A Multicenter, Randomized, Double-blind, Placebo-controlled, Phase 2 Study to Investigate the Efficacy and Safety of BGE-175 in Hospitalized Adults With COVID-19
et al., NCT04705597, NCT04705597, May 2022
RCT 194 hospitalized COVID-19 patients showing no significant differences with asapiprant (BGE-175).
Standard of Care (SOC) for COVID-19 in the study country,
the USA, is very poor with very low average efficacy for approved treatments1.
Only expensive, high-profit treatments were approved for early treatment. Low-cost treatments were excluded, reducing the probability of early treatment due to access and cost barriers, and eliminating complementary and synergistic benefits seen with many low-cost treatments.
|
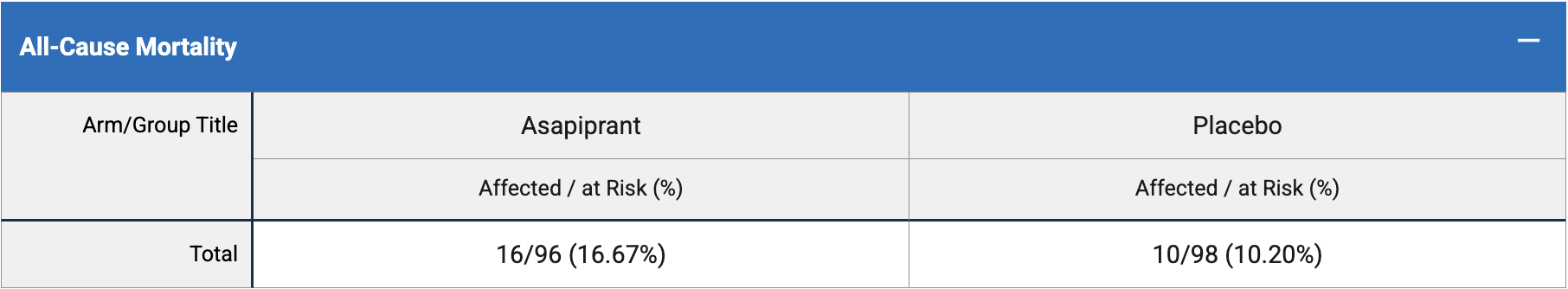
risk of death, 63.3% higher, RR 1.63, p = 0.21, treatment 16 of 96 (16.7%), control 10 of 98 (10.2%).
|
|
risk of ICU admission, 32.1% higher, RR 1.32, p = 0.37, treatment 22 of 96 (22.9%), control 17 of 98 (17.3%).
|
|
risk of severe case, 24.0% higher, RR 1.24, p = 0.36, treatment 34 of 96 (35.4%), control 28 of 98 (28.6%).
|
|
time to improvement, 33.3% higher, relative time 1.33, p = 0.17, treatment 8.0 [6.0-10.0] n=96, control 6.0 [5.0-9.0] n=98.
|
|
risk of progression, 23.0% higher, RR 1.23, p = 0.43, treatment 31 of 94 (33.0%), control 26 of 97 (26.8%), death or respiratory failure.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Wilkerson et al., 19 May 2022, Double Blind Randomized Controlled Trial, placebo-controlled, USA, preprint, 1 author, critical, trial NCT04705597 (history).
Contact: patrick@bioagelabs.com.