Antiviral Activity of Repurposing Ivermectin against a Panel of 30 Clinical SARS-CoV-2 Strains Belonging to 14 Variants
et al., Pharmaceuticals, doi:10.3390/ph15040445, Apr 2022
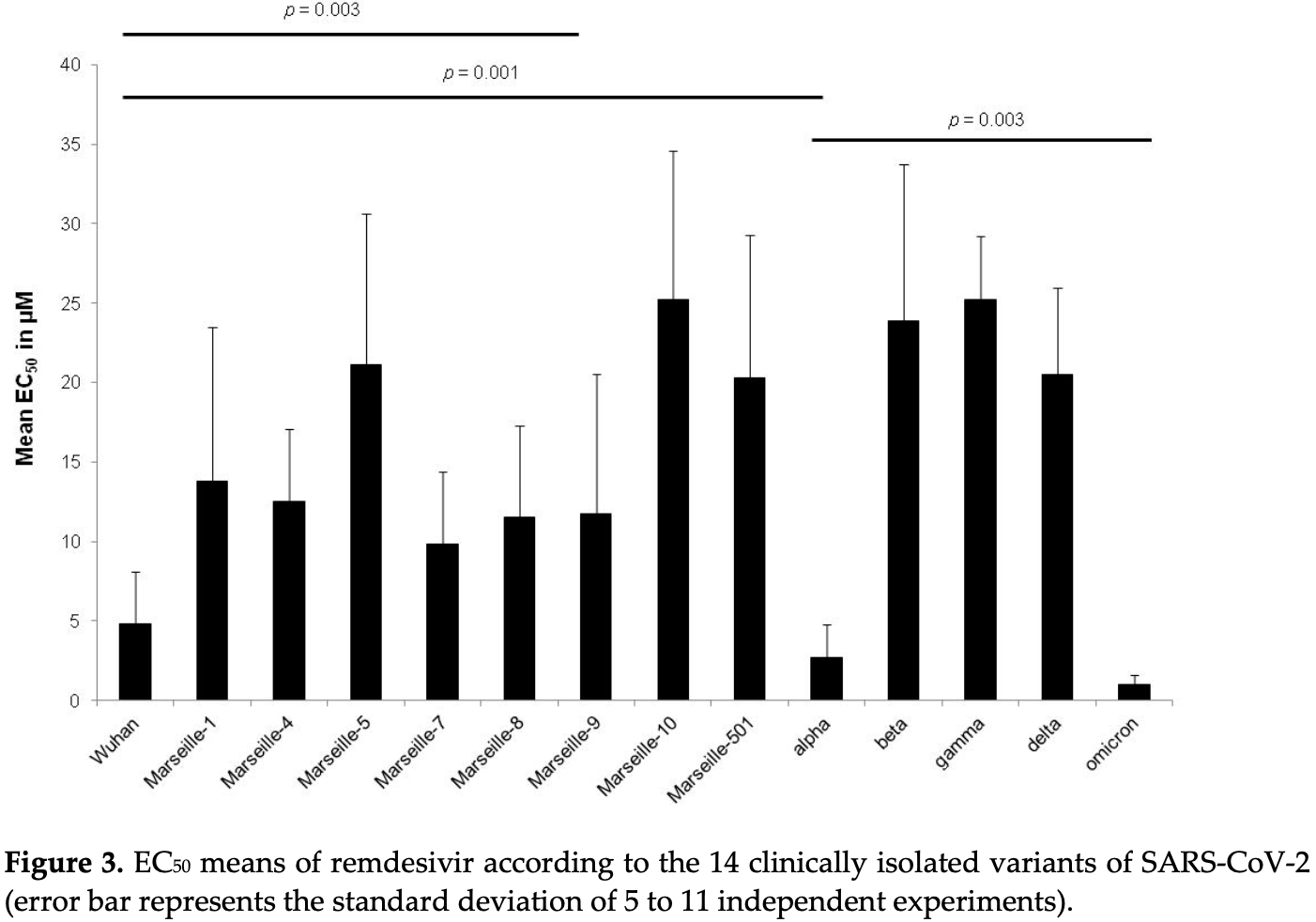
In vitro study with 30 COVID-19 strains from 14 variants, showing stronger efficacy with ivermectin compared to CQ and remdesivir, and relatively homogeneous efficacy with ivermectin regardless of strain/variant, in contrast to results for CQ and remdesivir.
Remdesivir showed better efficacy for omicron compared to most other variants.
Gérard, Zhou, Wu, Kamo, Choi, Kim show increased risk of acute kidney injury, Leo, Briciu, Muntean, Petrov, Arch show increased risk of liver injury, Negru, Cheng, Mohammed, Kwok, Zhu show increased risk of cardiac disorders, and Kwok, Merches, Akinci, Tang, Bagheri show increased risk of mitochondrial toxicity with remdesivir.
Study covers ivermectin, HCQ, and remdesivir.
1.
Gérard et al., Remdesivir and Acute Renal Failure: A Potential Safety Signal From Disproportionality Analysis of the WHO Safety Database, Clinical Pharmacology & Therapeutics, doi:10.1002/cpt.2145.
2.
Zhou et al., Acute Kidney Injury and Drugs Prescribed for COVID-19 in Diabetes Patients: A Real-World Disproportionality Analysis, Frontiers in Pharmacology, doi:10.3389/fphar.2022.833679.
3.
Wu et al., Acute Kidney Injury Associated With Remdesivir: A Comprehensive Pharmacovigilance Analysis of COVID-19 Reports in FAERS, Frontiers in Pharmacology, doi:10.3389/fphar.2022.692828.
4.
Kamo et al., Association of Antiviral Drugs for the Treatment of COVID-19 With Acute Renal Failure, In Vivo, doi:10.21873/invivo.13637.
5.
Choi et al., Comparative effectiveness of combination therapy with nirmatrelvir–ritonavir and remdesivir versus monotherapy with remdesivir or nirmatrelvir–ritonavir in patients hospitalised with COVID-19: a target trial emulation study, The Lancet Infectious Diseases, doi:10.1016/S1473-3099(24)00353-0.
6.
Kim et al., Investigating the Safety Profile of Fast‐Track COVID‐19 Drugs Using the FDA Adverse Event Reporting System Database: A Comparative Observational Study, Pharmacoepidemiology and Drug Safety, doi:10.1002/pds.70043.
7.
Leo et al., Hepatocellular liver injury in hospitalized patients affected by COVID-19: Presence of different risk factors at different time points, Digestive and Liver Disease, doi:10.1016/j.dld.2021.12.014.
8.
Briciu et al., Evolving Clinical Manifestations and Outcomes in COVID-19 Patients: A Comparative Analysis of SARS-CoV-2 Variant Waves in a Romanian Hospital Setting, Pathogens, doi:10.3390/pathogens12121453.
9.
