol. metabolism, doi:10.1210/jc.2018-02736
Wiernsperger, Al-Salameh, Cariou, Lalau, Protection by metformin against severe Covid-19: an in-depth mechanistic analysis, Diabetes & metabolism, doi:10.1016/j.diabet.2022.101359
Wilcock, Bailey, Accumulation of metformin by tissues of the normal and diabetic mouse. Xenobiotica; fate foreign Compd, Biol. Syst, doi:10.3109/00498259409043220
Wu, Bound, Standfield, Gedulin, Jones et al., Effects of rectal administration of taurocholic acid on glucagon-like peptide-1 and peptide YY secretion in healthy humans, Diabetes, Obes. metabolism, doi:10.1111/dom.12043
Wu, Bound, Standfield, Jones, Horowitz et al., Effects of taurocholic acid on glycemic, glucagon-like peptide-1, and insulin responses to small intestinal glucose infusion in healthy humans, J. Clin. Endocrinol. metabolism, doi:10.1210/jc.2012-3961
Wu, Esteve, Tremaroli, Khan, Caesar et al., Metformin alters the gut microbiome of individuals with treatment-naive type 2 diabetes, contributing to the therapeutic effects of the drug, Nat. Med, doi:10.1038/nm.4345
Wu, Xie, Wu, Jones, Horowitz et al., Metformin reduces the rate of small intestinal glucose absorption in type 2 diabetes, Diabetes, Obes. metabolism, doi:10.1111/dom.12812
Xian, Liu, Rundberg Nilsson, Gatchalian, Crother et al., Metformin inhibition of mitochondrial ATP and DNA synthesis abrogates NLRP3 inflammasome activation and pulmonary inflammation, Immunity, doi:10.1016/j.immuni.2021.05.004
Xiang, Liu, Alterations of the gut microbiota in coronavirus disease 2019 and its therapeutic potential, World J. gastroenterology, doi:10.3748/wjg.v28.i47.6689
Yadav, Jain, Sinha, Antidiabetic effect of probiotic dahi containing Lactobacillus acidophilus and Lactobacillus casei in high fructose fed rats, Nutrition, doi:10.1016/j.nut.2006.09.002
Yang, Ding, Wang, Gu, Zhang et al., Metformin regulates of Th17/treg cell balance and reduces hyperplastic synovium via activating AMPK and inhibiting mTOR in a collagen-induced arthritis rat model, J. Int. J. Clin. Exp. Med
Yeoh, Zuo, Lui, Zhang, Liu et al., Gut microbiota composition reflects disease severity and dysfunctional immune responses in patients with COVID-19, Gut, doi:10.1136/gutjnl-2020-323020
Zhang, Wang, Yang, Xu, Liang et al., Response of gut microbiota in type 2 diabetes to hypoglycemic agents, Endocrine, doi:10.1007/s12020-019-02041-5
Zhong, Ren, Lu, Fang, Hou et al., Distinct gut metagenomics and metaproteomics signatures in prediabetics and treatment-naïve type 2 diabetics, doi:10.1016/j.ebiom.2019.08.048
DOI record:
{
"DOI": "10.3389/fmolb.2023.1260633",
"ISSN": [
"2296-889X"
],
"URL": "http://dx.doi.org/10.3389/fmolb.2023.1260633",
"abstract": "<jats:p>This review article explores the potential of metformin, a medication commonly used for type 2 diabetes, as an antiviral and anti-inflammatory agent in the context of coronavirus disease 2019 (COVID-19). Metformin has demonstrated inhibitory effects on the growth of SARS-CoV-2 in cell culture models and has shown promising results in reducing viral load and achieving undetectable viral levels in clinical trials. Additionally, metformin exhibits anti-inflammatory properties by reducing the production of pro-inflammatory cytokines and modulating immune cell function, which may help prevent cytokine storms associated with severe COVID-19. The drug’s ability to regulate the balance between pro-inflammatory Th17 cells and anti-inflammatory Treg cells suggests its potential in mitigating inflammation and restoring T cell functionality. Furthermore, metformin’s modulation of the gut microbiota, particularly changes in bacterial taxa and the production of short-chain fatty acids, may contribute to its therapeutic effects. The interplay between metformin, bile acids, the gut microbiome, glucagon-like peptide-1 secretion, and glycemic control has implications for the management of diabetes and potential interventions in COVID-19. By refreshing the current evidence, this review highlights the potential of metformin as a therapeutic option in the management of COVID-19, while also exploring its effects on the gut microbiome and immunometabolism.</jats:p>",
"alternative-id": [
"10.3389/fmolb.2023.1260633"
],
"author": [
{
"affiliation": [],
"family": "Petakh",
"given": "Pavlo",
"sequence": "first"
},
{
"affiliation": [],
"family": "Kamyshna",
"given": "Iryna",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Kamyshnyi",
"given": "Aleksandr",
"sequence": "additional"
}
],
"container-title": "Frontiers in Molecular Biosciences",
"container-title-short": "Front. Mol. Biosci.",
"content-domain": {
"crossmark-restriction": true,
"domain": [
"frontiersin.org"
]
},
"created": {
"date-parts": [
[
2023,
10,
10
]
],
"date-time": "2023-10-10T06:13:36Z",
"timestamp": 1696918416000
},
"deposited": {
"date-parts": [
[
2023,
10,
10
]
],
"date-time": "2023-10-10T06:13:42Z",
"timestamp": 1696918422000
},
"indexed": {
"date-parts": [
[
2024,
2,
12
]
],
"date-time": "2024-02-12T10:42:00Z",
"timestamp": 1707734520738
},
"is-referenced-by-count": 1,
"issued": {
"date-parts": [
[
2023,
10,
10
]
]
},
"license": [
{
"URL": "https://creativecommons.org/licenses/by/4.0/",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2023,
10,
10
]
],
"date-time": "2023-10-10T00:00:00Z",
"timestamp": 1696896000000
}
}
],
"link": [
{
"URL": "https://www.frontiersin.org/articles/10.3389/fmolb.2023.1260633/full",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "1965",
"original-title": [],
"prefix": "10.3389",
"published": {
"date-parts": [
[
2023,
10,
10
]
]
},
"published-online": {
"date-parts": [
[
2023,
10,
10
]
]
},
"publisher": "Frontiers Media SA",
"reference": [
{
"DOI": "10.1007/s00125-012-2593-2",
"article-title": "Rectal taurocholate increases L cell and insulin secretion, and decreases blood glucose and food intake in obese type 2 diabetic volunteers",
"author": "Adrian",
"doi-asserted-by": "publisher",
"first-page": "2343",
"journal-title": "Diabetologia",
