Ivermectin for patients admitted to an ICU with COVID-19: REMAP-CAP randomized controlled trial
et al., Critical Care Medicine, doi:10.1097/CCM.0000000000007134 (partial results released 6/13/2024), REMAP-CAP, NCT02735707, Jun 2024
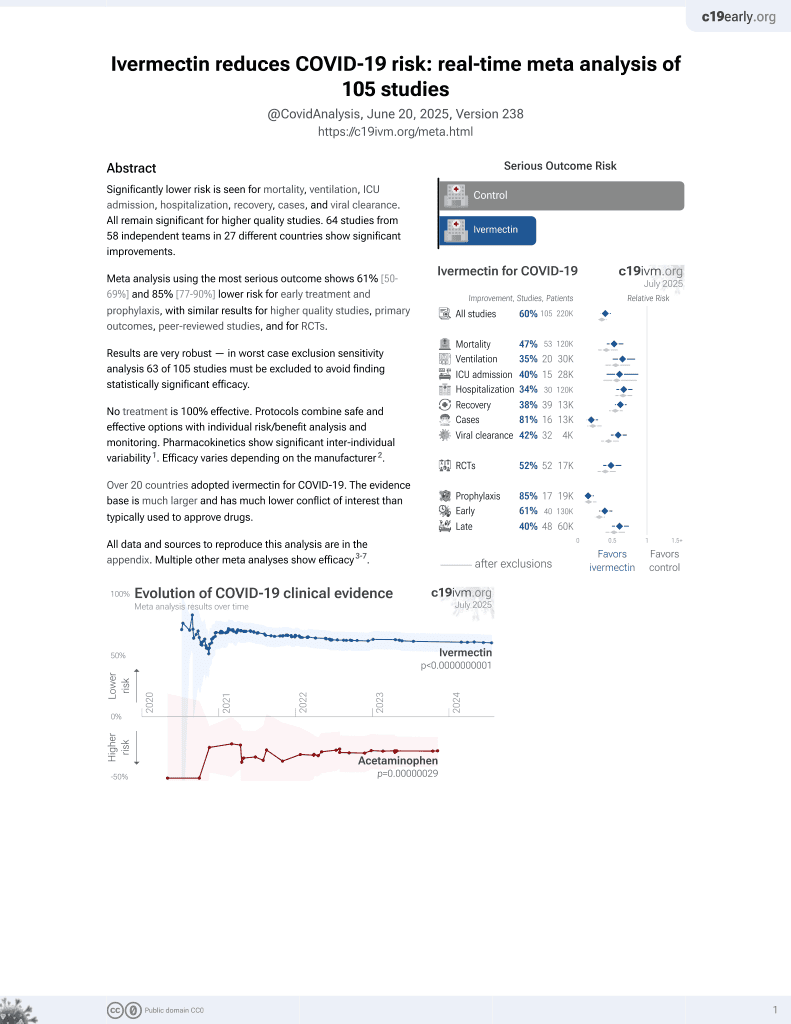
Ivermectin for COVID-19
4th treatment shown to reduce risk in
August 2020, now with p < 0.00000000001 from 106 studies, recognized in 24 countries.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
Early terminated REMAP-CAP results delayed
553 days (1,247 days for the more accurate concurrent ivermectin-specific analysis),
showing no significant differences with very low dose, poor administration, very late
treatment of late-stage patients.
Results trend towards benefit for non-critical patients in the primary analysis,
and for both groups in the more accurate concurrent ivermectin-specific analysis.
There are many critical issues as below. Two post-completion SAPs show
significant changes with the second version removing adjustment for severity, which significantly
favors the control group due to the severe baseline imbalance.
Responses: authors have not responded to any of these issues.
CRITICALNo severity adjustment; in protocol, removed in second post-hoc SAP.
The trial protocol includes adjustment for baseline severity1, which is important because the response-adaptive randomization may result in severe imbalance. Siddiqui et al. report that there were two post-completion statistical analysis plans with the second one removing adjustment for baseline severity. The first one, 5 months after completion, includes baseline disease state in adjustments3 - authors do not specify the state but baseline data includes APACHE II scores and PaO2/FiO2 (state here could not be critical/non-critical because those are already analyzed separately). This SAP has "FINAL" in the name, however a second post-completion SAP4, dated over a year after completion, deletes the baseline disease state adjustment, thereby favoring the control group which had lower baseline severity.
CRITICALBaseline severity favors control.