Muntean et al., Effects of COVID-19 on the Liver and Mortality in Patients with SARS-CoV-2 Pneumonia Caused by Delta and Non-Delta Variants: An Analysis in a Single Centre, Pharmaceuticals, doi:10.3390/ph17010003.
10.
Petrov et al., The Effect of Potentially Hepatotoxic Medicinal Products on Alanine Transaminase Levels in COVID-19 Patients: A Case–Control Study, Safety and Risk of Pharmacotherapy, doi:10.30895/2312-7821-2025-458.
11.
Arch et al., Evaluation of the effectiveness of remdesivir in treating severe COVID-19 using data from the ISARIC WHO Clinical Characterisation Protocol UK: a prospective, national cohort study, medRxiv, doi:10.1101/2021.06.18.21259072.
12.
Negru et al., Comparative Pharmacovigilance Analysis of Approved and Repurposed Antivirals for COVID-19: Insights from EudraVigilance Data, Biomedicines, doi:10.3390/biomedicines13061387.
13.
Cheng et al., Cardiovascular Safety of COVID-19 Treatments: A Disproportionality Analysis of Adverse Event Reports from the WHO VigiBase, Infectious Diseases and Therapy, doi:10.1007/s40121-025-01225-z.
14.
Mohammed et al., Bradycardia associated with remdesivir treatment in coronavirus disease 2019 patients: A propensity score-matched analysis, Medicine, doi:10.1097/MD.0000000000044501.
15.
Kwok et al., Remdesivir induces persistent mitochondrial and structural damage in human induced pluripotent stem cell-derived cardiomyocytes, Cardiovascular Research, doi:10.1093/cvr/cvab311.
16.
Zhu et al., Cardiovascular Risks of COVID-19 Therapeutics: Integrated Analysis of FAERS, Electronic Health Records, and Transcriptomics, Pharmaceuticals, doi:10.3390/ph19040574.
17.
Merches et al., The potential of remdesivir to affect function, metabolism and proliferation of cardiac and kidney cells in vitro, Archives of Toxicology, doi:10.1007/s00204-022-03306-1.
18.
Akinci et al., Elucidation of remdesivir cytotoxicity pathways through genome-wide CRISPR-Cas9 screening and transcriptomics, bioRxiv, doi:10.1101/2020.08.27.270819.
Delandre et al., 2 Apr 2022, peer-reviewed, 12 authors.
In vitro studies are an important part of preclinical research, however results may be very different in vivo.
Antiviral Activity of Repurposing Ivermectin against a Panel of 30 Clinical SARS-CoV-2 Strains Belonging to 14 Variants
Pharmaceuticals, doi:10.3390/ph15040445
Over the past two years, several variants of SARS-CoV-2 have emerged and spread all over the world. However, infectivity, clinical severity, re-infection, virulence, transmissibility, vaccine responses and escape, and epidemiological aspects have differed between SARS-CoV-2 variants. Currently, very few treatments are recommended against SARS-CoV-2. Identification of effective drugs among repurposing FDA-approved drugs is a rapid, efficient and low-cost strategy against SARS-CoV-2. One of those drugs is ivermectin. Ivermectin is an antihelminthic agent that previously showed in vitro effects against a SARS-CoV-2 isolate (Australia/VI01/2020 isolate) with an IC50 of around 2 µM. We evaluated the in vitro activity of ivermectin on Vero E6 cells infected with 30 clinically isolated SARS-CoV-2 strains belonging to 14 different variants, and particularly 17 strains belonging to six variants of concern (VOC) (variants related to Wuhan, alpha, beta, gamma, delta and omicron). The in vitro activity of ivermectin was compared to those of chloroquine and remdesivir. Unlike chloroquine (EC50 from 4.3 ± 2.5 to 29.3 ± 5.2 µM) or remdesivir (EC50 from 0.4 ± 0.3 to 25.2 ± 9.4 µM), ivermectin showed a relatively homogeneous in vitro activity against SARS-CoV-2 regardless of the strains or variants (EC50 from 5.1 ± 0.5 to 6.7 ± 0.4 µM), except for one omicron strain (EC50 = 1.3 ± 0.5 µM). Ivermectin (No. EC50 = 219, mean EC50 = 5.7 ± 1.0 µM) was, overall, more potent in vitro than chloroquine (No. EC50 = 214, mean EC50 = 16.1 ± 9.0 µM) (p = 1.3 × 10 −34 ) and remdesivir (No. EC50 = 201, mean EC50 = 11.9 ± 10.0 µM) (p = 1.6 × 10 −13 ). These results should be interpreted with caution regarding the potential use of ivermectin in SARS-CoV-2-infected patients: it is difficult to translate in vitro study results into actual clinical treatment in patients.
Supplementary Materials: The following supporting information can be downloaded at: www.mdpi.com/article/10.3390/ph15040445/s1, Table S1 : List of nucleotide and amino acid changes associated with the different SARS-CoV-2 variants.
Conflicts of Interest: The authors declare no conflicts of interest. The findings and conclusion of this report are those of the authors and do not represent the views of the Ministère des Armées and Ministère de l'Enseignement Supérieur, de la Recherche et de l'Innovation.
