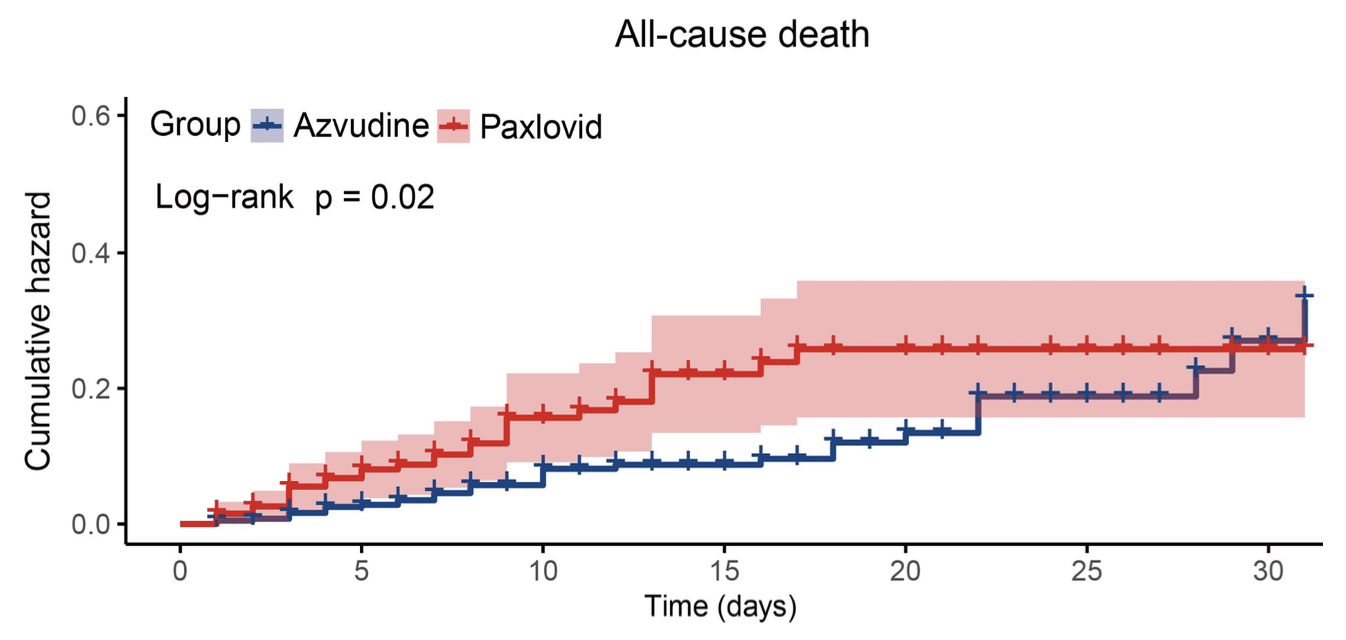
Efficacy and safety of azvudine versus nirmatrelvir/ritonavir in cancer patients with COVID-19
et al., Scientific Reports, doi:10.1038/s41598-025-85677-w, Mar 2025
PSM retrospective 596 cancer patients with COVID-19 showing that azvudine significantly reduced all-cause mortality and composite disease progression compared to paxlovid.
Resistance. Variants may be resistant to paxlovid1-9. Use may promote the emergence of variants that weaken host immunity and potentially contribute to long COVID10. Confounding by contraindication. Hoertel et al. find that over 50% of patients that died had a contraindication for the use of Paxlovid11. Retrospective studies that do not exclude contraindicated patients may significantly overestimate efficacy. Black box warning. The FDA notes that severe, life-threatening, and/or fatal adverse reactions due to drug interactions have been reported in patients treated with paxlovid12. Kidney and liver injury. Studies show significantly increased risk of acute kidney injury13 and liver injury14,15. Viral rebound. Studies show significantly increased risk of replication-competent viral rebound16-18.
Study covers azvudine and paxlovid.
1.
Zhou et al., Nirmatrelvir-resistant SARS-CoV-2 variants with high fitness in an infectious cell culture system, Science Advances, doi:10.1126/sciadv.add7197.
2.
Moghadasi et al., Rapid resistance profiling of SARS-CoV-2 protease inhibitors, npj Antimicrobials and Resistance, doi:10.1038/s44259-023-00009-0.
3.
Jochmans et al., The Substitutions L50F, E166A, and L167F in SARS-CoV-2 3CLpro Are Selected by a Protease Inhibitor In Vitro and Confer Resistance To Nirmatrelvir, mBio, doi:10.1128/mbio.02815-22.