"key": "B1",
"volume": "55",
"year": "2012"
},
{
"DOI": "10.3389/fcvm.2021.644095",
"article-title": "COVID-19 in relation to hyperglycemia and diabetes mellitus",
"author": "Al-Kuraishy",
"doi-asserted-by": "publisher",
"first-page": "644095",
"journal-title": "J Front. Cardiovasc. Med.",
"key": "B2",
"volume": "8",
"year": "2021"
},
{
"DOI": "10.1016/j.ebiom.2022.104430",
"article-title": "The gut microbiota and metabolome are associated with diminished COVID-19 vaccine-induced antibody responses in immunosuppressed inflammatory bowel disease patients",
"author": "Alexander",
"doi-asserted-by": "publisher",
"first-page": "104430",
"journal-title": "EBioMedicine",
"key": "B3",
"volume": "88",
"year": "2023"
},
{
"DOI": "10.1007/s00125-018-4550-1",
"article-title": "Aberrant intestinal microbiota in individuals with prediabetes",
"author": "Allin",
"doi-asserted-by": "publisher",
"first-page": "810",
"journal-title": "J. Diabetol.",
"key": "B4",
"volume": "61",
"year": "2018"
},
{
"DOI": "10.3389/fimmu.2023.1080043",
"article-title": "Gut and airway microbiota dysbiosis and their role in COVID-19 and long-COVID",
"author": "Ancona",
"doi-asserted-by": "publisher",
"first-page": "1080043",
"journal-title": "Front. Immunol.",
"key": "B5",
"volume": "14",
"year": "2023"
},
{
"DOI": "10.1111/febs.15375",
"article-title": "A Guide to COVID-19: a global pandemic caused by the novel coronavirus SARS-CoV-2",
"author": "Atzrodt",
"doi-asserted-by": "publisher",
"first-page": "3633",
"journal-title": "FEBS J.",
"key": "B6",
"volume": "287",
"year": "2020"
},
{
"DOI": "10.3389/fphar.2022.784459",
"article-title": "Diabetes, Metformin and the Clinical Course of Covid-19: outcomes, Mechanisms and Suggestions on the Therapeutic Use of Metformin",
"author": "Bailey",
"doi-asserted-by": "publisher",
"first-page": "784459",
"journal-title": "Front. Pharmacol.",
"key": "B7",
"volume": "13",
"year": "2022"
},
{
"DOI": "10.1111/j.1476-5381.1992.tb09093.x",
"article-title": "Effect of metformin on glucose metabolism in the splanchnic bed",
"author": "Bailey",
"doi-asserted-by": "publisher",
"first-page": "1009",
"journal-title": "Br. J. Pharmacol.",
"key": "B8",
"volume": "105",
"year": "1992"
},
{
"DOI": "10.1007/s00394-016-1317-7",
"article-title": "Improvement in glucose tolerance and insulin sensitivity by probiotic strains of Indian gut origin in high-fat diet-fed C57BL/6J mice",
"author": "Balakumar",
"doi-asserted-by": "publisher",
"first-page": "279",
"journal-title": "Eur. J. Nutr.",
"key": "B9",
"volume": "57",
"year": "2018"
},
{
"DOI": "10.1371/journal.pone.0194171",
"article-title": "Gut microbiota varies by opioid use, circulating leptin and oxytocin in African American men with diabetes and high burden of chronic disease",
"author": "Barengolts",
"doi-asserted-by": "publisher",
"first-page": "e0194171",
"journal-title": "PLoS One",
"key": "B10",
"volume": "13",
"year": "2018"
},
{
"DOI": "10.1016/j.cmet.2017.09.019",
"article-title": "Metformin Alters Upper Small Intestinal Microbiota that Impact a Glucose-SGLT1-Sensing Glucoregulatory Pathway",
"author": "Bauer",
"doi-asserted-by": "publisher",
"first-page": "101",
"journal-title": "Cell Metab.",
"key": "B11",
"volume": "27",
"year": "2018"
},
{
"DOI": "10.1186/s40168-021-01193-9",
"article-title": "Metabolite-based dietary supplementation in human type 1 diabetes is associated with microbiota and immune modulation",
"author": "Bell",
"doi-asserted-by": "publisher",
"first-page": "9",
"journal-title": "Microbiome",
"key": "B12",
"volume": "10",
"year": "2022"
},
{
"DOI": "10.1101/2023.06.06.23290989",
"article-title": "Metformin reduces SARS-CoV-2 in a Phase 3 Randomized Placebo Controlled Clinical Trial",
"author": "Bramante",
"doi-asserted-by": "publisher",
"first-page": "23290989",
"journal-title": "medRxiv.",
"key": "B13",
"volume": "2023",
"year": "2023"
},
{
"DOI": "10.1111/dom.13080",
"article-title": "The bile acid-sequestering resin sevelamer eliminates the acute GLP-1 stimulatory effect of endogenously released bile acids in patients with type 2 diabetes",
"author": "Brønden",
"doi-asserted-by": "publisher",
"first-page": "362",
"journal-title": "Diabetes, Obes. metabolism",
"key": "B14",
"volume": "20",
"year": "2018"
},
{
"DOI": "10.1210/jc.2017-01091",
"article-title": "Single-Dose Metformin Enhances Bile Acid-Induced Glucagon-Like Peptide-1 Secretion in Patients With Type 2 Diabetes",
"author": "Brønden",
"doi-asserted-by": "publisher",
"first-page": "4153",
"journal-title": "J. Clin. Endocrinol. metabolism",
"key": "B15",
"volume": "102",
"year": "2017"
},
{
"DOI": "10.1007/s00125-019-4848-7",
"article-title": "Metformin-induced changes of the gut microbiota in healthy young men: results of a non-blinded, one-armed intervention study",
"author": "Bryrup",
"doi-asserted-by": "publisher",
"first-page": "1024",
"journal-title": "Diabetologia",
"key": "B16",
"volume": "62",
"year": "2019"
},
{
"DOI": "10.1161/CIRCRESAHA.116.308445",
"article-title": "Anti-Inflammatory Effects of Metformin Irrespective of Diabetes Status",
"author": "Cameron",
"doi-asserted-by": "publisher",
"first-page": "652",
"journal-title": "Circ. Res.",
"key": "B17",
"volume": "119",
"year": "2016"
},
{
"DOI": "10.1017/S0007114516001045",
"article-title": "Modulation of gut microbiota dysbioses in type 2 diabetic patients by macrobiotic Ma-Pi 2 diet",
"author": "Candela",
"doi-asserted-by": "publisher",
"first-page": "80",
"journal-title": "Br. J. Nutr.",
"key": "B18",
"volume": "116",
"year": "2016"
},
{
"DOI": "10.1101/2023.06.06.23290989",
"article-title": "Metformin reduces SARS-CoV-2 in a Phase 3 Randomized Placebo Controlled Clinical Trial",
"author": "Carolyn",
"doi-asserted-by": "publisher",
"first-page": "23290989",
"key": "B19",
"volume": "2023",
"year": "2023"
},
{
"DOI": "10.3390/v14071538",
"article-title": "Galectin-9 and Interferon-Gamma Are Released by Natural Killer Cells upon Activation with Interferon-Alpha and Orchestrate the Suppression of Hepatitis C Virus Infection",