References
Adhikari, Banerjee, Baidya, Ghosh, Jha, Ligand-based quantitative structural assessements of SARS-CoV-2 3CLpro inhibitors: An analysis in light of structure-based multi-molecular modeling evidences, J. Mol. Struct
Al-Awaida, Al Hourani, Swedan, Nimer, Alzoughool et al., Correlates of SARS-CoV-2 variants on death, case incidence and case fatality ratio among the continents for the period of 1 December 2020 to 15, Genes
Amrane, Tissot-Dupont, Doudier, Eldin, Hocquart et al., Rapid viral diagnosis and ambulatory management of suspected COVID-19 cases presenting at the infectious disease referral hospital in Marseille, France, -January 31st to March 1st, 2020: A respiratory virus snapshot, Travel Med. Infect. Dis
Andreani, Le Bideau, Duflot, Jardot, Rolland et al., In vitro testing of hydroxychloroquine and azithromycin on SARS-CoV-2 shows synergistic effect, Microb. Pathog
Arshed, Pertinez, Box, Tatham, Rajoli et al., priorisation of anti-SARS-CoV-2 drug repurposing opportunities based on plasma and target site concentrations derived from their established human pharmacokinetics, Clin. Pharmacol. Ther
Arévalo, Pagotto, Porfido, Daghero, Segovia et al., Ivermectin reduced in vivo coronavirus in a mouse experimental model, Sci. Rep
Audus, Knaub, Guillot, Schaeffer, The effect of protein binding on ivermectin uptake by bovine brain microvessel endothelial cell, Vet. Res. Commun
Azam, Taban, Eid, Iqbal, Alam et al., An insilico analysis of ivermectin interaction with potential SARS-CoV-2 targets and host nuclear importin α, J. Biomol. Struct. Dyn
Baildya, Ghosh, Chattopadhyay, Inhibitory capacity of chloroquine against SARS-CoV-2 by effective binding with angiotensin converting enzyme-2 receptor: An insight from molecular docking and MD-simulation studies, J. Mol. Struct
Bello, Elucidation of the inhibitory activity of ivermectin with host nuclear importin α and several SARS-CoV-2 targets, J. Biomol. Struct. Dyn
Braz, Silveira, Marinho, De Moraes, Filho et al., In silico study of azithromycin, chloroquine and hydroxychloroquine and their potential mechanisms of action against SARS-CoV-2 infection, Int. J. Antimicrob. Agents
Brown, Won, Graham, Dinnon, Sims et al., Broad spectrum antiviral remdesivir inhibits human endemic and zoonotic deltacoronavirus with a highly divergent RNA dependent RNA polymerase, Antiviral Res
Caly, Druce, Catton, Jans, Wagstaff, The FDA-approved drug ivermectin inhibits the replication of SARS-CoV-2 in vitro, Anviral Res
Caly, Wagstaff, Jans, Nuclear trafficking of proteins from RNA viruses: Potential target for antivirals, Antiviral Res
Camprubi, Almuedo-Riera, Marti-Soler, Soriano, Hurtado et al., Lack of efficacy of stanadrd doses of ivermectin in severe COVID-19 patients, PLoS ONE
Chaccour, Abizanda, Irigoyen-Barrio, Casellas, Aldaz et al., Nebulized ivermectin for COVID-19 and other respiratory diseases, a proof of concept, does-ranging study in rats, Sci. Rep
Chaccour, Casellas, Matteo, Pineda, Fernandez-Montero et al., The effect of eraly treatment with ivermectin on viral load, symptoms and humoral response in patients with non-severe COVID-19: A pilot, doule-blind, placebo-controlled, randomized clinical trial, EClinicalMedicine
Choudhury, Das, Patra, Bhattacharya, Ghosh et al., Exploring the binding efficacy of ivermectin against the key proteins of SARS-CoV-2 pathogenesis: An in silico approach, Future Virol
Choy, Wong, Kaewpreedee, Sia, Chen et al., lopinavir, emetine, and homoharringtonine inhibit SARS-CoV-2 replication in vitro, Antivir. Res
Colson, Levasseur, Delerce, Pinault, Dudouet et al., Spreading of new SARS-CoV-2 N501Y spike variant in a new lineage, Clin. Microb. Infect
Colson, Levasseur, Gautret, Fenollar, Hoang et al., Introduction into the Marseille geographical area of a mild SARS-CoV-2 variant originating from sub-Saharan Africa: An investigational study, Travel Med. Infect. Dis
Colson, Rolain, Lagier, Brouqui, Raoult, Chloroquine and hydroxychloroquine as available weapons to fight COVID-19, Int. J. Antimicrob. Agents
Dao, Hoang, Colson, Lagier, Million et al., SARS-CoV-2 infectivity and severity of COVID-19 according to SARS-CoV-2 variants: Current evidence, J. Clin. Med
Dao, Hoang, Nguyen, Delerce, Chaudet et al., Clinical outcomes in COVID-19 patients infected with different SARS-CoV-2 variants in Marseille, France, Clin. Microbiol. Infect
Deshpande, Tiwari, Nyayanit, Modak, In silico molecular docking analysis for repurposing therapeutics against multiple proteins from SARS-CoV-2, Eur. J. Pharmacol
Devaux, Rolain, Colson, Raoult, New insights on the antiviral effects of chloroquine against coronavirus: What to expect for COVID-19, Int. J. Antimicrob. Agents
Dittmar, Lee, Whig, Segrist, Li et al., Drug repurposing screens reveal cell-type-specific entry and FDA-approved drugs active against SARS-CoV-2, Cell Rep
Drayman, Jones, Azizi, Froggatt, Tan et al., Drug repurposing screen identifies masitinib as a 3CLpro inhibitor that blocks replication of SARS-CoV-2 in vitro, bioRxiv, doi:10.1101/2020.08.31.274639
Elfiky, Azzam, Shafaa, The anti-HCV, Sofosbuvir, versus the anti-EBOV Remdesivir against SARS-CoV-2 RNA dependent RNA polymerase in silico, Mol. Divers
Errecalde, Lifschitz, Veccioli, Ceballos, Errecalde et al., Safety and phramacokinetic assessments of a novel ivermectin nasal spray formulation in a pig model, J. Pharm. Sci
Eweas, Alhossary, Abdel-Moneim, Molecular docking reveals ivermectin and remdesivir as potential repurposed drugs against SARS-CoV-2, Front. Microbiol
Fantini, Di Scala, Chahinian, Yahi, Structural and molecular modelling studies reveal a new mechanism of action of chloroquine and hydroxychloroquine against SARS-CoV-2 infection, Int. J. Antimicrob. Agents
Fantini, Yahi, Colson, Chahinian, La Scola et al., The puzzling mutational landscape of the SARS-2-variant Omicron, J. Med. Virol, doi:10.1002/jmv.27577
