A phase 2a double-blind, placebo-controlled randomized trial of the SARS-CoV-2-specific monoclonal antibody AER002 in people with Long COVID
et al., medRxiv, doi:10.64898/2026.03.07.26347857 (results released 10/28/25), outSMART-LC, NCT05877508, Oct 2025
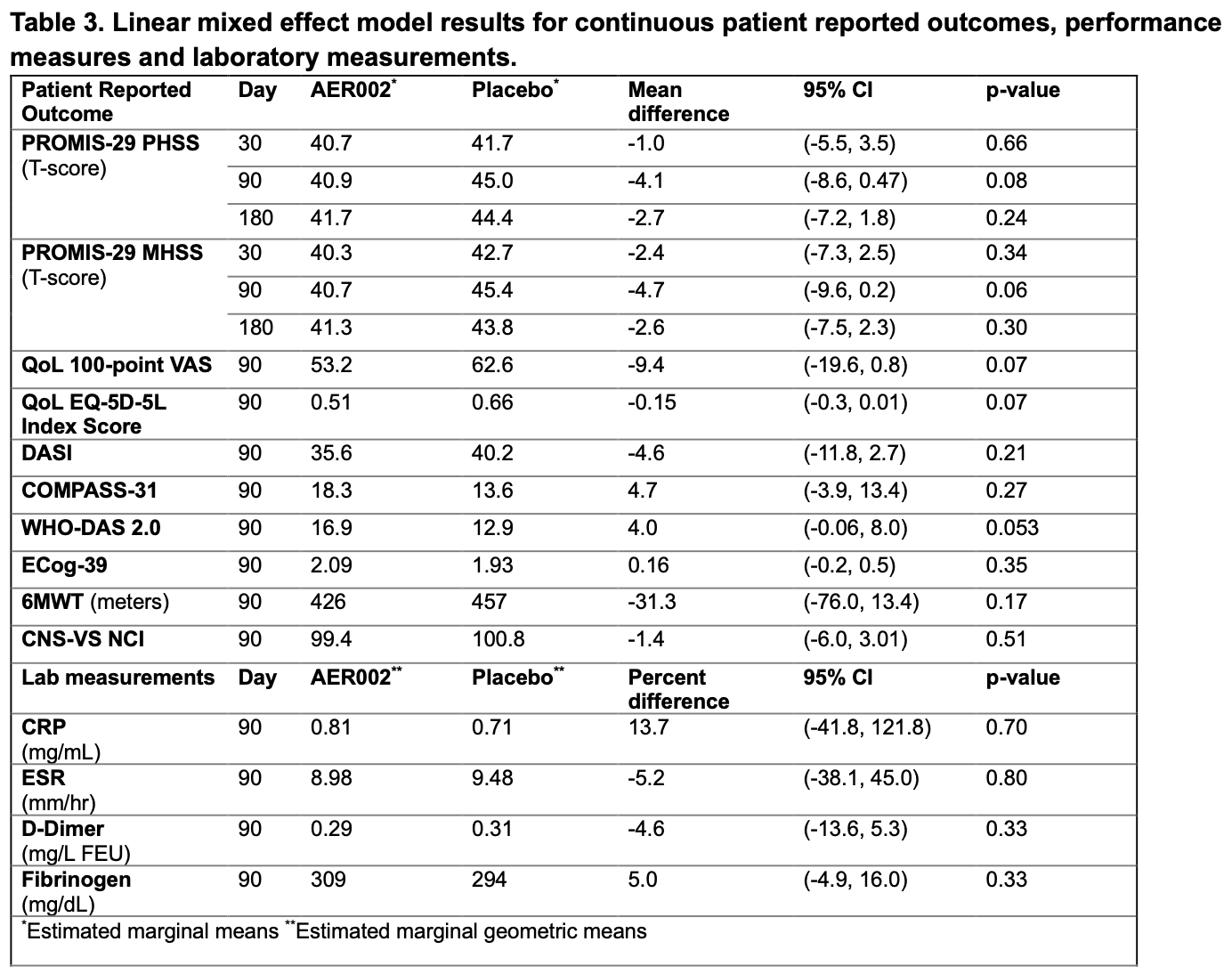
RCT 36 long COVID outpatients showing no significant clinical benefit with the AER002 monoclonal antibody compared to placebo.
