Famotidine Use Is Not Associated With 30-day Mortality: A Coarsened Exact Match Study in 7158 Hospitalized Patients With Coronavirus Disease 2019 From a Large Healthcare System
et al., Gastroenterology, doi:10.1053/j.gastro.2020.10.011, Feb 2021
Famotidine for COVID-19
28th treatment shown to reduce risk in
October 2021, now with p = 0.00072 from 29 studies, recognized in 2 countries.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
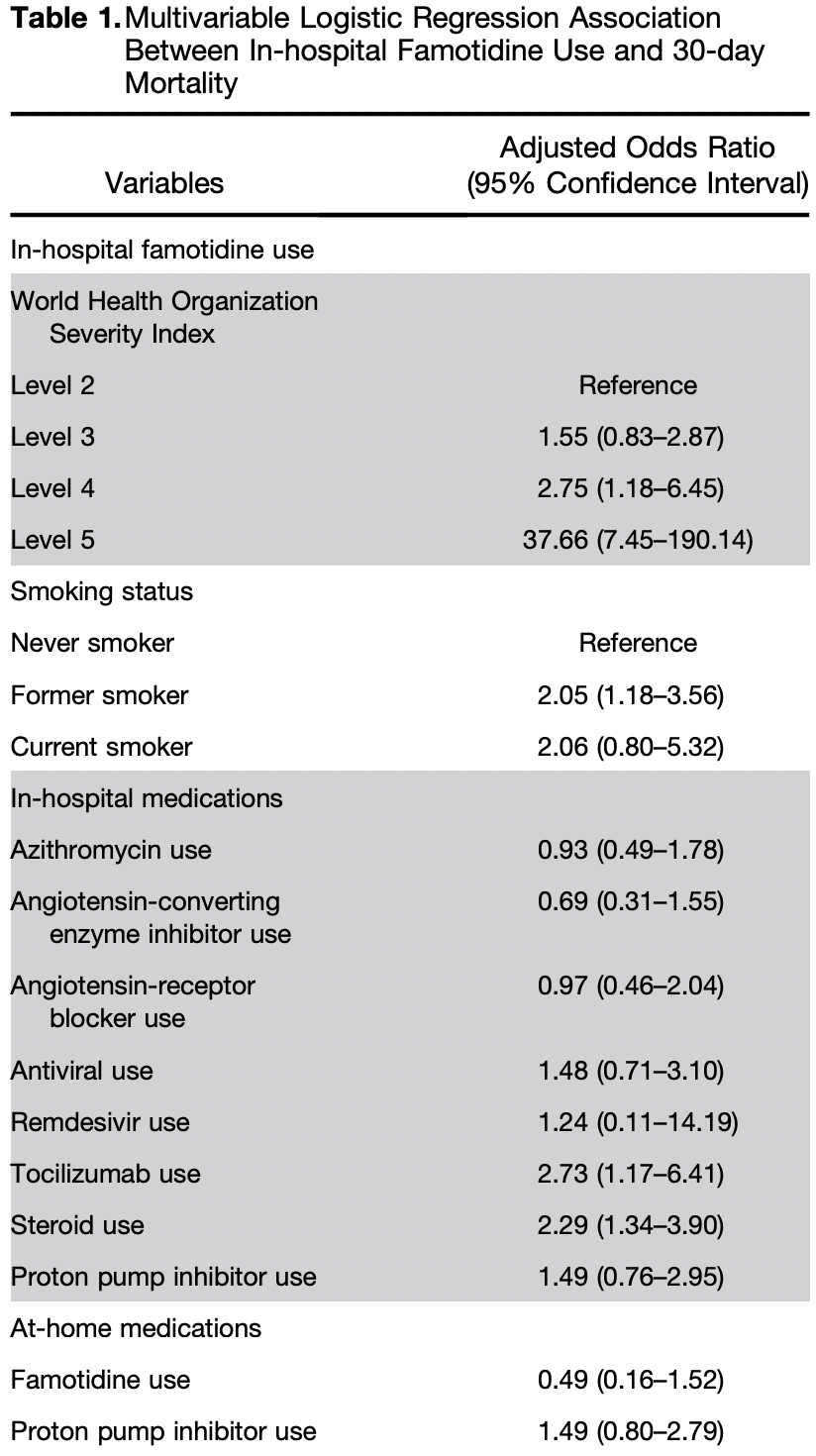
Retrospective 7,158 hospitalized COVID-19 patients in the USA, showing higher risk or mortality with in-hospital famotidine use, but lower risk when there was pre-existing at-home use, without statistical significance in both cases.