Fathizadeh, Afshar, Masoudi, Gholizadeh, Asgharzadeh et al., SARS-CoV-2 (COVID-19) vaccines structure, mechanisms and effectiveness: A review, Int. J. Biol. Macromol, doi:10.1016/j.ijbiomac.2021.08.076
Flanagan, Macintyre, Mcintyre, Nelson, SARS-CoV-2 vaccines: Where are we now?, J. Allergy Clin. Immunol. Pract, doi:10.1016/j.laip.2021.07.016
Fournier, Colson, Levasseur, Devaux, Gautret et al., Emergence and outcomes of the SARS-CoV-2 'Marseille-4' variant, Int. J. Infect. Dis
Galan, Santos, Asato, Araujo, De Lima Moreira et al., Phase 2 randomized study on chloroquine, hydroxychloroquine or ivermectin in hospitalized patients with severe manifestations of SARS-CoV-2 infection, Pathog. Glob. Health
Gautret, Houhamdi, Nguyen, Hoang, Giraud-Gatineau et al., Does SARS-CoV-2 re-infection depend on virus variant?, Clin. Microb. Infect
Gendrot, Andreani, Boxberger, Jardot, Fonta et al., Antimalarial drugs inhibit the replication of SARS-CoV-2: An in vitro evaluation, Travel Med. Infect. Dis
Gendrot, Andreani, Duflot, Boxberger, Le Bideau et al., Methylene blue inhibits replication of SARS-CoV-2 in vitro, Int. J. Antimicrob. Agents
Gendrot, Andreani, Jardot, Hutter, Delandre et al., In vitro antiviral activity of doxycycline against SARS-CoV-2, Molecules
Gendrot, Duflot, Boxberger, Delandre, Jardot et al., Antimalarial artemisinin-based combination therapies (ACT) and COVID-19 in Africa: In vitro inhibition of SARS-CoV-2 replication by mefloquine-artesunate, Int. J. Infect. Dis
Gendrot, Jardot, Delandre, Boxberger, Andreani et al., In vitro evaluation of the antiviral activity of methylene blue alone or in combination against SARS-CoV-2, J. Clin. Med
Gonzalez-Paz, Hurtado-Leon, Lossada, Fernandez-Materan, Vera-Villalobas et al., Comparative study of the interaction of ivermectin with proteins of interest associated with SARS-CoV-2: A computational and biophysical approach, Biophys. Chem
Gordon, Tchesnokov, Woolner, Perry, Feng et al., Remdesivir is a direct-acting antiviral that inhibits RNA-dependent RNA polymerase from severe acute respiratory syndrome coronavirus 2 with high potency, J. Biol. Chem
Gorial, Mashhadani, Sayaly, Dakhil, Almashhadani et al., Effectiveness of Ivermectin as add-on therapy in COVID-19 management, medRxiv, doi:10.1101/2020.07.07.20145979
Haribabu, Garisetti, Malekshah, Srividya, Gayathri et al., Design and synthesis of heteroclic azole based bioactive compounds: Molecular structures, quantum simulation, and mechanistic studies through docking as multi-target inhibitors of QARS-CoV-2 and cytotoxicity, J. Mol. Struct
Heidary, Gharebaghi, Ivermectin: A systematic review from antiviral effects to COVID-19 complementary regimen, J. Antibiot
Hill, Garratt, Levi, Falconner, Ellis et al., Meta-analysis of randomized trials of ivermectin to treat SARS-CoV-2 infection, Open Forum Infect. Dis
Hill, Mirchandani, Pilkington, Ivermectin for COVID-19: Adressing potential bias and medical fraud, Open Forum Infect. Dis
Hoang, Colson, Levasseur, Delerce, Lagier et al., Clinical outcomes in patients infected with different SARS-CoV-2 variants at one hospital during three phases of the COVID-19 epidemic in Marseille. France, Infect. Genet. Evol
Holwerda, V'kovski, Wider, Thiel, Djikman, Identification of an antiviral compound from the pandemic response box that efficiently inhibits SARS-CoV-2 infection in vitro, Microorganisms
Ikegame, Siddiquey, Hung, Haas, Brambilla et al., Neutralizing activity of Sputnik V vaccine sera against SARS-CoV-2 variants, Nat. Commun
Isaac-Lam, Molecular modeling of the interaction of ligands with ACE2-SARS-CoV-2 spike protein complex, Silico Pharmacol
Izcovich, Peiris, Ragusa, Tortosa, Rda et al., Bias as a source of inconsistency in ivermectin trials for COVID-19: A systematic review. Ivermectin's suggested benefits are mainly based on potentially biased results, J. Clin. Epidemiol
Jaafar, Boschi, Aherfi, Bancod, Le Bideau et al., High individual heterogeneity of neutralizing activities against the original strain and nine different variants of SARS-CoV-2, Viruses
Jans, Martin, Wagstaff, Inhibitors of nuclear transport, Curr. Opin. Cell Biol
Jermain, Hanafin, Cao, Lifschitz, Lanusse et al., Development of a minimal physiologically-based pharmacokinetic model to simulate lung exposure in humans following oral administration of ivermectin for COVID-19 drug repurposing, J. Pharm. Sci
Kato, Matsuyama, Kawase, Hishiki, Katoh et al., Antiviral activities of mycophenolic acid and IMD-0354 against SARS-CoV-2, Microbiol. Immunol
Kern, Schöning, Chaccour, Hammann, Modeling of SARS-CoV-2 treatment effects for informed drug repurposing, Front. Pharmacol
Kirti, Roy, Pattadar, Ray, Agarwal et al., Ivermectin as a potential treatment for mild to moderate COVID-19-A double blind randomized placebo-controlled trial, medRxiv, doi:10.1101/2021.01.05.21249310
Ko, Jeon, Ryu, Kim, Comparative analysis of antiviral efficacy of FDA-approved drugs against SARS-CoV-2 in human lung cells, J. Med. Virol
Kumar, Sarma, Kaur, Prajapat, Bhattacharyya et al., Clinically relevant cell culture models and their significance in isolation, pathogenesis, vaccine development, repurposing and screening of nex drugs for SARS-CoV-2: A systematic review, Tissue Cell
Lehrer, Rheinstein, Ivermectin docks to the SARS-CoV-2 spike receptor-binding domain attached to ACE, Vivo
Lim, Hor, Tay, Jelani, Tan et al., Efficacy of ivermectin treatment on disease progression among adults with mild to moderate COVID-19 and comorbidities: The I-TECH randomized clinical trial, JAMA Intern. Med, doi:10.1001/jamainternmed.2022.0189