Standard of Care (SOC) for COVID-19 in the study country,
the USA, is very poor with very low average efficacy for approved treatments1.
Only expensive, high-profit treatments were approved for early treatment. Low-cost treatments were excluded, reducing the probability of early treatment due to access and cost barriers, and eliminating complementary and synergistic benefits seen with many low-cost treatments.
|
risk of long COVID, 6.5% higher, RR 1.06, p = 0.25, treatment 41.7 [39.1-44.3] n=24, control 44.4 [40.7-48.0] n=12, relative PROMIS-29 score.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Peluso et al., 28 Oct 2025, Double Blind Randomized Controlled Trial, placebo-controlled, USA, preprint, median age 42.0, 40 authors, study period July 2023 - April 2024, trial NCT05877508 (history) (outSMART-LC).
Contact: michael.peluso@ucsf.edu.
Abstract: medRxiv preprint doi: https://doi.org/10.64898/2026.03.07.26347857; this version posted March 9, 2026. The copyright holder for this preprint
(which was not certified by peer review) is the author/funder, who has granted medRxiv a license to display the preprint in perpetuity.
It is made available under a CC-BY 4.0 International license .
1
2
3
4
5
6
7
8
9
10
11
12
13
14
15
16
17
18
19
20
21
22
23
24
25
26
27
28
29
30
31
32
33
34
35
36
37
38
39
40
41
42
43
44
45
46
47
48
49
50
A phase 2a double-blind, placebo-controlled randomized trial of the SARS-CoV-2-specific
monoclonal antibody AER002 in people with Long COVID
Michael J. Peluso1*^, Dylan Ryder1*, Thomas Dalhuisen1, Danny Hoi Tsun Chu2, Meghann C. Williams1,
Antonio E. Rodriguez1, Brian LaFranchi3, Joanna Vinden3, Emily A. Fehrman1, Beatrice Huang1, Rebecca
Hoh1, Kofi A. Asare1, Kathleen Bellon Pizarro1, Mohammad Rahman2, Emilio de Narvaez3, Mark M. Painter5,
E. John Wherry5, Zoe N. Swank6, Louise L. Hansen6, David R. Walt6, Yoshinori Fukazawa3, Anisha Sekar7,
Steven E. Bellan7, Holly Tieu8, Josephat Asiago8, Prakash Bhuyan8, Rajeev Venkayya8, Robert R. Flavell9,
Henry VanBrocklin9, J. Daniel Kelly10, Priscilla Y. Hsue11, Matthew S. Durstenfeld4, Peter W. Hunt3, Leonard
Calabrese12, Ma Somsouk13, Jeffrey N. Martin10, David V. Glidden10, Amelia N. Deitchman2, Timothy J.
Henrich3, Steven G. Deeks1
*Co-first authors
1
Division of HIV, Infectious Diseases, and Global Medicine, University of California, San Francisco, San
Francisco, CA, USA
2
Department of Clinical Pharmacy, University of California, San Francisco, San Francisco, CA, USA
3
Division of Experimental Medicine, University of California, San Francisco, San Francisco, CA, USA
4
Division of Cardiology, University of California, San Francisco, San Francisco, CA, USA
5
University of Pennsylvania, Philadelphia, PA, USA
6
Department of Pathology, Brigham & Women’s Hospital, Harvard Medical School, Boston, MA, USA
7
Patient-Led Research Collaborative
8
Aerium Therapeutics, Boston, MA, USA
9
Department of Radiology and Biomedical Imaging, University of California, San Francisco, San Francisco,
CA, USA
10
Department of Epidemiology and Biostatistics, University of California, San Francisco, San Francisco, CA,
USA
11
Division of Cardiology, University of California, Los Angeles, CA, USA
12
Cleveland Clinic, Cleveland, OH, USA
13
Division of Gastroenterology, University of California, San Francisco, San Francisco, CA, USA
^Corresponding Author:
Michael J. Peluso, MD
2540 23rd Street, #4807
San Francisco CA 94110
michael.peluso@ucsf.edu
NOTE: This preprint reports new research that has not been certified by peer review and should not be used to guide clinical practice.