Randomization resulted in 58% more critical patients at baseline in the treatment group (38 vs. 24). Baseline APACHE II scores were higher in both critical and non-critical groups (11 vs. 9 and 7 vs. 6). 25% of critical treatment patients had APACHE II ≥17 while the corresponding value for the control group is 12. The difference in expected mortality for APACHE II 17 vs. 12 is about 2x. The PaO2/FiO2 medians show severe ARDS for treatment versus moderate ARDS for control.
CRITICALCONSORT guidelines not followed.
CONSORT guidelines state that if a severe baseline imbalance occurs, authors should transparently report it and provide an adjusted analysis.
CRITICALConcurrent cohort results better.
Table S7 shows results for the concurrent ivermectin-specific cohort. These results should be more accurate because the controls were randomized at the same sites in the same time window under the same conditions as the ivermectin patients, eliminating cross-domain and cross-period confounding that the pooled analysis must correct for through model assumptions about exchangeability across the broader REMAP-CAP cohort. Significant time-related confounding is expected - the ivermectin domain ran June 2021-September 2022, while non-concurrent controls span earlier pandemic periods with different variants, vaccination rates, ICU strain, and standards of care - and the pooled analysis must correct for these differences through additive time, site, and domain effects whose assumptions cannot be verified in a 150-patient three-country study. With samples this small, the data are insufficient to support the cross-domain exchangeability and interaction-free adjustments that the pooled model requires, so the simpler concurrent-control comparison is more appropriate.
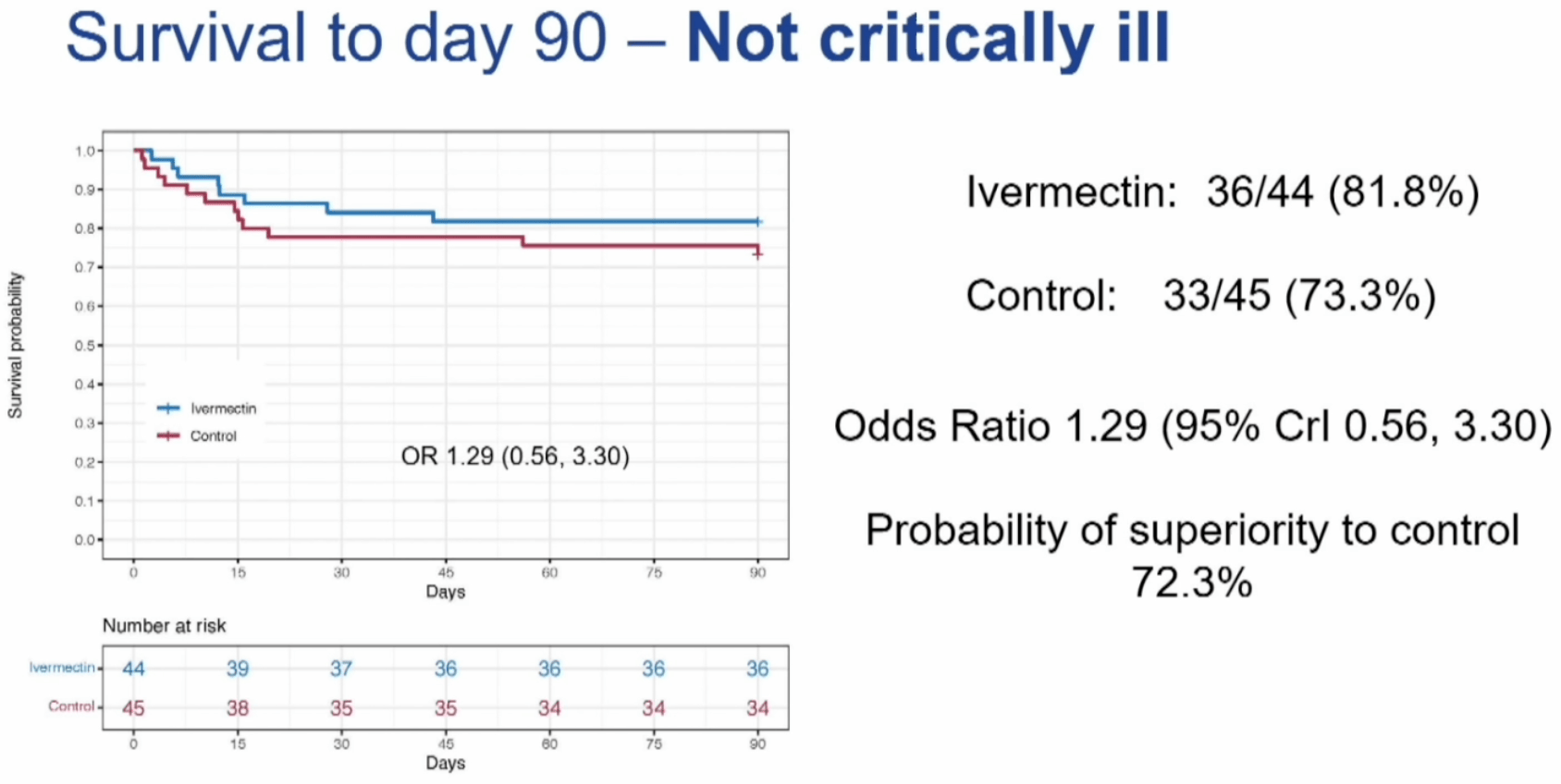
CRITICAL32% lower mortality for non-critical patients.