Lima-Morales, Mendez-Hernandez, Flores, Osorno-Romero, Sancho-Hernandez et al., Effectiveness of multidrug therapy consisting of Ivermectin, Azithromycin, Montelukast, and Acethylsalicylic acid to prevent hospitalization and death among ambulatory COVID-19 cases in Tlaxcala, Mexico, Int. J. Infect. Dis
Lopez-Medina, Lopez, Hurtado, Davalos, Ramirez et al., Effect if ivermectin on time to resolution of symptoms among adults with mild COVID-19, JAMA
Low, Yip, Lal, Repositioning ivermectin for COVID-19 treatment: Molecular mechanisms of action against SARS-CoV-2 replication, Biochim. Biophys. Acta Mol. Basis Dis
Mahmud, Rahman, Alam, Ahmed, Kabir et al., Ivermectin in combination with doxycycline for treating COVID-19 symptoms: A randomized trial, J. Int. Med. Res
Marciniec, Beberok, Boryczka, Wrzesniok, The application of in silico experimental model in the assessement of ciprofloxacin and levofloxacin interaction with man SARS-CoV-2 targets: S-, E-and TMPRSS2 proteins, RNA-dependent RNA polymerase and papain-like protease (PLpro)-Preliminary molecular docking analysis, Pharmacol. Rep
Mody, Ho, Wills, Mawri, Lawson et al., Identification of 3-chymothrypsin like protease (3CLpro) inhibitors as potentail anti-SARS-CoV-2 agents, Commun. Biol
Mohan, Tiwari, Suri, Mittal, Patel et al., Single-dose oral ivermectin in mild and moderate COVID-19 (RIVET-COV): A single-centre randomized, placebo-controlled trial, J. Infect. Chemother
Mohapatra, Perekhoda, Azam, Suleiman, Sarangi et al., Computational investigations of three main drugs and their comparison with synthesized compounds as potent inhibitors of SARS-CoV-2 main protease (Mpro): DFT, QSAR, molecular docking, and in silico toxicity analysis, J. King Saud Univ. Sci
Ozer, Goksu, Conception, Ulker, Balderas et al., Effectiveness and safety of ivermectin in COVID-19 patients: A prospecive study at a safety-net hospital, J. Med. Virol, doi:10.1002/jmv.27469
Parvez, Karim, Hasan, Jaman, Karim et al., Prediction of potential inhibitors for RNA-dependent RNA polymerase of SARS-CoV-2 using comprehensive drug repurposing and molecular docking approach, Int. J. Biol. Macromol
Pena-Silva, Duffull, Steer, Jaramillo-Rincon, Gwee et al., Pharmacokinetic considerations on the repurposing of ivermectin for treatment of COVID-19, Br. J. Clin. Pharmacol
Pires De Souza, Le Bideau, Boschi, Ferreira, Wurtz et al., Emerging SARS-CoV-2 genotypes show different replication patterns in human pulmonary and intestnal epithelial cells, Viruses
Planas, Saunders, Maes, Guivel-Benhassine, Planchais et al., Considerable escape of SARS-CoV-2 Omicron to antibody neutralization, Nature, doi:10.1038/s41586-021-04389-z
Rajter, Sherman, Fatteh, Vogel, Sacks et al., Use of ivermectin is associated with lower mortality in hospitalized patients with coronavirus disease, Chest
Ribaudo, Coghi, Yang, Ng, Mastinu et al., Computational and experimental insights on the interaction of artemisinin, dihydroartemisinin and chloroquine with SARS-CoV-2 spike prtein receptor-binding domain (RBD), Nat. Prod. Res
Saha, Raihan, The binding mechanism of ivermectin and levosalbutamol with spike protein of SARS-CoV-2, Struct. Chem
Sanyaolu, Okorie, Marinkovic, Haider, Abbasi et al., The emerging SARS-CoV-2 variants of concern, Ther. Adv. Infect. Dis
Sarkar, Thakur, Ghadge, Rath, Computational studies reveal Fluorine based quinilines to be potent inhibitors for proteins involved in SARS-CoV-2 assembly, J. Fluor. Chem
Schmith, Zhou, Lohmer, The approved dose of ivermectin alone is not the ideal dose for the treatment of COVID-19, Clin. Pharmacol. Ther
Scola, Lavrad, Fournier, Colson, Lacoste et al., SARS-CoV-2 variant from India to Marseille: The still active role of ports in the introduction of epidemics, Travel Med. Infect. Dis
Segatory, Garona, Caligiuri, Bizzotto, Lavignolle et al., Effect of ivermectin and atorvastatin on nuclear localization of importin Alpha and drug target expression profiling in host cells from nasopharyngeal swabs of SARS-CoV-2-positive patients, Viruses
Sencanski, Perovic, Pajovic, Adzic, Paessler et al., Drug repurposing for candidate SARS-CoV-2 main protease inhibitors by a novel in silico methods, Molecules
Singh, Pandit, Mcarthur, Banerjee, Mossman, Evolutionary trajectory of SARS-CoV-2 and emerging variants, Virol. J
Tada, Zhou, Samanovic, Dcosta, Cornelius et al., Comparison of neutralizing antibody titers elicited by mRNA and adenoviral vector vaccine against SARS-CoV-2 variants, bioRxiv, doi:10.1101/2021.07.19.452771
Tan, Tan, Chu, Chow, Combination treatment with remdesivir and ivermectin exerts highly synergistic and potent antiviral activity against murine coronavirus infection, Front. Cell. Infect. Microbiol
Tregoning, Flight, Higham, Wang, Pierce, Progress of the COVID-19 vaccine effort: Viruses, vaccines and variants versus efficacy, effectiveness and escape, Nat. Rev. Immunol
Tripathi, Upadhyay, Singh, Raghavendhar, Bhardwaj et al., Sreening and evaluation of approved drugs as inhibitors of main protease of SARS-CoV-2, Int. J. Biol. Macromol
Udofia, Gbayo, Oloba-Whenu, Ogunbayo, Isanbor, In silico studies of selected multi-drug targeting against 3CLpro and nsp12 RNA-dependent RNA-polymerase proteins of SARS-CoV-2 and SARS-CoV, Netw. Model. Anal. Health Inform. Bioinform
Vallejos, Zoni, Bangher, Villamandos, Bobadilla et al., Ivermectin to prevent hospitalizations in patients with COVD-19 (IVERCOR-COVID19) a randomized, double-blind, placebo-controlled trial, BMC Infect. Dis
Wagstaff, Sivakumaran, Heaton, Harrich, Jans, Ivermectin is a specific inhibitor of importin α/βmediated nuclear import able to inhibit replication of HIV-1 and dengue virus, Biochem. J
Wang, Cao, Zhang, Yang, Liu et al., Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCov) in vitro, Cell Res
Wang, Chan, Wang, Li, Zhao et al., Orally administered bismuth drug together with N-acetyl cysteine as a broad-spectrum anti-coronavirus therapy, Chem. Sci