medRxiv preprint doi: https://doi.org/10.64898/2026.03.07.26347857; this version posted March 9, 2026. The copyright holder for this preprint
(which was not certified by peer review) is the author/funder, who has granted medRxiv a license to display the preprint in perpetuity.
It is made available under a CC-BY 4.0 International license .
51
52
53
54
55
56
57
58
59
60
61
62
63
64
65
66
67
68
69
70
ABSTRACT
Long COVID is a disabling chronic illness with no proven treatments. Persistence of SARS-CoV-2 has been
proposed as a biological driver of the disease. We conducted a placebo-controlled, double-blind, 2:1
randomized mechanistic trial of the SARS-CoV-2-specific monoclonal antibody AER002 in 36 participants
who met the World Health..
DOI record:
{
"DOI": "10.64898/2026.03.07.26347857",
"URL": "http://dx.doi.org/10.64898/2026.03.07.26347857",
"abstract": "<jats:title>ABSTRACT</jats:title>\n <jats:p>Long COVID is a disabling chronic illness with no proven treatments. Persistence of SARS-CoV-2 has been proposed as a biological driver of the disease. We conducted a placebo-controlled, double-blind, 2:1 randomized mechanistic trial of the SARS-CoV-2-specific monoclonal antibody AER002 in 36 participants who met the World Health Organization case definition of Long COVID. After baseline characterization, participants received a single infusion and were followed for 360 days. The primary endpoint was the PROMIS-29 Physical Health Summary Score (PHSS) at 90 days; secondary and exploratory endpoints included patient-reported and objective measures of physical, cognitive, and neurologic function as well as blood-, imaging-, and tissue-based biomarkers. While AER002 was safe and well tolerated, no significant differences in physical health, quality of life, objective measures of physical function or cognition, or blood-based biomarkers were demonstrated between the treatment and control arms. In a post-hoc analysis, participants with a lower baseline SARS-CoV-2 antibody level and higher drug exposure were more likely to express a perceived treatment benefit based on the Patient Global Impression of Change scale (p<0.05 for anti-S, S1, and RBD). Although AER002 was not efficacious in this proof-of-concept study of people with broadly defined Long COVID, our findings could inform recruitment or dosing strategies employed in future trials using monoclonal antibodies to target viral persistence as a driver of Long COVID.</jats:p>",
"accepted": {
"date-parts": [
[
2026,
3,
9
]
]
},
"author": [
{
"affiliation": [
{
"name": "Division of HIV, Infectious Diseases, and Global Medicine, University of California, San Francisco, San Francisco, CA, USA"
}
],
"family": "Peluso",
"given": "Michael J.",
"sequence": "first"
},
{
"affiliation": [
{
"name": "Division of HIV, Infectious Diseases, and Global Medicine, University of California, San Francisco, San Francisco, CA, USA"
}
],
"family": "Ryder",
"given": "Dylan",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Division of HIV, Infectious Diseases, and Global Medicine, University of California, San Francisco, San Francisco, CA, USA"
}
],
"family": "Dalhuisen",
"given": "Thomas",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Clinical Pharmacy, University of California, San Francisco, San Francisco, CA, USA"
}
],
"family": "Chu",
"given": "Danny Hoi Tsun",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Division of HIV, Infectious Diseases, and Global Medicine, University of California, San Francisco, San Francisco, CA, USA"
}
],
"family": "Williams",
"given": "Meghann C.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Division of HIV, Infectious Diseases, and Global Medicine, University of California, San Francisco, San Francisco, CA, USA"
}
],
"family": "Rodriguez",
"given": "Antonio E.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Division of Experimental Medicine, University of California, San Francisco, San Francisco, CA, USA"
}
],
"family": "LaFranchi",
"given": "Brian",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Division of Experimental Medicine, University of California, San Francisco, San Francisco, CA, USA"
}
],
"family": "Vinden",
"given": "Joanna",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Division of HIV, Infectious Diseases, and Global Medicine, University of California, San Francisco, San Francisco, CA, USA"