4.
Lopez et al., SARS-CoV-2 Resistance to Small Molecule Inhibitors, Current Clinical Microbiology Reports, doi:10.1007/s40588-024-00229-6.
5.
Zvornicanin et al., Molecular Mechanisms of Drug Resistance and Compensation in SARS-CoV-2 Main Protease: The Interplay Between E166 and L50, bioRxiv, doi:10.1101/2025.01.24.634813.
6.
Vukovikj et al., Impact of SARS-CoV-2 variant mutations on susceptibility to monoclonal antibodies and antiviral drugs: a non-systematic review, April 2022 to October 2024, Eurosurveillance, doi:10.2807/1560-7917.ES.2025.30.10.2400252.
7.
Deschenes et al., Functional and structural characterization of treatment-emergent nirmatrelvir resistance mutations at low frequencies in the main protease (Mpro) reveals a unique evolutionary route for SARS-CoV-2 to gain resistance, The Journal of Infectious Diseases, doi:10.1093/infdis/jiaf294.
8.
Zhou (B) et al., SARS-CoV-2 Mpro inhibitor ensitrelvir: asymmetrical cross-resistance with nirmatrelvir and emerging resistance hotspots, Emerging Microbes & Infections, doi:10.1080/22221751.2025.2552716.
9.
Chen et al., SARS-CoV-2 3CLpro mutations T21I and E166A confer differential resistance to simnotrelvir, bofutrelvir, and ensitrelvir, Journal of Virology, doi:10.1128/jvi.02223-25.
10.
Thomas et al., Nirmatrelvir-Resistant Mutations in SARS-CoV-2 Mpro Enhance Host Immune Evasion via Cleavage of NF-κB Essential Modulator, bioRxiv, doi:10.1101/2024.10.18.619137.
11.
Hoertel et al., Prevalence of Contraindications to Nirmatrelvir-Ritonavir Among Hospitalized Patients With COVID-19 at Risk for Progression to Severe Disease, JAMA Network Open, doi:10.1001/jamanetworkopen.2022.42140.
12.
FDA, Fact sheet for healthcare providers: emergency use authorization for paxlovid, www.fda.gov/media/155050/download.
13.
Kamo et al., Association of Antiviral Drugs for the Treatment of COVID-19 With Acute Renal Failure, In Vivo, doi:10.21873/invivo.13637.
14.
Wang et al., Development and validation of a nomogram to assess the occurrence of liver dysfunction in patients with COVID-19 pneumonia in the ICU, BMC Infectious Diseases, doi:10.1186/s12879-025-10684-1.
15.
Siby et al., Temporal Trends in Serious Adverse Events Associated with Oral Antivirals During the COVID-19 Pandemic: Insights from the FAERS Database (2020–2023), Open Forum Infectious Diseases, doi:10.1093/ofid/ofaf695.1825.
16.
Edelstein et al., SARS-CoV-2 virologic rebound with nirmatrelvir-ritonavir therapy, medRxiv, doi:10.1101/2023.06.23.23288598.
Jia et al., 31 Mar 2025, retrospective, China, peer-reviewed, 16 authors, study period 5 December, 2022 - 31 January, 2023.
Contact: hxliang@zzu.edu.cn, johnyuem@zzu.edu.cn, fccrenzg@zzu.edu.cn.
Efficacy and safety of azvudine versus nirmatrelvir/ritonavir in cancer patients with COVID-19
Scientific Reports, doi:10.1038/s41598-025-85677-w
Cancer significantly contributes to the unfavorable prognosis of coronavirus disease 2019 (COVID-19) patients. The efficacy and safety of azvudine and nirmatrelvir/ritonavir (Paxlovid) in cancer patients with COVID-19 remain uncertain. Therefore, we designed a comprehensive retrospective study encompassing clinical data of 32,864 hospitalized COVID-19 patients, 691 of whom were cancer patients treated with azvudine and 200 were cancer patients treated with Paxlovid. After 2:1 propensity score matching, 397 patients in the azvudine group and 199 patients in the Paxlovid group were enrolled. Cox regression analysis revealed the risk of all-cause death (HR: 1.84, 95% CI: 1.059-3.182, P = 0.030) and composite disease progression (HR: 1.70, 95% CI: 1.043-2.757, P = 0.033) were greater in the Paxlovid group than in the azvudine group. Two sensitivity analyses confirmed the robustness of our findings. The safety analysis of adverse events revealed no statistically significant differences between the two groups. In conclusion, we carried out the first analysis to compare the efficacy and safety of azvudine and Paxlovid in cancer patients with COVID-19 and demonstrated that azvudine significantly reduced the risk of all-cause death and composite disease progression among cancer patients with COVID-19 compared with Paxlovid.