"author": "Carreca",
"doi-asserted-by": "publisher",
"first-page": "1538",
"journal-title": "Viruses",
"key": "B20",
"volume": "14",
"year": "2022"
},
{
"DOI": "10.1136/gutjnl-2019-318424",
"article-title": "Dietary supplementation with inulin-propionate ester or inulin improves insulin sensitivity in adults with overweight and obesity with distinct effects on the gut microbiota, plasma metabolome and systemic inflammatory responses: a randomised cross-over trial",
"author": "Chambers",
"doi-asserted-by": "publisher",
"first-page": "1430",
"journal-title": "Gut",
"key": "B21",
"volume": "68",
"year": "2019"
},
{
"DOI": "10.1136/gutjnl-2014-307913",
"article-title": "Effects of targeted delivery of propionate to the human colon on appetite regulation, body weight maintenance and adiposity in overweight adults",
"author": "Chambers",
"doi-asserted-by": "publisher",
"first-page": "1744",
"journal-title": "Gut",
"key": "B22",
"volume": "64",
"year": "2015"
},
{
"DOI": "10.3390/microorganisms8010094",
"article-title": "Characterization of the gut microbiota of individuals at different T2D stages reveals a complex relationship with the host",
"author": "Chávez-Carbajal",
"doi-asserted-by": "publisher",
"first-page": "94",
"journal-title": "Microorganisms",
"key": "B23",
"volume": "8",
"year": "2020"
},
{
"DOI": "10.3390/vaccines10071122",
"article-title": "Association between Recent Usage of Antibiotics and Immunogenicity within Six Months after COVID-19 Vaccination",
"author": "Cheung",
"doi-asserted-by": "publisher",
"first-page": "1122",
"journal-title": "Vaccines",
"key": "B24",
"volume": "10",
"year": "2022"
},
{
"DOI": "10.1016/j.nut.2018.11.019",
"article-title": "The alteration of gut microbiota in newly diagnosed type 2 diabetic patients",
"author": "Chen",
"doi-asserted-by": "publisher",
"first-page": "51",
"journal-title": "Nutrition",
"key": "B25",
"year": "2019"
},
{
"DOI": "10.21203/rs.3.rs-513461/v1",
"article-title": "SARS-CoV-2 Infection Causes Dopaminergic Neuron Senescence",
"author": "Chen",
"doi-asserted-by": "publisher",
"first-page": "513461",
"journal-title": "Res. square",
"key": "B26",
"year": "2021"
},
{
"DOI": "10.3389/fimmu.2021.733921",
"article-title": "Metformin Suppresses Monocyte Immunometabolic Activation by SARS-CoV-2 Spike Protein Subunit 1",
"author": "Cory",
"doi-asserted-by": "publisher",
"first-page": "733921",
"journal-title": "Front. Immunol.",
"key": "B27",
"volume": "12",
"year": "2021"
},
{
"DOI": "10.1038/s41385-019-0160-6",
"article-title": "Microbes, metabolites, and the gut-lung axis",
"author": "Dang",
"doi-asserted-by": "publisher",
"first-page": "843",
"journal-title": "Mucosal Immunol.",
"key": "B28",
"volume": "12",
"year": "2019"
},
{
"DOI": "10.2337/dc16-1324",
"article-title": "Metformin is associated with higher relative abundance of mucin-degrading Akkermansia muciniphila and several short-chain fatty acid–producing microbiota in the gut",
"author": "De La Cuesta-Zuluaga",
"doi-asserted-by": "publisher",
"first-page": "54",
"journal-title": "Diabetes care",
"key": "B29",
"volume": "40",
"year": "2017"
},
{
"DOI": "10.3389/fimmu.2021.635471",
"article-title": "Microbiota Modulation of the Gut-Lung Axis in COVID-19",
"author": "de Oliveira",
"doi-asserted-by": "publisher",
"first-page": "635471",
"journal-title": "Front. Immunol.",
"key": "B30",
"volume": "12",
"year": "2021"
},
{
"DOI": "10.1194/jlr.R036012",
"article-title": "The role of short-chain fatty acids in the interplay between diet, gut microbiota, and host energy metabolism",
"author": "den Besten",
"doi-asserted-by": "publisher",
"first-page": "2325",
"journal-title": "J. lipid Res.",
"key": "B31",
"volume": "54",
"year": "2013"
},
{
"DOI": "10.1016/j.apsb.2015.01.004",
"article-title": "Bile acid nuclear receptor FXR and digestive system diseases",
"author": "Ding",
"doi-asserted-by": "publisher",
"first-page": "135",
"journal-title": "Acta Pharm. Sin. B",
"key": "B32",
"volume": "5",
"year": "2015"
},
{
"DOI": "10.1371/journal.pone.0003974",
"article-title": "Bitter taste receptors influence glucose homeostasis",
"author": "Dotson",
"doi-asserted-by": "publisher",
"first-page": "e3974",
"journal-title": "PLoS One",
"key": "B33",
"volume": "3",
"year": "2008"
},
{
"article-title": "Metformin mitigates autoimmune insulitis by inhibiting Th1 and Th17 responses while promoting Treg production",
"author": "Duan",
"first-page": "2393",
"journal-title": "Am. J. Transl. Res.",
"key": "B34",
"volume": "11",
"year": "2019"
},
{
"DOI": "10.1038/nm.3787",
"article-title": "Metformin activates a duodenal Ampk-dependent pathway to lower hepatic glucose production in rats",
"author": "Duca",
"doi-asserted-by": "publisher",
"first-page": "506",
"journal-title": "Nat. Med.",
"key": "B35",
"volume": "21",
"year": "2015"
},
{
"DOI": "10.1152/ajpcell.00442.2020",
"article-title": "The H(+)-ATPase (V-ATPase): from proton pump to signaling complex in health and disease",
"author": "Eaton",
"doi-asserted-by": "publisher",
"first-page": "C392",
"journal-title": "Am. J. physiology Cell physiology",
"key": "B36",
"volume": "320",
"year": "2021"
},
{
"DOI": "10.1016/j.nut.2011.08.013",
"article-title": "Probiotic yogurt improves antioxidant status in type 2 diabetic patients",
"author": "Ejtahed",
"doi-asserted-by": "publisher",
"first-page": "539",
"journal-title": "Nutrition",
"key": "B37",
"volume": "28",
"year": "2012"
},
{
"DOI": "10.1136/gutjnl-2011-301955",
"article-title": "The gut microbiota elicits a profound metabolic reorientation in the mouse jejunal mucosa during conventionalisation",
"author": "El Aidy",
"doi-asserted-by": "publisher",
"first-page": "1306",
"journal-title": "Gut",
"key": "B38",
"volume": "62",
"year": "2013"
},
{
"DOI": "10.2337/dc22-2539",
"article-title": "Metformin for Treatment of Acute COVID-19: systematic Review of Clinical Trial Data Against SARS-CoV-2",
"author": "Erickson",