While authors describe the result as negative, there was 32% lower mortality for non-critical patients, despite greater baseline severity in the treatment group, very low dose, poor administration, and very late treatment. 18% died in the treatment group versus 27% for the control group.
CRITICALExpected failure based on design.
In the protocol authors note that previous trials used 0.2mg/kg - 0.6mg/kg and they note that a dose-response effect has been seen, however they chose the lowest dose 0.2mg/kg, and further do not specify administration with food5, suggesting that the expected outcome is failure. The presentation acknowledges that the dose is too low and not expected to be effective. They also note poor absorption in severe disease and presence of ileus, exarcerbated in critical illness, and they note studies show improved absorption with ivermectin oral solution. However, administration was done with tablets dissolved in water, which is noted to be a poor choice.
CRITICALDesign for ICU patients - inappropriate outcome.
The organ support-free days (OSFD) primary outcome was designed for ICU patients. The outcome assumes patients start at risk of needing organ support and the question is whether they receive it and how long. For a critically ill ICU patient already on organ support, this makes immediate sense - every day off support is a day of recovery. For a non-ICU ward patient on supplemental oxygen, the outcome is more degenerate: most of these patients will never need organ support regardless of treatment, so the OSFD scale collapses toward the maximum (22) for most patients. Looking at Table 2, the median OSFD for noncritically ill patients is 22 in both arms. Figure 3B confirms 68-70% of noncritically ill patients were at the ceiling. The posterior probability of superiority on this outcome is computed from a sample that is ceiling-dominated.
CRITICALRemdesivir use.
Remdesivir use was higher in the treatment group which adds risk given the ICU population and 90 day outcomes. Meta regression with followup duration for remdesivir shows declining efficacy with harm past ~60 days followup, which may reflect antiviral efficacy being offset by side effects6. Authors do not adjust for remdesivir use. The journal paper confirms this - Tables S8 and S9 show better results for patients not treated with remdesivir.
CRITICALBias in closing and presentation.
The trial was closed with an incorrect statement on the evidence base at the time, suggesting significant bias7.
CRITICALInappropriate termination.
The trial was terminated early when the concurrent ivermectin-specific analysis showed 72.6% and 72.3% probability of superiority for the critical and non-critical groups respectively, percentages that underestimate superiority at the time due to failure to adjust for baseline severity.
CRITICALPost hoc SAP one year post completion.
As above, authors created a post-hoc statistical analysis plan. The first one is dated 19 February 20233, long after the trial completed, and the second one is dated 16 October 2023, over a year after completion on September 9, 20224.
CRITICALResults withheld for years.
Results were withheld for 553 days (end of followup to CCR24 presentation), with the more accurate concurrent ivermectin-specific analysis being withheld for 1,247 days (end of followup until journal publication). After completion there were two post-hoc statistical analysis plans that made changes in outcomes and analysis, raising concern that delay was required to make changes in the outcomes and analysis that favor a preferred outcome.
CRITICALInconsistent blinding claims.
The second post-hoc SAP states that "The authors of this document are blinded to all individual data other than publicly disclosed results", however the authors include Scott Berry, Lindsay Berry, and Elizabeth Lorenzi of Berry Consultants, who were members of the Statistical Analysis Committee (SAC). The SAC is described in the first post-hoc SAP as "unblinded to all interventions and domains as part of their role for REMAP-CAP" in order to run the analyses that updated response-adaptive randomization weights and evaluated statistical triggers. The reference to the SAC and its unblinded role was removed from the second SAP.
CRITICALClaim of no analysis prior to second SAP impossible.