Author contributions Bohan Jia, Junyi Sun, Di Zhu, Ling Wang and Xiaobo Hu contributed equally to this work. Hongxia Liang, Zujiang Yu and Zhigang Ren conceived and designed the study; Guowu Qian, Donghua Zhang, Silin Li, Hong Luo, Shixi Zhang, Guotao Li and Guangming Li managed the patients and collected the data; Di Zhu, Haiyu Wang, Ling Wang and Xiaobo Hu analyzed the data; Bohan Jia, Junyi Sun wrote the manuscript; All authors reviewed and approved the manuscript.
Competing interests The authors declare no competing interests.
Additional information
Supplementary Information The online version contains supplementary material available at h t t p s : / / d o i . o r g / 1 0 . 1 0 3 8 / s 4 1 5 9 8 -0 2 5 -8 5 6 7 7 -w . Correspondence and requests for materials should be addressed to H.L., Z.Y. or Z.R. Reprints and permissions information is available at www.nature.com/reprints. Publisher's note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations. Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to..
References
Amani, Amani, Azvudine versus paxlovid in COVID-19: a systematic review and meta-analysis, Rev. Med. Virol
Amani, Amani, Effectiveness and safety of azvudine in COVID-19: a systematic review and meta-analysis, PLoS One
Anwar, Nguyen, Nagasaka, Ou, Chan, Overview of drug-drug interactions between ritonavir-boosted nirmatrelvir (paxlovid) and targeted therapy and supportive care for lung cancer, JTO Clin. Res. Rep
Bray, Laversanne, Weiderpass, Soerjomataram, The ever-increasing importance of cancer as a leading cause of premature death worldwide, Cancer
Dai, Patients with cancer appear more vulnerable to SARS-CoV-2: a multicenter study during the COVID-19 outbreak, Cancer Discov
Guan, Clinical characteristics of coronavirus disease 2019 in China, N. Engl. J. Med
Guermazi, Arvanitis, Vieira, Warner, Farmakiotis, Oral antivirals for COVID-19 among patients with cancer, Res. Square, doi:10.21203/rs.3.rs-3876022/v1
Hammond, Oral nirmatrelvir for high-risk, nonhospitalized adults with covid-19, N Engl. J. Med
Jung, Altered pathogenesis of porcine respiratory coronavirus in pigs due to immunosuppressive effects of dexamethasone: implications for corticosteroid use in treatment of severe acute respiratory syndrome coronavirus, J. Virol
Kamboj, Sepkowitz, Nosocomial infections in patients with cancer, Lancet Oncol
Lee, COVID-19 mortality in patients with cancer on chemotherapy or other anticancer treatments: a prospective cohort study, Lancet (Lond. Engl.)
Lee, Effects of early corticosteroid treatment on plasma SARS-associated coronavirus RNA concentrations in adult patients, J. Clin. Virol
Li, A retrospective analysis of azvudine in patients with COVID-19 and pre-existing cancer, J. Cancer
Liang, Cancer patients in SARS-CoV-2 infection: a nationwide analysis in China, Lancet Oncol
Ma, Corticosteroid use in the treatment of COVID-19: a Multicenter Retrospective Study in Hunan, China, Front. Pharmacol
Ma, Yin, Qian, Wu, Clinical characteristics and prognosis in cancer patients with COVID-19: a single center's retrospective study, J. Infect
Najjar-Debbiny, Effectiveness of paxlovid in reducing severe coronavirus disease 2019 and mortality in high-risk patients, Clin. Infect. Dis
Pardoll, Cancer and the immune system: basic concepts and targets for intervention, Semin. Oncol
Ren, A randomized, open-label, controlled clinical trial of azvudine tablets in the treatment of mild and common COVID-19, a pilot study, Adv. Sci. (Weinh.)
