Effects of graphene photothermal adjuvant therapy in patients infected with the Omicron BF.7 variant 2022: a prospective randomized controlled trial
et al., Scientific Reports, doi:10.1038/s41598-026-50266-y, NCT05651815, Apr 2026
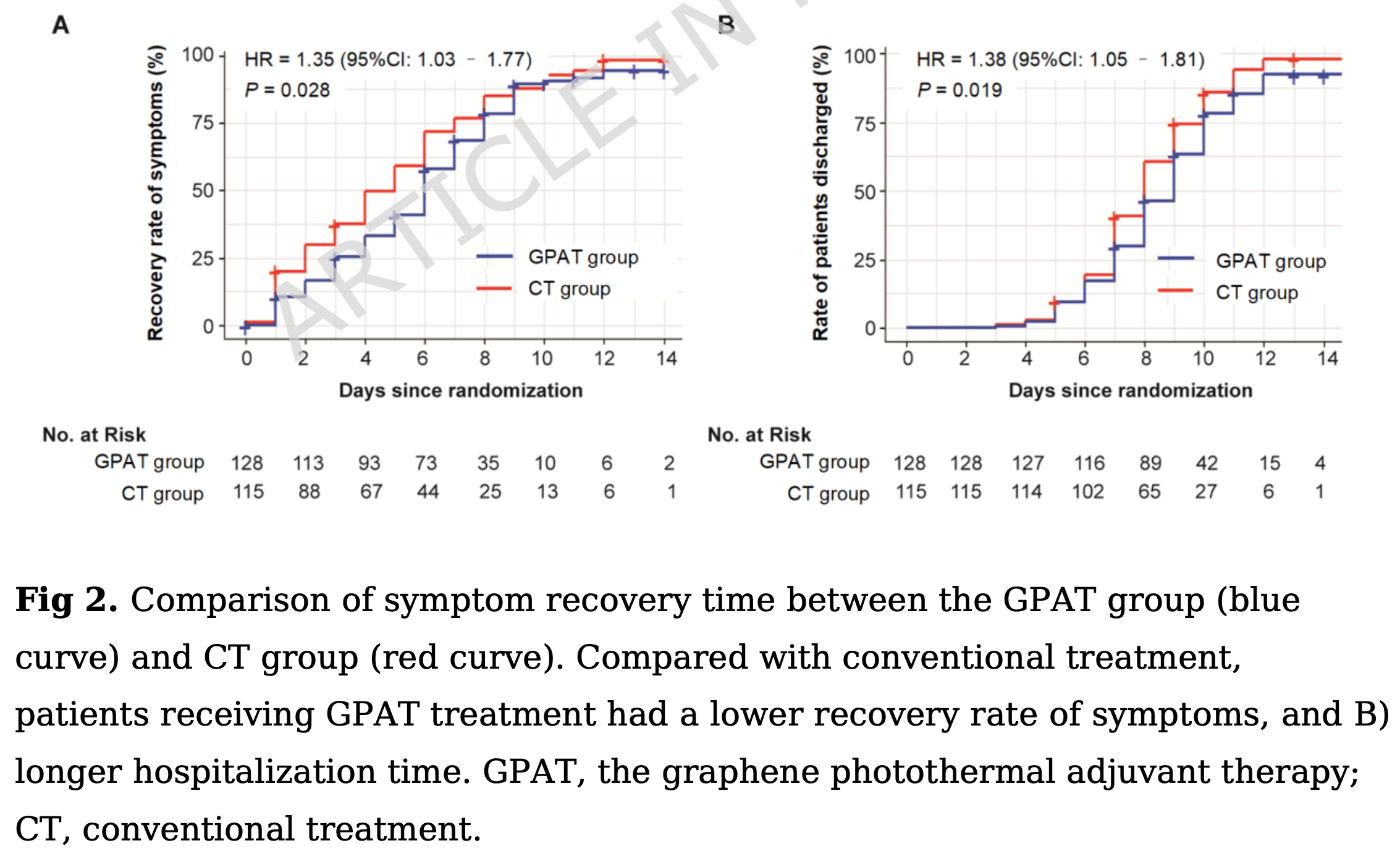
RCT 243 mildly symptomatic hospitalized COVID-19 patients (Omicron BF.7 variant) in China showing increased risk with graphene photothermal adjuvant therapy (GPAT).