The second post-hoc SAP claims "The SAP was subsequently revised (Version 2, 29 September 2023) before any analysis was conducted...", however REMAP-CAP is an adaptive platform trial that requires analysis to operate. For example, allocation ratios "were updated in May 2022 following an adaptive analysis to weight assignment to the intervention that appeared most favorable at that time" which requires outcome analysis, and the termination decision requires analysis. The platform-wide adaptive analyses run continuously throughout the trial by Berry Consultants update randomization weights based on ongoing outcome data. The first post-hoc SAP explicitly described this: "there will be analyses conducted by the Statistical Analysis Committee (SAC) using additional randomizations and unblinding of other randomizations. The SAC is unblinded to all interventions and domains."
CRITICALPopulation change.
The second post-hoc SAP changes the analysis population from global to IIP-only (India, Ireland and Pakistan), reducing the model's ability to handle baseline imbalance. In the first post-hoc SAP, covariate effects (age, sex, time, site) would have been estimated from a much larger pool - hundreds to thousands of patients across all REMAP-CAP countries - giving more stable estimates of how those factors affect outcomes. In the second post-hoc SAP, covariate effects come from the 538 IIP patients (333 critical + 205 noncritical), a much smllaer amount of data to learn the covariate structure.
CRITICALAllocation inconsistency.
According to Table S2, the adaptive randomization proportion for ivermectin began at 1.0:1.0 and was subsequently updated to 1.0:2.0 in May 2022, however the results report >60% allocated to ivermectin which is inconsistent. In an open-label trial, such deviations can indicate a breakdown in allocation concealment, where investigators might bypass the sequence or selectively enroll patients when they know ivermectin will be assigned.
CRITICALModel penalizes noncritical efficacy.
Authors specify using a hierarchical prior that shrinks the estimates for critically and noncritically ill patients toward an overall intervention effect. However, because antivirals are mechanistically expected to be more effective early in the disease course and may be ineffective when used during very late-stage critical illness, mathematically pooling these biologically distinct populations will attenuate a genuine early treatment benefit. This unfair penalization of the noncritical efficacy signal is compounded by the very small sample size and severe baseline imbalances in the critical cohort, which forces the model to rely heavily on a blended average, and is likely to obscure true phase-dependent treatment effects.
CRITICALModel accuracy.
The accuracy of the primary analysis is fundamentally limited by the very small sample size, particularly when evaluating the Organ Support-Free Days (OSFD) outcome. Because OSFD is a 24-level ordinal scale, the cumulative logistic model must calculate distinct baseline probability thresholds for nearly every day of recovery or death. With only 61 critically ill patients distributed across these 24 possible outcomes, the data is far too sparse to support such a highly parameterized model, resulting in significant mathematical uncertainty. To prevent the model from failing entirely, it is forced to artificially collapse empty outcome categories and rely heavily on rigid statistical assumptions such as proportional odds. The final efficacy estimates may be driven more by mathematical compromises than by the observed patient outcomes.
CRITICALBaseline imbalance not mentioned.
In addition to not adjusting for the severe baseline imbalance, authors do not even mention this imbalance, suggesting significant bias.
SERIOUS>20% missing treatment data.
According to Table S5, in the non-critically ill group, the denominators for key co-interventions show only 32/44 and 35/45 patients with data. >20% missing basic treatment data is unusual and unexplained.
SERIOUSInconsistent co-intervention counts.
In the critically ill group, Table S5 shows 29/37 patients received remdesivir, while Table S8 shows 21/37 received remdesivir at baseline. Table S5 may include use after baseline, however Table 1 shows 29/37 patients received remdesivir at baseline.
SERIOUSBMI missing data.
Baseline BMI data is strangely separated from other baseline data in Table S4, and shows very high missing data rates.
SERIOUSMisleading conclusion.
The main conclusion does not reflect the 72.3% probability of superiority for survival in the non-critically ill group, or the 72.6% and 72.3% probability of superiority for the critical and non-critical groups with the more accurate concurrent ivermectin-specific analysis, or the severe baseline imbalance which would result in even higher probability of superiority if adjusted for.
SERIOUSPrimary outcome changed.
The primary outcome in the protocol is 90 day mortality1. It's unclear why this was changed in the antiviral domain8 to organ support-free days. Authors do not report 90 day mortality at all, only 90 day survival which shows a lower relative change for the observed trend of lower mortality in non-critical patients. For noncritical patients that mostly survive, reporting mortality instead of survival is preferred.
This is the 51st of 54 COVID-19 RCTs for ivermectin, which collectively show efficacy with p=0.00000004.
This is the 103rd of 106 COVID-19 controlled studies for ivermectin, which collectively show efficacy with p<0.0000000001.