Sica, Massarotti, Myeloid suppressor cells in cancer and autoimmunity, J. Autoimmun
Sun, Mechanistic insight into antiretroviral potency of 2'-deoxy-2'-β-fluoro-4'-azidocytidine (FNC) with a long-lasting effect on HIV-1 prevention, J. Med. Chem
Sun, Oral azvudine for hospitalised patients with COVID-19 and pre-existing conditions: a retrospective cohort study, EClinicalMedicine
Sung, GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries, Cancer J. Clin
Tobaiqy, Therapeutic management of patients with COVID-19: a systematic review, Infect. Prev. Pract
V'kovski, Kratzel, Steiner, Stalder, Thiel, Coronavirus biology and replication: implications for SARS-CoV-2, Nat. Rev. Microbiol
Vaishampayan, Parchment, Jasti, Hussain, Taxanes: an overview of the pharmacokinetics and pharmacodynamics, Urology
Vineis, Wild, Global cancer patterns: causes and prevention, Lancet
Wang, Therapeutic response to corticosteroids in a critically ill patient with COVID-19: a case report, Med. (Baltim)
Yang, Rao, Structural biology of SARS-CoV-2 and implications for therapeutic development, Nat. Rev. Microbiol
Yao, Ding, Burchell, Wolf, Friedberg, Detoxication of vinca alkaloids by human P450 CYP3A4-mediated metabolism: implications for the development of drug resistance, J. Pharmacol. Exp. Ther
Zhang, Azvudine is a thymus-homing anti-SARS-CoV-2 drug effective in treating COVID-19 patients, Signal. Transduct. Target. Therapy
Zhang, Clinical characteristics of COVID-19-infected cancer patients: a retrospective case study in three hospitals within Wuhan, China, Ann. Oncol
Zhang, NKG2A is a NK cell exhaustion checkpoint for HCV persistence, Nat. Commun
DOI record:
{
"DOI": "10.1038/s41598-025-85677-w",
"ISSN": [
"2045-2322"
],
"URL": "http://dx.doi.org/10.1038/s41598-025-85677-w",
"alternative-id": [
"85677"
],
"article-number": "11022",
"assertion": [
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "Received",
"name": "received",
"order": 1,
"value": "2 August 2024"
},
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "Accepted",
"name": "accepted",
"order": 2,
"value": "6 January 2025"
},
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "First Online",
"name": "first_online",
"order": 3,
"value": "31 March 2025"
},
{
"group": {
"label": "Competing interests",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 1,
"value": "The authors declare no competing interests."
}
],
"author": [
{
"affiliation": [],
"family": "Jia",
"given": "Bohan",
"sequence": "first"
},
{
"affiliation": [],
"family": "Sun",
"given": "Junyi",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Zhu",
"given": "Di",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Wang",
"given": "Ling",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Hu",
"given": "Xiaobo",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Wang",
"given": "Haiyu",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Qian",
"given": "Guowu",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Zhang",
"given": "Donghua",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Li",
"given": "Silin",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Luo",
"given": "Hong",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Zhang",
"given": "Shixi",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Li",
"given": "Guotao",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Li",
"given": "Guangming",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Liang",
"given": "Hongxia",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Yu",
"given": "Zujiang",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ren",
"given": "Zhigang",
"sequence": "additional"
}
],
"container-title": "Scientific Reports",
"container-title-short": "Sci Rep",
"content-domain": {
"crossmark-restriction": false,
"domain": [
"link.springer.com"
]
},
"created": {
"date-parts": [
[
2025,
3,
31
]
],
"date-time": "2025-03-31T19:42:46Z",
"timestamp": 1743450166000