"doi-asserted-by": "publisher",
"first-page": "1432",
"journal-title": "Diabetes Care",
"key": "B39",
"volume": "46",
"year": "2023"
},
{
"DOI": "10.1016/j.diabres.2020.108282",
"article-title": "A proposed mechanism for the possible therapeutic potential of Metformin in COVID-19",
"author": "Esam",
"doi-asserted-by": "publisher",
"first-page": "108282",
"journal-title": "Diabetes Res. Clin. Pract.",
"key": "B40",
"volume": "167",
"year": "2020"
},
{
"DOI": "10.1038/s41598-021-87707-9",
"article-title": "The antiviral effect of metformin on zika and dengue virus infection",
"author": "Farfan-Morales",
"doi-asserted-by": "publisher",
"first-page": "8743",
"journal-title": "Sci. Rep.",
"key": "B41",
"volume": "11",
"year": "2021"
},
{
"DOI": "10.1038/nature15766",
"article-title": "Disentangling type 2 diabetes and metformin treatment signatures in the human gut microbiota",
"author": "Forslund",
"doi-asserted-by": "publisher",
"first-page": "262",
"journal-title": "Nature",
"key": "B42",
"volume": "528",
"year": "2015"
},
{
"DOI": "10.3389/fimmu.2021.686240",
"article-title": "Plasma Markers of Disrupted Gut Permeability in Severe COVID-19 Patients",
"author": "Giron",
"doi-asserted-by": "publisher",
"first-page": "686240",
"journal-title": "Front. Immunol.",
"key": "B43",
"volume": "12",
"year": "2021"
},
{
"DOI": "10.1007/s10482-020-01474-7",
"article-title": "Human gut microbiota/microbiome in health and diseases: a review",
"author": "Gomaa",
"doi-asserted-by": "publisher",
"first-page": "2019",
"journal-title": "Antonie Leeuwenhoek",
"key": "B44",
"volume": "113",
"year": "2020"
},
{
"DOI": "10.2337/db11-1029",
"article-title": "Na+-d-glucose Cotransporter SGLT1 is Pivotal for Intestinal Glucose Absorption and Glucose-Dependent Incretin Secretion",
"author": "Gorboulev",
"doi-asserted-by": "publisher",
"first-page": "187",
"journal-title": "Diabetes",
"key": "B45",
"volume": "61",
"year": "2011"
},
{
"DOI": "10.2337/db11-1029",
"article-title": "Na(+)-D-glucose cotransporter SGLT1 is pivotal for intestinal glucose absorption and glucose-dependent incretin secretion",
"author": "Gorboulev",
"doi-asserted-by": "publisher",
"first-page": "187",
"journal-title": "Diabetes",
"key": "B46",
"volume": "61",
"year": "2012"
},
{
"DOI": "10.1038/s41586-020-2286-9",
"article-title": "A SARS-CoV-2 protein interaction map reveals targets for drug repurposing",
"author": "Gordon",
"doi-asserted-by": "publisher",
"first-page": "459",
"journal-title": "Nature",
"key": "B47",
"volume": "583",
"year": "2020"
},
{
"DOI": "10.3389/fimmu.2022.841532",
"article-title": "Ingestion, Immunity, and Infection: nutrition and Viral Respiratory Tract Infections",
"author": "Govers",
"doi-asserted-by": "publisher",
"first-page": "841532",
"journal-title": "Front. Immunol.",
"key": "B48",
"volume": "13",
"year": "2022"
},
{
"DOI": "10.3390/biomedicines11010179",
"article-title": "Impact of COVID-19 on the Microbiome and Inflammatory Status of Type 2 Diabetes Patients",
"author": "Gradisteanu Pircalabioru",
"doi-asserted-by": "publisher",
"first-page": "179",
"journal-title": "Biomedicines",
"key": "B49",
"volume": "11",
"year": "2023"
},
{
"DOI": "10.4239/wjd.v6.i4.598",
"article-title": "Type 2 diabetes mellitus: from a metabolic disorder to an inflammatory condition",
"author": "Hameed",
"doi-asserted-by": "publisher",
"first-page": "598",
"journal-title": "World J. diabetes",
"key": "B50",
"volume": "6",
"year": "2015"
},
{
"DOI": "10.1002/jmv.27965",
"article-title": "SARS-CoV-2 ORF10 antagonizes STING-dependent interferon activation and autophagy",
"author": "Han",
"doi-asserted-by": "publisher",
"first-page": "5174",
"journal-title": "J. Med. Virol.",
"key": "B51",
"volume": "94",
"year": "2022"
},
{
"DOI": "10.1111/dom.12648",
"article-title": "Effect of chenodeoxycholic acid and the bile acid sequestrant colesevelam on glucagon-like peptide-1 secretion",
"author": "Hansen",
"doi-asserted-by": "publisher",
"first-page": "571",
"journal-title": "Diabetes, Obes. metabolism",
"key": "B52",
"volume": "18",
"year": "2016"
},
{
"DOI": "10.1016/j.jdiacomp.2017.01.011",
"article-title": "Bile acid sequestrants for glycemic control in patients with type 2 diabetes: A systematic review with meta-analysis of randomized controlled trials",
"author": "Hansen",
"doi-asserted-by": "publisher",
"first-page": "918",
"journal-title": "J. diabetes its Complicat.",
"key": "B53",
"volume": "31",
"year": "2017"
},
{
"DOI": "10.1038/361362a0",
"article-title": "Pancreatic beta-cells are rendered glucose-competent by the insulinotropic hormone glucagon-like peptide-1(7-37)",
"author": "Holz",
"doi-asserted-by": "publisher",
"first-page": "362",
"journal-title": "Nature",
"key": "B54",
"volume": "361",
"year": "1993"
},
{
"DOI": "10.3389/fcimb.2021.646348",
"author": "Huang",
"doi-asserted-by": "crossref",
"key": "B55",
"volume-title": "Dysbiosis and Implication of the Gut Microbiota in Diabetic Retinopathy",
"year": "2021"
},
{
"DOI": "10.3389/fendo.2021.587801",
"article-title": "Metformin and Covid-19: focused Review of Mechanisms and Current Literature Suggesting Benefit",
"author": "Ibrahim",
"doi-asserted-by": "publisher",
"first-page": "587801",
"journal-title": "Front. Endocrinol. (Lausanne).",
"key": "B56",
"volume": "12",
"year": "2021"
},
{
"DOI": "10.1007/s11357-020-00261-6",
"article-title": "A geroscience perspective on immune resilience and infectious diseases: a potential case for metformin",
"author": "Justice",
"doi-asserted-by": "publisher",
"first-page": "1093",
"journal-title": "GeroScience",
"key": "B57",
"volume": "43",
"year": "2021"
},
{
"DOI": "10.1016/j.biopha.2021.112230",
"article-title": "Metformin to decrease COVID-19 severity and mortality: molecular mechanisms and therapeutic potential",
"author": "Kamyshnyi",
"doi-asserted-by": "publisher",
"first-page": "112230",
"journal-title": "Biomed. Pharmacother.",
"key": "B58",
"volume": "144",
"year": "2021"
},
{
"DOI": "10.1038/nature12198",
