Decreased Plasma Concentration of Hydrogen Sulfide in Hospitalized COVID-19 Patients: A Novel Determinant of Mortality?
et al., Antioxidants, doi:10.3390/antiox15030307, Feb 2026
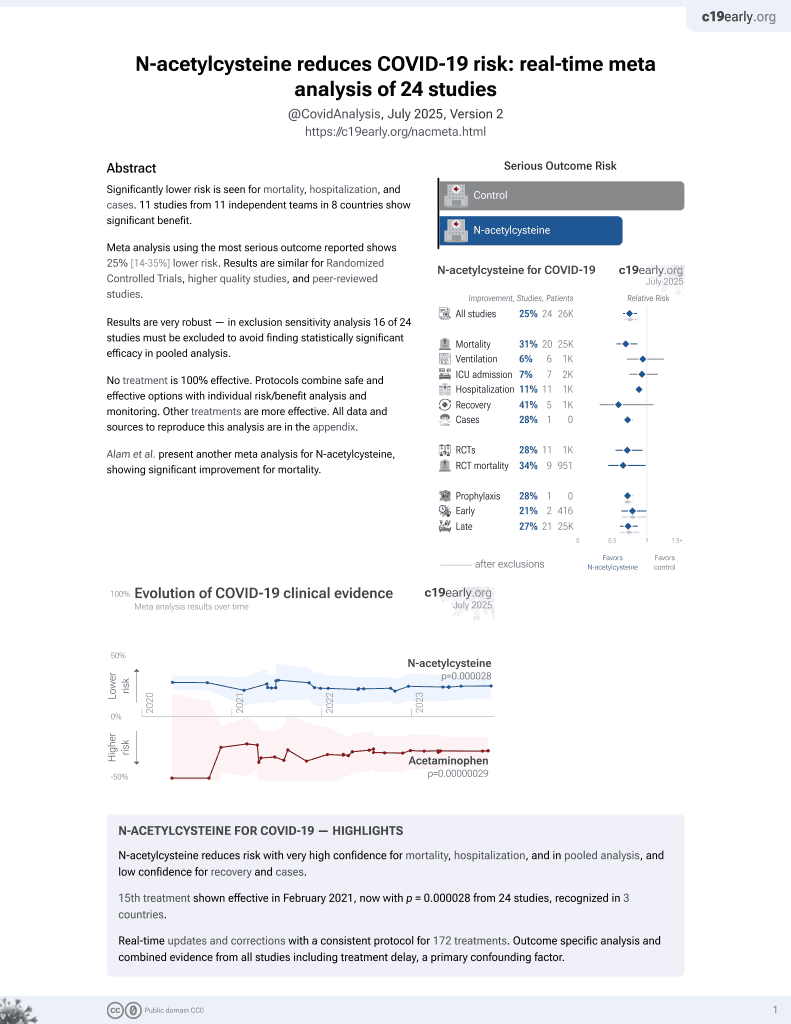
15th treatment shown to reduce risk in
February 2021, now with p = 0.0000032 from 25 studies, recognized in 3 countries.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
Retrospective 80 hospitalized COVID-19 patients (40 survivors, 40 non-survivors) and 40 non-COVID-19 controls showing that lower plasma hydrogen sulfide (H2S) levels at admission were significantly associated with in-hospital mortality. In vitro experiments demonstrated that N-acetylcysteine (NAC) dose-dependently reduced ROS and lipid peroxidation in THP-1 and small airway epithelial cells, and increased both GSH and H2S production, suggesting NAC may be beneficial in COVID-19 patients with inadequate H2S response.
15 preclinical studies support the efficacy of N-acetylcysteine for COVID-19:
Severe COVID-19 is marked by endotheliopathy with elevated von Willebrand factor (VWF) levels and platelet/VWF-rich microthrombi; N-acetylcysteine can reduce VWF multimers and lyse VWF-dependent clots in vivo, potentially helping to alleviate thrombosis associated with COVID-1911-13.
N-acetylcysteine shows dose-dependent inhibition of SARS-CoV-25,8,10 , shows anti-inflammatory and immunomodulatory effects against SARS-CoV-2-induced immune responses in combination with bromelain7, suppressed virus-induced reactive oxygen species and blocked viral replication in a humanized mouse model and in human lung cells6, may limit COVID-19 induced cardiac damage by boosting cellular antioxidant defenses and potentially mitigating the oxidative stress caused by spike protein-induced ROS production in cardiac fibroblasts4, and reduces disulfide bonds in proteins and exhibits antioxidant properties that may inhibit viral replication and modulate inflammatory responses3.
NAC may be beneficial for COVID-19 by replenishing glutathione stores and reinforcing the glutathione peroxidase-4 pathway to inhibit ferroptosis, an oxidative stress-induced cell death pathway implicated in COVID-1914.
NAC reinforces glutathione levels, reduces ROS, and minimizes ferroptosis and cytokine storm15.
1.
Agamah et al., Network-based multi-omics-disease-drug associations reveal drug repurposing candidates for COVID-19 disease phases, ScienceOpen, doi:10.58647/DRUGARXIV.PR000010.v1.
2.
Stranieri et al., Decreased Plasma Concentration of Hydrogen Sulfide in Hospitalized COVID-19 Patients: A Novel Determinant of Mortality?, Antioxidants, doi:10.3390/antiox15030307.
3.
Reis et al., Antiviral effect of Bromelain combined with acetylcysteine against SARS-CoV-2 Omicron variant, Scientific Reports, doi:10.1038/s41598-025-92242-y.
4.
Van Tin et al., Spike Protein of SARS-CoV-2 Activates Cardiac Fibrogenesis through NLRP3 Inflammasomes and NF-κB Signaling, Cells, doi:10.3390/cells13161331.
5.