Standard of Care (SOC) for COVID-19 in the study country,
the USA, is very poor with very low average efficacy for approved treatments1.
Only expensive, high-profit treatments were approved for early treatment. Low-cost treatments were excluded, reducing the probability of early treatment due to access and cost barriers, and eliminating complementary and synergistic benefits seen with many low-cost treatments.
Study covers remdesivir and famotidine.
|
risk of death, 51.0% lower, OR 0.49, p = 0.22, treatment 351, control 6,807, adjusted per study, with home use, multivariable, day 30, RR approximated with OR.
|
|
risk of death, 59.0% higher, OR 1.59, p = 0.09, treatment 410, control 746, adjusted per study, hospital use only, multivariable, RR approximated with OR, late treatment result.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Yeramaneni et al., 28 Feb 2021, retrospective, USA, peer-reviewed, 6 authors, study period 11 February, 2020 - 8 May, 2020.
Famotidine Use Is Not Associated With 30-day Mortality: A Coarsened Exact Match Study in 7158 Hospitalized Patients With Coronavirus Disease 2019 From a Large Healthcare System
Gastroenterology, doi:10.1053/j.gastro.2020.10.011
P revious reports have found that in-hospital famotidine use in coronavirus disease 2019 (COVID-19) patients was associated with reduced risk of death or intubation. 1, 2 In 1 of these studies the authors proposed that famotidine inhibits the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) protease, 3-chymotrypsin-like protease, that is essential for breakdown of the immature SARS-CoV-2 protein particles that contribute to the inflammatory response seen in some COVID-19-infected individuals, 1 which in turn can lead to acute respiratory distress syndrome, multiorgan dysfunction, physiologic deterioration, and death. 3 In a global pandemic with a lack of US Food and Drug Administration-approved targeted therapeutic agents, identification and repurposing of well-established drugs with a proven track record of safety, affordability, and widespread availability are necessary. 4 The purpose of this study was to evaluate the reported protective effect of famotidine on mortality in hospitalized COVID-19 patients.
Supplementary Material Note: To access the supplementary material accompanying this article, visit the online version of Gastroenterology at www.gastrojournal.org and at https://doi.org/10.1053/ j.gastro.2020.10.011.
Conflicts of interest The authors disclose no conflicts.
References
Dr, Luther, Boulevard, Suite 800
Freedberg, None, Gastroenterology
Iacus, None, Political Analysis
Mather, None, Am J Gastroenterol
Rogosnitzky, None, JMIR Public Health Surveill
Wang, None, Lancet
Zhou, None, Lancet
DOI record:
{
"DOI": "10.1053/j.gastro.2020.10.011",
"ISSN": [
"0016-5085"
],
"URL": "http://dx.doi.org/10.1053/j.gastro.2020.10.011",
"alternative-id": [
"S0016508520352495"
],
"assertion": [
{
"label": "This article is maintained by",
"name": "publisher",
"value": "Elsevier"
},
{
"label": "Article Title",
"name": "articletitle",
"value": "Famotidine Use Is Not Associated With 30-day Mortality: A Coarsened Exact Match Study in 7158 Hospitalized Patients With Coronavirus Disease 2019 From a Large Healthcare System"
},
{
"label": "Journal Title",
"name": "journaltitle",
"value": "Gastroenterology"
},
{
"label": "CrossRef DOI link to publisher maintained version",
"name": "articlelink",
"value": "https://doi.org/10.1053/j.gastro.2020.10.011"
},
{
"label": "Content Type",
"name": "content_type",
"value": "article"
},
{
"label": "Copyright",
"name": "copyright",
"value": "© 2021 by the AGA Institute"
}
],
"author": [
{
"ORCID": "http://orcid.org/0000-0002-1411-2359",
"affiliation": [],
"authenticated-orcid": false,
"family": "Yeramaneni",
"given": "Samrat",
"sequence": "first"
},
{
"affiliation": [],
"family": "Doshi",
"given": "Pratik",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Sands",
