A Retrospective Observational Study on COVID-19 Patients Receiving Treatment with Nirmatrelvir/Ritonavir (PAXLOVID)
et al., Pulmonary Therapy, doi:10.1007/s41030-025-00316-z, Oct 2025
Retrospective 3,011 outpatients in Bahrain showing lower hospitalization with paxlovid treatment. However, the study has significant methodological limitations that likely substantially overestimate paxlovid's benefit:
Contraindicated/sicker patients likely contaminate the control group: authors do not appear to have excluded all contraindicated patients in the control group (and likely do not have the data to do so), therefore patients with severe liver disease, severe CKD, or major drug interactions may have been placed in the "declined" group rather than excluded, systematically loading the control arm with higher-risk patients.
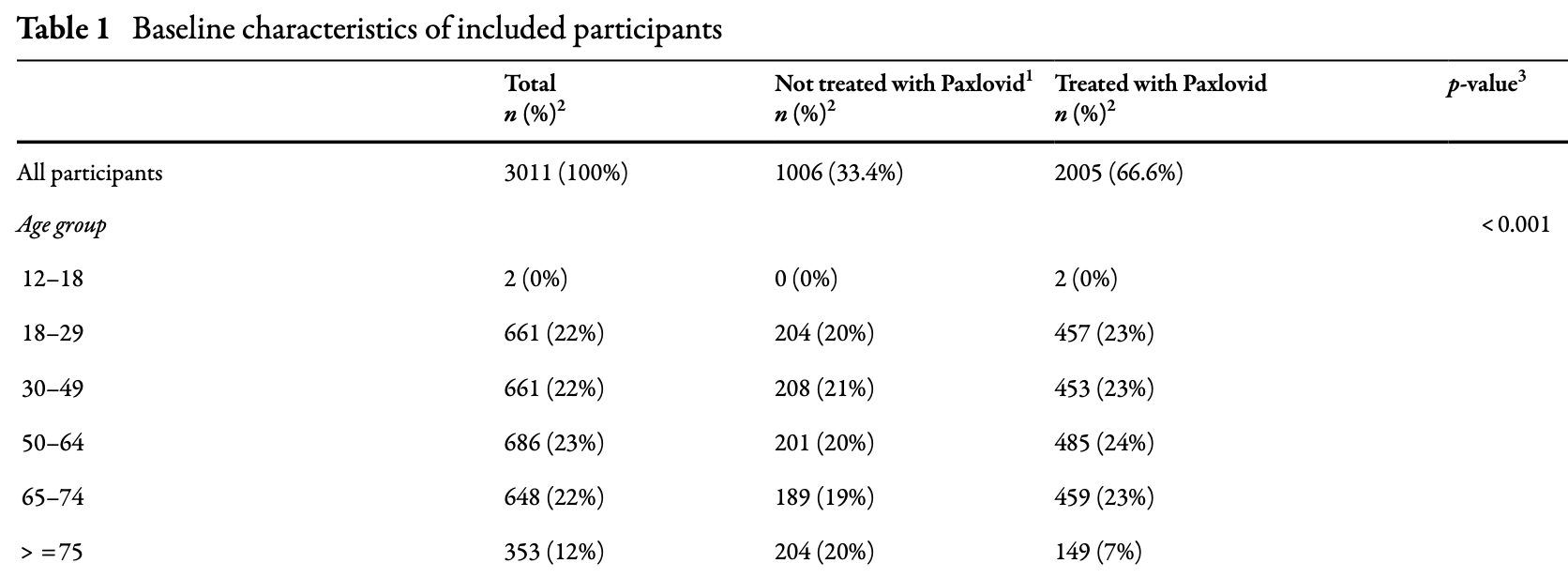
Severe confounding by indication inadequately adjusted: the untreated group had far more cardiopulmonary disease (50% vs 31%), more patients aged ≥75 (20% vs 7%), and more high-risk WHO classification (10% vs 5%), yet the primary analysis adjusted only for a crude 3-level WHO risk variable. The propensity score sensitivity analysis attenuated several findings to non-significance, confirming confounding explains a substantial portion of the effect.
The abstract claims no difference in age, vaccination, or viral load between groups, but Tables 1-2 show highly significant differences (age p<0.001; vaccination p<0.001), and viral load data doesn't appear anywhere in the paper.
Seelction bias: treated patients were younger and more vaccinated. The primary models adjusted only for coarse WHO risk categories, not for age or vaccination.
Undisclosed conflicts of interest: seven of fourteen authors are Pfizer employees, Pfizer funded the study and contributed to design, interpretation, writing, and submission - yet all authors declare "nothing to disclose."
Asymmetric follow-up methods: treated patients received structured day-5 telephone follow-up; untreated patients had only passive EMR review, creating differential outcome ascertainment.