Chaopreecha et al., Andrographolide attenuates SARS-CoV-2 infection via an up-regulation of glutamate-cysteine ligase catalytic subunit (GCLC), Phytomedicine, doi:10.1016/j.phymed.2024.156279.
6.
Frasson et al., Identification of druggable host dependency factors shared by multiple SARS-CoV-2 variants of concern, Journal of Molecular Cell Biology. doi:10.1093/jmcb/mjae004, academic.oup.com/jmcb/advance-article/doi/10.1093/jmcb/mjae004/7596546.
7.
Ferreira et al., Taming the SARS-CoV-2-mediated proinflammatory response with BromAc®, Frontiers in Immunology, doi:10.3389/fimmu.2023.1308477.
8.
La Maestra et al., Inhibition of the Cell Uptake of Delta and Omicron SARS-CoV-2 Pseudoviruses by N-Acetylcysteine Irrespective of the Oxidoreductive Environment, Cells, doi:10.3390/cells11203313.
9.
Goc et al., Inhibitory effects of specific combination of natural compounds against SARS-CoV-2 and its Alpha, Beta, Gamma, Delta, Kappa, and Mu variants, European Journal of Microbiology and Immunology, doi:10.1556/1886.2021.00022.
10.
Akhter et al., The Combination of Bromelain and Acetylcysteine (BromAc) Synergistically Inactivates SARS-CoV-2, Viruses, doi:10.3390/v13030425.
11.
Martinez de Lizarrondo et al., Potent Thrombolytic Effect of N-Acetylcysteine on Arterial Thrombi, Circulation, doi:10.1161/CIRCULATIONAHA.117.027290.
12.
Chen et al., N-acetylcysteine reduces the size and activity of von Willebrand factor in human plasma and mice, Journal of Clinical Investigation, doi:10.1172/JCI41062.
13.
Goshua et al., Endotheliopathy in COVID-19-associated coagulopathy: evidence from a single-centre, cross-sectional study, The Lancet Haematology, doi:10.1016/S2352-3026(20)30216-7.
Stranieri et al., 28 Feb 2026, retrospective, USA, peer-reviewed, 9 authors, study period April 2020 - May 2021.
Contact: annamaria.frattapasini@univr.it (corresponding author), chiara.stranieri@univr.it, edoardogiuseppe.dileo@univr.it, arianna.barbieri@studenti.univr.it, antonio.randon@studenti.univr.it, luciano.cominacini@univr.it, elisa.danese@univr.it, roberta.poffe@aovr.veneto.it, laura.pighi@univr.it.
Abstract:
Article
Decreased Plasma Concentration of Hydrogen Sulfide in Hospitalized COVID-19 Patients: A Novel Determinant of Mortality?
Chiara Stranieri 1 , Edoardo Giuseppe Di Leo 1 , Elisa Danese 2 , Roberta Poffe 2 , Arianna Barbieri 1 , Laura Pighi 2 , Antonio Randon 1 , Luciano Cominacini 1 and Anna Maria Fratta Pasini 1, *
- 1 Section of Internal Medicine D, Department of Medicine, University of Verona, P. le L.A. Scuro, 37134 Verona, Italy; chiara.stranieri@univr.it (C.S.); edoardogiuseppe.dileo@univr.it (E.G.D.L.); arianna.barbieri@studenti.univr.it (A.B.); antonio.randon@studenti.univr.it (A.R.); luciano.cominacini@univr.it (L.C.)
- 2 Section of Clinical Biochemistry, Department of Engineering for Innovation Medicine, University of Verona, 37134 Verona, Italy; elisa.danese@univr.it (E.D.); roberta.poffe@aovr.veneto.it (R.P.); laura.pighi@univr.it (L.P.)
* Correspondence: annamaria.frattapasini@univr.it; Tel.: +39-045-8124749
Abstract
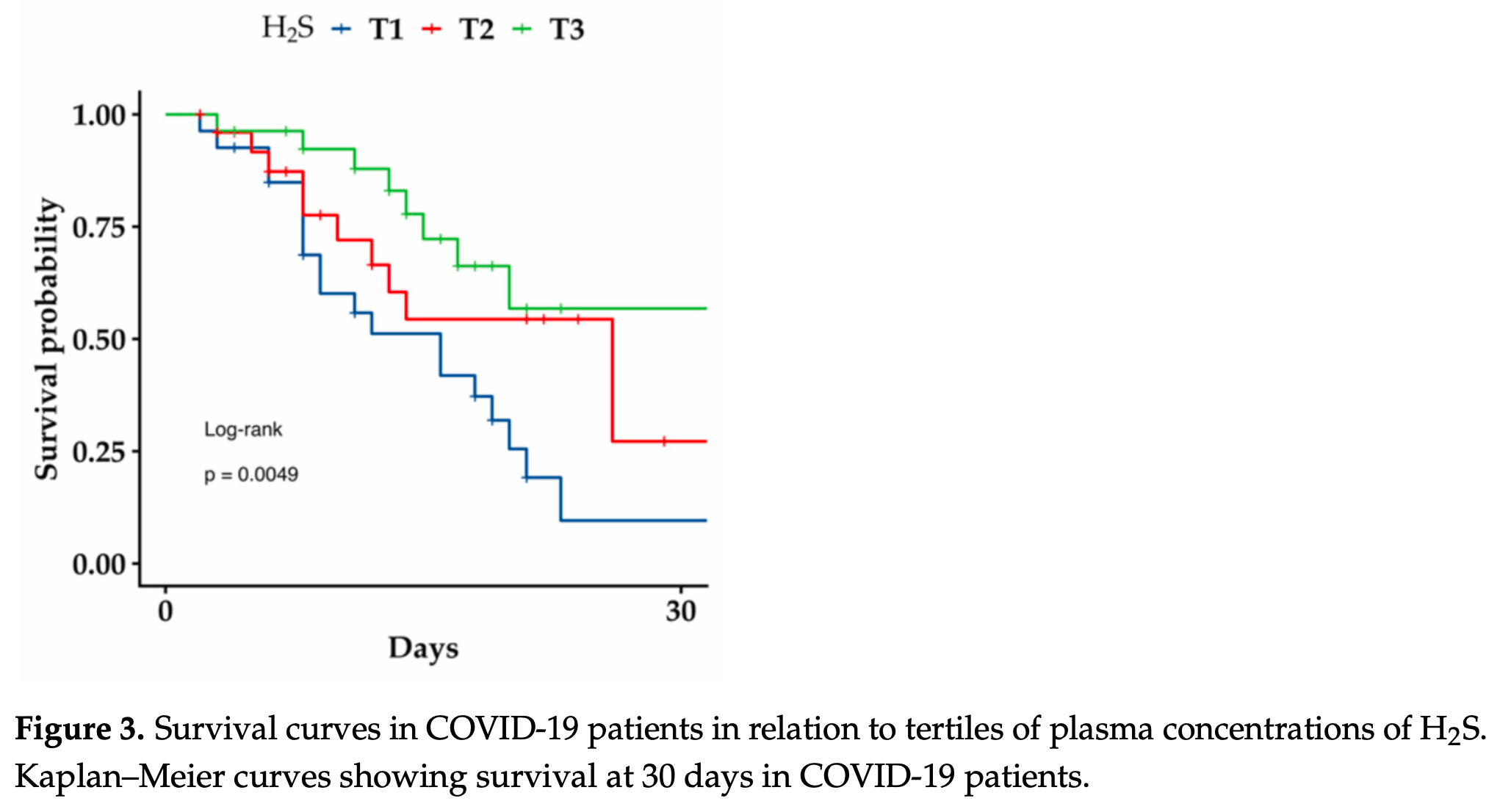
In this study, we first focused on measuring H2S and oxidative stress as indicators of in-hospital mortality observed within 24 h from admission in hospitalized non-survivor and survivor patients affected by COVID-19. Then, we analyzed whether N-acetylcysteine (NAC) can increase H2S and GSH concentrations in different cell lines. H2S levels were significantly increased in all COVID-19 patients (both survivors and non-survivors) compared to non-COVID-19 subjects ( p = 0.0016), but non-survivors showed significantly lower H2S plasma levels than survivors ( p = 0.008). Oxidative stress measured as circulating malondialdehyde (MDA) resulted in lower levels in non-COVID-19 subjects than in the two COVID-19 patient groups ( p = 0.03). However, non-survivors had significantly higher plasma MDA than survivors ( p = 0.0001). A Kaplan-Meier curve for H2S indicates a markedly reduced survival probability in COVID-19 patients with lower H2S levels (logrank p = 0.004). NAC activity significantly reduced reactive oxygen species and lipid peroxidation induced by tert-butyl hydroperoxide in cultured cells ( p from <0.01 to <0.001). Furthermore, NAC increased the cellular production of H2S ( p < 0.01) and GSH ( p < 0.01). These findings indicate the important prognostic role of H2S in COVID-19 patients at hospital admission and that NAC might be helpful in all clinical situations characterized by low levels of H2S.