}
],
"family": "Fehrman",
"given": "Emily A.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Division of HIV, Infectious Diseases, and Global Medicine, University of California, San Francisco, San Francisco, CA, USA"
}
],
"family": "Huang",
"given": "Beatrice",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Division of HIV, Infectious Diseases, and Global Medicine, University of California, San Francisco, San Francisco, CA, USA"
}
],
"family": "Hoh",
"given": "Rebecca",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Division of HIV, Infectious Diseases, and Global Medicine, University of California, San Francisco, San Francisco, CA, USA"
}
],
"family": "Asare",
"given": "Kofi A.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Division of HIV, Infectious Diseases, and Global Medicine, University of California, San Francisco, San Francisco, CA, USA"
}
],
"family": "Pizarro",
"given": "Kathleen Bellon",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Clinical Pharmacy, University of California, San Francisco, San Francisco, CA, USA"
}
],
"family": "Rahman",
"given": "Mohammad",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Division of Experimental Medicine, University of California, San Francisco, San Francisco, CA, USA"
}
],
"family": "de Narvaez",
"given": "Emilio",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "University of Pennsylvania, Philadelphia, PA, USA"
}
],
"family": "Painter",
"given": "Mark M.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "University of Pennsylvania, Philadelphia, PA, USA"
}
],
"family": "Wherry",
"given": "E. John",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Pathology, Brigham & Women’s Hospital, Harvard Medical School, Boston, MA, USA"
}
],
"family": "Swank",
"given": "Zoe N.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Pathology, Brigham & Women’s Hospital, Harvard Medical School, Boston, MA, USA"
}
],
"family": "Hansen",
"given": "Louise L.",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0002-5524-7348",
"affiliation": [
{
"name": "Department of Pathology, Brigham & Women’s Hospital, Harvard Medical School, Boston, MA, USA"
}
],
"authenticated-orcid": false,
"family": "Walt",
"given": "David R.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Division of Experimental Medicine, University of California, San Francisco, San Francisco, CA, USA"
}
],
"family": "Fukazawa",
"given": "Yoshinori",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Patient-Led Research Collaborative"
}
],
"family": "Sekar",
"given": "Anisha",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Patient-Led Research Collaborative"
}
],
"family": "Bellan",
"given": "Steven E.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Aerium Therapeutics, Boston, MA, USA"
}
],
"family": "Tieu",
"given": "Holly",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Aerium Therapeutics, Boston, MA, USA"
}
],
"family": "Asiago",
"given": "Josephat",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Aerium Therapeutics, Boston, MA, USA"
}
],
"family": "Bhuyan",
"given": "Prakash",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Aerium Therapeutics, Boston, MA, USA"
}
],
"family": "Venkayya",
"given": "Rajeev",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Radiology and Biomedical Imaging, University of California, San Francisco, San Francisco, CA, USA"
}
],
"family": "Flavell",
"given": "Robert R.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Radiology and Biomedical Imaging, University of California, San Francisco, San Francisco, CA, USA"
}
],
"family": "VanBrocklin",
"given": "Henry",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Epidemiology and Biostatistics, University of California, San Francisco, San Francisco, CA, USA"
}
],
"family": "Daniel Kelly",
"given": "J.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Division of Cardiology, University of California, Los Angeles, CA, USA"
}
],
"family": "Hsue",
"given": "Priscilla Y.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Division of Cardiology, University of California, San Francisco, San Francisco, CA, USA"
}
],
"family": "Durstenfeld",
"given": "Matthew S.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Division of Experimental Medicine, University of California, San Francisco, San Francisco, CA, USA"