Retrospective registration and long publication delay: the study was registered approximately 2 years after data collection, with a 3-year gap to submission, removing safeguards against outcome switching and raising selective reporting concerns.
Table 2 arithmetic error: the low-risk category total is reported as 327 but the treated + untreated columns sum to only 177, a discrepancy of 150 patients.
Adverse event reporting gaps: adverse events were collected for only 1,427 of 2,005 treated patients (71%) with no explanation, and the 85% dysgeusia rate is an order of magnitude higher than clinical trial literature.
Reasons for non-compliance (n=160+125+117=402) exceed the number of non-compliant patients (n=277) without explanation.
Overall, the confounding issues could plausibly account for the entire observed treatment effect.
Resistance. Variants may be resistant to paxlovid1-9. Use may promote the emergence of variants that weaken host immunity and potentially contribute to long COVID10. Confounding by contraindication. Hoertel et al. find that over 50% of patients that died had a contraindication for the use of Paxlovid11. Retrospective studies that do not exclude contraindicated patients may significantly overestimate efficacy. Black box warning. The FDA notes that severe, life-threatening, and/or fatal adverse reactions due to drug interactions have been reported in patients treated with paxlovid12. Kidney and liver injury. Studies show significantly increased risk of acute kidney injury13 and liver injury14,15. Viral rebound. Studies show significantly increased risk of replication-competent viral rebound16-18.
1.
Zhou et al., Nirmatrelvir-resistant SARS-CoV-2 variants with high fitness in an infectious cell culture system, Science Advances, doi:10.1126/sciadv.add7197.
2.
Moghadasi et al., Rapid resistance profiling of SARS-CoV-2 protease inhibitors, npj Antimicrobials and Resistance, doi:10.1038/s44259-023-00009-0.
3.
Jochmans et al., The Substitutions L50F, E166A, and L167F in SARS-CoV-2 3CLpro Are Selected by a Protease Inhibitor In Vitro and Confer Resistance To Nirmatrelvir, mBio, doi:10.1128/mbio.02815-22.
4.
Lopez et al., SARS-CoV-2 Resistance to Small Molecule Inhibitors, Current Clinical Microbiology Reports, doi:10.1007/s40588-024-00229-6.
5.
Zvornicanin et al., Molecular Mechanisms of Drug Resistance and Compensation in SARS-CoV-2 Main Protease: The Interplay Between E166 and L50, bioRxiv, doi:10.1101/2025.01.24.634813.
6.
Vukovikj et al., Impact of SARS-CoV-2 variant mutations on susceptibility to monoclonal antibodies and antiviral drugs: a non-systematic review, April 2022 to October 2024, Eurosurveillance, doi:10.2807/1560-7917.ES.2025.30.10.2400252.
7.
Deschenes et al., Functional and structural characterization of treatment-emergent nirmatrelvir resistance mutations at low frequencies in the main protease (Mpro) reveals a unique evolutionary route for SARS-CoV-2 to gain resistance, The Journal of Infectious Diseases, doi:10.1093/infdis/jiaf294.
8.
Zhou (B) et al., SARS-CoV-2 Mpro inhibitor ensitrelvir: asymmetrical cross-resistance with nirmatrelvir and emerging resistance hotspots, Emerging Microbes & Infections, doi:10.1080/22221751.2025.2552716.
9.
Chen et al., SARS-CoV-2 3CLpro mutations T21I and E166A confer differential resistance to simnotrelvir, bofutrelvir, and ensitrelvir, Journal of Virology, doi:10.1128/jvi.02223-25.
10.
Thomas et al., Nirmatrelvir-Resistant Mutations in SARS-CoV-2 Mpro Enhance Host Immune Evasion via Cleavage of NF-κB Essential Modulator, bioRxiv, doi:10.1101/2024.10.18.619137.
11.
Hoertel et al., Prevalence of Contraindications to Nirmatrelvir-Ritonavir Among Hospitalized Patients With COVID-19 at Risk for Progression to Severe Disease, JAMA Network Open, doi:10.1001/jamanetworkopen.2022.42140.
12.
FDA, Fact sheet for healthcare providers: emergency use authorization for paxlovid, www.fda.gov/media/155050/download.
13.
Kamo et al., Association of Antiviral Drugs for the Treatment of COVID-19 With Acute Renal Failure, In Vivo, doi:10.21873/invivo.13637.
14.
Wang et al., Development and validation of a nomogram to assess the occurrence of liver dysfunction in patients with COVID-19 pneumonia in the ICU, BMC Infectious Diseases, doi:10.1186/s12879-025-10684-1.
15.
Siby et al., Temporal Trends in Serious Adverse Events Associated with Oral Antivirals During the COVID-19 Pandemic: Insights from the FAERS Database (2020–2023), Open Forum Infectious Diseases, doi:10.1093/ofid/ofaf695.1825.