Keywords: COVID-19; glutathione; hydrogen sulfide; N-acetylcysteine; oxidative stress
DOI record:
{
"DOI": "10.3390/antiox15030307",
"ISSN": [
"2076-3921"
],
"URL": "http://dx.doi.org/10.3390/antiox15030307",
"abstract": "<jats:p>In this study, we first focused on measuring H2S and oxidative stress as indicators of in-hospital mortality observed within 24 h from admission in hospitalized non-survivor and survivor patients affected by COVID-19. Then, we analyzed whether N-acetylcysteine (NAC) can increase H2S and GSH concentrations in different cell lines. H2S levels were significantly increased in all COVID-19 patients (both survivors and non-survivors) compared to non-COVID-19 subjects (p = 0.0016), but non-survivors showed significantly lower H2S plasma levels than survivors (p = 0.008). Oxidative stress measured as circulating malondialdehyde (MDA) resulted in lower levels in non-COVID-19 subjects than in the two COVID-19 patient groups (p = 0.03). However, non-survivors had significantly higher plasma MDA than survivors (p = 0.0001). A Kaplan–Meier curve for H2S indicates a markedly reduced survival probability in COVID-19 patients with lower H2S levels (log-rank p = 0.004). NAC activity significantly reduced reactive oxygen species and lipid peroxidation induced by tert-butyl hydroperoxide in cultured cells (p from <0.01 to <0.001). Furthermore, NAC increased the cellular production of H2S (p < 0.01) and GSH (p < 0.01). These findings indicate the important prognostic role of H2S in COVID-19 patients at hospital admission and that NAC might be helpful in all clinical situations characterized by low levels of H2S.</jats:p>",
"alternative-id": [
"antiox15030307"
],
"author": [
{
"ORCID": "https://orcid.org/0009-0005-4377-6647",
"affiliation": [
{
"name": "Section of Internal Medicine D, Department of Medicine, University of Verona, P. le L.A. Scuro, 37134 Verona, Italy"
}
],
"authenticated-orcid": false,
"family": "Stranieri",
"given": "Chiara",
"sequence": "first"
},
{
"ORCID": "https://orcid.org/0009-0003-2742-1358",
"affiliation": [
{
"name": "Section of Internal Medicine D, Department of Medicine, University of Verona, P. le L.A. Scuro, 37134 Verona, Italy"
}
],
"authenticated-orcid": false,
"family": "Di Leo",
"given": "Edoardo Giuseppe",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0002-2454-0410",
"affiliation": [
{
"name": "Section of Clinical Biochemistry, Department of Engineering for Innovation Medicine, University of Verona, 37134 Verona, Italy"
}
],
"authenticated-orcid": false,
"family": "Danese",
"given": "Elisa",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Section of Clinical Biochemistry, Department of Engineering for Innovation Medicine, University of Verona, 37134 Verona, Italy"
}
],
"family": "Poffe",
"given": "Roberta",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Section of Internal Medicine D, Department of Medicine, University of Verona, P. le L.A. Scuro, 37134 Verona, Italy"
}
],
"family": "Barbieri",
"given": "Arianna",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0003-3603-9495",
"affiliation": [
{
"name": "Section of Clinical Biochemistry, Department of Engineering for Innovation Medicine, University of Verona, 37134 Verona, Italy"
}
],
"authenticated-orcid": false,
"family": "Pighi",
