Ursodeoxycholic acid treatment did not show protective effect for severe COVID-19 outcomes – a nationwide register study
et al., BMC Public Health, doi:10.1186/s12889-026-26908-1, Apr 2026
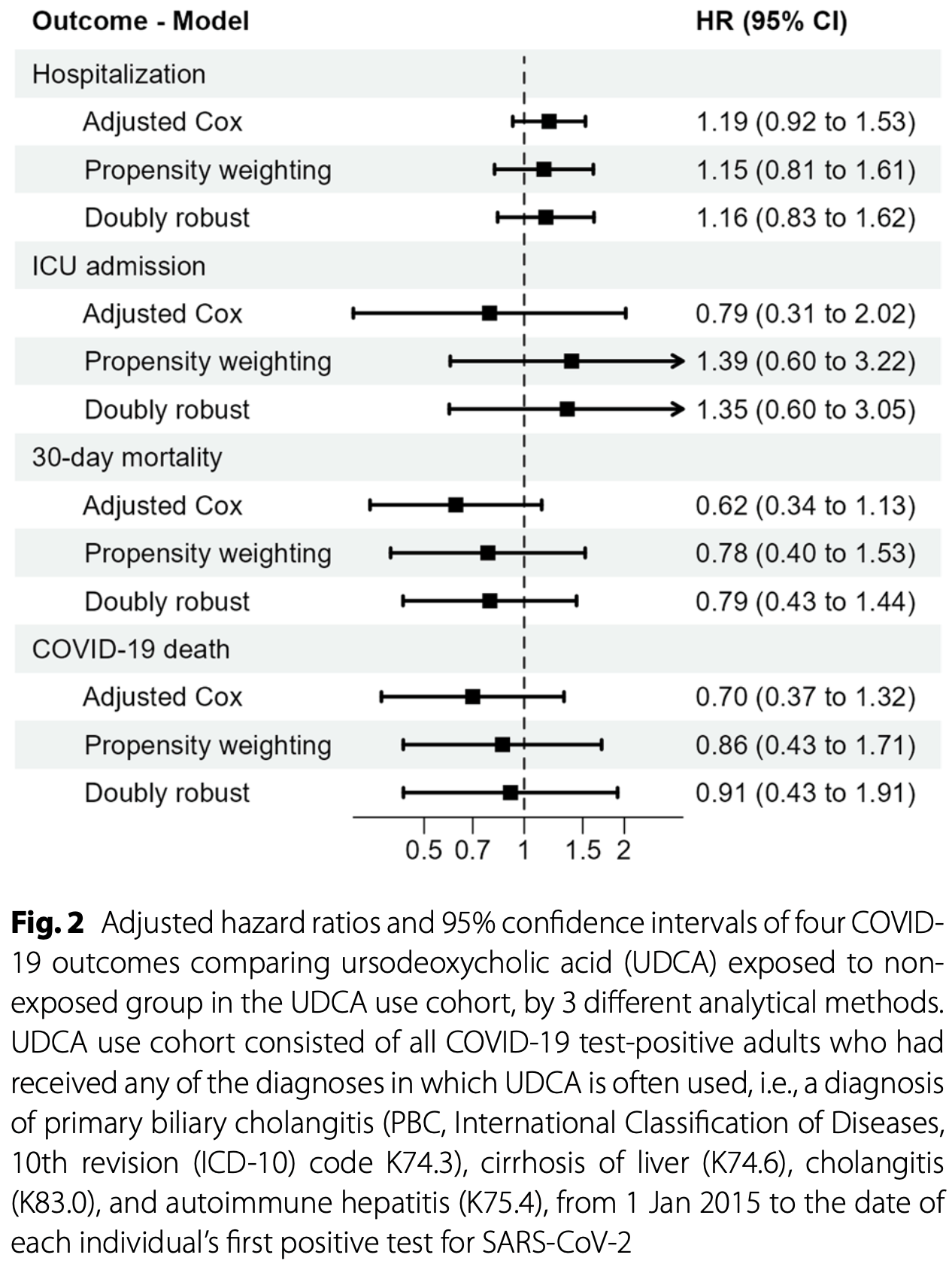
Retrospective nationwide register-based cohort study of 4,471 Swedish adults with liver/biliary diseases, showing no significant protective effect of ursodeoxycholic acid (UDCA) against severe COVID-19 outcomes. The study used a current-users vs. non-users design rather than a new-users design, which the authors acknowledge is suboptimal for causal inference and may introduce bias.