},
"deposited": {
"date-parts": [
[
2025,
3,
31
]
],
"date-time": "2025-03-31T19:43:48Z",
"timestamp": 1743450228000
},
"funder": [
{
"DOI": "10.13039/501100012166",
"award": [
"2023YFC3043514",
"2022YFC2303100"
],
"doi-asserted-by": "publisher",
"id": [
{
"asserted-by": "publisher",
"id": "10.13039/501100012166",
"id-type": "DOI"
}
],
"name": "National Key Research and Development Program of China"
},
{
"award": [
"ZYCXTD2023002",
"QNCXTD2023002"
],
"name": "Scientific Research and Innovation Team of The First Affiliated Hospital of Zhengzhou University"
},
{
"name": "2024 Special Project of the National Key Laboratory of Innovative Drugs for Antiviral Infectious Diseases"
}
],
"indexed": {
"date-parts": [
[
2025,
3,
31
]
],
"date-time": "2025-03-31T20:10:22Z",
"timestamp": 1743451822445,
"version": "3.40.3"
},
"is-referenced-by-count": 0,
"issue": "1",
"issued": {
"date-parts": [
[
2025,
3,
31
]
]
},
"journal-issue": {
"issue": "1",
"published-online": {
"date-parts": [
[
2025,
12
]
]
}
},
"language": "en",
"license": [
{
"URL": "https://creativecommons.org/licenses/by-nc-nd/4.0",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2025,
3,
31
]
],
"date-time": "2025-03-31T00:00:00Z",
"timestamp": 1743379200000
}
},
{
"URL": "https://creativecommons.org/licenses/by-nc-nd/4.0",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2025,
3,
31
]
],
"date-time": "2025-03-31T00:00:00Z",
"timestamp": 1743379200000
}
}
],
"link": [
{
"URL": "https://www.nature.com/articles/s41598-025-85677-w.pdf",
"content-type": "application/pdf",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://www.nature.com/articles/s41598-025-85677-w",
"content-type": "text/html",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://www.nature.com/articles/s41598-025-85677-w.pdf",
"content-type": "application/pdf",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "297",
"original-title": [],
"prefix": "10.1038",
"published": {
"date-parts": [
[
2025,
3,
31
]
]
},
"published-online": {
"date-parts": [
[
2025,
3,
31
]
]
},
"publisher": "Springer Science and Business Media LLC",
"reference": [
{
"DOI": "10.1002/cncr.33587",
"author": "F Bray",
"doi-asserted-by": "crossref",
"first-page": "3029",
"journal-title": "Cancer",
"key": "85677_CR1",
"unstructured": "Bray, F., Laversanne, M., Weiderpass, E. & Soerjomataram, I. The ever-increasing importance of cancer as a leading cause of premature death worldwide. Cancer 127, 3029–3030 (2021).",
"volume": "127",
"year": "2021"
},
{
"DOI": "10.3322/caac.21660",
"author": "H Sung",
"doi-asserted-by": "crossref",
"first-page": "209",
"journal-title": "Cancer J. Clin.",
"key": "85677_CR2",
"unstructured": "Sung, H. et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. Cancer J. Clin. 71, 209–249 (2021).",
"volume": "71",
"year": "2021"
},
{
"DOI": "10.1016/S0140-6736(13)62224-2",
"author": "P Vineis",
"doi-asserted-by": "crossref",
"first-page": "549",
"journal-title": "Lancet",
"key": "85677_CR3",
"unstructured": "Vineis, P. & Wild, C. P. Global cancer patterns: causes and prevention. Lancet 383, 549–557 (2014).",
"volume": "383",
"year": "2014"
},
{
"DOI": "10.1053/j.seminoncol.2015.05.003",
"author": "D Pardoll",
"doi-asserted-by": "crossref",
"first-page": "523",
"journal-title": "Semin. Oncol.",
"key": "85677_CR4",
"unstructured": "Pardoll, D. Cancer and the immune system: basic concepts and targets for intervention. Semin. Oncol. 42, 523–538 (2015).",
"volume": "42",
"year": "2015"
},
{
"DOI": "10.1016/j.jaut.2017.07.010",
"author": "A Sica",
"doi-asserted-by": "crossref",
"first-page": "117",
"journal-title": "J. Autoimmun.",
"key": "85677_CR5",
"unstructured": "Sica, A. & Massarotti, M. Myeloid suppressor cells in cancer and autoimmunity. J. Autoimmun. 85, 117–125 (2017).",
"volume": "85",
"year": "2017"
},
{
"DOI": "10.1016/S0140-6736(20)31173-9",
"author": "LY Lee",
"doi-asserted-by": "crossref",
"first-page": "1919",