"given": "Laura",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0009-0003-7433-6551",
"affiliation": [
{
"name": "Section of Internal Medicine D, Department of Medicine, University of Verona, P. le L.A. Scuro, 37134 Verona, Italy"
}
],
"authenticated-orcid": false,
"family": "Randon",
"given": "Antonio",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Section of Internal Medicine D, Department of Medicine, University of Verona, P. le L.A. Scuro, 37134 Verona, Italy"
}
],
"family": "Cominacini",
"given": "Luciano",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0001-7661-7785",
"affiliation": [
{
"name": "Section of Internal Medicine D, Department of Medicine, University of Verona, P. le L.A. Scuro, 37134 Verona, Italy"
}
],
"authenticated-orcid": false,
"family": "Fratta Pasini",
"given": "Anna Maria",
"sequence": "additional"
}
],
"container-title": "Antioxidants",
"container-title-short": "Antioxidants",
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2026,
3,
2
]
],
"date-time": "2026-03-02T14:55:01Z",
"timestamp": 1772463301000
},
"deposited": {
"date-parts": [
[
2026,
3,
2
]
],
"date-time": "2026-03-02T15:14:15Z",
"timestamp": 1772464455000
},
"funder": [
{
"name": "University of Verona, Italy, School of Medicine"
}
],
"indexed": {
"date-parts": [
[
2026,
3,
2
]
],
"date-time": "2026-03-02T16:00:24Z",
"timestamp": 1772467224467,
"version": "3.50.1"
},
"is-referenced-by-count": 0,
"issue": "3",
"issued": {
"date-parts": [
[
2026,
2,
28
]
]
},
"journal-issue": {
"issue": "3",
"published-online": {
"date-parts": [
[
2026,
3
]
]
}
},
"language": "en",
"license": [
{
"URL": "https://creativecommons.org/licenses/by/4.0/",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2026,
2,
28
]
],
"date-time": "2026-02-28T00:00:00Z",
"timestamp": 1772236800000
}
}
],
"link": [
{
"URL": "https://www.mdpi.com/2076-3921/15/3/307/pdf",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "1968",
"original-title": [],
"page": "307",
"prefix": "10.3390",
"published": {
"date-parts": [
[
2026,
2,
28
]
]
},
"published-online": {
"date-parts": [
[
2026,
2,
28
]
]
},
"publisher": "MDPI AG",
"reference": [
{
"key": "ref_1",
"unstructured": "(2025, October 21). Weekly Epidemiological Update on COVID-19-1 September 2023. Available online: https://www.who.int/publications/m/item/weekly-epidemiological-update-on-covid-19---1-september-2023."
},
{
"DOI": "10.1152/physrev.00028.2021",
"article-title": "Physiological Roles of Hydrogen Sulfide in Mammalian Cells, Tissues, and Organs",
"author": "Cirino",
"doi-asserted-by": "crossref",
"first-page": "31",
"journal-title": "Physiol. Rev.",
"key": "ref_2",
"volume": "103",
"year": "2023"
},
{
"DOI": "10.1074/jbc.M808026200",
"article-title": "H2S Biogenesis by Human Cystathionine Gamma-Lyase Leads to the Novel Sulfur Metabolites Lanthionine and Homolanthionine and Is Responsive to the Grade of Hyperhomocysteinemia",
"author": "Chiku",
"doi-asserted-by": "crossref",
"first-page": "11601",
"journal-title": "J. Biol. Chem.",
"key": "ref_3",
"volume": "284",
"year": "2009"
},
{
"DOI": "10.1074/jbc.M109.010868",
"article-title": "Relative Contributions of Cystathionine β-Synthase and γ-Cystathionase to H2S Biogenesis via Alternative Trans-Sulfuration Reactions",
"author": "Singh",
"doi-asserted-by": "crossref",
"first-page": "22457",
"journal-title": "J. Biol. Chem.",
"key": "ref_4",
"volume": "284",
"year": "2009"
},
{
"DOI": "10.3390/biom14070740",
"doi-asserted-by": "crossref",
"key": "ref_5",
"unstructured": "Sun, X., Wu, S., Mao, C., Qu, Y., Xu, Z., Xie, Y., Jiang, D., and Song, Y. (2024). Therapeutic Potential of Hydrogen Sulfide in Ischemia and Reperfusion Injury. Biomolecules, 14."
},
{
"DOI": "10.3389/fendo.2022.934231",
"doi-asserted-by": "crossref",
"key": "ref_6",
"unstructured": "Zhu, C., Liu, Q., Li, X., Wei, R., Ge, T., Zheng, X., Li, B., Liu, K., and Cui, R. (2022). Hydrogen Sulfide: A New Therapeutic Target in Vascular Diseases. Front. Endocrinol., 13."