"article-title": "Gut metagenome in European women with normal, impaired and diabetic glucose control",
"author": "Karlsson",
"doi-asserted-by": "publisher",
"first-page": "99",
"journal-title": "Nature",
"key": "B59",
"volume": "498",
"year": "2013"
},
{
"DOI": "10.3389/fphar.2021.726707",
"article-title": "Metformin Exerts Anti-inflammatory and Mucus Barrier Protective Effects by Enriching Akkermansia muciniphila in Mice With Ulcerative Colitis",
"author": "Ke",
"doi-asserted-by": "publisher",
"first-page": "726707",
"journal-title": "Front. Pharmacol.",
"key": "B60",
"volume": "12",
"year": "2021"
},
{
"DOI": "10.1016/j.diabres.2017.07.015",
"article-title": "Metformin treatment significantly enhances intestinal glucose uptake in patients with type 2 diabetes: results from a randomized clinical trial",
"author": "Koffert",
"doi-asserted-by": "publisher",
"first-page": "208",
"journal-title": "Diabetes Res. Clin. Pract.",
"key": "B61",
"volume": "131",
"year": "2017"
},
{
"DOI": "10.2337/db14-0807",
"article-title": "Molecular mechanisms of glucose-stimulated GLP-1 secretion from perfused rat small intestine",
"author": "Kuhre",
"doi-asserted-by": "publisher",
"first-page": "370",
"journal-title": "Diabetes",
"key": "B62",
"volume": "64",
"year": "2015"
},
{
"DOI": "10.1016/j.molmet.2018.03.007",
"article-title": "Bile acids are important direct and indirect regulators of the secretion of appetite- and metabolism-regulating hormones from the gut and pancreas",
"author": "Kuhre",
"doi-asserted-by": "publisher",
"first-page": "84",
"journal-title": "Mol. Metab.",
"key": "B63",
"volume": "11",
"year": "2018"
},
{
"DOI": "10.1016/j.cmet.2020.04.001",
"article-title": "Benefits of Metformin in Attenuating the Hallmarks of Aging",
"author": "Kulkarni",
"doi-asserted-by": "publisher",
"first-page": "15",
"journal-title": "Cell Metab.",
"key": "B64",
"volume": "32",
"year": "2020"
},
{
"DOI": "10.1016/j.celrep.2019.01.047",
"article-title": "Important Role of the GLP-1 Axis for Glucose Homeostasis after Bariatric Surgery",
"author": "Larraufie",
"doi-asserted-by": "publisher",
"first-page": "1399",
"journal-title": "Cell Rep.",
"key": "B65",
"volume": "26",
"year": "2019"
},
{
"DOI": "10.1371/journal.pone.0009085",
"article-title": "Gut microbiota in human adults with type 2 diabetes differs from non-diabetic adults",
"author": "Larsen",
"doi-asserted-by": "publisher",
"first-page": "e9085",
"journal-title": "PloS one",
"key": "B66",
"volume": "5",
"year": "2010"
},
{
"DOI": "10.3390/ijms22073566",
"article-title": "The Relationship between the Gut Microbiome and Metformin as a Key for Treating Type 2 Diabetes Mellitus",
"author": "Lee",
"doi-asserted-by": "publisher",
"first-page": "3566",
"journal-title": "Int. J. Mol. Sci.",
"key": "B67",
"volume": "22",
"year": "2021"
},
{
"DOI": "10.1016/0006-2952(95)02243-0",
"article-title": "Effect of metformin on SGLT1, GLUT2, and GLUT5 hexose transporter gene expression in small intestine from rats",
"author": "Lenzen",
"doi-asserted-by": "publisher",
"first-page": "893",
"journal-title": "Biochem. Pharmacol.",
"key": "B68",
"volume": "51",
"year": "1996"
},
{
"DOI": "10.1038/ncomms3384",
"article-title": "Microbiome remodelling leads to inhibition of intestinal farnesoid X receptor signalling and decreased obesity",
"author": "Li",
"doi-asserted-by": "publisher",
"first-page": "2384",
"journal-title": "Nat. Commun.",
"key": "B69",
"volume": "4",
"year": "2013"
},
{
"DOI": "10.3390/v14081774",
"article-title": "An Update on the Mutual Impact between SARS-CoV-2 Infection and Gut Microbiota",
"author": "Li",
"doi-asserted-by": "publisher",
"first-page": "1774",
"journal-title": "Viruses",
"key": "B70",
"volume": "14",
"year": "2022"
},
{
"article-title": "Effects of metformin on glucagon-like peptide-1 levels in obese patients with and without Type 2 diabetes",
"author": "Mannucci",
"first-page": "336",
"journal-title": "Diabetes, Nutr. metabolism",
"key": "B71",
"volume": "17",
"year": "2004"
},
{
"DOI": "10.1038/s41598-022-09639-2",
"article-title": "Metformin is associated with favorable outcomes in patients with COVID-19 and Type 2 diabetes mellitus",
"author": "Ma",
"doi-asserted-by": "publisher",
"first-page": "5553",
"journal-title": "J. Sci. Rep.",
"key": "B72",
"volume": "12",
"year": "2022"
},
{
"DOI": "10.1007/s00125-019-4816-2",
"article-title": "Effect of Roux-en-Y gastric bypass surgery on diabetes remission and complications in individuals with type 2 diabetes: a Danish population-based matched cohort study",
"author": "Madsen",
"doi-asserted-by": "publisher",
"first-page": "611",
"journal-title": "Diabetologia",
"key": "B73",
"volume": "62",
"year": "2019"
},
{
"DOI": "10.1126/science.284.5418.1362",
"article-title": "Identification of a nuclear receptor for bile acids",
"author": "Makishima",
"doi-asserted-by": "publisher",
"first-page": "1362",
"journal-title": "Science",
"key": "B74",
"volume": "284",
"year": "1999"
},
{
"DOI": "10.1038/s42003-022-03789-9",
"article-title": "The spike of SARS-CoV-2 promotes metabolic rewiring in hepatocytes",
"author": "Mercado-Gómez",
"doi-asserted-by": "publisher",
"first-page": "827",
"journal-title": "Commun. Biol.",
"key": "B75",
"volume": "5",
"year": "2022"
},
{
"DOI": "10.3389/fendo.2021.718942",
"article-title": "A Critical Review of the Evidence That Metformin Is a Putative Anti-Aging Drug That Enhances Healthspan and Extends Lifespan",
"author": "Mohammed",
"doi-asserted-by": "publisher",
"first-page": "718942",
"journal-title": "Front. Endocrinol. (Lausanne)",
"key": "B76",
"volume": "12",
"year": "2021"
},
{
"DOI": "10.3390/microorganisms11020452",
"article-title": "Association between Gut Microbiota and SARS-CoV-2 Infection and Vaccine Immunogenicity",
"author": "Ng",
"doi-asserted-by": "publisher",
"first-page": "452",
"journal-title": "Microorganisms",
"key": "B77",
"volume": "11",
"year": "2023"
},
{
"DOI": "10.1016/j.clim.2020.108464",