16.
Edelstein et al., SARS-CoV-2 virologic rebound with nirmatrelvir-ritonavir therapy, medRxiv, doi:10.1101/2023.06.23.23288598.
Murad et al., 6 Oct 2025, retrospective, Bahrain, peer-reviewed, 14 authors, study period February 2020 - September 2021.
Contact: mmurad@rcsi.com, satkin@rcsi-mub.com, psulaiman@rcsi-mub.com, dr.abehzad@gmail.com, dr.aamalmohamed@gmail.com, florencelefebvredhellencourt@gmail.com, lu.wang4@pfizer.com, moe.kyaw@pfizer.com, jean.joury@pfizer.com, mohamed.abdelaziz@pfizer.com, hammam.haridy@pfizer.com, julia.spinardi@pfizer.com, fionaboland@rcsi.ie, manaf.alqahtani@rms.bh.
A Retrospective Observational Study on COVID-19 Patients Receiving Treatment with Nirmatrelvir/Ritonavir (PAXLOVID)
Pulmonary Therapy, doi:10.1007/s41030-025-00316-z
Introduction: This study was undertaken in COVID-19 patients treated with and without Paxlovid™ (Paxlovid) in an outpatient setting to determine hospitalization from the community. Methods: This retrospective secondary data observational cohort study was conducted between February and November 2022. All patients diagnosed by polymerase chain reaction for COVID-19 and at risk of COVID-19 disease progression were offered Paxlovid in the outpatient setting. Potential associations between Paxlovid use and likelihood of hospitalization, antibiotic use, and other clinical outcomes were explored using regression models as appropriate
Funding. This study was conducted as a collaboration between RCSI Medical University of Bahrain and Pfizer. The study was funded by Pfizer grant number 76181715. The funder contributed to the study design, to the interpretation of the data, to the writing of the manuscript, and to the decision to submit the manuscript for publication and payment of rapid service fees. Representatives of Pfizer were not involved in the collection, management, and analysis of data. Data Availability. The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request. Upon request, and subject to review, the study sponsor, RCSI Medical University of Bahrain, will provide data that support the findings of this study. Subject to certain criteria, conditions, and exceptions, RCSI Medical University of Bahrain may also provide access to the related individual de-identified participant data. Contact RCSI Medical University of Bahrain for more information.
Declarations
Ethics approval. The study was conducted according to the guidelines and approved by the National COVID-19 Taskforce ethical Pulm Ther approval, obtained in June 2023 for this retrospective study (CRT-COVID2022-160). A waiver which aligns with FDA guidelines that permits us to use the participants' information for this study, including for publication purposes, was obtained from the National COVID-19 Taskforce ethics committee, allowing us to proceed without..
References
Al-Qahtani, Alali, Abdulrahman, Alsayyad, Otoom et al., The prevalence of asymptomatic and symptomatic COVID-19 in a cohort of quarantined subjects, Int J Infect Dis
Al-Shami, Ahmed, Alzoubi, Motivators and barriers towards clinical research participation: a population-based survey from an Arab MENA country, PLoS One
Bernal, Da Silva, Musungaie, Kovalchuk, Gonzalez et al., Molnupiravir for oral treatment of covid-19 in nonhospitalized patients, New Engl J Med
Cai, Yan, Liu, Li, Ding et al., Paxlovid for hospitalized COVID-19 patients with chronic kidney disease, Antiviral Res
Cdc, Centers for disease control and prevention
Choi, Wan, Wong, Chan, Chu et al., Comparative effectiveness of combination therapy with nirmatrelvir-ritonavir and remdesivir versus monotherapy with remdesivir or nirmatrelvir-ritonavir in patients hospitalised with COVID-19: a target trial emulation study, Lancet Infect Dis
Docherty, Harrison, Green, Hardwick, Pius et al., Features of 20 133 UK patients in hospital with covid-19 using the ISARIC WHO Clinical Characterisation Protocol: prospective observational cohort study, BMJ
Gavaghan, Bergroth, Alexander-Parrish, Yang, Draica, Effectiveness of nirmatrelvir-ritonavir for the prevention of COVID-19-related hospitalization and mortality: a systematic literature review, Am J Ther
Gupta, Gonzalez-Rojas, Juarez, Casal, Moya et al., Early treatment for Covid-19 with SARS-CoV-2 neutralizing antibody sotrovimab, N Engl J Med
Hammond, Leister-Tebbe, Gardner, Abreu, Wisemandle, Oral nirmatrelvir for high-risk, nonhospitalized adults with Covid-19, N Engl J Med