},
{
"DOI": "10.3390/ijms19124121",
"doi-asserted-by": "crossref",
"key": "ref_7",
"unstructured": "Abdulle, A.E., Van Goor, H., and Mulder, D.J. (2018). Hydrogen Sulfide: A Therapeutic Option in Systemic Sclerosis. Int. J. Mol. Sci., 19."
},
{
"DOI": "10.1038/s42003-019-0431-5",
"article-title": "Non-Enzymatic Hydrogen Sulfide Production from Cysteine in Blood Is Catalyzed by Iron and Vitamin B6",
"author": "Yang",
"doi-asserted-by": "crossref",
"first-page": "194",
"journal-title": "Commun. Biol.",
"key": "ref_8",
"volume": "2",
"year": "2019"
},
{
"DOI": "10.1089/ars.2020.8247",
"article-title": "N-Acetylcysteine and Hydrogen Sulfide in Coronavirus Disease 2019",
"author": "Bourgonje",
"doi-asserted-by": "crossref",
"first-page": "1207",
"journal-title": "Antioxid. Redox Signal.",
"key": "ref_9",
"volume": "35",
"year": "2021"
},
{
"DOI": "10.1371/journal.ppat.1013164",
"doi-asserted-by": "crossref",
"key": "ref_10",
"unstructured": "Agrawal, R., Pal, V.K., Suhas, K.S., Menon, G.J., Singh, I.R., Malhotra, N., Naren, C.S., Ganesh, K., Rajmani, R.S., and Seshasayee, A.S.N. (2025). Hydrogen Sulfide (H2S) Coordinates Redox Balance, Carbon Metabolism, and Mitochondrial Bioenergetics to Suppress SARS-CoV-2 Infection. PLoS Pathog., 21."
},
{
"DOI": "10.7150/thno.48076",
"article-title": "SARS-CoV-2 Pandemic and Research Gaps: Understanding SARS-CoV-2 Interaction with the ACE2 Receptor and Implications for Therapy",
"author": "Datta",
"doi-asserted-by": "crossref",
"first-page": "7448",
"journal-title": "Theranostics",
"key": "ref_11",
"volume": "10",
"year": "2020"
},
{
"DOI": "10.1161/CIRCRESAHA.120.317015",
"article-title": "Angiotensin-Converting Enzyme 2: SARS-CoV-2 Receptor and Regulator of the Renin-Angiotensin System",
"author": "Gheblawi",
"doi-asserted-by": "crossref",
"first-page": "1456",
"journal-title": "Circ. Res.",
"key": "ref_12",
"volume": "126",
"year": "2020"
},
{
"DOI": "10.1007/s10096-020-04138-6",
"article-title": "COVID-19: Angiotensin-Converting Enzyme 2 (ACE2) Expression and Tissue Susceptibility to SARS-CoV-2 Infection",
"author": "Beyerstedt",
"doi-asserted-by": "crossref",
"first-page": "905",
"journal-title": "Eur. J. Clin. Microbiol. Infect. Dis.",
"key": "ref_13",
"volume": "40",
"year": "2021"
},
{
"DOI": "10.1016/j.mce.2024.112309",
"article-title": "Angiotensin II: Role in Oxidative Stress, Endothelial Dysfunction, and Diseases",
"author": "Ajoolabady",
"doi-asserted-by": "crossref",
"first-page": "112309",
"journal-title": "Mol. Cell. Endocrinol.",
"key": "ref_14",
"volume": "592",
"year": "2024"
},
{
"DOI": "10.3390/cells11213336",
"doi-asserted-by": "crossref",
"key": "ref_15",
"unstructured": "Bhullar, S.K., and Dhalla, N.S. (2022). Angiotensin II-Induced Signal Transduction Mechanisms for Cardiac Hypertrophy. Cells, 11."
},
{
"DOI": "10.3389/fphys.2017.00782",
"doi-asserted-by": "crossref",
"key": "ref_16",
"unstructured": "Lin, Y., Zeng, H., Gao, L., Gu, T., Wang, C., and Zhang, H. (2017). Hydrogen Sulfide Attenuates Atherosclerosis in a Partially Ligated Carotid Artery Mouse Model via Regulating Angiotensin Converting Enzyme 2 Expression. Front. Physiol., 8."
},
{
"DOI": "10.1073/pnas.2003138117",
"article-title": "Cell Entry Mechanisms of SARS-CoV-2",
"author": "Shang",
"doi-asserted-by": "crossref",
"first-page": "11727",
"journal-title": "Proc. Natl. Acad. Sci. USA",
"key": "ref_17",
"volume": "117",
"year": "2020"
},
{
"DOI": "10.1152/ajpcell.00187.2020",
"article-title": "H2S as a Potential Defense against COVID-19?",
"author": "Yang",
"doi-asserted-by": "crossref",
"first-page": "C244",
"journal-title": "Am. J. Physiol.-Cell Physiol.",
"key": "ref_18",
"volume": "319",
"year": "2020"
},
{
"DOI": "10.1038/srep41029",
"article-title": "Broad-Range Antiviral Activity of Hydrogen Sulfide Against Highly Pathogenic RNA Viruses",
"author": "Bazhanov",
"doi-asserted-by": "crossref",
"first-page": "41029",
"journal-title": "Sci. Rep.",
"key": "ref_19",
"volume": "7",
"year": "2017"
},
{
"DOI": "10.1002/iub.1740",
"article-title": "Hydrogen Sulfide in Physiology and Pathogenesis of Bacteria and Viruses",
"author": "Pal",
"doi-asserted-by": "crossref",
"first-page": "393",
"journal-title": "IUBMB Life",
"key": "ref_20",
"volume": "70",
"year": "2018"
},
{
"DOI": "10.1128/JVI.00264-15",
"article-title": "Role of Hydrogen Sulfide in Paramyxovirus Infections",
"author": "Li",
"doi-asserted-by": "crossref",
"first-page": "5557",
"journal-title": "J. Virol.",
"key": "ref_21",
"volume": "89",
"year": "2015"
},
{
"DOI": "10.3390/v10050249",
"doi-asserted-by": "crossref",
"key": "ref_22",
"unstructured": "Bazhanov, N., Ivanciuc, T., Wu, H., Garofalo, M., Kang, J., Xian, M., and Casola, A. (2018). Thiol-Activated Hydrogen Sulfide Donors Antiviral and Anti-Inflammatory Activity in Respiratory Syncytial Virus Infection. Viruses, 10."