All patients received nebulized N-acetylcysteine and the interaction with GPAT is potentially negative. N-acetylcysteine (NAC) is a potent antioxidant/reducing agent which works by donating thiol groups, scavenging reactive oxygen species (ROS), and replenishing glutathione. One of the proposed mechanisms by which far-infrared and photobiomodulation exert anti-inflammatory effects is through mild ROS modulation and mitochondrial signaling (cytochrome c oxidase activation, transient ROS bursts that then trigger downstream anti-inflammatory pathways). If far-infrared's benefit depends partly on a controlled ROS signal, a strong systemic antioxidant like NAC given concurrently could blunt that signal. This concern has been raised in exercise physiology and photobiomodulation literature - high-dose antioxidants can attenuate the hormetic benefits of mild oxidative stimuli. Additionally, acetylcysteine nebulization liquefies mucus and can cause bronchospasm or increased secretions in some patients. If GPAT involves inhaling warm steam (authors mention "inhaling hot steam"), there may be increased airway secretions or transient irritation prolonging cough, stuffy nose, and dry/itchy throat - matching two of the symptoms that recovered more slowly in the GPAT arm.
Standard of Care (SOC) for COVID-19 in the study country,
China, is poor with low average efficacy for approved treatments1.
|
risk of hospitalization, 38.0% higher, HR 1.38, p = 0.02, treatment 128, control 115.
|
|
risk of no recovery, 35.0% higher, HR 1.35, p = 0.03, treatment 128, control 115.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
An et al., 28 Apr 2026, Randomized Controlled Trial, China, peer-reviewed, 15 authors, study period 22 November, 2022 - 13 December, 2022, trial NCT05651815 (history).
Contact: zhang.jj@vip.163.com, zhangkj@nwu.edu.cn.
Graphene photothermal adjuvant therapy (GPAT) is a device-based physical intervention that utilizes a graphene electrothermal film to deliver far-infrared radiation, investigated for its potential to modulate inflammatory immune response.
Abstract: ## Scientific Reports
Article in Press
Effects of graphene photothermal adjuvant therapy in patients infected with the Omicron BF.7 variant 2022: a prospective randomized controlled trial
Received: 4 July 2024
Accepted: 20 April 2026
Cite this article as: An C., Hao L., Li Y. et al. Effects of graphene photothermal adjuvant therapy in patients infected with the Omicron BF.7 variant 2022: a prospective randomized controlled trial. Sci Rep (2026). https:// doi.org/10.1038/s41598-026-50266-y
ARTICLE IN PRESS
Caiyan An, Lingfang Hao, Yanfei Li, Rui Wu, Xia Li, Jianqiang Zhou, Zhiheng Chang, Xianliang Ren, Haijiang Zheng, Bo Chen, Huijun Wang, Weihua Zheng, Yali Yan, Kejin Zhang & Junjing Zhang
We are providing an unedited version of this manuscript to give early access to its findings. Before final publication, the manuscript will undergo further editing. Please note there may be errors present which affect the content, and all legal disclaimers apply.
If this paper is publishing under a Transparent Peer Review model then Peer Review reports will publish with the final article.
© The Author(s) 2026. Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
Effects of Graphene Photothermal Adjuvant Therapy in Patients Infected with the Omicron BF.7 Variant 2022: A Prospective Randomized Controlled Trial
Caiyan An, 1# Lingfang Hao, 2# Yanfei Li, 1# Rui Wu, 3 Xia Li, 4 Jianqiang Zhou, 5 Zhiheng Chang, 6 Xianliang Ren, 7 Haijiang Zheng, 8 Bo Chen, 9 Huijun Wang, 1 Weihua Zheng, 1 Yali Yan, 1 Kejin Zhang, 10* Junjing Zhang 1*
1 Inner Mongolia Key Laboratory of Allergic Diseases, Foundational and Translational Medical Research Center, Department of Allergy and General Surgery, Hohhot First Hospital, Hohhot 010030, China