Healey, Sheikh, Daines, Vasileiou, Symptoms and signs of long COVID: a rapid review and meta-analysis, J Glob Health
Iketani, Mohri, Culbertson, Hong, Duan et al., Multiple pathways for SARS-CoV-2 resistance to Nirmatrelvir, Nature
Marzi, Vakil, Bahmanyar, Zarenezhad, Paxlovid: mechanism of action, synthesis, and in silico study, Biomed Res Int
Matsushita, Ding, Kou, Hu, Chen et al., The relationship of COVID-19 severity with cardiovascular disease and its traditional risk factors: a systematic review and meta-analysis, Glob Heart
Mutair, Layqah, Alhassan, Alkhalifah, Almossabeh et al., Estimated cost of treating hospitalized COVID-19 patients in Saudi Arabia, Sci Rep
Najjar-Debbiny, Gronich, Weber, Khoury, Amar et al., Effectiveness of Paxlovid in reducing severe coronavirus disease 2019 and mortality in high-risk patients, Clin Infect Dis
Nguyen, Chinn, Nahmias, Yuen, Kirby et al., Outcomes and mortality among adults hospitalized with COVID-19 at US medical centers, JAMA Netw Open
Nih, The COVID-19 Treatment Guideline Panel's Statement on Therapies for High-Risk, Nonhospitalized Patients with Mild to Moderate COVID-19, National Institute of Health
Petrilli, Jones, Yang, Rajagopalan, Donnell et al., Factors associated with hospital admission and critical illness among 5279 people with coronavirus disease 2019 in New York City: prospective cohort study, BMJ
Shah, Joyce, Plumb, Sahakian, Feldstein et al., Paxlovid associated with decreased hospitalization rate among adults with COVID-19 -United States, April-September 2022, MMWR Morb Mortal Wkly Rep
Shah, Joyce, Plumb, Sahakian, Feldstein et al., Paxlovid associated with decreased hospitalization rate among adults with COVID-19 -United States, April-September 2022, MMWR Morb Mortal Wkly Rep
Souza, Carrasco, Rojas-Cortés, Barbosa, Bambirra et al., Effectiveness of nirmatrelvir-ritonavir for the treatment of Pulm Ther patients with mild to moderate COVID-19 and at high risk of hospitalization: systematic review and meta-analyses of observational studies, PLoS One
Vangeel, Chiu, Jonghe, Maes, Slechten et al., Remdesivir, molnupiravir and nirmatrelvir remain active against SARS-CoV-2 Omicron and other variants of concern, Antivir Res
Wang, Berger, Davis, Kaelber, Volkow et al., COVID-19 rebound after Paxlovid and Molnupiravir during January
Zaidi, Iskander, Ahmed, Jaber, Paz et al., A rare case of Paxlovid-induced pancreatitis, Cureus, doi:10.7759/cureus.36528
Zhang, Li, Zhou, Liu, Wang et al., Cost-effectiveness of Paxlovid in reducing severe COVID-19 and mortality in China, Front Public Health, doi:10.3389/fpubh.2023.1174879
Zhu, Yin, Chen, Liu, Yang, Appraisal of evidence reliability and applicability of Paxlovid as treatment for SARS-COV-2 infection: a systematic review, Rev Med Virol, doi:10.1002/rmv.2476
DOI record:
{
"DOI": "10.1007/s41030-025-00316-z",
"ISSN": [
"2364-1754",
"2364-1746"
],
"URL": "http://dx.doi.org/10.1007/s41030-025-00316-z",
"alternative-id": [
"316"
],
"assertion": [
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "Received",
"name": "received",
"order": 1,
"value": "27 July 2025"
},
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "Accepted",
"name": "accepted",
"order": 2,
"value": "2 September 2025"
},
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "First Online",
"name": "first_online",
"order": 3,
"value": "6 October 2025"
},
{
"group": {
"label": "Declarations",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 1
},
{
"group": {
"label": "Conflict of Interest",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 2,
"value": "Mariam M. Murad, Stephen L. Atkin, Pearl W. Wasif, Alwaleed Abdulaziz Behzad, Aamal M. J. Abdulla Husain, Florence Lefebvre d’Hellencourt, Jean Joury, Mohamed Abdel Aziz, Hammam Haridy, Julia Spinardi, Lu Wang, Fiona Boland, Moe H. Kyaw, and Manaf Al-Qahtani have nothing to disclose."
},
{
"group": {
"label": "Ethics approval",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 3,
"value": "The study was conducted according to the guidelines and approved by the National COVID-19 Taskforce ethical approval, obtained in June 2023 for this retrospective study (CRT-COVID2022-160). A waiver which aligns with FDA guidelines that permits us to use the participants’ information for this study, including for publication purposes, was obtained from the National COVID-19 Taskforce ethics committee, allowing us to proceed without additional consent."
