RECOVER-VITAL: A Platform Protocol for Evaluation of Interventions for Viral Persistence, Viral Reactivation, and Immune Dysregulation in Post-Acute Sequelae of SARS-CoV-2 Infection (PASC)
et al., NCT05965726, RECOVER-VITAL, NCT05965726, Mar 2026
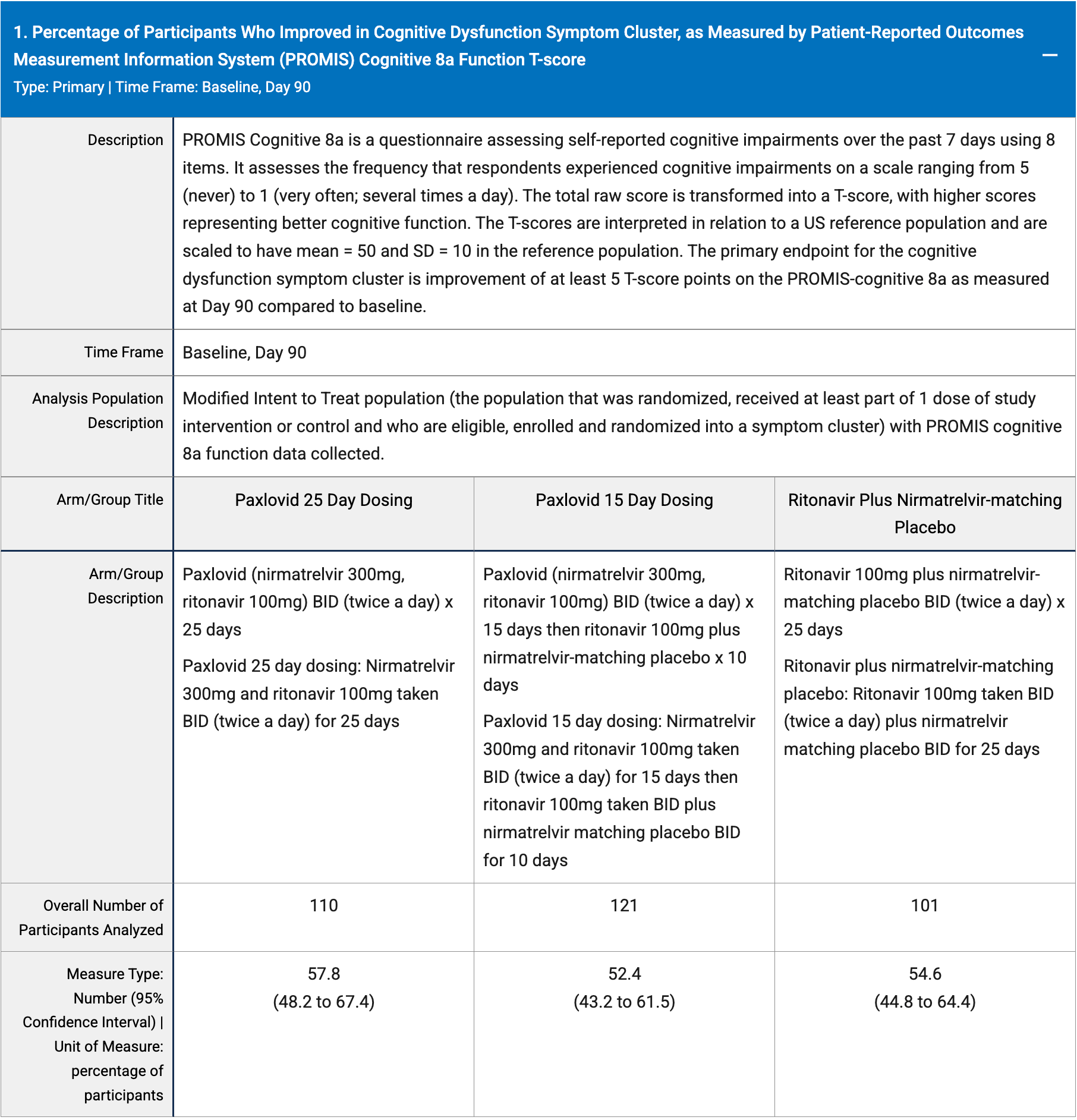
RCT 964 patients showing no difference in improvement of long COVID with 15-day or 25-day paxlovid compared with placebo.
Results appeared in the registry 15 months after primary completion, with no publication or announcement1.
While authors have not published the results, in the same month as the results appeared in the registry, they published a design paper that states: “While the STOP-PASC and PAX LC trials did not demonstrate a positive effect on Long COVID, the RECOVER-VITAL study remains a critical addition to the field and still has potential for demonstrating effect...”2.
Standard of Care (SOC) for COVID-19 in the study country,
the USA, is very poor with very low average efficacy for approved treatments3.
Only expensive, high-profit treatments were approved for early treatment. Low-cost treatments were excluded, reducing the probability of early treatment due to access and cost barriers, and eliminating complementary and synergistic benefits seen with many low-cost treatments.
|
risk of no improvement, 8.9% higher, RR 1.09, p = 0.22, treatment 121, control 110, 25 day, all symptom clusters.
|
|
risk of no improvement, 7.0% lower, RR 0.93, p = 0.65, treatment 42.2 [32.6-51.8] n=110, control 45.4 [35.6-55.2] n=101, cognitive dysfunction, 25 day.
|
|
risk of no improvement, 21.0% higher, RR 1.21, p = 0.30, treatment 36.9 [28.5-45.3] n=121, control 30.5 [21.7-39.3] n=110, autonomic dysfunction, 25 day.
|
|
risk of no improvement, 11.7% higher, RR 1.12, p = 0.20, treatment 74.5 [66.4-82.7] n=112, control 66.7 [58.2-75.2] n=114, exercise intolerance, 25 day.
|
|
risk of no improvement, 0.4% higher, RR 1.00, p = 0.96, treatment 121, control 101, 15 day, all symptom clusters.
|
|
risk of no improvement, 4.8% higher, RR 1.05, p = 0.75, treatment 47.6 [38.5-56.8] n=121, control 45.4 [35.6-55.2] n=101, cognitive dysfunction, 15 day.
|
|
risk of no improvement, 0.3% higher, RR 1.00, p = 0.99, treatment 30.6 [21.7-39.5] n=103, control 30.5 [21.7-39.3] n=110, autonomic dysfunction, 15 day.
|
|
risk of no improvement, 1.3% lower, RR 0.99, p = 0.89, treatment 65.8 [56.7-74.8] n=105, control 66.7 [58.2-75.2] n=114, exercise intolerance, 15 day.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Zimmerman et al., 27 Mar 2026, Double Blind Randomized Controlled Trial, placebo-controlled, USA, preprint, 1 author, trial NCT05965726 (history) (RECOVER-VITAL).
Contact: kanecia.zimmerman@duke.edu.