}
],
"family": "Hunt",
"given": "Peter W.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Cleveland Clinic, Cleveland, OH, USA"
}
],
"family": "Calabrese",
"given": "Leonard",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Division of Gastroenterology, University of California, San Francisco, San Francisco, CA, USA"
}
],
"family": "Somsouk",
"given": "Ma",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Epidemiology and Biostatistics, University of California, San Francisco, San Francisco, CA, USA"
}
],
"family": "Martin",
"given": "Jeffrey N.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Epidemiology and Biostatistics, University of California, San Francisco, San Francisco, CA, USA"
}
],
"family": "Glidden",
"given": "David V.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Clinical Pharmacy, University of California, San Francisco, San Francisco, CA, USA"
}
],
"family": "Deitchman",
"given": "Amelia N.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Division of Experimental Medicine, University of California, San Francisco, San Francisco, CA, USA"
}
],
"family": "Henrich",
"given": "Timothy J.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Division of HIV, Infectious Diseases, and Global Medicine, University of California, San Francisco, San Francisco, CA, USA"
}
],
"family": "Deeks",
"given": "Steven G.",
"sequence": "additional"
}
],
"container-title": [],
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2026,
3,
11
]
],
"date-time": "2026-03-11T16:00:15Z",
"timestamp": 1773244815000
},
"deposited": {
"date-parts": [
[
2026,
3,
11
]
],
"date-time": "2026-03-11T16:00:19Z",
"timestamp": 1773244819000
},
"group-title": "Infectious Diseases (except HIV/AIDS)",
"indexed": {
"date-parts": [
[
2026,
3,
12
]
],
"date-time": "2026-03-12T10:19:09Z",
"timestamp": 1773310749974,
"version": "3.50.1"
},
"institution": [
{
"name": "medRxiv"
}
],
"is-referenced-by-count": 0,
"issued": {
"date-parts": [
[
2026,
3,
9
]
]
},
"license": [
{
"URL": "http://creativecommons.org/licenses/by/4.0/",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2026,
3,
9
]
],
"date-time": "2026-03-09T00:00:00Z",
"timestamp": 1773014400000
}
}
],
"link": [
{
"URL": "https://syndication.highwire.org/content/doi/10.64898/2026.03.07.26347857",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "54368",
"original-title": [],
"posted": {
"date-parts": [
[
2026,
3,
9
]
]
},
"prefix": "10.64898",
"published": {
"date-parts": [
[
2026,
3,
9
]
]
},
"publisher": "openRxiv",
"reference": [
{
"key": "2026031109000665000_2026.03.07.26347857v1.1",
"unstructured": "National Academies of Sciences E, Medicine. A Long COVID Definition: A Chronic, Systemic Disease State with Profound Consequences. Washington, DC: The National Academies Press, 2024."
},
{
"DOI": "10.1038/s41591-024-03173-6",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.2"
},
{
"DOI": "10.1001/jamahealthforum.2022.1809",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.3"
},
{
"DOI": "10.1038/s41579-022-00846-2",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.4"
},
{
"DOI": "10.1016/j.cell.2024.07.054",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.5"
},
{
"DOI": "10.1038/s41590-023-01601-2",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.6"
},
{
"DOI": "10.1093/cid/ciac722",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.7"
},
{
"DOI": "10.1016/j.cmi.2024.09.001",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.8"
},
{
"DOI": "10.1016/S1473-3099(24)00211-1",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.9"
},
{
"DOI": "10.1093/ofid/ofaf515",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.10"
},
{
"DOI": "10.1038/s41586-021-03207-w",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.11"
},
{
"DOI": "10.1053/j.gastro.2022.04.037",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.12"
},
{
"DOI": "10.1038/s41586-022-05542-y",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.13"
},
{
"DOI": "10.1136/gutjnl-2021-324280",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.14"
},
{
"DOI": "10.1126/scitranslmed.adk3295",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.15"
},
{
"DOI": "10.1016/S1473-3099(24)00171-3",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.16"
},
{