}
],
"author": [
{
"ORCID": "https://orcid.org/0009-0008-8006-996X",
"affiliation": [],
"authenticated-orcid": false,
"family": "Murad",
"given": "Mariam M.",
"sequence": "first"
},
{
"affiliation": [],
"family": "Atkin",
"given": "Stephen L.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Wasif",
"given": "Pearl W.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Behzad",
"given": "Alwaleed Abdulaziz",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Husain",
"given": "Aamal M. J. Abdulla",
"sequence": "additional"
},
{
"affiliation": [],
"family": "d’Hellencourt",
"given": "Florence Lefebvre",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Joury",
"given": "Jean",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Aziz",
"given": "Mohamed Abdel",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Haridy",
"given": "Hammam",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Spinardi",
"given": "Julia",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Wang",
"given": "Lu",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Boland",
"given": "Fiona",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Kyaw",
"given": "Moe H.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Al-Qahtani",
"given": "Manaf",
"sequence": "additional"
}
],
"container-title": "Pulmonary Therapy",
"container-title-short": "Pulm Ther",
"content-domain": {
"crossmark-restriction": false,
"domain": [
"link.springer.com"
]
},
"created": {
"date-parts": [
[
2025,
10,
6
]
],
"date-time": "2025-10-06T14:10:05Z",
"timestamp": 1759759805000
},
"deposited": {
"date-parts": [
[
2025,
10,
6
]
],
"date-time": "2025-10-06T14:10:10Z",
"timestamp": 1759759810000
},
"funder": [
{
"DOI": "10.13039/100004319",
"award": [
"76181715"
],
"doi-asserted-by": "publisher",
"id": [
{
"asserted-by": "publisher",
"id": "10.13039/100004319",
"id-type": "DOI"
}
],
"name": "Pfizer"
}
],
"indexed": {
"date-parts": [
[
2025,
10,
6
]
],
"date-time": "2025-10-06T14:43:59Z",
"timestamp": 1759761839613,
"version": "build-2065373602"
},
"is-referenced-by-count": 0,
"issued": {
"date-parts": [
[
2025,
10,
6
]
]
},
"language": "en",
"license": [
{
"URL": "https://creativecommons.org/licenses/by-nc/4.0",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2025,
10,
6
]
],
"date-time": "2025-10-06T00:00:00Z",
"timestamp": 1759708800000
}
},
{
"URL": "https://creativecommons.org/licenses/by-nc/4.0",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2025,
10,
6
]
],
"date-time": "2025-10-06T00:00:00Z",
"timestamp": 1759708800000
}
}
],
"link": [
{
"URL": "https://link.springer.com/content/pdf/10.1007/s41030-025-00316-z.pdf",
"content-type": "application/pdf",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://link.springer.com/article/10.1007/s41030-025-00316-z/fulltext.html",
"content-type": "text/html",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://link.springer.com/content/pdf/10.1007/s41030-025-00316-z.pdf",
"content-type": "application/pdf",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "297",
"original-title": [],
"prefix": "10.1007",
"published": {
"date-parts": [
[
2025,
10,
6
]
]
},
"published-online": {
"date-parts": [
[
2025,
10,
6
]
]
},
"publisher": "Springer Science and Business Media LLC",
"reference": [
{
"key": "316_CR1",
"unstructured": ": World Health Organization; 2020 [Available from: https://www.euro.who.int/en/healthtopics/health-emergencies/coronavirus-covid19/news/news/2020/01/2019-ncovoutbreak-is-an-emergency-of-international-concern."
},
{
"key": "316_CR2",
"unstructured": "WHO Situation Report 51 Coronavirus disease 2019 (COVID-19) : World Health Organization; 2019 [Available from: https://www.euro.who.int/en/healthtopics/health-emergencies/coronavirus-covid19/news/news/2020/01/2019-ncovoutbreak-is-an-emergency-of-international-concern."
},
{
"DOI": "10.1016/j.ijid.2020.10.091",
"author": "M Al-Qahtani",
"doi-asserted-by": "publisher",
"first-page": "285",
"journal-title": "Int J Infect Dis",
"key": "316_CR3",
"unstructured": "Al-Qahtani M, AlAli S, AbdulRahman A, Salman Alsayyad A, Otoom S, Atkin SL. The prevalence of asymptomatic and symptomatic COVID-19 in a cohort of quarantined subjects. Int J Infect Dis. 2021;102:285–8.",
"volume": "102",
"year": "2021"
},
{
"DOI": "10.1136/bmj.m1985",
"author": "AB Docherty",
"doi-asserted-by": "publisher",
"journal-title": "BMJ",
"key": "316_CR4",
"unstructured": "Docherty AB, Harrison EM, Green CA, Hardwick HE, Pius R, Norman L, et al. Features of 20 133 UK patients in hospital with covid-19 using the ISARIC WHO Clinical Characterisation Protocol: prospective observational cohort study. BMJ. 2020;369:m1985.",
"volume": "369",
"year": "2020"
},
{
"key": "316_CR5",
"unstructured": "Estimating excess mortality due to the COVID-19 pandemic: a systematic analysis of COVID-19-related mortality, 2020–21. Lancet. 2022;399 (10334):1513–36."