The doubly robust estimator in the UDCA use cohort is likely the most reliable of the estimates made.
Confounding concerns include:
Prevalent-user bias - comparing current users to non-users (rather than new-users to non-users) means anyone who had early bad outcomes on UDCA is already filtered out of the exposed group.
Adherence misclassification - dispensing ≠ ingestion.
Residual confounding by indication - even with balance on measured covariates, UDCA-exposed and unexposed groups differ structurally on why they're treated (disease severity, specialist access, treatment eligibility decisions) in ways the data does not capture.
Residual confounding by liver disease mix - PBC is 53% of exposed vs 2.7% of unexposed; cirrhosis is 16% vs 38%. Weighting balances the marginal proportions but can't fully remove differences in disease stage/severity within each category.
Healthy-adherer effect - people who fill prescriptions reliably tend to have better health behaviors generally.
|
risk of death, 9.0% lower, HR 0.91, p = 0.82, treatment 833, control 3,638, adjusted per study, doubly robust analysis, COVID-19 mortality.
|
|
risk of death, 21.0% lower, HR 0.79, p = 0.45, treatment 833, control 3,638, adjusted per study, doubly robust analysis, 30-day mortality, day 30.
|
|
risk of ICU admission, 35.0% higher, HR 1.35, p = 0.48, treatment 833, control 3,638, adjusted per study, doubly robust analysis.
|
|
risk of hospitalization, 16.0% higher, HR 1.16, p = 0.39, treatment 833, control 3,638, adjusted per study, doubly robust analysis.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Li et al., 11 Apr 2026, retrospective, Sweden, peer-reviewed, 5 authors, study period 1 January, 2020 - 31 October, 2023.
Contact: huiqi.li@gu.se.
Abstract: ## RESEARCH
Open Access
Ursodeoxycholic acid treatment did not show protective effect for severe COVID-19 outcomes - a nationwide register study
Huiqi Li 1* , Yiyi Xu 1 , Brian Kirui 1 , Ailiana Santosa 1 and Fredrik Nyberg 1
Abstract
Background Ursodeoxycholic acid (UDCA), used for treating cholestasis, was reported to protect against severe COVID-19 outcomes. We aimed to assess this potential effect using nationwide Swedish register data.
Method We included all SARS-CoV-2 test-positive subjects aged ≥ 18 years during 2020-2023, with previous diagnosis of potentially UDCA-treated diseases, including primary biliary cholangitis, liver cirrhosis, autoimmune hepatitis, or cholangitis. Subjects who had filled a UDCA prescription within 6 months before testing positive were considered exposed. Subjects were followed from test-positivity to the earliest of analyzed outcome (COVID-19related hospitalization, 30-day all-cause mortality, or COVID-19-related death), one year of follow-up, emigration, death, or end of 2023. Control for potential confounding (vaccination status, COVID-19 waves, prior comorbidities, and sociodemographics) was done by adjustment, propensity score weighting (PSW), and doubly robust analysis. Cox regression was used to estimate hazard ratios (HR) with 95% confidence intervals (95%CI).
Results The study cohort consisted of 833 UDCA-exposed and 3638 non-exposed individuals. The adjusted Cox analysis showed potentially decreased risks (non-significant) among UDCA-exposed for 30-day mortality (HR 0.62, 95%CI 0.34-1.13) and COVID-19 death (0.70, 0.37-1.32), but a smaller non-significant increased risk for hospitalization (1.19, 0.92-1.53). PSW achieved an adequate balance between exposed and non-exposed and showed similar results for 30-day mortality (0.78, 0.40-1.53), COVID-19 death (0.86, 0.43-1.71), and hospitalization (1.15, 0.81-1.61). The doubly robust method showed similar results as PSW.