"given": "Kenneth",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Cooper",
"given": "Mandelin",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Kurbegov",
"given": "Dax",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Fromell",
"given": "Gregg",
"sequence": "additional"
}
],
"container-title": [
"Gastroenterology"
],
"content-domain": {
"crossmark-restriction": true,
"domain": [
"clinicalkey.jp",
"clinicalkey.com",
"gastrojournal.org",
"clinicalkey.es",
"clinicalkey.com.au",
"clinicalkey.fr",
"elsevier.com",
"sciencedirect.com"
]
},
"created": {
"date-parts": [
[
2020,
10,
12
]
],
"date-time": "2020-10-12T05:38:45Z",
"timestamp": 1602481125000
},
"deposited": {
"date-parts": [
[
2021,
3,
20
]
],
"date-time": "2021-03-20T07:51:40Z",
"timestamp": 1616226700000
},
"funder": [
{
"DOI": "10.13039/100016646",
"doi-asserted-by": "publisher",
"name": "HCA Healthcare"
}
],
"indexed": {
"date-parts": [
[
2021,
12,
23
]
],
"date-time": "2021-12-23T22:27:20Z",
"timestamp": 1640298440528
},
"is-referenced-by-count": 17,
"issn-type": [
{
"type": "print",
"value": "0016-5085"
}
],
"issue": "3",
"issued": {
"date-parts": [
[
2021,
2
]
]
},
"journal-issue": {
"issue": "3",
"published-print": {
"date-parts": [
[
2021,
2
]
]
}
},
"language": "en",
"license": [
{
"URL": "https://www.elsevier.com/tdm/userlicense/1.0/",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2021,
2,
1
]
],
"date-time": "2021-02-01T00:00:00Z",
"timestamp": 1612137600000
}
}
],
"link": [
{
"URL": "https://api.elsevier.com/content/article/PII:S0016508520352495?httpAccept=text/xml",
"content-type": "text/xml",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://api.elsevier.com/content/article/PII:S0016508520352495?httpAccept=text/plain",
"content-type": "text/plain",
"content-version": "vor",
"intended-application": "text-mining"
}
],
"member": "78",
"original-title": [],
"page": "919-921.e3",
"prefix": "10.1053",
"published": {
"date-parts": [
[
2021,
2
]
]
},
"published-print": {
"date-parts": [
[
2021,
2
]
]
},
"publisher": "Elsevier BV",
"reference": [
{
"DOI": "10.1053/j.gastro.2020.05.053",
"author": "Freedberg",
"doi-asserted-by": "crossref",
"first-page": "1129",
"journal-title": "Gastroenterology",
"key": "10.1053/j.gastro.2020.10.011_bib1",
"volume": "159",
"year": "2020"
},
{
"DOI": "10.14309/ajg.0000000000000832",
"author": "Mather",
"doi-asserted-by": "crossref",
"first-page": "1617",
"journal-title": "Am J Gastroenterol",
"key": "10.1053/j.gastro.2020.10.011_bib2",
"volume": "115",
"year": "2020"
},
{
"DOI": "10.1016/S0140-6736(20)30566-3",
"author": "Zhou",
"doi-asserted-by": "crossref",
"first-page": "1054",
"journal-title": "Lancet",
"key": "10.1053/j.gastro.2020.10.011_bib3",
"volume": "395",
"year": "2020"
},
{
"DOI": "10.2196/19199",
"author": "Rogosnitzky",
"doi-asserted-by": "crossref",
"journal-title": "JMIR Public Health Surveill",
"key": "10.1053/j.gastro.2020.10.011_bib4",
"volume": "6",
"year": "2020"
},
{
"DOI": "10.1093/pan/mpr013",
"author": "Iacus",
"doi-asserted-by": "crossref",
"first-page": "1",
"journal-title": "Political Analysis",
"key": "10.1053/j.gastro.2020.10.011_bib5",
"volume": "20",
"year": "2012"
},
{
"DOI": "10.1016/S0140-6736(20)31022-9",
"author": "Wang",
"doi-asserted-by": "crossref",
"first-page": "1569",
"journal-title": "Lancet",
"key": "10.1053/j.gastro.2020.10.011_bib6",
"volume": "395",
"year": "2020"
},
{
"journal-title": "N Engl J Med",
"key": "10.1053/j.gastro.2020.10.011_bib7",
"year": "2020"
}
],
"reference-count": 7,
"references-count": 7,
"relation": {},
"score": 1,
"short-container-title": [
"Gastroenterology"
],
"short-title": [],
"source": "Crossref",
"subject": [
"Gastroenterology",
"Hepatology"
],
"subtitle": [],
"title": [
"Famotidine Use Is Not Associated With 30-day Mortality: A Coarsened Exact Match Study in 7158 Hospitalized Patients With Coronavirus Disease 2019 From a Large Healthcare System"
],
"type": "journal-article",
"update-policy": "http://dx.doi.org/10.1016/elsevier_cm_policy",
"volume": "160"
}