},
{
"key": "316_CR6",
"unstructured": "CDC. Estimated disease burden of COVID-19. Centers for disease control and prevention. Data and Surveillance Web site: Centers for Disease Control and Prevention.; 2020 [updated october 2 2021]. Available from: Web site. https://www.cdc.gov/coronavirus/2019-ncov/cases-updates/burden."
},
{
"key": "316_CR7",
"unstructured": "NIH. COVID-19 Treatment Guidelines National Institutes of Health; 2021 [updated December 16 2021]. Available from: https://www.covid19treatmentguidelines.nih.gov/outpatient-management."
},
{
"key": "316_CR8",
"unstructured": "NIH. The COVID-19 Treatment Guideline Panel’s Statement on Therapies for High-Risk, Nonhospitalized Patients with Mild to Moderate COVID-19: National Institute of Health; 2021 [updated Updated December 30, 2021]. Available from: https://www.covid19treatmentguidelines.nih.gov/therapies/statement-on-therapies-for-high-risk-nonhospitalized-patients."
},
{
"DOI": "10.15585/mmwr.mm7148e2",
"author": "MM Shah",
"doi-asserted-by": "publisher",
"first-page": "1531",
"issue": "48",
"journal-title": "MMWR Morb Mortal Wkly Rep",
"key": "316_CR9",
"unstructured": "Shah MM, Joyce B, Plumb ID, Sahakian S, Feldstein LR, Barkley E, et al. Paxlovid associated with decreased hospitalization rate among adults with COVID-19 — United States, April–September 2022. MMWR Morb Mortal Wkly Rep. 2022;71(48):1531–7.",
"volume": "71",
"year": "2022"
},
{
"DOI": "10.1155/2022/7341493",
"author": "M Marzi",
"doi-asserted-by": "publisher",
"first-page": "1",
"journal-title": "Biomed Res Int",
"key": "316_CR10",
"unstructured": "Marzi M, Vakil MK, Bahmanyar M, Zarenezhad E. Paxlovid: mechanism of action, synthesis, and in silico study. Biomed Res Int. 2022;2022:1–16.",
"volume": "2022",
"year": "2022"
},
{
"DOI": "10.1056/NEJMoa2107934",
"author": "A Gupta",
"doi-asserted-by": "publisher",
"first-page": "1941",
"issue": "21",
"journal-title": "N Engl J Med",
"key": "316_CR11",
"unstructured": "Gupta A, Gonzalez-Rojas Y, Juarez E, Crespo Casal M, Moya J, Falci DR, et al. Early treatment for Covid-19 with SARS-CoV-2 neutralizing antibody sotrovimab. N Engl J Med. 2021;385(21):1941–50.",
"volume": "385",
"year": "2021"
},
{
"DOI": "10.1056/NEJMoa2116044",
"doi-asserted-by": "crossref",
"key": "316_CR12",
"unstructured": "Jayk Bernal A, Gomes da Silva MM, Musungaie DB, Kovalchuk E, Gonzalez A, Delos Reyes V, et al. Molnupiravir for oral treatment of covid-19 in nonhospitalized patients. New Engl J Med. 2022;386 (6):509–20."