2 Department of Oncology, Hohhot First Hospital, Hohhot 010030, China
3 Department of Infection Management, Hohhot First Hospital, Hohhot 010030, China
4 Department of Medical Laboratory Medicine, Hohhot First Hospital, Hohhot 010030, China
5 Department of Orthopedics and Joints, Hohhot First Hospital, Hohhot 010030, China
6 Department of Gastroenterology, Hohhot First Hospital, Hohhot 010030, China
ARTIC/E IN PRESS
7 Department of Cardiology, Hohhot First Hospital, Hohhot 010030, China
8 Department of Gastrointestinal Surgery, Hohhot First Hospital, Hohhot 010030, China
9 Department of Neurosurgery, Hohhot First Hospital, Hohhot 010030, China
10 Department of Biological Sciences, College of Life Science, ..
DOI record:
{
"DOI": "10.1038/s41598-026-50266-y",
"ISSN": [
"2045-2322"
],
"URL": "http://dx.doi.org/10.1038/s41598-026-50266-y",
"alternative-id": [
"50266"
],
"assertion": [
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "Received",
"name": "received",
"order": 1,
"value": "4 July 2024"
},
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "Accepted",
"name": "accepted",
"order": 2,
"value": "20 April 2026"
},
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "First Online",
"name": "first_online",
"order": 3,
"value": "28 April 2026"
},
{
"group": {
"label": "Declarations",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 1
},
{
"group": {
"label": "Ethics approval and consent to participate",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 2,
"value": "The study received ethical approval from the Hohhot First Hospital Ethics Committee (Permit Number: IRB2022075). All patients signed written informed consent."
},
{
"group": {
"label": "Competing interests",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 3,
"value": "The authors declare no competing interests."
}
],
"author": [
{
"affiliation": [],
"family": "An",
"given": "Caiyan",
"sequence": "first"
},
{
"affiliation": [],
"family": "Hao",
"given": "Lingfang",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Li",
"given": "Yanfei",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Wu",
"given": "Rui",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Li",
"given": "Xia",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Zhou",
"given": "Jianqiang",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Chang",
"given": "Zhiheng",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ren",
"given": "Xianliang",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Zheng",
"given": "Haijiang",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Chen",
"given": "Bo",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Wang",
"given": "Huijun",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Zheng",
"given": "Weihua",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Yan",
"given": "Yali",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Zhang",
"given": "Kejin",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Zhang",
"given": "Junjing",
"sequence": "additional"
}
],
"container-title": "Scientific Reports",
"container-title-short": "Sci Rep",
"content-domain": {
"crossmark-restriction": false,
"domain": [
"link.springer.com"
]
},
"created": {
"date-parts": [
[
2026,
4,
28
]
],
"date-time": "2026-04-28T12:43:40Z",
"timestamp": 1777380220000
},
"deposited": {
"date-parts": [
[
2026,
4,
28
]
],
"date-time": "2026-04-28T12:43:41Z",
"timestamp": 1777380221000
},
"indexed": {
"date-parts": [
[
2026,
4,
29
]
],
"date-time": "2026-04-29T08:14:40Z",
"timestamp": 1777450480154,
"version": "3.51.4"
},
"is-referenced-by-count": 0,
"issued": {
"date-parts": [
[
2026,
4,
28
]
]
},
"language": "en",
"license": [
{
"URL": "https://creativecommons.org/licenses/by-nc-nd/4.0",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2026,
4,
28
]
],
"date-time": "2026-04-28T00:00:00Z",
"timestamp": 1777334400000
}
},
{
"URL": "https://creativecommons.org/licenses/by-nc-nd/4.0",
"content-version": "am",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2026,
4,
28
]
],
"date-time": "2026-04-28T00:00:00Z",
"timestamp": 1777334400000
}
}
],
"link": [
{
"URL": "https://www.nature.com/articles/s41598-026-50266-y",
"content-type": "text/html",
"content-version": "vor",
"intended-application": "text-mining"
}
],
"member": "297",
"original-title": [],
"prefix": "10.1038",
"published": {
"date-parts": [
[
2026,
4,
28
]
]
},
"published-online": {
"date-parts": [
[
2026,
4,
28
]
]
},
"publisher": "Springer Science and Business Media LLC",
"reference-count": 0,
"references-count": 0,
"relation": {},
"resource": {
"primary": {
"URL": "https://www.nature.com/articles/s41598-026-50266-y"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "Effects of graphene photothermal adjuvant therapy in patients infected with the Omicron BF.7 variant 2022: a prospective randomized controlled trial",
"type": "journal-article",
"update-policy": "https://doi.org/10.1007/springer_crossmark_policy"
}