Ivermectin therapy is associated with changes in SARS-CoV-2 RNA load in asymptomatic patients: a randomized controlled trial
et al., Trials, doi:10.1186/s13063-026-09736-x, ChiCTR2000033627, May 2026
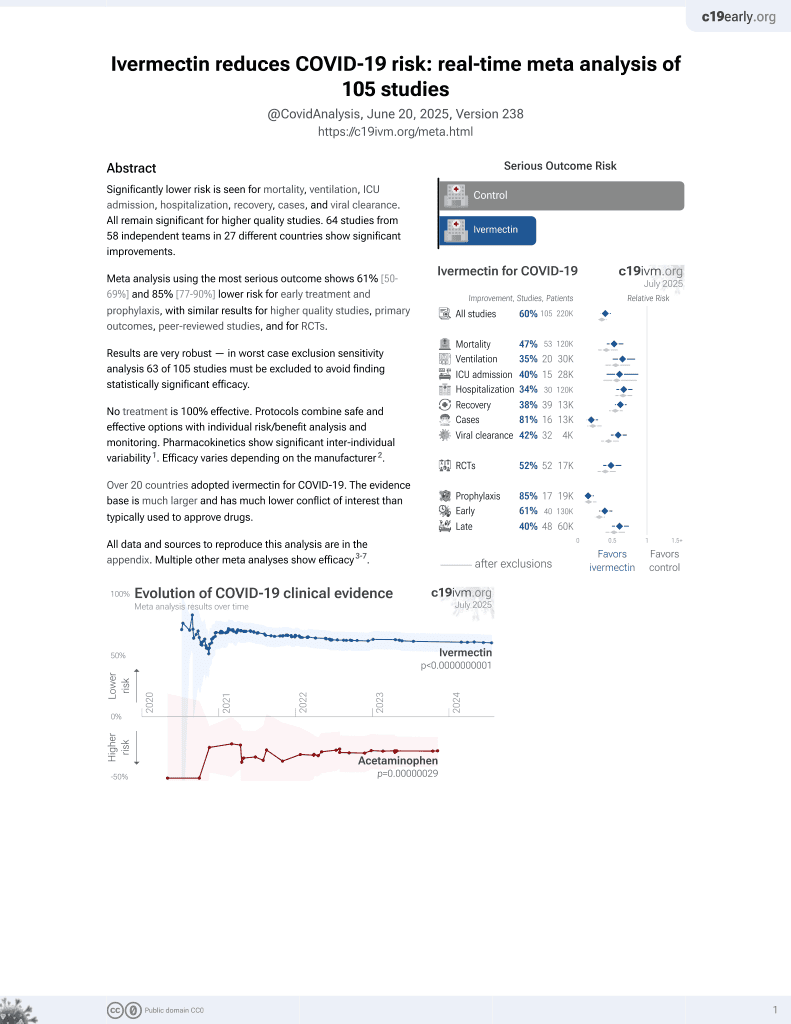
Ivermectin for COVID-19
4th treatment shown to reduce risk in
August 2020, now with p < 0.00000000001 from 106 studies, recognized in 24 countries.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
RCT of 126 asymptomatic PCR+ patients with high viral load, showing improved recovery and significantly lower viral load with ivermectin treatment. All patients received zind and vitamin C.
The trial selected for patients with high viral load that were still asymptomatic - i.e., patients were treated early before significant progression and had received a high viral load, which may be favorable conditions for the strong viral load improvement seen.
This is the 54th COVID-19 RCT for ivermectin, which collectively show efficacy with p=0.00000004.
This is the 106th COVID-19 controlled study for ivermectin, which collectively show efficacy with p<0.0000000001.
|
risk of hospitalization, 55.6% lower, RR 0.44, p = 0.24, treatment 4 of 63 (6.3%), control 9 of 63 (14.3%), NNT 13, day 4.
|
|
risk of no recovery, 30.9% lower, RR 0.69, p = 0.007, treatment 63, control 63, all symptoms combined.
|
|
risk of no recovery, 81.2% lower, RR 0.19, p = 0.002, treatment 3 of 63 (4.8%), control 16 of 63 (25.4%), NNT 4.8, day 4, fever.
|
|
risk of no recovery, 33.3% higher, RR 1.33, p = 0.52, treatment 16 of 63 (25.4%), control 12 of 63 (19.0%), day 4, cough.
|
|
risk of no recovery, 60.0% lower, RR 0.40, p = 0.44, treatment 2 of 63 (3.2%), control 5 of 63 (7.9%), NNT 21, day 4, sore throat.
|
|
risk of no recovery, 26.3% lower, RR 0.74, p = 0.42, treatment 14 of 63 (22.2%), control 19 of 63 (30.2%), NNT 13, day 4, dyspnea.
|
|
risk of no recovery, 33.3% lower, RR 0.67, p = 0.74, treatment 4 of 63 (6.3%), control 6 of 63 (9.5%), NNT 32, day 4, pneumonia.
|
|
risk of no recovery, 40.0% lower, RR 0.60, p = 0.02, treatment 21 of 63 (33.3%), control 35 of 63 (55.6%), NNT 4.5, day 4, headache.
|
|
risk of no recovery, 46.2% higher, RR 1.46, p = 0.31, treatment 19 of 63 (30.2%), control 13 of 63 (20.6%), day 4, anosmia.
|
|
risk of no recovery, 22.2% lower, RR 0.78, p = 0.36, treatment 21 of 63 (33.3%), control 27 of 63 (42.9%), NNT 10, day 4, myalgia.
|
|
risk of no recovery, 48.0% lower, RR 0.52, p = 0.03, treatment 13 of 63 (20.6%), control 25 of 63 (39.7%), NNT 5.2, day 4, loss of taste.
|
|
risk of no recovery, 46.7% lower, RR 0.53, p = 0.02, treatment 16 of 63 (25.4%), control 30 of 63 (47.6%), NNT 4.5, day 4, fatigue.
|
|
risk of no recovery, 42.9% lower, RR 0.57, p = 0.53, treatment 4 of 63 (6.3%), control 7 of 63 (11.1%), NNT 21, day 4, dizziness.
|
|
viral load, 61.9% lower, relative load 0.38, p < 0.001, treatment 8.38 [7.56-9.2] n=63, control 3.19 [2.36-4.02] n=63, relative ΔCt, day 4.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Mouawia et al., 1 May 2026, Randomized Controlled Trial, Lebanon, peer-reviewed, mean age 40.1, 8 authors, study period November 2021 - December 2021, dosage 12mg single dose, <65kg - 9mg, 65-84kg - 12 mg, ≥85kg - 15 mg, trial ChiCTR2000033627.
Contact: raadhoussamrh@gmail.com, houssam.raad@ul.edu.lb, houssein.mouawia@ul.edu.lb.