"article-title": "Targeting T-cell senescence and cytokine storm with rapamycin to prevent severe progression in COVID-19",
"author": "Omarjee",
"doi-asserted-by": "publisher",
"first-page": "108464",
"journal-title": "Clin. Immunol. Orl. Fla)",
"key": "B78",
"volume": "216",
"year": "2020"
},
{
"DOI": "10.1016/j.jbc.2022.101635",
"article-title": "Elimination of Aicardi–Goutières syndrome protein SAMHD1 activates cellular innate immunity and suppresses SARS-CoV-2 replication",
"author": "Oo",
"doi-asserted-by": "publisher",
"first-page": "101635",
"journal-title": "J. Biol. Chem.",
"key": "B79",
"volume": "298",
"year": "2022"
},
{
"DOI": "10.1007/s00125-012-2585-2",
"article-title": "Predominant role of active versus facilitative glucose transport for glucagon-like peptide-1 secretion",
"author": "Parker",
"doi-asserted-by": "publisher",
"first-page": "2445",
"journal-title": "Diabetologia",
"key": "B80",
"volume": "55",
"year": "2012"
},
{
"DOI": "10.1016/j.virusres.2022.199010",
"article-title": "Metformin suppresses SARS-CoV-2 in cell culture",
"author": "Parthasarathy",
"doi-asserted-by": "publisher",
"first-page": "199010",
"journal-title": "Virus Res.",
"key": "B81",
"volume": "323",
"year": "2022"
},
{
"DOI": "10.1042/bj2620881",
"article-title": "Hypoglycaemic effect of metformin in genetically obese (fa/fa) rats results from an increased utilization of blood glucose by intestine",
"author": "Pénicaud",
"doi-asserted-by": "publisher",
"first-page": "881",
"journal-title": "Biochem. J.",
"key": "B82",
"volume": "262",
"year": "1989"
},
{
"DOI": "10.1038/nature18309",
"article-title": "Acetate mediates a microbiome-brain-β-cell axis to promote metabolic syndrome",
"author": "Perry",
"doi-asserted-by": "publisher",
"first-page": "213",
"journal-title": "Nature",
"key": "B83",
"volume": "534",
"year": "2016"
},
{
"DOI": "10.5455/medarh.2022.76.329-332",
"article-title": "Effects of Metformin, Insulin on Hematological Parameters of COVID-19 Patients with Type 2 Diabetes",
"author": "Petakh",
"doi-asserted-by": "publisher",
"first-page": "329",
"journal-title": "Med. archives (Sarajevo, Bosnia Herzegovina)",
"key": "B84",
"volume": "76",
"year": ""
},
{
"DOI": "10.5603/dk.a2022.0055",
"article-title": "Association between Use of Metformin and Insulin with Hematological Parameters in COVID-19 Patients with Type 2 Diabetes: A Single Center, Cross-Sectional Study",
"author": "Petakh",
"doi-asserted-by": "publisher",
"first-page": "432",
"journal-title": "Clin. Diabetol.",
"key": "B85",
"volume": "11",
"year": ""
},
{
"DOI": "10.1016/j.molmet.2023.101805",
"article-title": "Effects of metformin on the gut microbiota: A systematic review",
"author": "Petakh",
"doi-asserted-by": "publisher",
"first-page": "101805",
"journal-title": "Mol. Metab.",
"key": "B86",
"volume": "77",
"year": ""
},
{
"DOI": "10.3390/v14030477",
"article-title": "Immunoregulatory Intestinal Microbiota and COVID-19 in Patients with Type Two Diabetes: A Double-Edged Sword",
"author": "Petakh",
"doi-asserted-by": "publisher",
"first-page": "477",
"journal-title": "J. Viruses",
"key": "B87",
"volume": "14",
"year": ""
},
{
"DOI": "10.3390/ph16060904",
"article-title": "Metformin Therapy Changes Gut Microbiota Alpha-Diversity in COVID-19 Patients with Type 2 Diabetes: the Role of SARS-CoV-2 Variants and Antibiotic Treatment",
"author": "Petakh",
"doi-asserted-by": "publisher",
"first-page": "904",
"journal-title": "Pharm. (Basel, Switz.",
"key": "B88",
"volume": "16",
"year": ""
},
{
"DOI": "10.3389/fcimb.2023.1142578",
"article-title": "Gut microbiota in patients with COVID-19 and type 2 diabetes: A culture-based method",
"author": "Petakh",
"doi-asserted-by": "publisher",
"first-page": "1142578",
"journal-title": "Front. Cell. Infect. Microbiol.",
"key": "B89",
"volume": "13",
"year": ""
},
{
"DOI": "10.15407/fz69.01.035",
"article-title": "Hematological features of patients with type 2 diabetes depending on the variant of SARS-COV-2",
"author": "Petakh",
"doi-asserted-by": "publisher",
"first-page": "35",
"journal-title": "Fiziolohichnyĭ Zhurnal",
"key": "B90",
"volume": "69",
"year": ""
},
{
"DOI": "10.1016/j.biopha.2023.114892",
"article-title": "The F/B ratio as a biomarker for inflammation in COVID-19 and T2D: impact of metformin",
"author": "Petakh",
"doi-asserted-by": "publisher",
"first-page": "114892",
"journal-title": "Biomed. Pharmacother.",
"key": "B91",
"volume": "163",
"year": ""
},
{
"DOI": "10.1038/nature11450",
"article-title": "A metagenome-wide association study of gut microbiota in type 2 diabetes",
"author": "Qin",
"doi-asserted-by": "publisher",
"first-page": "55",
"journal-title": "Nature",
"key": "B92",
"volume": "490",
"year": "2012"
},
{
"DOI": "10.1016/j.lana.2021.100142",
"article-title": "Effect of early treatment with metformin on risk of emergency care and hospitalization among patients with COVID-19: the TOGETHER randomized platform clinical trial",
"author": "Reis",
"doi-asserted-by": "publisher",
"first-page": "100142",
"journal-title": "Lancet reg. health Am.",
"key": "B93",
"volume": "6",
"year": "2022"
},
{
"DOI": "10.1186/1471-2180-10-16",
"article-title": "Metabolites produced by probiotic Lactobacilli rapidly increase glucose uptake by Caco-2 cells",
"author": "Rooj",
"doi-asserted-by": "publisher",
"first-page": "16",
"journal-title": "BMC Microbiol.",
"key": "B94",
"volume": "10",
"year": "2010"
},
{
"article-title": "Metformin-induced regulation of the intestinal D-glucose transporters",
"author": "Sakar",
"first-page": "301",
"journal-title": "J. physiology Pharmacol.",
"key": "B95",
"volume": "61",
"year": "2010"
},
{
"DOI": "10.3390/ph13090234",
"article-title": "Metformin: A Potential Therapeutic Tool for Rheumatologists",
"author": "Salvatore",
"doi-asserted-by": "publisher",
"first-page": "234",
"journal-title": "Pharm. (Basel, Switz.",
"key": "B96",
"volume": "13",
"year": "2020"
},
{
"DOI": "10.1111/dom.13869",
"article-title": "Mechanism of glucose-lowering by metformin in type 2 diabetes: role of bile acids",
