A Randomised, Single Blinded Study Investigating the Role of ' Black Elderberry (Sambucus Nigra) (Sambucol®) in the Treatment, Progression and Reduction of Symptoms in Participants With Coronavirus 19
et al., NCT05489770, BERRY, NCT05489770, May 2022
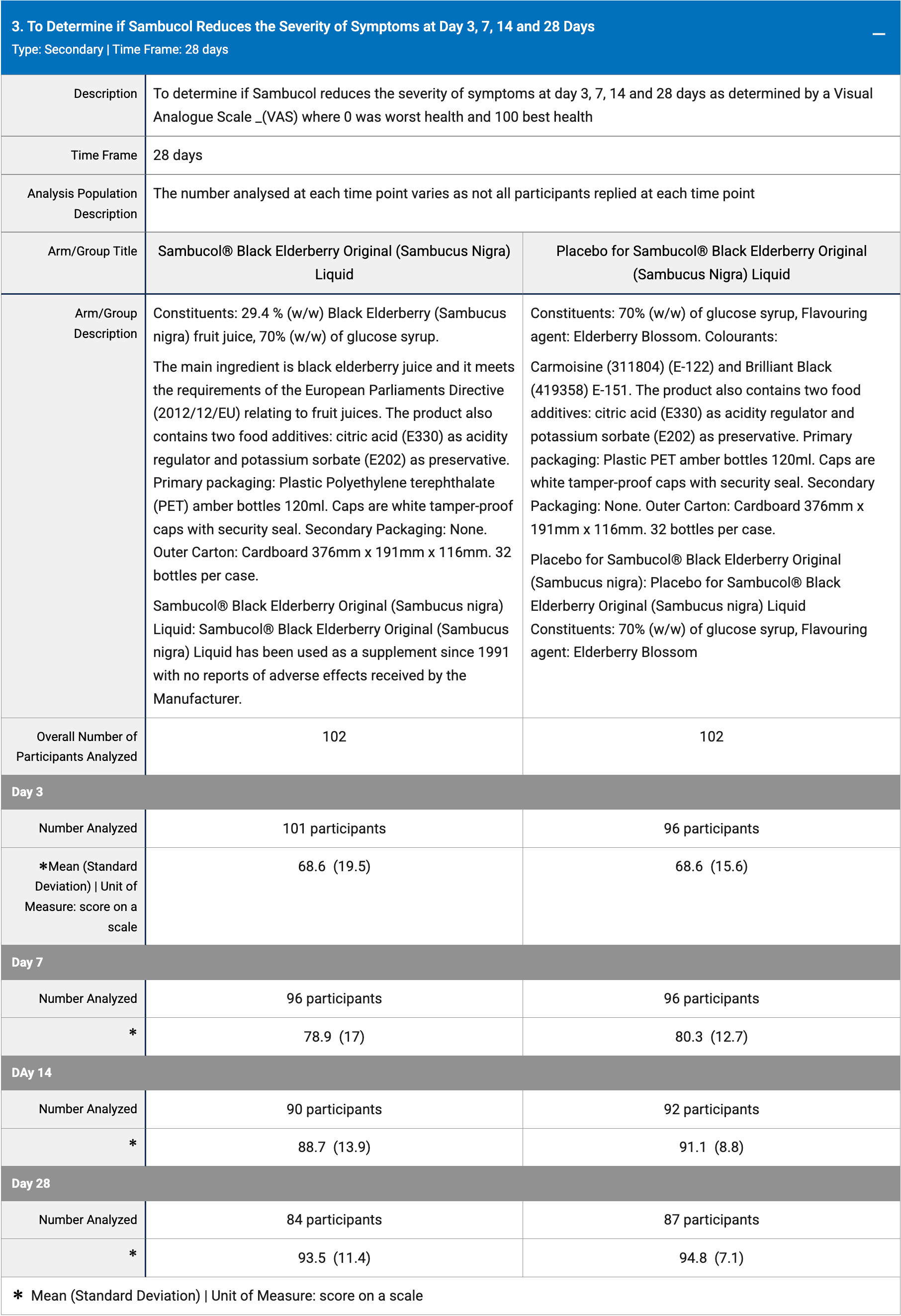
RCT 204 COVID-19 patients showing no significant differences with Sambucol® black elderberry (Sambucus nigra) treatment. Treatment delay is unknown.
Standard of Care (SOC) for COVID-19 in the study country,
the United Kingdom, is very poor with very low average efficacy for approved treatments1.
The United Kingdom focused on expensive high-profit treatments, approving only one low-cost early treatment, which required a prescription and had limited adoption. The high-cost prescription treatment strategy reduces the probability of early treatment due to access and cost barriers, and eliminates complementary and synergistic benefits seen with many low-cost treatments.
|
risk of no recovery, 1.4% higher, RR 1.01, p = 0.37, treatment mean 93.5 (±11.4) n=84, control mean 94.8 (±7.1) n=87, day 28.
|
|
risk of no recovery, 2.7% higher, RR 1.03, p = 0.16, treatment mean 88.7 (±13.9) n=90, control mean 91.1 (±8.8) n=92, day 14.
|
|
risk of no recovery, 2.6% higher, RR 1.03, p = 0.22, treatment mean 84.3 (±13.8) n=90, control mean 86.5 (±9.9) n=93, day 10.
|
|
risk of no recovery, 1.8% higher, RR 1.02, p = 0.52, treatment mean 78.9 (±17.0) n=96, control mean 80.3 (±12.7) n=96, day 7.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Evans et al., 16 May 2022, Double Blind Randomized Controlled Trial, placebo-controlled, United Kingdom, preprint, 1 author, trial NCT05489770 (history) (BERRY).
Contact: jane.povey2@nhs.net.
Elderberry (Sambucus nigra) is an orally administered botanical dietary supplement proposed as potentially beneficial for SARS-CoV-2 via antiviral and immunomodulatory mechanisms.