"DOI": "10.1016/s1473-3099(24)00769-2",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.17"
},
{
"DOI": "10.20411/pai.v7i1.518",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.18"
},
{
"DOI": "10.1038/s43856-024-00668-8",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.19"
},
{
"DOI": "10.14218/erhm.2022.00045",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.20"
},
{
"DOI": "10.1001/jamainternmed.2024.2007",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.21"
},
{
"DOI": "10.1016/S1473-3099(25)00073-8",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.22"
},
{
"DOI": "10.1126/scitranslmed.ado2101",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.23"
},
{
"DOI": "10.1038/s41586-020-2571-7",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.24"
},
{
"DOI": "10.1128/AAC.01285-13",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.25"
},
{
"DOI": "10.1128/aac.01020-16",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.26"
},
{
"DOI": "10.1126/scitranslmed.abl8124",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.27"
},
{
"DOI": "10.1038/s41577-021-00542-x",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.28"
},
{
"DOI": "10.1016/j.ajem.2023.09.051",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.29"
},
{
"DOI": "10.7759/cureus.96246",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.30"
},
{
"DOI": "10.1007/s40121-023-00908-9",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.31"
},
{
"key": "2026031109000665000_2026.03.07.26347857v1.32",
"unstructured": "Institute of Medicine. 2015. Beyond Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: Redefining an Illness. Washington, DC: The National Academies Press."
},
{
"DOI": "10.3390/diagnostics8030066",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.33"
},
{
"DOI": "10.1177/21501327211030413",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.34"
},
{
"DOI": "10.1007/s11136-018-1842-3",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.35"
},
{
"DOI": "10.1007/s11136-018-1842-3",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.36"
},
{
"DOI": "10.1016/j.jval.2019.02.009",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.37"
},
{
"DOI": "10.3109/07853890109002087",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.38"
},
{
"key": "2026031109000665000_2026.03.07.26347857v1.39",
"unstructured": "WHO Disability Assessment Schedule (WHODAS 2.0). Available at: https://www.who.int/standards/classifications/international-classification-of-functioning-disability-and-health/who-disability-assessment-schedule."
},
{
"DOI": "10.1016/S1473-3099(25)00665-6",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.40"
},
{
"DOI": "10.1016/j.jpain.2009.01.258",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.41"
},
{
"DOI": "10.1016/0002-9149(89)90496-7",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.42"
},
{
"DOI": "10.1164/ajrccm.166.1.at1102",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.43"
},
{
"key": "2026031109000665000_2026.03.07.26347857v1.44",
"unstructured": "Matos Casano HA , Ahmed I , Anjum F . Six-Minute Walk Test. StatPearls. Treasure Island (FL): StatPearls Publishing Copyright © 2025, StatPearls Publishing LLC.; 2025."
},
{
"DOI": "10.1037/0894-4105.22.4.531",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.45"
},
{
"DOI": "10.1016/j.acn.2006.05.007",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.46"
},
{
"DOI": "10.1016/j.mayocp.2012.10.013",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.47"
},
{
"key": "2026031109000665000_2026.03.07.26347857v1.48",
"unstructured": "International Guidelines for Active Standing Test Procedure. Available at: https://api.heartrhythmalliance.org/files/download/2fd746f6b5cd66daff21595af7d8b36b."
},
{
"DOI": "10.1039/c5an01238d",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.49"
},
{
"DOI": "10.1021/acs.jproteome.5c00571",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.50"
},
{
"DOI": "10.1214/aos/1074290335",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.51"
},
{
"DOI": "10.1016/j.cels.2015.12.004",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.52"
},
{
"key": "2026031109000665000_2026.03.07.26347857v1.53",
"unstructured": "Peluso MJea. Post-acute SARS-CoV-2 antigen persistence is associated with some but not all Long COVID symptoms. CROI. San Francisco, U.S.A.2025."