Conclusion This study did not provide evidence supporting protective effects for severe COVID-19 outcomes. However, the results are limited by the small cohort of patients treated with UDCA.
*Correspondence:
Huiqi Li huiqi.li@gu.se
1 School of Public Health and Community Medicine, Institute of Medicine,
Sahlgrenska Academy, University of Gothenburg, Box 469,
Gothenburg 40530 , Sweden
© The Author(s) 2026. Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit h t t p : / / c r e a t i v e c o m m o n s . o r g / l i c e n s e s / b y / 4 . 0 /.
DOI record:
{
"DOI": "10.1186/s12889-026-26908-1",
"ISSN": [
"1471-2458"
],
"URL": "http://dx.doi.org/10.1186/s12889-026-26908-1",
"alternative-id": [
"26908"
],
"article-number": "1240",
"assertion": [
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "Received",
"name": "received",
"order": 1,
"value": "15 November 2024"
},
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "Accepted",
"name": "accepted",
"order": 2,
"value": "1 March 2026"
},
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "First Online",
"name": "first_online",
"order": 3,
"value": "11 April 2026"
},
{
"group": {
"label": "Declarations",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 1
},
{
"group": {
"label": "Ethics approval and consent to participate",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 2,
"value": "The study is part of the SCIFI-PEARL project, and ethical approval was obtained from the Swedish Ethical Review Authority (Reference number 2020 − 01800 with subsequent amendments). As it is a register-based study, individual consent from participants was not required. The study was conducted in accordance with the Declaration of Helsinki."
},
{
"group": {
"label": "Consent for publication",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 3,
"value": "Not applicable."
},
{
"group": {
"label": "Competing interests",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 4,
"value": "FN, AS, HL report participation in a research project funded by Bayer (regulator-mandated phase IV study), with funds paid to the University of Gothenburg where they are employed (no personal fees) and with no relation to the work reported here. FN owns some AstraZeneca shares. All other authors have no competing interests to disclose."
}
],
"author": [
{
"affiliation": [],
"family": "Li",
"given": "Huiqi",
"sequence": "first"
},
{
"affiliation": [],
"family": "Xu",
"given": "Yiyi",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Kirui",
"given": "Brian",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Santosa",
"given": "Ailiana",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Nyberg",
"given": "Fredrik",
"sequence": "additional"
}
],
"container-title": "BMC Public Health",
"container-title-short": "BMC Public Health",
"content-domain": {
"crossmark-restriction": false,
"domain": [
"link.springer.com"
]
},
"created": {
"date-parts": [
[
2026,
4,
11
]
],
"date-time": "2026-04-11T07:33:10Z",
"timestamp": 1775892790000
},
"deposited": {
"date-parts": [
[
2026,
4,
16
]
],
"date-time": "2026-04-16T02:53:47Z",
"timestamp": 1776308027000
},
"funder": [
{
"DOI": "10.13039/501100005760",
"doi-asserted-by": "crossref",
"id": [
{
"asserted-by": "crossref",
"id": "10.13039/501100005760",
"id-type": "DOI"
}
],
"name": "University of Gothenburg"
}
],
"indexed": {
"date-parts": [
[
2026,
4,
16
]
],
"date-time": "2026-04-16T03:51:17Z",
"timestamp": 1776311477487,
"version": "3.50.1"
},
"is-referenced-by-count": 0,
"issue": "1",