"journal-title": "Lancet (Lond. Engl.)",
"key": "85677_CR6",
"unstructured": "Lee, L. Y. et al. COVID-19 mortality in patients with cancer on chemotherapy or other anticancer treatments: a prospective cohort study. Lancet (Lond. Engl.) 395, 1919–1926 (2020).",
"volume": "395",
"year": "2020"
},
{
"DOI": "10.1016/S1470-2045(09)70069-5",
"author": "M Kamboj",
"doi-asserted-by": "crossref",
"first-page": "589",
"journal-title": "Lancet Oncol.",
"key": "85677_CR7",
"unstructured": "Kamboj, M. & Sepkowitz, K. A. Nosocomial infections in patients with cancer. Lancet Oncol. 10, 589–597 (2009).",
"volume": "10",
"year": "2009"
},
{
"DOI": "10.1038/s41579-021-00630-8",
"author": "H Yang",
"doi-asserted-by": "crossref",
"first-page": "685",
"journal-title": "Nat. Rev. Microbiol.",
"key": "85677_CR8",
"unstructured": "Yang, H. & Rao, Z. Structural biology of SARS-CoV-2 and implications for therapeutic development. Nat. Rev. Microbiol. 19, 685–700 (2021).",
"volume": "19",
"year": "2021"
},
{
"DOI": "10.1038/s41579-020-00468-6",
"author": "P V’kovski",
"doi-asserted-by": "crossref",
"first-page": "155",
"journal-title": "Nat. Rev. Microbiol.",
"key": "85677_CR9",
"unstructured": "V’kovski, P., Kratzel, A., Steiner, S., Stalder, H. & Thiel, V. Coronavirus biology and replication: implications for SARS-CoV-2. Nat. Rev. Microbiol. 19, 155–170 (2021).",
"volume": "19",
"year": "2021"
},
{
"DOI": "10.1056/NEJMoa2002032",
"author": "WJ Guan",
"doi-asserted-by": "crossref",
"first-page": "1708",
"journal-title": "N. Engl. J. Med.",
"key": "85677_CR10",
"unstructured": "Guan, W. J. et al. Clinical characteristics of coronavirus disease 2019 in China. N. Engl. J. Med. 382, 1708–1720 (2020).",
"volume": "382",
"year": "2020"
},
{
"DOI": "10.1016/S1470-2045(20)30096-6",
"author": "W Liang",
"doi-asserted-by": "crossref",
"first-page": "335",
"journal-title": "Lancet Oncol.",
"key": "85677_CR11",
"unstructured": "Liang, W. et al. Cancer patients in SARS-CoV-2 infection: a nationwide analysis in China. Lancet Oncol. 21, 335–337 (2020).",
"volume": "21",
"year": "2020"
},
{
"DOI": "10.1016/j.annonc.2020.03.296",
"author": "L Zhang",
"doi-asserted-by": "crossref",
"first-page": "894",
"journal-title": "Ann. Oncol.",
"key": "85677_CR12",
"unstructured": "Zhang, L. et al. Clinical characteristics of COVID-19-infected cancer patients: a retrospective case study in three hospitals within Wuhan, China. Ann. Oncol. 31, 894–901 (2020).",
"volume": "31",
"year": "2020"
},
{
"DOI": "10.1056/NEJMoa2118542",
"author": "J Hammond",
"doi-asserted-by": "crossref",
"first-page": "1397",
"journal-title": "N Engl. J. Med.",
"key": "85677_CR13",
"unstructured": "Hammond, J. et al. Oral nirmatrelvir for high-risk, nonhospitalized adults with covid-19. N Engl. J. Med. 386, 1397–1408 (2022).",
"volume": "386",
"year": "2022"
},
{
"DOI": "10.1093/cid/ciac443",
"author": "R Najjar-Debbiny",
"doi-asserted-by": "crossref",
"first-page": "e342",
"journal-title": "Clin. Infect. Dis.",
"key": "85677_CR14",
"unstructured": "Najjar-Debbiny, R. et al. Effectiveness of paxlovid in reducing severe coronavirus disease 2019 and mortality in high-risk patients. Clin. Infect. Dis. 76, e342–e349 (2023).",
"volume": "76",
"year": "2023"
},
{
"DOI": "10.1021/acs.jmedchem.0c00940",
"author": "L Sun",
"doi-asserted-by": "crossref",
"first-page": "8554",
"journal-title": "J. Med. Chem.",
"key": "85677_CR15",
"unstructured": "Sun, L. et al. Mechanistic insight into antiretroviral potency of 2’-deoxy-2’-β-fluoro-4’-azidocytidine (FNC) with a long-lasting effect on HIV-1 prevention. J. Med. Chem. 63, 8554–8566 (2020).",
"volume": "63",
"year": "2020"
},
{
"key": "85677_CR16",
"unstructured": "National Medical Products Administratior. The National Medical Products Administration has conditionally approved the marketing of Azvudine Tablets (2024, accessed 26 Dec 2024). https://www.nmpa.gov.cn/zhuanti/cxylqx/cxypxx/20210721142223181.html."