Wang, Li, Rajpoot, Saqib, Yu et al., Comparative assessement of favipiravir and remdesivir againt human coronavirus NL63 in molecular docking and cell culture lodels, Sci. Rep
Warren, Jordan, Lo, Ray, Mackman et al., Therapeutic efficacy of the small molecule GS-5734 against Ebola virus in rhesus monkeys, Nature
Weston, Coleman, Haupt, Logue, Matthews et al., Broad anticoronavirus activity of Food and Drug Administration-approved drugs against SARS-CoV-2 in vitro and SARS-CoV in vivo, J. Virol
Willet, Grove, Maclean, Wilkie, Logan et al., The hyper-transmissible SARS-CoV-2 Omicron variant exhibite significant antigenic change, vaccine escape and a switch in cell entry mechanism, medRxiv, doi:10.1101/2022.01.03.21268111
Wu, Crich, Pegan, Lou, Hansen et al., Polyphenols as potential inhibitors of SARS-CoV-2 RNA dependent RNA polymerase (RdRp), Molecules
Wu, Zhao, Yu, Chen, Wang et al., A new coronavirus associated with human respiratory disease in China, Nature
Wurtz, Penant, Jardot, Duclos, La Scola, Culture of SARS-CoV-2 in a panel of laboratory cell lines, permissivity, and differences in growth profile, Eur. J. Clin. Microbiol. Infect. Dis
Yang, Atkinson, Fraser, Wang, Maher et al., Novel flavivirus antiviral that targets the host nuclear tranmport importin α/β1 heterodimer, Cells
Yang, Atkinson, Wang, Lee, Bogoyevitch et al., The broad spectrum antiviral ivermectin targets the host nuclear transport importin α/β1 heterodimer, Antiviral Res
Yuan, Yin, Meng, Chan, Ye et al., Clofazimine broadly inhibits coronaviruses including SARS-CoV-2, Nature
Zaidi, Dehgani-Mobaraki, The mechanisms of action against SARS-CoV-2: An evidence-based clinical review article, J. Antibiot
Zhan, Dowell, Shen, Lee, Chloroquine to fight COVID-19: A consideration of mechanisms and adverse effects?, Heliyon
Zhang, Liu, Cao, Xu, Wu et al., Comparative antiviral efficacy of viral protease inhibitors against the novel SARS-CoV-2 in vitro, Virol. Sin
DOI record:
{
"DOI": "10.3390/ph15040445",
"ISSN": [
"1424-8247"
],
"URL": "http://dx.doi.org/10.3390/ph15040445",
"abstract": "<jats:p>Over the past two years, several variants of SARS-CoV-2 have emerged and spread all over the world. However, infectivity, clinical severity, re-infection, virulence, transmissibility, vaccine responses and escape, and epidemiological aspects have differed between SARS-CoV-2 variants. Currently, very few treatments are recommended against SARS-CoV-2. Identification of effective drugs among repurposing FDA-approved drugs is a rapid, efficient and low-cost strategy against SARS-CoV-2. One of those drugs is ivermectin. Ivermectin is an antihelminthic agent that previously showed in vitro effects against a SARS-CoV-2 isolate (Australia/VI01/2020 isolate) with an IC50 of around 2 µM. We evaluated the in vitro activity of ivermectin on Vero E6 cells infected with 30 clinically isolated SARS-CoV-2 strains belonging to 14 different variants, and particularly 17 strains belonging to six variants of concern (VOC) (variants related to Wuhan, alpha, beta, gamma, delta and omicron). The in vitro activity of ivermectin was compared to those of chloroquine and remdesivir. Unlike chloroquine (EC50 from 4.3 ± 2.5 to 29.3 ± 5.2 µM) or remdesivir (EC50 from 0.4 ± 0.3 to 25.2 ± 9.4 µM), ivermectin showed a relatively homogeneous in vitro activity against SARS-CoV-2 regardless of the strains or variants (EC50 from 5.1 ± 0.5 to 6.7 ± 0.4 µM), except for one omicron strain (EC50 = 1.3 ± 0.5 µM). Ivermectin (No. EC50 = 219, mean EC50 = 5.7 ± 1.0 µM) was, overall, more potent in vitro than chloroquine (No. EC50 = 214, mean EC50 = 16.1 ± 9.0 µM) (p = 1.3 × 10−34) and remdesivir (No. EC50 = 201, mean EC50 = 11.9 ± 10.0 µM) (p = 1.6 × 10−13). These results should be interpreted with caution regarding the potential use of ivermectin in SARS-CoV-2-infected patients: it is difficult to translate in vitro study results into actual clinical treatment in patients.</jats:p>",
"alternative-id": [
"ph15040445"
],
"author": [
{
"ORCID": "http://orcid.org/0000-0002-8794-8923",
"affiliation": [],
"authenticated-orcid": false,
"family": "Delandre",
"given": "Océane",
"sequence": "first"
},
{
"ORCID": "http://orcid.org/0000-0002-2322-5427",
"affiliation": [],
"authenticated-orcid": false,
"family": "Gendrot",
"given": "Mathieu",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Jardot",
"given": "Priscilla",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Le Bideau",
"given": "Marion",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-7740-0518",
"affiliation": [],
"authenticated-orcid": false,
"family": "Boxberger",
"given": "Manon",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Boschi",
"given": "Céline",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Fonta",
"given": "Isabelle",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Mosnier",
"given": "Joel",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Hutter",
"given": "Sébastien",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Levasseur",
"given": "Anthony",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0001-8006-7704",
"affiliation": [],
"authenticated-orcid": false,
"family": "La Scola",
"given": "Bernard",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-2360-3803",
"affiliation": [],
"authenticated-orcid": false,
"family": "Pradines",
"given": "Bruno",
"sequence": "additional"
}
],
"container-title": "Pharmaceuticals",
"container-title-short": "Pharmaceuticals",
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2022,
4,
3
]
],
"date-time": "2022-04-03T06:59:52Z",
"timestamp": 1648969192000
},
"deposited": {
"date-parts": [
[
2022,
4,
5
]
],
"date-time": "2022-04-05T10:25:31Z",
"timestamp": 1649154331000
},
"funder": [
{
"DOI": "10.13039/501100001665",
"award": [
"Investissement d'avenir ANR-10-IAHU-03"
],
"doi-asserted-by": "publisher",