"issued": {
"date-parts": [
[
2026,
4,
11
]
]
},
"journal-issue": {
"issue": "1",
"published-online": {
"date-parts": [
[
2026,
12
]
]
}
},
"language": "en",
"license": [
{
"URL": "https://creativecommons.org/licenses/by/4.0",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2026,
4,
11
]
],
"date-time": "2026-04-11T00:00:00Z",
"timestamp": 1775865600000
}
},
{
"URL": "https://creativecommons.org/licenses/by/4.0",
"content-version": "vor",
"delay-in-days": 5,
"start": {
"date-parts": [
[
2026,
4,
16
]
],
"date-time": "2026-04-16T00:00:00Z",
"timestamp": 1776297600000
}
}
],
"link": [
{
"URL": "https://link.springer.com/article/10.1186/s12889-026-26908-1",
"content-type": "text/html",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://link.springer.com/content/pdf/10.1186/s12889-026-26908-1.pdf",
"content-type": "application/pdf",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://link.springer.com/content/pdf/10.1186/s12889-026-26908-1.pdf",
"content-type": "application/pdf",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "297",
"original-title": [],
"prefix": "10.1186",
"published": {
"date-parts": [
[
2026,
4,
11
]
]
},
"published-online": {
"date-parts": [
[
2026,
4,
11
]
]
},
"publisher": "Springer Science and Business Media LLC",
"reference": [
{
"DOI": "10.1038/s41586-022-05594-0",
"author": "T Brevini",
"doi-asserted-by": "publisher",
"first-page": "134",
"journal-title": "Nature",
"key": "26908_CR1",
"unstructured": "Brevini T, Maes M, Webb GJ, John BV, Fuchs CD, Buescher G, et al. FXR inhibition may protect from SARS-CoV-2 infection by reducing ACE2. Nature. 2023;615:134–42. https://doi.org/10.1038/s41586-022-05594-0.",
"volume": "615",
"year": "2023"
},
{
"DOI": "10.1177/17562848241265782",
"author": "S Drazilova",
"doi-asserted-by": "publisher",
"first-page": "175628482412657",
"journal-title": "Ther Adv Gastroenterol",
"key": "26908_CR2",
"unstructured": "Drazilova S, Koky T, Macej M, Janicko M, Simkova D, Jarcuska P. The treatment of primary biliary cholangitis: from shadow to light. Ther Adv Gastroenterol. 2024;17:17562848241265782. https://doi.org/10.1177/17562848241265782.",
"volume": "17",
"year": "2024"
},
{
"DOI": "10.1111/joim.13630",
"author": "BV John",
"doi-asserted-by": "publisher",
"first-page": "636",
"journal-title": "J Intern Med",
"key": "26908_CR3",
"unstructured": "John BV, Bastaich D, Webb G, Brevini T, Moon A, Ferreira RD, et al. Ursodeoxycholic acid is associated with a reduction in SARS-CoV-2 infection and reduced severity of COVID-19 in patients with cirrhosis. J Intern Med. 2023;293:636–47. https://doi.org/10.1111/joim.13630.",
"volume": "293",
"year": "2023"
},
{
"DOI": "10.3389/fcimb.2023.1178590",
"author": "Y Li",
"doi-asserted-by": "publisher",
"first-page": "1178590",
"journal-title": "Front Cell Infect Microbiol",
"key": "26908_CR4",
"unstructured": "Li Y, Zhu N, Cui X, Lin Y, Li X. Protective effect of ursodeoxycholic acid on COVID-19 in patients with chronic liver disease. Front Cell Infect Microbiol. 2023;13:1178590. https://doi.org/10.3389/fcimb.2023.1178590.",
"volume": "13",
"year": "2023"
},
{
"DOI": "10.1093/qjmed/hcad254",
"doi-asserted-by": "publisher",
"key": "26908_CR5",
"unstructured": "Hu L, Zhang H, Huang C, Shen T, Feng Z, Mu F, et al. Effect of Ursodeoxycholic Acid on Preventing SARS-CoV-2 Infection in Patients With Liver Transplantation: a multicenter retrospective cohort study. QJM Mon J Assoc Physicians. 2023;hcad254. https://doi.org/10.1093/qjmed/hcad254."