},
{
"DOI": "10.1038/s41392-020-0191-1",
"article-title": "Immune Response in COVID-19: Addressing a Pharmacological Challenge by Targeting Pathways Triggered by SARS-CoV-2",
"author": "Catanzaro",
"doi-asserted-by": "crossref",
"first-page": "84",
"journal-title": "Signal Transduct. Target. Ther.",
"key": "ref_23",
"volume": "5",
"year": "2020"
},
{
"DOI": "10.3390/cells13161331",
"doi-asserted-by": "crossref",
"key": "ref_24",
"unstructured": "Van Tin, H., Rethi, L., Higa, S., Kao, Y.-H., and Chen, Y.-J. (2024). Spike Protein of SARS-CoV-2 Activates Cardiac Fibrogenesis through NLRP3 Inflammasomes and NF-κB Signaling. Cells, 13."
},
{
"DOI": "10.1042/CS20190514",
"article-title": "Endogenous Hydrogen Sulfide Sulfhydrates IKKβ at Cysteine 179 to Control Pulmonary Artery Endothelial Cell Inflammation",
"author": "Zhang",
"doi-asserted-by": "crossref",
"first-page": "2045",
"journal-title": "Clin. Sci.",
"key": "ref_25",
"volume": "133",
"year": "2019"
},
{
"DOI": "10.3390/ijms23094818",
"doi-asserted-by": "crossref",
"key": "ref_26",
"unstructured": "Zhao, H., Liu, H., Yang, Y., and Wang, H. (2022). The Role of H2S Regulating NLRP3 Inflammasome in Diabetes. Int. J. Mol. Sci., 23."
},
{
"DOI": "10.33549/physiolres.934928",
"article-title": "Hydrogen Sulfide Alleviates Lipopolysaccharide-Induced Myocardial Injury through TLR4-NLRP3 Pathway",
"author": "Xia",
"doi-asserted-by": "crossref",
"first-page": "15",
"journal-title": "Physiol. Res.",
"key": "ref_27",
"volume": "72",
"year": "2023"
},
{
"DOI": "10.3390/antiox12081644",
"doi-asserted-by": "crossref",
"key": "ref_28",
"unstructured": "Huang, D., Jing, G., and Zhu, S. (2023). Regulation of Mitochondrial Respiration by Hydrogen Sulfide. Antioxid. Basel Switz., 12."
},
{
"DOI": "10.1016/j.redox.2020.101772",
"article-title": "Effects of Hydrogen Sulfide on Mitochondrial Function and Cellular Bioenergetics",
"author": "Paul",
"doi-asserted-by": "crossref",
"first-page": "101772",
"journal-title": "Redox Biol.",
"key": "ref_29",
"volume": "38",
"year": "2021"
},
{
"DOI": "10.1016/j.bbrc.2013.02.131",
"article-title": "Oxidative Stress Suppresses the Cellular Bioenergetic Effect of the 3-Mercaptopyruvate Sulfurtransferase/Hydrogen Sulfide Pathway",
"author": "Asimakopoulou",
"doi-asserted-by": "crossref",
"first-page": "401",
"journal-title": "Biochem. Biophys. Res. Commun.",
"key": "ref_30",
"volume": "433",
"year": "2013"
},
{
"DOI": "10.1073/pnas.1717877115",
"article-title": "Golgi Stress Response Reprograms Cysteine Metabolism to Confer Cytoprotection in Huntington’s Disease",
"author": "Sbodio",
"doi-asserted-by": "crossref",
"first-page": "780",
"journal-title": "Proc. Natl. Acad. Sci. USA",
"key": "ref_31",
"volume": "115",
"year": "2018"
},
{
"DOI": "10.1089/ars.2019.7824",
"article-title": "Golgi Stress Response, Hydrogen Sulfide Metabolism, and Intracellular Calcium Homeostasis",
"author": "Zhang",
"doi-asserted-by": "crossref",
"first-page": "583",
"journal-title": "Antioxid. Redox Signal.",
"key": "ref_32",
"volume": "32",
"year": "2020"
},
{
"DOI": "10.1074/jbc.C116.742213",
"article-title": "Heme-Dependent Metabolite Switching Regulates H2S Synthesis in Response to Endoplasmic Reticulum (ER) Stress",
"author": "Kabil",
"doi-asserted-by": "crossref",
"first-page": "16418",
"journal-title": "J. Biol. Chem.",
"key": "ref_33",
"volume": "291",
"year": "2016"
},
{
"DOI": "10.1016/j.cell.2014.11.048",
"article-title": "Endogenous Hydrogen Sulfide Production Is Essential for Dietary Restriction Benefits",
"author": "Hine",
"doi-asserted-by": "crossref",
"first-page": "132",
"journal-title": "Cell",
"key": "ref_34",
"volume": "160",
"year": "2015"
},
{
"DOI": "10.1016/j.neuint.2010.01.004",
"article-title": "Glutathione Depletion Causes a JNK and p38MAPK-Mediated Increase in Expression of Cystathionine-γ-Lyase and Upregulation of the Transsulfuration Pathway in C6 Glioma Cells",
"author": "Kandil",
"doi-asserted-by": "crossref",
"first-page": "611",
"journal-title": "Neurochem. Int.",
"key": "ref_35",
"volume": "56",
"year": "2010"
},
{
"article-title": "Oxidative Stress as a Signal to Up-Regulate Gamma-Cystathionase in the Fetal-to-Neonatal Transition in Rats",
"author": "Pereda",
"first-page": "1010",
"journal-title": "Cell. Mol. Biol.",
"key": "ref_36",
"volume": "53",
"year": "2007"
},
{
"DOI": "10.1016/j.clim.2020.108544",
"article-title": "Therapeutic Blockade of Inflammation in Severe COVID-19 Infection with Intravenous N-Acetylcysteine",
"author": "Ibrahim",
"doi-asserted-by": "crossref",
"first-page": "108544",
"journal-title": "Clin. Immunol.",
"key": "ref_37",
"volume": "219",
"year": "2020"
},
{
"DOI": "10.2174/18734316MTEyyNzY6y",
"article-title": "The Potential Mechanism of N-Acetylcysteine in Treating COVID-19",
"author": "Zhou",
"doi-asserted-by": "crossref",
"first-page": "1584",
"journal-title": "Curr. Pharm. Biotechnol.",
"key": "ref_38",
"volume": "22",
"year": "2021"
},
{
"DOI": "10.3390/ijms232314518",
"doi-asserted-by": "crossref",
"key": "ref_39",
"unstructured": "Milara, J., Martínez-Expósito, F., Montero, P., Roger, I., Bayarri, M.A., Ribera, P., Oishi-Konari, M.N., Alba-García, J.R., Zapater, E., and Cortijo, J. (2022). N-Acetylcysteine Reduces Inflammasome Activation Induced by SARS-CoV-2 Proteins In Vitro. Int. J. Mol. Sci., 23."