},
{
"author": "Z Ren",
"first-page": "e2001435",
"journal-title": "Adv. Sci. (Weinh.)",
"key": "85677_CR17",
"unstructured": "Ren, Z. et al. A randomized, open-label, controlled clinical trial of azvudine tablets in the treatment of mild and common COVID-19, a pilot study. Adv. Sci. (Weinh.) 7, e2001435 (2020).",
"volume": "7",
"year": "2020"
},
{
"DOI": "10.1038/s41392-021-00835-6",
"author": "JL Zhang",
"doi-asserted-by": "crossref",
"first-page": "414",
"journal-title": "Signal. Transduct. Target. Therapy",
"key": "85677_CR18",
"unstructured": "Zhang, J. L. et al. Azvudine is a thymus-homing anti-SARS-CoV-2 drug effective in treating COVID-19 patients. Signal. Transduct. Target. Therapy 6, 414 (2021).",
"volume": "6",
"year": "2021"
},
{
"DOI": "10.1158/2159-8290.CD-20-0422",
"author": "M Dai",
"doi-asserted-by": "crossref",
"first-page": "783",
"journal-title": "Cancer Discov.",
"key": "85677_CR19",
"unstructured": "Dai, M. et al. Patients with cancer appear more vulnerable to SARS-CoV-2: a multicenter study during the COVID-19 outbreak. Cancer Discov. 10, 783–791 (2020).",
"volume": "10",
"year": "2020"
},
{
"DOI": "10.1016/j.jinf.2020.04.006",
"author": "J Ma",
"doi-asserted-by": "crossref",
"first-page": "318",
"journal-title": "J. Infect.",
"key": "85677_CR20",
"unstructured": "Ma, J., Yin, J., Qian, Y. & Wu, Y. Clinical characteristics and prognosis in cancer patients with COVID-19: a single center’s retrospective study. J. Infect. 81, 318–356 (2020).",
"volume": "81",
"year": "2020"
},
{
"DOI": "10.1002/rmv.2551",
"author": "B Amani",
"doi-asserted-by": "crossref",
"first-page": "e2551",
"journal-title": "Rev. Med. Virol.",
"key": "85677_CR21",
"unstructured": "Amani, B. & Amani, B. Azvudine versus paxlovid in COVID-19: a systematic review and meta-analysis. Rev. Med. Virol. 34, e2551 (2024).",
"volume": "34",
"year": "2024"
},
{
"DOI": "10.1371/journal.pone.0298772",
"author": "B Amani",
"doi-asserted-by": "crossref",
"first-page": "e0298772",
"journal-title": "PLoS One",
"key": "85677_CR22",
"unstructured": "Amani, B. & Amani, B. Effectiveness and safety of azvudine in COVID-19: a systematic review and meta-analysis. PLoS One 19, e0298772 (2024).",
"volume": "19",
"year": "2024"
},
{
"DOI": "10.7150/jca.91530",
"author": "F Li",
"doi-asserted-by": "crossref",
"first-page": "2442",
"journal-title": "J. Cancer",
"key": "85677_CR23",
"unstructured": "Li, F. et al. A retrospective analysis of azvudine in patients with COVID-19 and pre-existing cancer. J. Cancer 15, 2442–2447 (2024).",
"volume": "15",
"year": "2024"
},
{
"DOI": "10.21203/rs.3.rs-3876022/v1",
"doi-asserted-by": "publisher",
"key": "85677_CR24",
"unstructured": "Guermazi, D., Arvanitis, P., Vieira, K., Warner, J. L. & Farmakiotis, D. Oral antivirals for COVID-19 among patients with cancer. Res. Square. https://doi.org/10.21203/rs.3.rs-3876022/v1 (2024)."
},
{
"DOI": "10.1038/s41467-019-09212-y",
"author": "C Zhang",
"doi-asserted-by": "crossref",
"first-page": "1507",
"journal-title": "Nat. Commun.",
"key": "85677_CR25",
"unstructured": "Zhang, C. et al. NKG2A is a NK cell exhaustion checkpoint for HCV persistence. Nat. Commun. 10, 1507 (2019).",
"volume": "10",
"year": "2019"
},
{
"author": "K Anwar",
"first-page": "100452",
"journal-title": "JTO Clin. Res. Rep.",
"key": "85677_CR26",
"unstructured": "Anwar, K., Nguyen, L., Nagasaka, M., Ou, S. H. I. & Chan, A. Overview of drug-drug interactions between ritonavir-boosted nirmatrelvir (paxlovid) and targeted therapy and supportive care for lung cancer. JTO Clin. Res. Rep. 4, 100452 (2023).",
"volume": "4",
"year": "2023"
},
{
"DOI": "10.1016/S0090-4295(99)00451-3",
"author": "U Vaishampayan",
"doi-asserted-by": "crossref",
"first-page": "22",
"journal-title": "Urology",
"key": "85677_CR27",
"unstructured": "Vaishampayan, U., Parchment, R. E., Jasti, B. R. & Hussain, M. Taxanes: an overview of the pharmacokinetics and pharmacodynamics. Urology 54, 22–29 (1999).",