},
{
"DOI": "10.1111/joim.13711",
"author": "L Ojeda-Fernández",
"doi-asserted-by": "publisher",
"journal-title": "J Intern Med",
"key": "26908_CR6",
"unstructured": "Ojeda-Fernández L, Baviera M, Macaluso G, Schena S, Tettamanti M, Cartabia M, et al. UDCA treatment against COVID-19: Do we have enough clinical evidence for drug repurposing? J Intern Med. 2023. https://doi.org/10.1111/joim.13711.",
"year": "2023"
},
{
"DOI": "10.1111/liv.15660",
"author": "T Liu",
"doi-asserted-by": "publisher",
"first-page": "1950",
"journal-title": "Liver Int Off J Int Assoc Study Liver",
"key": "26908_CR7",
"unstructured": "Liu T, Wang J-S. Ursodeoxycholic acid administration did not reduce susceptibility to SARS-CoV-2 infection in children. Liver Int Off J Int Assoc Study Liver. 2023;43:1950–4. https://doi.org/10.1111/liv.15660.",
"volume": "43",
"year": "2023"
},
{
"DOI": "10.3390/v15081738",
"author": "F Colapietro",
"doi-asserted-by": "publisher",
"first-page": "1738",
"journal-title": "Viruses",
"key": "26908_CR8",
"unstructured": "Colapietro F, Angelotti G, Masetti C, Shiffer D, Pugliese N, De Nicola S, et al. Ursodeoxycholic Acid Does Not Improve COVID-19 Outcome in Hospitalized Patients. Viruses. 2023;15:1738. https://doi.org/10.3390/v15081738.",
"volume": "15",
"year": "2023"
},
{
"DOI": "10.2147/CLEP.S312742",
"author": "F Nyberg",
"doi-asserted-by": "publisher",
"first-page": "649",
"journal-title": "Clin Epidemiol",
"key": "26908_CR9",
"unstructured": "Nyberg F, Franzén S, Lindh M, Vanfleteren L, Hammar N, Wettermark B, et al. Swedish Covid-19 Investigation for Future Insights - A Population Epidemiology Approach Using Register Linkage (SCIFI-PEARL). Clin Epidemiol. 2021;13:649–59. https://doi.org/10.2147/CLEP.S312742.",
"volume": "13",
"year": "2021"
},
{
"key": "26908_CR10",
"unstructured": "World Bank Country and Lending Groups – World Bank Data Help Desk. 2023. https://datahelpdesk.worldbank.org/knowledgebase/articles/906519-world-bank-country-and-lending-groups. Accessed 15 Nov 2023."
},
{
"DOI": "10.1007/978-3-319-19425-7",
"doi-asserted-by": "crossref",
"key": "26908_CR11",
"unstructured": "Harrell FE Jr. Regression Modeling Strategies: With Applications to Linear Models, Logistic and Ordinal Regression, and Survival Analysis. Second ed. Berlin: Springer; 2015."
},
{
"DOI": "10.1136/bmj.l5657",
"author": "RJ Desai",
"doi-asserted-by": "publisher",
"first-page": "l5657",
"journal-title": "BMJ",
"key": "26908_CR12",
"unstructured": "Desai RJ, Franklin JM. Alternative approaches for confounding adjustment in observational studies using weighting based on the propensity score: a primer for practitioners. BMJ. 2019;367:l5657. https://doi.org/10.1136/bmj.l5657.",
"volume": "367",
"year": "2019"
},
{
"key": "26908_CR13",
"unstructured": "Ridgeway G, McCaffrey D, Morral A, Cefalu M, Burgette L, Griffin BA. Toolkit for Weighting and Analysis of Nonequivalent Groups: A guide to the twang package. 2023."