"name": "French National Research Agency"
},
{
"award": [
"COVID-19"
],
"name": "Institut hospitalo-universitaire Méditerranée Infection"
}
],
"indexed": {
"date-parts": [
[
2024,
2,
9
]
],
"date-time": "2024-02-09T13:19:30Z",
"timestamp": 1707484770021
},
"is-referenced-by-count": 6,
"issue": "4",
"issued": {
"date-parts": [
[
2022,
4,
2
]
]
},
"journal-issue": {
"issue": "4",
"published-online": {
"date-parts": [
[
2022,
4
]
]
}
},
"language": "en",
"license": [
{
"URL": "https://creativecommons.org/licenses/by/4.0/",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2022,
4,
2
]
],
"date-time": "2022-04-02T00:00:00Z",
"timestamp": 1648857600000
}
}
],
"link": [
{
"URL": "https://www.mdpi.com/1424-8247/15/4/445/pdf",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "1968",
"original-title": [],
"page": "445",
"prefix": "10.3390",
"published": {
"date-parts": [
[
2022,
4,
2
]
]
},
"published-online": {
"date-parts": [
[
2022,
4,
2
]
]
},
"publisher": "MDPI AG",
"reference": [
{
"DOI": "10.1038/s41586-020-2008-3",
"doi-asserted-by": "publisher",
"key": "ref1"
},
{
"DOI": "10.1186/s12985-021-01633-w",
"doi-asserted-by": "publisher",
"key": "ref2"
},
{
"DOI": "10.1177/20499361211024372",
"doi-asserted-by": "publisher",
"key": "ref3"
},
{
"DOI": "10.1016/j.jaip.2021.07.016",
"doi-asserted-by": "publisher",
"key": "ref4"
},
{
"DOI": "10.1016/j.ijbiomac.2021.08.076",
"doi-asserted-by": "publisher",
"key": "ref5"
},
{
"DOI": "10.1016/j.tmaid.2020.101873",
"doi-asserted-by": "publisher",
"key": "ref6"
},
{
"DOI": "10.1016/j.ijid.2020.08.032",
"doi-asserted-by": "publisher",
"key": "ref7"
},
{
"DOI": "10.1038/s41422-020-0282-0",
"doi-asserted-by": "publisher",
"key": "ref8"
},
{
"DOI": "10.3390/microorganisms8121872",
"doi-asserted-by": "publisher",
"key": "ref9"
},
{
"DOI": "10.1016/j.micpath.2020.104228",
"doi-asserted-by": "publisher",
"key": "ref10"
},
{
"DOI": "10.3390/molecules25215064",
"doi-asserted-by": "publisher",
"key": "ref11"
},
{
"DOI": "10.1016/j.antiviral.2020.104786",
"doi-asserted-by": "publisher",
"key": "ref12"
},
{
"DOI": "10.1007/s12250-020-00288-1",
"doi-asserted-by": "publisher",
"key": "ref13"
},
{
"DOI": "10.1038/s41586-021-03431-4",
"doi-asserted-by": "publisher",
"key": "ref14"
},
{
"DOI": "10.1128/JVI.01218-20",
"doi-asserted-by": "publisher",
"key": "ref15"
},
{
"DOI": "10.1101/2020.08.31.274639",
"doi-asserted-by": "publisher",
"key": "ref16"
},
{
"DOI": "10.1016/j.celrep.2021.108959",
"doi-asserted-by": "publisher",
"key": "ref17"
},
{
"DOI": "10.1111/1348-0421.12828",
"doi-asserted-by": "publisher",
"key": "ref18"
},
{
"DOI": "10.1002/jmv.26397",
"doi-asserted-by": "publisher",
"key": "ref19"
},
{
"DOI": "10.1016/j.ijantimicag.2020.106202",
"doi-asserted-by": "publisher",
"key": "ref20"
},
{
"DOI": "10.3390/jcm10143007",
"doi-asserted-by": "publisher",
"key": "ref21"
},
{
"DOI": "10.1039/D1SC04515F",
"doi-asserted-by": "publisher",
"key": "ref22"
},
{
"DOI": "10.1038/s41429-020-0336-z",
"doi-asserted-by": "publisher",
"key": "ref23"
},
{
"DOI": "10.1038/s41598-021-86679-0",
"doi-asserted-by": "publisher",
"key": "ref24"
},
{
"DOI": "10.3389/fcimb.2021.700502",
"doi-asserted-by": "publisher",
"key": "ref25"
},
{
"DOI": "10.1016/j.antiviral.2020.104787",
"doi-asserted-by": "publisher",
"key": "ref26"
},
{
"DOI": "10.1016/j.cmi.2021.05.029",
"doi-asserted-by": "publisher",
"key": "ref27"
},
{
"DOI": "10.1016/j.cmi.2021.06.029",
"doi-asserted-by": "publisher",
"key": "ref28"
},
{
"DOI": "10.3390/jcm10122635",
"doi-asserted-by": "publisher",
"key": "ref29"
},
{
"DOI": "10.1038/s41467-021-24909-9",
"doi-asserted-by": "publisher",
"key": "ref30"
},
{
"DOI": "10.1038/s41577-021-00592-1",
"doi-asserted-by": "publisher",
"key": "ref31"
},
{
"DOI": "10.3390/genes12071061",
"doi-asserted-by": "publisher",
"key": "ref32"
},
{
"DOI": "10.1101/2021.07.19.452771",
"doi-asserted-by": "publisher",
"key": "ref33"
},
{
"DOI": "10.3390/v13112177",
"doi-asserted-by": "publisher",
"key": "ref34"
},
{
"DOI": "10.1002/jmv.27577",
"doi-asserted-by": "publisher",
"key": "ref35"
},
{
"DOI": "10.3390/v14010023",
"doi-asserted-by": "publisher",
"key": "ref36"
},
{
"DOI": "10.1038/s41586-021-04389-z",
"doi-asserted-by": "publisher",
"key": "ref37"
},
{
"article-title": "The mechanisms of action against SARS-CoV-2: An evidence-based clinical review article",
"author": "Zaidi",
"first-page": "1",
"journal-title": "J. Antibiot.",
"key": "ref38",
"volume": "21",
"year": "2021"
},
{
"DOI": "10.1016/j.antiviral.2012.06.008",
"doi-asserted-by": "publisher",
"key": "ref39"
},
{
"DOI": "10.3390/cells8030281",
"doi-asserted-by": "publisher",
"key": "ref40"
},
{
"DOI": "10.1016/j.ceb.2019.01.001",
"doi-asserted-by": "publisher",
"key": "ref41"
},
{
"DOI": "10.1042/BJ20120150",
"doi-asserted-by": "publisher",
"key": "ref42"
},
{
"DOI": "10.1016/j.antiviral.2020.104760",
"doi-asserted-by": "publisher",
"key": "ref43"
},
{
"DOI": "10.1016/j.bbadis.2021.166294",
"doi-asserted-by": "publisher",
"key": "ref44"
},
{
"DOI": "10.1080/07391102.2020.1841028",
"doi-asserted-by": "publisher",
"key": "ref45"
},
{
"DOI": "10.1080/07391102.2021.1911857",
"doi-asserted-by": "publisher",
"key": "ref46"
},
{
"DOI": "10.1016/j.bpc.2021.106677",
"doi-asserted-by": "publisher",
"key": "ref47"
},
{
"DOI": "10.2217/fvl-2020-0342",
"doi-asserted-by": "publisher",
"key": "ref48"
},
{
"DOI": "10.1007/s13721-021-00299-2",
"doi-asserted-by": "publisher",
"key": "ref49"
},
{
"DOI": "10.1016/j.ijbiomac.2020.09.098",
"doi-asserted-by": "publisher",
"key": "ref50"
},
{
"DOI": "10.3389/fmicb.2020.592908",
"doi-asserted-by": "publisher",
"key": "ref51"
},
{
"DOI": "10.1038/nature17180",
"doi-asserted-by": "publisher",
"key": "ref52"
},
{
"DOI": "10.1074/jbc.RA120.013679",
"doi-asserted-by": "publisher",
"key": "ref53"
},
{
"DOI": "10.3390/molecules25173830",