"author": "Sansome",
"doi-asserted-by": "publisher",
"first-page": "141",
"journal-title": "Diabetes, Obes. metabolism",
"key": "B97",
"volume": "22",
"year": "2020"
},
{
"DOI": "10.1002/(SICI)1096-9136(199808)15:8<651::AID-DIA628>3.0.CO;2-A",
"article-title": "Effect of metformin on bile salt circulation and intestinal motility in type 2 diabetes mellitus",
"author": "Scarpello",
"doi-asserted-by": "publisher",
"first-page": "651",
"journal-title": "Diabet. Med. a J. Br. Diabet. Assoc.",
"key": "B98",
"volume": "15",
"year": "1998"
},
{
"DOI": "10.1172/jci.insight.148003",
"article-title": "Ex vivo SARS-CoV-2 infection of human lung reveals heterogeneous host defense and therapeutic responses",
"author": "Schaller",
"doi-asserted-by": "publisher",
"first-page": "e148003",
"journal-title": "JCI insight",
"key": "B99",
"volume": "6",
"year": "2021"
},
{
"DOI": "10.1016/j.diabet.2020.07.006",
"article-title": "Metformin and COVID-19: from cellular mechanisms to reduced mortality",
"author": "Scheen",
"doi-asserted-by": "publisher",
"first-page": "423",
"journal-title": "J. Diabetes Metab.",
"key": "B100",
"volume": "46",
"year": "2020"
},
{
"DOI": "10.1016/j.diabres.2020.108183",
"article-title": "Metformin in COVID-19: a possible role beyond diabetes",
"author": "Sharma",
"doi-asserted-by": "publisher",
"first-page": "108183",
"journal-title": "Diabetes Res. Clin. Pract.",
"key": "B101",
"volume": "164",
"year": "2020"
},
{
"DOI": "10.1016/j.heliyon.2023.e18954",
"article-title": "Metformin promotes cGAS/STING signaling pathway activation by blocking AKT phosphorylation in gastric cancer",
"author": "Shen",
"doi-asserted-by": "publisher",
"first-page": "e18954",
"journal-title": "Heliyon",
"key": "B102",
"volume": "9",
"year": "2023"
},
{
"DOI": "10.3390/microorganisms8091360",
"article-title": "Akkermansia muciniphila is Negatively Correlated with Hemoglobin A1c in Refractory Diabetes",
"author": "Shih",
"doi-asserted-by": "publisher",
"first-page": "1360",
"journal-title": "Microorganisms",
"key": "B103",
"volume": "8",
"year": "2020"
},
{
"DOI": "10.1111/imr.12170",
"article-title": "Regulatory T-cell homeostasis: steady-state maintenance and modulation during inflammation",
"author": "Smigiel",
"doi-asserted-by": "publisher",
"first-page": "40",
"journal-title": "Immunol. Rev.",
"key": "B104",
"volume": "259",
"year": "2014"
},
{
"DOI": "10.1002/jmv.28175",
"article-title": "SARS-CoV-2 ORF3a inhibits cGAS-STING-mediated autophagy flux and antiviral function",
"author": "Su",
"doi-asserted-by": "publisher",
"first-page": "e28175",
"journal-title": "J. Med. Virol.",
"key": "B105",
"volume": "95",
"year": "2023"
},
{
"DOI": "10.1038/s41591-018-0222-4",
"article-title": "Gut microbiota and intestinal FXR mediate the clinical benefits of metformin",
"author": "Sun",
"doi-asserted-by": "publisher",
"first-page": "1919",
"journal-title": "Nat. Med.",
"key": "B106",
"volume": "24",
"year": "2018"
},
{
"DOI": "10.1038/s41418-021-00916-7",
"article-title": "SARS-CoV-2 non-structural protein 6 triggers NLRP3-dependent pyroptosis by targeting ATP6AP1",
"author": "Sun",
"doi-asserted-by": "publisher",
"first-page": "1240",
"journal-title": "Cell death Differ.",
"key": "B107",
"volume": "29",
"year": ""
},
{
"DOI": "10.1186/s12916-021-02212-0",
"article-title": "Gut microbiome alterations and gut barrier dysfunction are associated with host immune homeostasis in COVID-19 patients",
"author": "Sun",
"doi-asserted-by": "publisher",
"first-page": "24",
"journal-title": "BMC Med.",
"key": "B108",
"volume": "20",
"year": ""
},
{
"DOI": "10.3390/microorganisms10091764",
"article-title": "Immunomodulatory Effects of Probiotics on COVID-19 Infection by Targeting the Gut-Lung Axis Microbial Cross-Talk",
"author": "Synodinou",
"doi-asserted-by": "publisher",
"first-page": "1764",
"journal-title": "Microorganisms",
"key": "B109",
"volume": "10",
"year": "2022"
},
{
"DOI": "10.3390/cells8040323",
"article-title": "The Potential Use of Metformin, Dipyridamole, N-Acetylcysteine and Statins as Adjunctive Therapy for Systemic Lupus Erythematosus",
"author": "Tan",
"doi-asserted-by": "publisher",
"first-page": "323",
"journal-title": "Syst. Lupus Erythematosus. Cells.",
"key": "B110",
"volume": "8",
"year": "2019"
},
{
"DOI": "10.1016/j.cmet.2009.08.001",
"article-title": "TGR5-mediated bile acid sensing controls glucose homeostasis",
"author": "Thomas",
"doi-asserted-by": "publisher",
"first-page": "167",
"journal-title": "Cell Metab.",
"key": "B111",
"volume": "10",
"year": "2009"
},
{
"DOI": "10.4049/jimmunol.1801651",
"article-title": "Metformin Inhibits the Type 1 IFN Response in Human CD4(+) T Cells",
"author": "Titov",
"doi-asserted-by": "publisher",
"first-page": "338",
"journal-title": "J. Immunol.",
"key": "B112",
"volume": "203",
"year": "2019"
},
{
"DOI": "10.1038/ncomms8629",
"article-title": "Farnesoid X receptor inhibits glucagon-like peptide-1 production by enteroendocrine L cells",
"author": "Trabelsi",
"doi-asserted-by": "publisher",
"first-page": "7629",
"journal-title": "Nat. Commun.",
"key": "B113",
"volume": "6",
"year": "2015"
},
{
"DOI": "10.1016/j.metabol.2022.155223",
"article-title": "Metformin: is it a drug for all reasons and diseases?",
"author": "Triggle",
"doi-asserted-by": "publisher",
"first-page": "155223",
"journal-title": "Metabolism Clin. Exp.",
"key": "B114",
"volume": "133",
"year": "2022"
},
{
"DOI": "10.18632/oncotarget.20248",
"article-title": "Metformin activates type I interferon signaling against HCV via activation of adenosine monophosphate-activated protein kinase",
"author": "Tsai",
"doi-asserted-by": "publisher",
"first-page": "91928",
"journal-title": "Oncotarget",
"key": "B115",
"volume": "8",
"year": "2017"
},
{
"DOI": "10.1016/j.biopha.2022.113223",
"article-title": "Treatment with metformin glycinate reduces SARS-CoV-2 viral load: an in vitro model and randomized, double-blind, Phase IIb clinical trial",
"author": "Ventura-López",