},
{
"DOI": "10.1016/j.jclinepi.2013.01.013",
"doi-asserted-by": "publisher",
"key": "26908_CR14",
"unstructured": "Stuart EA, Lee BK, Leacy FP. Prognostic score-based balance measures can be a useful diagnostic for propensity score methods in comparative effectiveness research. J Clin Epidemiol. 2013;66(8 Suppl):S84-S90.e1. https://doi.org/10.1016/j.jclinepi.2013.01.013"
},
{
"DOI": "10.3389/fped.2021.616381",
"author": "A Di Giorgio",
"doi-asserted-by": "publisher",
"first-page": "616381",
"journal-title": "Front Pediatr",
"key": "26908_CR15",
"unstructured": "Di Giorgio A, Hartleif S, Warner S, Kelly D. COVID-19 in Children With Liver Disease. Front Pediatr. 2021;9:616381. https://doi.org/10.3389/fped.2021.616381.",
"volume": "9",
"year": "2021"
},
{
"DOI": "10.1016/j.jpeds.2020.04.026",
"author": "E Nicastro",
"doi-asserted-by": "publisher",
"first-page": "231",
"journal-title": "J Pediatr",
"key": "26908_CR16",
"unstructured": "Nicastro E, Mazza A, Gervasoni A, Di Giorgio A, D’Antiga L. A Pediatric Emergency Department Protocol to Avoid Intrahospital Spread of SARS-CoV-2 during the Outbreak in Bergamo, Italy. J Pediatr. 2020;222:231–5. https://doi.org/10.1016/j.jpeds.2020.04.026.",
"volume": "222",
"year": "2020"
},
{
"DOI": "10.1016/j.jhep.2023.06.011",
"author": "Z Li",
"doi-asserted-by": "publisher",
"first-page": "e157",
"journal-title": "J Hepatol",
"key": "26908_CR17",
"unstructured": "Li Z, Hu Y, Zou B. The vaccine-response in patients with cirrhosis after COVID-19 vaccination: A systematic analysis of 168,245 patients with cirrhosis. J Hepatol. 2023;79:e157–62. https://doi.org/10.1016/j.jhep.2023.06.011.",
"volume": "79",
"year": "2023"
},
{
"key": "26908_CR18",
"unstructured": "Hofmeister M, Stone EC, Okasako-Schmucker DL, Henry MC, Kumasaka J, Fellow O, et al. Brief Summary of Findings on the Association Between Underlying Liver Diseases and Severe COVID-19 Outcomes. 2025. https://www.cdc.gov/covid/media/pdfs/2025/02/B-ChronicLiverReview_Final.pdf"
},
{
"DOI": "10.1016/j.biopha.2022.113568",
"author": "P Li",
"doi-asserted-by": "publisher",
"first-page": "113568",
"journal-title": "Biomed Pharmacother Biomedecine Pharmacother",
"key": "26908_CR19",
"unstructured": "Li P, Liu Y, Cheng Z, Yu X, Li Y. COVID-19-associated liver injury: Clinical characteristics, pathophysiological mechanisms and treatment management. Biomed Pharmacother Biomedecine Pharmacother. 2022;154:113568. https://doi.org/10.1016/j.biopha.2022.113568.",
"volume": "154",
"year": "2022"
},
{
"DOI": "10.1097/EDE.0b013e3181875e61",
"author": "MA Hernán",
"doi-asserted-by": "publisher",
"first-page": "766",
"journal-title": "Epidemiol Camb Mass",
"key": "26908_CR20",
"unstructured": "Hernán MA, Alonso A, Logan R, Grodstein F, Michels KB, Willett WC, et al. Observational studies analyzed like randomized experiments: an application to postmenopausal hormone therapy and coronary heart disease. Epidemiol Camb Mass. 2008;19:766–79. https://doi.org/10.1097/EDE.0b013e3181875e61.",
"volume": "19",
"year": "2008"
}
],
"reference-count": 20,
"references-count": 20,
"relation": {},
"resource": {
"primary": {
"URL": "https://link.springer.com/10.1186/s12889-026-26908-1"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "Ursodeoxycholic acid treatment did not show protective effect for severe COVID-19 outcomes – a nationwide register study",
"type": "journal-article",
"update-policy": "https://doi.org/10.1007/springer_crossmark_policy",
"volume": "26"
}