},
{
"DOI": "10.1016/j.chembiol.2018.01.011",
"article-title": "N-Acetyl Cysteine Functions as a Fast-Acting Antioxidant by Triggering Intracellular H2S and Sulfane Sulfur Production",
"author": "Takano",
"doi-asserted-by": "crossref",
"first-page": "447",
"journal-title": "Cell Chem. Biol.",
"key": "ref_40",
"volume": "25",
"year": "2018"
},
{
"DOI": "10.1097/SHK.0000000000001562",
"article-title": "Serum Hydrogen Sulfide and Outcome Association in Pneumonia by the SARS-CoV-2 Coronavirus",
"author": "Renieris",
"doi-asserted-by": "crossref",
"first-page": "633",
"journal-title": "Shock",
"key": "ref_41",
"volume": "54",
"year": "2020"
},
{
"DOI": "10.14715/cmb/2025.71.5.13",
"article-title": "COVID-19 Clinical Outcomes and N-Acetylcysteine (CoViNAC Study): A GRADE Compliant Meta-Analysis of Randomized Controlled Trials with Molecular Docking and Dynamics Simulation Studies with Mpro of SARS-CoV-2",
"author": "Varikasuvu",
"doi-asserted-by": "crossref",
"first-page": "95",
"journal-title": "Cell. Mol. Biol.",
"key": "ref_42",
"volume": "71",
"year": "2025"
},
{
"DOI": "10.3390/v17030359",
"doi-asserted-by": "crossref",
"key": "ref_43",
"unstructured": "Fratta Pasini, A.M., Stranieri, C., Di Leo, E.G., Bertolone, L., Aparo, A., Busti, F., Castagna, A., Vianello, A., Chesini, F., and Friso, S. (2025). Identification of Early Biomarkers of Mortality in COVID-19 Hospitalized Patients: A LASSO-Based Cox and Logistic Approach. Viruses, 17."
},
{
"DOI": "10.1016/j.freeradbiomed.2011.01.025",
"article-title": "Measurement of Plasma Hydrogen Sulfide in Vivo and in Vitro",
"author": "Shen",
"doi-asserted-by": "crossref",
"first-page": "1021",
"journal-title": "Free Radic. Biol. Med.",
"key": "ref_44",
"volume": "50",
"year": "2011"
},
{
"article-title": "Optimized THP-1 Differentiation Is Required for the Detection of Responses to Weak Stimuli",
"author": "Park",
"first-page": "45",
"journal-title": "Inflamm. Res. Off. J. Eur. Histamine Res. Soc. Al",
"key": "ref_45",
"volume": "56",
"year": "2007"
},
{
"DOI": "10.3390/antiox9040349",
"doi-asserted-by": "crossref",
"key": "ref_46",
"unstructured": "Peserico, D., Stranieri, C., Garbin, U., Mozzini, C.C., Danese, E., Cominacini, L., and Fratta Pasini, A.M. (2020). Ezetimibe Prevents Ischemia/Reperfusion-Induced Oxidative Stress and Up-Regulates Nrf2/ARE and UPR Signaling Pathways. Antioxidants, 9."