"doi-asserted-by": "publisher",
"first-page": "113223",
"journal-title": "Biomed. Pharmacother. = Biomedecine Pharmacother.",
"key": "B116",
"volume": "152",
"year": "2022"
},
{
"DOI": "10.1038/s41392-022-00986-0",
"article-title": "Alterations in microbiota of patients with COVID-19: potential mechanisms and therapeutic interventions",
"author": "Wang",
"doi-asserted-by": "publisher",
"first-page": "143",
"journal-title": "Signal Transduct. Target. Ther.",
"key": "B117",
"volume": "7",
"year": "2022"
},
{
"DOI": "10.1007/s00253-020-10689-7",
"article-title": "A comparative study of microbial community and functions of type 2 diabetes mellitus patients with obesity and healthy people",
"author": "Wang",
"doi-asserted-by": "publisher",
"first-page": "7143",
"journal-title": "Appl. Microbiol. Biotechnol.",
"key": "B118",
"volume": "104",
"year": "2020"
},
{
"DOI": "10.1210/jc.2018-02736",
"article-title": "Gastric Emptying in Patients With Well-Controlled Type 2 Diabetes Compared With Young and Older Control Subjects Without Diabetes",
"author": "Watson",
"doi-asserted-by": "publisher",
"first-page": "3311",
"journal-title": "J. Clin. Endocrinol. metabolism",
"key": "B119",
"volume": "104",
"year": "2019"
},
{
"DOI": "10.1016/j.diabet.2022.101359",
"article-title": "Protection by metformin against severe Covid-19: an in-depth mechanistic analysis",
"author": "Wiernsperger",
"doi-asserted-by": "publisher",
"first-page": "101359",
"journal-title": "Diabetes & metabolism",
"key": "B120",
"volume": "48",
"year": "2022"
},
{
"DOI": "10.3109/00498259409043220",
"article-title": "Accumulation of metformin by tissues of the normal and diabetic mouse",
"author": "Wilcock",
"doi-asserted-by": "publisher",
"first-page": "49",
"journal-title": "Xenobiotica; fate foreign Compd. Biol. Syst.",
"key": "B121",
"volume": "24",
"year": "1994"
},
{
"DOI": "10.1038/nm.4345",
"article-title": "Metformin alters the gut microbiome of individuals with treatment-naive type 2 diabetes, contributing to the therapeutic effects of the drug",
"author": "Wu",
"doi-asserted-by": "publisher",
"first-page": "850",
"journal-title": "Nat. Med.",
"key": "B122",
"volume": "23",
"year": ""
},
{
"DOI": "10.1111/dom.12043",
"article-title": "Effects of rectal administration of taurocholic acid on glucagon-like peptide-1 and peptide YY secretion in healthy humans",
"author": "Wu",
"doi-asserted-by": "publisher",
"first-page": "474",
"journal-title": "Diabetes, Obes. metabolism",
"key": "B123",
"volume": "15",
"year": ""
},
{
"DOI": "10.1210/jc.2012-3961",
"article-title": "Effects of taurocholic acid on glycemic, glucagon-like peptide-1, and insulin responses to small intestinal glucose infusion in healthy humans",
"author": "Wu",
"doi-asserted-by": "publisher",
"first-page": "E718",
"journal-title": "J. Clin. Endocrinol. metabolism",
"key": "B124",
"volume": "98",
"year": ""
},
{
"DOI": "10.1111/dom.12812",
"article-title": "Metformin reduces the rate of small intestinal glucose absorption in type 2 diabetes",
"author": "Wu",
"doi-asserted-by": "publisher",
"first-page": "290",
"journal-title": "Diabetes, Obes. metabolism",
"key": "B125",
"volume": "19",
"year": ""
},
{
"DOI": "10.1016/j.immuni.2021.05.004",
"article-title": "Metformin inhibition of mitochondrial ATP and DNA synthesis abrogates NLRP3 inflammasome activation and pulmonary inflammation",
"author": "Xian",
"doi-asserted-by": "publisher",
"first-page": "1463",
"journal-title": "Immunity",
"key": "B126",
"volume": "54",
"year": "2021"
},
{
"DOI": "10.3748/wjg.v28.i47.6689",
"article-title": "Alterations of the gut microbiota in coronavirus disease 2019 and its therapeutic potential",
"author": "Xiang",
"doi-asserted-by": "publisher",
"first-page": "6689",
"journal-title": "World J. gastroenterology",
"key": "B127",
"volume": "28",
"year": "2022"
},
{
"DOI": "10.1016/j.nut.2006.09.002",
"article-title": "Antidiabetic effect of probiotic dahi containing Lactobacillus acidophilus and Lactobacillus casei in high fructose fed rats",
"author": "Yadav",
"doi-asserted-by": "publisher",
"first-page": "62",
"journal-title": "Nutrition",
"key": "B128",
"volume": "23",
"year": "2007"
},
{
"article-title": "Metformin regulates of Th17/treg cell balance and reduces hyperplastic synovium via activating AMPK and inhibiting mTOR in a collagen-induced arthritis rat model",
"author": "Yang",
"first-page": "11479",
"journal-title": "J. Int. J. Clin. Exp. Med.",
"key": "B129",
"volume": "10",
"year": "2017"
},
{
"DOI": "10.1136/gutjnl-2020-323020",
"article-title": "Gut microbiota composition reflects disease severity and dysfunctional immune responses in patients with COVID-19",
"author": "Yeoh",
"doi-asserted-by": "publisher",
"first-page": "698",
"journal-title": "Gut",
"key": "B130",
"volume": "70",
"year": "2021"
},
{
"DOI": "10.1007/s12020-019-02041-5",
"article-title": "Response of gut microbiota in type 2 diabetes to hypoglycemic agents",
"author": "Zhang",
"doi-asserted-by": "publisher",
"first-page": "485",
"journal-title": "Endocrine",
"key": "B131",
"volume": "66",
"year": "2019"
},
{
"DOI": "10.1016/j.ebiom.2019.08.048",
"article-title": "Distinct gut metagenomics and metaproteomics signatures in prediabetics and treatment-naïve type 2 diabetics",
"author": "Zhong",
"doi-asserted-by": "publisher",
"first-page": "373",
"journal-title": "eBioMedicine",
"key": "B132",
"volume": "47",
"year": "2019"
}
],
"reference-count": 132,
"references-count": 132,
"relation": {},
"resource": {
"primary": {
"URL": "https://www.frontiersin.org/articles/10.3389/fmolb.2023.1260633/full"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [
"Biochemistry, Genetics and Molecular Biology (miscellaneous)",
"Molecular Biology",
"Biochemistry"
],
"subtitle": [],
"title": "Unveiling the potential pleiotropic effects of metformin in treating COVID-19: a comprehensive review",
"type": "journal-article",
"update-policy": "http://dx.doi.org/10.3389/crossmark-policy",
"volume": "10"
}