"volume": "54",
"year": "1999"
},
{
"DOI": "10.1016/S0022-3565(24)39081-0",
"author": "D Yao",
"doi-asserted-by": "crossref",
"first-page": "387",
"journal-title": "J. Pharmacol. Exp. Ther.",
"key": "85677_CR28",
"unstructured": "Yao, D., Ding, S., Burchell, B., Wolf, C. R. & Friedberg, T. Detoxication of vinca alkaloids by human P450 CYP3A4-mediated metabolism: implications for the development of drug resistance. J. Pharmacol. Exp. Ther. 294, 387–395 (2000).",
"volume": "294",
"year": "2000"
},
{
"DOI": "10.1016/j.infpip.2020.100061",
"author": "M Tobaiqy",
"doi-asserted-by": "crossref",
"first-page": "100061",
"journal-title": "Infect. Prev. Pract.",
"key": "85677_CR29",
"unstructured": "Tobaiqy, M. et al. Therapeutic management of patients with COVID-19: a systematic review. Infect. Prev. Pract. 2, 100061 (2020).",
"volume": "2",
"year": "2020"
},
{
"DOI": "10.1097/MD.0000000000021597",
"author": "K Wang",
"doi-asserted-by": "crossref",
"first-page": "e21597",
"journal-title": "Med. (Baltim).",
"key": "85677_CR30",
"unstructured": "Wang, K. et al. Therapeutic response to corticosteroids in a critically ill patient with COVID-19: a case report. Med. (Baltim). 99, e21597 (2020).",
"volume": "99",
"year": "2020"
},
{
"DOI": "10.1001/jama.2020.17023",
"author": "WHO Rapid Evidence Appraisal for COVID-19 Therapies (REACT) Working Group",
"doi-asserted-by": "crossref",
"first-page": "1330",
"journal-title": "Jama",
"key": "85677_CR31",
"unstructured": "WHO Rapid Evidence Appraisal for COVID-19 Therapies (REACT) Working Group. Association between administration of systemic corticosteroids and mortality among critically ill patients with COVID-19: a meta-analysis. Jama 324, 1330–1341 (2020).",
"volume": "324",
"year": "2020"
},
{
"DOI": "10.1128/JVI.01702-07",
"author": "K Jung",
"doi-asserted-by": "crossref",
"first-page": "13681",
"journal-title": "J. Virol.",
"key": "85677_CR32",
"unstructured": "Jung, K. et al. Altered pathogenesis of porcine respiratory coronavirus in pigs due to immunosuppressive effects of dexamethasone: implications for corticosteroid use in treatment of severe acute respiratory syndrome coronavirus. J. Virol. 81, 13681–13693 (2007).",
"volume": "81",
"year": "2007"
},
{
"DOI": "10.1016/j.jcv.2004.07.006",
"author": "N Lee",
"doi-asserted-by": "crossref",
"first-page": "304",
"journal-title": "J. Clin. Virol.",
"key": "85677_CR33",
"unstructured": "Lee, N. et al. Effects of early corticosteroid treatment on plasma SARS-associated coronavirus RNA concentrations in adult patients. J. Clin. Virol. 31, 304–309 (2004).",
"volume": "31",
"year": "2004"
},
{
"DOI": "10.3389/fphar.2020.01198",
"author": "Y Ma",
"doi-asserted-by": "crossref",
"first-page": "1198",
"journal-title": "Front. Pharmacol.",
"key": "85677_CR34",
"unstructured": "Ma, Y. et al. Corticosteroid use in the treatment of COVID-19: a Multicenter Retrospective Study in Hunan, China. Front. Pharmacol. 11, 1198 (2020).",
"volume": "11",
"year": "2020"
},
{
"DOI": "10.1016/j.eclinm.2023.101981",
"author": "Y Sun",
"doi-asserted-by": "crossref",
"first-page": "101981",
"journal-title": "EClinicalMedicine",
"key": "85677_CR35",
"unstructured": "Sun, Y. et al. Oral azvudine for hospitalised patients with COVID-19 and pre-existing conditions: a retrospective cohort study. EClinicalMedicine 59, 101981 (2023).",
"volume": "59",
"year": "2023"
}
],
"reference-count": 35,
"references-count": 35,
"relation": {},
"resource": {
"primary": {
"URL": "https://www.nature.com/articles/s41598-025-85677-w"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "Efficacy and safety of azvudine versus nirmatrelvir/ritonavir in cancer patients with COVID-19",
"type": "journal-article",
"update-policy": "https://doi.org/10.1007/springer_crossmark_policy",
"volume": "15"
}
jia2