},
{
"DOI": "10.1016/j.antiviral.2022.105252",
"author": "L Vangeel",
"doi-asserted-by": "publisher",
"journal-title": "Antivir Res",
"key": "316_CR13",
"unstructured": "Vangeel L, Chiu W, De Jonghe S, Maes P, Slechten B, Raymenants J, et al. Remdesivir, molnupiravir and nirmatrelvir remain active against SARS-CoV-2 Omicron and other variants of concern. Antivir Res. 2022;198:105252.",
"volume": "198",
"year": "2022"
},
{
"DOI": "10.1371/journal.pone.0284006",
"author": "KM Souza",
"doi-asserted-by": "publisher",
"issue": "10",
"journal-title": "PLoS One",
"key": "316_CR14",
"unstructured": "Souza KM, Carrasco G, Rojas-Cortés R, Michel Barbosa M, Bambirra EHF, Castro JL, et al. Effectiveness of nirmatrelvir-ritonavir for the treatment of patients with mild to moderate COVID-19 and at high risk of hospitalization: systematic review and meta-analyses of observational studies. PLoS One. 2023;18(10):e0284006.",
"volume": "18",
"year": "2023"
},
{
"DOI": "10.1097/MJT.0000000000001744",
"author": "AS Cha-Silva",
"doi-asserted-by": "publisher",
"first-page": "e246",
"issue": "3",
"journal-title": "Am J Ther",
"key": "316_CR15",
"unstructured": "Cha-Silva AS, Gavaghan MB, Bergroth T, Alexander-Parrish R, Yang J, Draica F, et al. Effectiveness of nirmatrelvir-ritonavir for the prevention of COVID-19-related hospitalization and mortality: a systematic literature review. Am J Ther. 2024;31(3):e246–57.",
"volume": "31",
"year": "2024"
},
{
"DOI": "10.1136/bmj.m1966",
"author": "CM Petrilli",
"doi-asserted-by": "publisher",
"journal-title": "BMJ",
"key": "316_CR16",
"unstructured": "Petrilli CM, Jones SA, Yang J, Rajagopalan H, O’Donnell L, Chernyak Y, et al. Factors associated with hospital admission and critical illness among 5279 people with coronavirus disease 2019 in New York City: prospective cohort study. BMJ. 2020;369:m1966.",
"volume": "369",
"year": "2020"
},
{
"DOI": "10.1001/jamanetworkopen.2021.0417",
"author": "NT Nguyen",
"doi-asserted-by": "publisher",
"issue": "3",
"journal-title": "JAMA Netw Open",
"key": "316_CR17",
"unstructured": "Nguyen NT, Chinn J, Nahmias J, Yuen S, Kirby KA, Hohmann S, et al. Outcomes and mortality among adults hospitalized with COVID-19 at US medical centers. JAMA Netw Open. 2021;4(3):e210417.",
"volume": "4",
"year": "2021"
},
{
"DOI": "10.1038/s41598-022-26042-z",
"author": "A Al Mutair",
"doi-asserted-by": "publisher",
"first-page": "21487",
"issue": "1",
"journal-title": "Sci Rep",
"key": "316_CR18",
"unstructured": "Al Mutair A, Layqah L, Alhassan B, Alkhalifah S, Almossabeh M, AlSaleh T, et al. Estimated cost of treating hospitalized COVID-19 patients in Saudi Arabia. Sci Rep. 2022;12(1):21487.",
"volume": "12",
"year": "2022"
},
{
"DOI": "10.1093/cid/ciac443",
"author": "R Najjar-Debbiny",
"doi-asserted-by": "publisher",
"first-page": "e342",
"issue": "3",
"journal-title": "Clin Infect Dis",
"key": "316_CR19",
"unstructured": "Najjar-Debbiny R, Gronich N, Weber G, Khoury J, Amar M, Stein N, et al. Effectiveness of Paxlovid in reducing severe coronavirus disease 2019 and mortality in high-risk patients. Clin Infect Dis. 2023;76(3):e342–9.",
"volume": "76",
"year": "2023"
},
{
"DOI": "10.5334/gh.814",
"author": "K Matsushita",
"doi-asserted-by": "publisher",
"first-page": "64",
"issue": "1",
"journal-title": "Glob Heart",
"key": "316_CR20",
"unstructured": "Matsushita K, Ding N, Kou M, Hu X, Chen M, Gao Y, et al. The relationship of COVID-19 severity with cardiovascular disease and its traditional risk factors: a systematic review and meta-analysis. Glob Heart. 2020;15(1):64.",
"volume": "15",
"year": "2020"
},
{
"key": "316_CR21",
"unstructured": "Site: CfDCaPDSW. Underlying conditions and the higher risk for severe COVID-19: Centers for Disease Control and Prevention; 2024. [Available from: https://www.cdc.gov/covid/hcp/clinical-care/underlying-conditions.html."