},
{
"DOI": "10.1161/ATVBAHA.107.142695",
"article-title": "Enhanced Levels of Oxidized Low-Density Lipoprotein Prime Monocytes to Cytokine Overproduction via Upregulation of CD14 and Toll-like Receptor 4 in Unstable Angina",
"author": "Anselmi",
"doi-asserted-by": "crossref",
"first-page": "1991",
"journal-title": "Arterioscler. Thromb. Vasc. Biol.",
"key": "ref_47",
"volume": "27",
"year": "2007"
},
{
"article-title": "Assessment of Cell Viability",
"author": "Johnson",
"first-page": "1",
"journal-title": "Curr. Protoc. Cytom.",
"key": "ref_48",
"volume": "64",
"year": "2013"
},
{
"DOI": "10.2174/1389201017666160808160513",
"article-title": "Cell Proliferation and Cytotoxicity Assays",
"author": "Adan",
"doi-asserted-by": "crossref",
"first-page": "1213",
"journal-title": "Curr. Pharm. Biotechnol.",
"key": "ref_49",
"volume": "17",
"year": "2016"
},
{
"DOI": "10.1002/cbf.4007",
"article-title": "Cell Viability and Cytotoxicity Assays: Biochemical Elements and Cellular Compartments",
"author": "Khalef",
"doi-asserted-by": "crossref",
"first-page": "e4007",
"journal-title": "Cell Biochem. Funct.",
"key": "ref_50",
"volume": "42",
"year": "2024"
},
{
"DOI": "10.1016/0022-1759(95)00072-I",
"article-title": "A Novel Assay for Apoptosis. Flow Cytometric Detection of Phosphatidylserine Expression on Early Apoptotic Cells Using Fluorescein Labelled Annexin V",
"author": "Vermes",
"doi-asserted-by": "crossref",
"first-page": "39",
"journal-title": "J. Immunol. Methods",
"key": "ref_51",
"volume": "184",
"year": "1995"
},
{
"DOI": "10.1002/(SICI)1097-0320(19960601)24:2<131::AID-CYTO5>3.0.CO;2-M",
"article-title": "A Novel Assay to Measure Loss of Plasma Membrane Asymmetry during Apoptosis of Adherent Cells in Culture",
"author": "Ramaekers",
"doi-asserted-by": "crossref",
"first-page": "131",
"journal-title": "Cytometry",
"key": "ref_52",
"volume": "24",
"year": "1996"
},
{
"DOI": "10.3390/molecules30153095",
"doi-asserted-by": "crossref",
"key": "ref_53",
"unstructured": "Di Leo, E.G., Stranieri, C., Zoccatelli, G., Bellumori, M., Zonfrillo, B., Cominacini, L., and Fratta Pasini, A.M. (2025). Olive Pomace Extract Acts as a New Potent Ferroptosis Inhibitor in Human Cells. Molecules, 30."
},
{
"DOI": "10.1080/10495398.2019.1654485",
"article-title": "Efficiency of CellROX Deep Red ® and CellROX Orange ® Fluorescent Probes in Identifying Reactive Oxygen Species in Sperm Samples from High and Low Fertility Bulls",
"author": "Celeghini",
"doi-asserted-by": "crossref",
"first-page": "77",
"journal-title": "Anim. Biotechnol.",
"key": "ref_54",
"volume": "32",
"year": "2021"
},
{
"DOI": "10.1016/S0891-5849(02)00848-1",
"article-title": "C11-BODIPY(581/591), an Oxidation-Sensitive Fluorescent Lipid Peroxidation Probe: (Micro)Spectroscopic Characterization and Validation of Methodology",
"author": "Drummen",
"doi-asserted-by": "crossref",
"first-page": "473",
"journal-title": "Free Radic. Biol. Med.",
"key": "ref_55",
"volume": "33",
"year": "2002"
},
{
"DOI": "10.1016/S0014-5793(99)00696-1",
"article-title": "Ratio-Fluorescence Microscopy of Lipid Oxidation in Living Cells Using C11-BODIPY581/591",
"author": "Pap",
"doi-asserted-by": "crossref",
"first-page": "278",
"journal-title": "FEBS Lett.",
"key": "ref_56",
"volume": "453",
"year": "1999"
},
{
"DOI": "10.1002/bmc.4854",
"doi-asserted-by": "crossref",
"key": "ref_57",
"unstructured": "Enomoto, A.C., Schneider, E., McKinnon, T., Goldfine, H., and Levy, M.A. (2020). Validation of a Simplified Procedure for Convenient and Rapid Quantification of Reduced and Oxidized Glutathione in Human Plasma by Liquid Chromatography Tandem Mass Spectrometry Analysis. Biomed. Chromatogr. BMC, 34."
},
{
"article-title": "Power Comparisons of Shapiro-Wilk, Kolmogorov-Smirnov, Lilliefors and Anderson-Darling Tests",
"author": "Razali",
"first-page": "21",
"journal-title": "J. Stat. Model. Anal.",
"key": "ref_58",
"volume": "2",
"year": "2011"
},
{
"DOI": "10.1007/s10528-013-9624-7",
"article-title": "Regulation of Cystathionine γ-Lyase in Mammalian Cells by Hypoxia",
"author": "Wang",
"doi-asserted-by": "crossref",
"first-page": "29",
"journal-title": "Biochem. Genet.",
"key": "ref_59",
"volume": "52",
"year": "2014"
},
{
"DOI": "10.3892/ijmm.2014.1788",
"article-title": "The Binding Site for the Transcription Factor, NF-κB, on the Cystathionine γ-Lyase Promoter Is Critical for LPS-induced Cystathionine γ-Lyase Expression",
"author": "Wang",
"doi-asserted-by": "crossref",
"first-page": "639",
"journal-title": "Int. J. Mol. Med.",
"key": "ref_60",
"volume": "34",
"year": "2014"
},
{
"DOI": "10.1016/j.molcel.2011.10.021",
"article-title": "Hydrogen Sulfide-Linked Sulfhydration of NF-κB Mediates Its Antiapoptotic Actions",
"author": "Sen",
"doi-asserted-by": "crossref",
"first-page": "13",
"journal-title": "Mol. Cell",
"key": "ref_61",
"volume": "45",
"year": "2012"
}
],
"reference-count": 61,
"references-count": 61,
"relation": {},
"resource": {
"primary": {
"URL": "https://www.mdpi.com/2076-3921/15/3/307"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "Decreased Plasma Concentration of Hydrogen Sulfide in Hospitalized COVID-19 Patients: A Novel Determinant of Mortality?",
"type": "journal-article",
"update-policy": "https://doi.org/10.3390/mdpi_crossmark_policy",
"volume": "15"
}