},
{
"DOI": "10.1073/pnas.1710519114",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.54"
},
{
"DOI": "10.3389/fimmu.2024.1386607",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.55"
},
{
"DOI": "10.1016/j.smim.2024.101873",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.56"
},
{
"DOI": "10.3389/fnins.2025.1627462",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.57"
},
{
"DOI": "10.1001/jamanetworkopen.2024.36874",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.58"
},
{
"DOI": "10.1093/cid/ciae205",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.59"
},
{
"DOI": "10.1037/0021-9010.69.2.334",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.60"
},
{
"DOI": "10.1001/jama.1955.02960340022006",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.61"
},
{
"DOI": "10.1056/NEJMra1907805",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.62"
},
{
"DOI": "10.1038/nrn3976",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.63"
},
{
"DOI": "10.1038/s41586-024-07469-y",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.64"
},
{
"DOI": "10.1001/jamanetworkopen.2020.13196",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.65"
},
{
"DOI": "10.1177/23743735221077514",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.66"
},
{
"DOI": "10.1016/j.cct.2021.106503",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.67"
},
{
"DOI": "10.1093/ofid/ofab640",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.68"
},
{
"DOI": "10.1016/s1473-3099(21)00703-9",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.69"
},
{
"DOI": "10.7326/annals-24-00464",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.70"
},
{
"key": "2026031109000665000_2026.03.07.26347857v1.71",
"unstructured": "Spritzer, K. L. & Hays, R. D. Calculating Physical and Mental Health Summary Scores for PROMIS-29 v2.0 and v2.1. Available at: https://www.healthmeasures.net/media/kunena/attachments/257/PROMIS29_Scoring_08082018.pdf."
},
{
"key": "2026031109000665000_2026.03.07.26347857v1.72",
"unstructured": "EuroQol Value sets. EuroQol https://euroqol.org/information-and-support/resources/value-sets/."
},
{
"DOI": "10.1007/s11136-022-03180-5",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.73"
},
{
"DOI": "10.1111/j.1365-2796.2011.02428.x",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.74"
},
{
"key": "2026031109000665000_2026.03.07.26347857v1.75",
"unstructured": "Bell DS . The Doctor’s Guide to Chronic Fatigue Syndrome: Understanding, Treating and Living with CFIDS. Boston: Da Capo Lifelong Books; 1995."
},
{
"DOI": "10.1002/ajmg.c.31552",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.76"
},
{
"DOI": "10.3390/jcm10153216",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.77"
},
{
"key": "2026031109000665000_2026.03.07.26347857v1.78",
"unstructured": "Coastal Integrative Medicine. Mast Cell Activation Disease Questionnaire. Available at: https://www.coastalintegrativemedicine.com/storage/app/media/neurodevelopmental/mast-cell-activation-questionnaire.pdf."
},
{
"DOI": "10.1136/pgmj.2007.058198",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.79"
},
{
"DOI": "10.1038/s41551-020-00611-x",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.80"
},
{
"DOI": "10.1093/infdis/jiad131",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.81"
},
{
"key": "2026031109000665000_2026.03.07.26347857v1.82",
"unstructured": "Lenth R (2025). _emmeans: Estimated Marginal Means, aka Least-Squares Means_. R package version 1.11.2, <https://CRAN.R-project.org/package=emmeans>."
},
{
"key": "2026031109000665000_2026.03.07.26347857v1.83",
"unstructured": "R Core Team (2024). _R: A Language and Environment for Statistical Computing_. R Foundation for Statistical Computing, Vienna, Austria. <https://www.R-project.org/>."
},
{
"DOI": "10.1038/s41596-024-01020-z",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.84"
},
{
"DOI": "10.1093/genetics/iyad031",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.85"
},
{
"DOI": "10.1111/bcp.12451",
"doi-asserted-by": "publisher",
"key": "2026031109000665000_2026.03.07.26347857v1.86"
}
],
"reference-count": 86,
"references-count": 86,
"relation": {},
"resource": {
"primary": {
"URL": "http://medrxiv.org/lookup/doi/10.64898/2026.03.07.26347857"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"subtype": "preprint",
"title": "A phase 2a double-blind, placebo-controlled randomized trial of the SARS-CoV-2-specific monoclonal antibody AER002 in people with Long COVID",
"type": "posted-content"
}