},
{
"DOI": "10.15585/mmwr.mm7148e2",
"author": "MM Shah",
"doi-asserted-by": "publisher",
"first-page": "1531",
"issue": "48",
"journal-title": "MMWR Morb Mortal Wkly Rep",
"key": "316_CR22",
"unstructured": "Shah MM, Joyce B, Plumb ID, Sahakian S, Feldstein LR, Barkley E, et al. Paxlovid associated with decreased hospitalization rate among adults with COVID-19 - United States, April-September 2022. MMWR Morb Mortal Wkly Rep. 2022;71(48):1531–7.",
"volume": "71",
"year": "2022"
},
{
"DOI": "10.1016/S1473-3099(24)00353-0",
"author": "MH Choi",
"doi-asserted-by": "publisher",
"first-page": "1213",
"issue": "11",
"journal-title": "Lancet Infect Dis",
"key": "316_CR23",
"unstructured": "Choi MH, Wan EYF, Wong ICK, Chan EWY, Chu WM, Tam AR, et al. Comparative effectiveness of combination therapy with nirmatrelvir-ritonavir and remdesivir versus monotherapy with remdesivir or nirmatrelvir-ritonavir in patients hospitalised with COVID-19: a target trial emulation study. Lancet Infect Dis. 2024;24(11):1213–24.",
"volume": "24",
"year": "2024"
},
{
"DOI": "10.7189/jogh.12.05014",
"author": "Q Healey",
"doi-asserted-by": "publisher",
"first-page": "05014",
"journal-title": "J Glob Health",
"key": "316_CR24",
"unstructured": "Healey Q, Sheikh A, Daines L, Vasileiou E. Symptoms and signs of long COVID: a rapid review and meta-analysis. J Glob Health. 2022;12:05014.",
"volume": "12",
"year": "2022"
},
{
"DOI": "10.1056/NEJMoa2118542",
"author": "J Hammond",
"doi-asserted-by": "publisher",
"first-page": "1397",
"issue": "15",
"journal-title": "N Engl J Med",
"key": "316_CR25",
"unstructured": "Hammond J, Leister-Tebbe H, Gardner A, Abreu P, Bao W, Wisemandle W, et al. Oral nirmatrelvir for high-risk, nonhospitalized adults with Covid-19. N Engl J Med. 2022;386(15):1397–408.",
"volume": "386",
"year": "2022"
},
{
"DOI": "10.7759/cureus.36528",
"author": "SMH Zaidi",
"doi-asserted-by": "publisher",
"journal-title": "Cureus",
"key": "316_CR26",
"unstructured": "Zaidi SMH, Iskander PA, Ahmed K, Jaber F, Paz M, Khan A, et al. A rare case of Paxlovid-induced pancreatitis. Cureus. 2023. https://doi.org/10.7759/cureus.36528.",
"year": "2023"
},
{
"DOI": "10.1016/j.antiviral.2023.105659",
"author": "H Cai",
"doi-asserted-by": "publisher",
"journal-title": "Antiviral Res",
"key": "316_CR27",
"unstructured": "Cai H, Yan J, Liu S, Li P, Ding L, Zhan Y, et al. Paxlovid for hospitalized COVID-19 patients with chronic kidney disease. Antiviral Res. 2023;216:105659.",
"volume": "216",
"year": "2023"
},
{
"DOI": "10.1038/s41586-022-05514-2",
"author": "S Iketani",
"doi-asserted-by": "publisher",
"first-page": "558",
"issue": "7944",
"journal-title": "Nature",
"key": "316_CR28",
"unstructured": "Iketani S, Mohri H, Culbertson B, Hong SJ, Duan Y, Luck MI, et al. Multiple pathways for SARS-CoV-2 resistance to Nirmatrelvir. Nature. 2022;613(7944):558–64.",
"volume": "613",
"year": "2022"
},
{
"DOI": "10.1101/2022.06.21.22276724",
"doi-asserted-by": "crossref",
"key": "316_CR29",
"unstructured": "Wang L, Berger NA, Davis PB, Kaelber DC, Volkow ND, Xu R. COVID-19 rebound after Paxlovid and Molnupiravir during January-June 2022. medRxiv: the preprint server for health sciences. 2022."
},
{
"DOI": "10.3389/fpubh.2023.1174879",
"author": "W Zhang",
"doi-asserted-by": "publisher",
"journal-title": "Front Public Health",
"key": "316_CR30",
"unstructured": "Zhang W, Li L, Zhou Z, Liu Q, Wang G, Liu D. Cost-effectiveness of Paxlovid in reducing severe COVID-19 and mortality in China. Front Public Health. 2023. https://doi.org/10.3389/fpubh.2023.1174879.",
"year": "2023"
},
{
"DOI": "10.1002/rmv.2476",
"author": "C Zhu",
"doi-asserted-by": "publisher",
"journal-title": "Rev Med Virol",
"key": "316_CR31",
"unstructured": "Zhu C, Yin J, Chen X, Liu M, Yang S. Appraisal of evidence reliability and applicability of Paxlovid as treatment for SARS-COV-2 infection: a systematic review. Rev Med Virol. 2023. https://doi.org/10.1002/rmv.2476.",
"year": "2023"
},
{
"DOI": "10.1371/journal.pone.0270300",
"author": "KM Al-Shami",
"doi-asserted-by": "publisher",
"issue": "6",
"journal-title": "PLoS One",
"key": "316_CR32",
"unstructured": "Al-Shami KM, Ahmed WS, Alzoubi KH. Motivators and barriers towards clinical research participation: a population-based survey from an Arab MENA country. PLoS One. 2022;17(6):e0270300.",
"volume": "17",
"year": "2022"
}
],
"reference-count": 32,
"references-count": 32,
"relation": {},
"resource": {
"primary": {
"URL": "https://link.springer.com/10.1007/s41030-025-00316-z"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "A Retrospective Observational Study on COVID-19 Patients Receiving Treatment with Nirmatrelvir/Ritonavir (PAXLOVID)",
"type": "journal-article",
"update-policy": "https://doi.org/10.1007/springer_crossmark_policy"
}