"doi-asserted-by": "publisher",
"key": "ref54"
},
{
"DOI": "10.1038/s42003-020-01577-x",
"doi-asserted-by": "publisher",
"key": "ref55"
},
{
"DOI": "10.1016/j.jksus.2020.101315",
"doi-asserted-by": "publisher",
"key": "ref56"
},
{
"DOI": "10.21873/invivo.12134",
"doi-asserted-by": "publisher",
"key": "ref57"
},
{
"DOI": "10.1007/s11224-021-01776-0",
"doi-asserted-by": "publisher",
"key": "ref58"
},
{
"DOI": "10.1016/j.ijantimicag.2020.105960",
"doi-asserted-by": "publisher",
"key": "ref59"
},
{
"DOI": "10.1016/j.molstruc.2021.129891",
"doi-asserted-by": "publisher",
"key": "ref60"
},
{
"DOI": "10.1080/14786419.2021.1925894",
"doi-asserted-by": "publisher",
"key": "ref61"
},
{
"DOI": "10.1007/s40203-021-00114-w",
"doi-asserted-by": "publisher",
"key": "ref62"
},
{
"DOI": "10.1016/j.molstruc.2021.131782",
"doi-asserted-by": "publisher",
"key": "ref63"
},
{
"DOI": "10.1016/j.jfluchem.2021.109865",
"doi-asserted-by": "publisher",
"key": "ref64"
},
{
"DOI": "10.1007/s43440-021-00282-8",
"article-title": "The application of in silico experimental model in the assessement of ciprofloxacin and levofloxacin interaction with man SARS-CoV-2 targets: S-, E- and TMPRSS2 proteins, RNA-dependent RNA polymerase and papain-like protease (PLpro)—Preliminary molecular docking analysis",
"author": "Marciniec",
"doi-asserted-by": "crossref",
"first-page": "1765",
"journal-title": "Pharmacol. Rep.",
"key": "ref65",
"volume": "73",
"year": "2021"
},
{
"DOI": "10.1016/j.ijbiomac.2020.08.166",
"doi-asserted-by": "publisher",
"key": "ref66"
},
{
"DOI": "10.1016/j.ijantimicag.2020.106119",
"doi-asserted-by": "publisher",
"key": "ref67"
},
{
"DOI": "10.1101/2022.01.03.21268111",
"doi-asserted-by": "publisher",
"key": "ref68"
},
{
"DOI": "10.1016/j.ijantimicag.2020.105932",
"doi-asserted-by": "publisher",
"key": "ref69"
},
{
"DOI": "10.1016/j.ijantimicag.2020.105938",
"doi-asserted-by": "publisher",
"key": "ref70"
},
{
"DOI": "10.1016/j.heliyon.2020.e04900",
"doi-asserted-by": "publisher",
"key": "ref71"
},
{
"DOI": "10.1016/j.antiviral.2019.104541",
"doi-asserted-by": "publisher",
"key": "ref72"
},
{
"DOI": "10.3390/molecules26247438",
"doi-asserted-by": "publisher",
"key": "ref73"
},
{
"DOI": "10.1038/s41598-021-02972-y",
"doi-asserted-by": "publisher",
"key": "ref74"
},
{
"DOI": "10.1007/s11030-020-10178-z",
"doi-asserted-by": "publisher",
"key": "ref75"
},
{
"DOI": "10.1016/j.ejphar.2020.173430",
"doi-asserted-by": "publisher",
"key": "ref76"
},
{
"DOI": "10.1016/j.molstruc.2021.132041",
"doi-asserted-by": "publisher",
"key": "ref77"
},
{
"DOI": "10.1007/BF01839186",
"doi-asserted-by": "publisher",
"key": "ref78"
},
{
"DOI": "10.1111/bcp.14476",
"doi-asserted-by": "publisher",
"key": "ref79"
},
{
"DOI": "10.1016/j.xphs.2020.08.024",
"doi-asserted-by": "publisher",
"key": "ref80"
},
{
"DOI": "10.1002/cpt.1889",
"doi-asserted-by": "publisher",
"key": "ref81"
},
{
"DOI": "10.3389/fphar.2021.625678",
"doi-asserted-by": "publisher",
"key": "ref82"
},
{
"DOI": "10.1002/cpt.1909",
"doi-asserted-by": "publisher",
"key": "ref83"
},
{
"DOI": "10.3390/v13102084",
"doi-asserted-by": "publisher",
"key": "ref84"
},
{
"DOI": "10.1177/03000605211013550",
"doi-asserted-by": "publisher",
"key": "ref85"
},
{
"DOI": "10.1101/2020.07.07.20145979",
"doi-asserted-by": "publisher",
"key": "ref86"
},
{
"DOI": "10.1038/s41598-020-74084-y",
"doi-asserted-by": "publisher",
"key": "ref87"
},
{
"DOI": "10.1016/j.xphs.2021.01.017",
"doi-asserted-by": "publisher",
"key": "ref88"
},
{
"DOI": "10.1371/journal.pone.0242184",
"doi-asserted-by": "publisher",
"key": "ref89"
},
{
"DOI": "10.1080/20477724.2021.1890887",
"doi-asserted-by": "publisher",
"key": "ref90"
},
{
"DOI": "10.1101/2021.01.05.21249310",
"doi-asserted-by": "publisher",
"key": "ref91"
},
{
"DOI": "10.1186/s12879-021-06348-5",
"doi-asserted-by": "publisher",
"key": "ref92"
},
{
"DOI": "10.1001/jama.2021.3071",
"doi-asserted-by": "publisher",
"key": "ref93"
},
{
"DOI": "10.1016/j.jiac.2021.08.021",
"doi-asserted-by": "publisher",
"key": "ref94"
},
{
"DOI": "10.1001/jamainternmed.2022.0189",
"doi-asserted-by": "publisher",
"key": "ref95"
},
{
"DOI": "10.1016/j.ijid.2021.02.014",
"doi-asserted-by": "publisher",
"key": "ref96"
},
{
"DOI": "10.1016/j.eclinm.2020.100720",
"doi-asserted-by": "publisher",
"key": "ref97"
},
{
"DOI": "10.1002/jmv.27469",
"doi-asserted-by": "publisher",
"key": "ref98"
},
{
"DOI": "10.1016/j.chest.2020.10.009",
"doi-asserted-by": "publisher",
"key": "ref99"
},
{
"DOI": "10.1093/ofid/ofab645",
"doi-asserted-by": "publisher",
"key": "ref100"
},
{
"DOI": "10.1016/j.jclinepi.2021.12.018",
"doi-asserted-by": "publisher",
"key": "ref101"
},
{
"DOI": "10.1093/ofid/ofab358",
"doi-asserted-by": "publisher",
"key": "ref102"
},
{
"DOI": "10.1016/j.meegid.2021.105092",
"doi-asserted-by": "publisher",
"key": "ref103"
},
{
"DOI": "10.1016/j.tmaid.2021.101980",
"doi-asserted-by": "publisher",
"key": "ref104"
},
{
"DOI": "10.1016/j.ijid.2021.03.068",
"doi-asserted-by": "publisher",
"key": "ref105"
},
{
"DOI": "10.1016/j.cmi.2021.05.006",
"doi-asserted-by": "publisher",
"key": "ref106"
},
{
"DOI": "10.1016/j.tmaid.2021.102085",
"doi-asserted-by": "publisher",
"key": "ref107"
},
{
"DOI": "10.1007/s10096-020-04106-0",
"doi-asserted-by": "publisher",
"key": "ref108"
},
{
"DOI": "10.1016/j.tice.2021.101497",
"doi-asserted-by": "publisher",
"key": "ref109"
},
{
"DOI": "10.1016/j.tmaid.2020.101632",
"doi-asserted-by": "publisher",
"key": "ref110"
}
],
"reference-count": 110,
"references-count": 110,
"relation": {},
"resource": {
"primary": {
"URL": "https://www.mdpi.com/1424-8247/15/4/445"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "Antiviral Activity of Repurposing Ivermectin against a Panel of 30 Clinical SARS-CoV-2 Strains Belonging to 14 Variants",
"type": "journal-article",
"volume": "15"
}
delandre