This study is excluded in the after exclusion results of meta-analysis:
baseline severity favors control, post-hoc outcome and SAP changes, see discussion.
|
risk of death, 25.6% lower, RR 0.74, p = 0.40, treatment 81, control 69, adjusted per study, combined.
|
|
risk of death, 26.5% lower, OR 0.74, p = 0.57, treatment 44, control 45, adjusted per study, inverted to make OR<1 favor treatment, concurrent, non-critical, day 90, Table S7, RR approximated with OR.
|
|
risk of death, 24.8% lower, OR 0.75, p = 0.55, treatment 37, control 24, adjusted per study, inverted to make OR<1 favor treatment, concurrent, critical, day 90, Table S7, RR approximated with OR.
|
|
risk of death, 14.9% lower, RR 0.85, p = 0.64, treatment 81, control 69, adjusted per study, combined.
|
|
risk of death, 22.5% lower, OR 0.78, p = 0.59, treatment 44, control 45, adjusted per study, inverted to make OR<1 favor treatment, non-critical, day 90, RR approximated with OR.
|
|
risk of death, 5.7% lower, OR 0.94, p = 0.91, treatment 37, control 24, adjusted per study, inverted to make OR<1 favor treatment, critical, day 90, RR approximated with OR.
|
|
intubation, ECMO, death, 23.7% lower, OR 0.76, p = 0.57, treatment 44, control 45, adjusted per study, inverted to make OR<1 favor treatment, non-critical, RR approximated with OR.
|
|
intubation, ECMO, death, 16.0% lower, OR 0.84, p = 0.72, treatment 37, control 24, adjusted per study, inverted to make OR<1 favor treatment, critical, RR approximated with OR.
|
|
organ support-free days, 3.8% lower, OR 0.96, p = 0.93, treatment 44, control 45, inverted to make OR<1 favor treatment, non-critical, RR approximated with OR.
|
|
organ support-free days, 6.4% higher, OR 1.06, p = 0.89, treatment 37, control 24, inverted to make OR<1 favor treatment, critical, RR approximated with OR.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
1.
static1.squarespace.com, static1.squarespace.com/static/5cde3c7d9a69340001d79ffe/t/5e82b62eda9c1c714065efc0/1585624649154/REMAP-CAP+Core+Protocol+V3+-+10+July+2019_WM.pdf.
2.
Siddiqui et al., Desperate Times and Good Science: Lessons From Randomized, Embedded, Multifactorial Adaptive Platform Trial for Community-Acquired Pneumonia (REMAP-CAP), Critical Care Medicine, doi:10.1097/CCM.0000000000007160.
Hashmi et al., 13 Jun 2024, Randomized Controlled Trial, Pakistan, peer-reviewed, 83 authors, study period 11 June, 2021 - 9 September, 2022, dosage 200μg/kg days 1-5, trial NCT02735707 (history) (REMAP-CAP).
Abstract: ## CLINICAL INVESTIGATION
Ivermectin for Critically and Noncritically Ill Hospitalized Patients With COVID-19: Randomized, Embedded, Multifactorial Adaptive Platform Trial for CommunityAcquired Pneumonia (REMAP-CAP)
OBJECTIVE: To determine whether ivermectin improves outcomes for critically and noncritically ill hospitalized patients with COVID-19.
DESIGN: An ongoing international, multifactorial, adaptive platform, randomized, controlled trial.
SETTING: Hospitals in Pakistan, India, and Ireland between June 11, 2021, and September 9, 2022.
PATIENTS: Critically and noncritically ill patients.
INTERVENTIONS: Randomized to ivermectin or no ivermectin (control).
MEASUREMENTS AND MAIN RESULTS: The primary outcome was respiratory and cardiovascular organ support-free days, assessed on an ordinal scale combining in-hospital death (assigned a value of -1) and days free of organ support through day 21 in survivors. Analyses used a Bayesian cumulative logistic model. Enrollment was closed for operational futility, following external evidence suggesting no benefit with ivermectin in nonhospitalized patients with COVID-19. Among 61 critically ill patients, the median number of organ support-free days was -1, indicating death was the most common vital outcome (interquartile range [IQR], -1 to 17), for the ivermectin group and -1 (IQR, -1 to 17 .25) for the control group (adjusted proportional odds ratio [OR], 0.94; 95% credible interval [CrI], 0.40-2.07) and the posterior probability of superiority to control was 44.2%. Among 89 noncritically ill patients, the median number of organ support-free days was 22 (IQR, 18.5-22) for ivermectin and 22 (IQR, 16-22) for control (adjusted proportional OR, 1.04; 95% CrI, 0.48-2.34) and the posterior probability of superiority was 53.7%. Among critically ill patients, hospital survival was 35.1% (13/37) for ivermectin and 37 .5% (9/24) for control (adjusted OR, 1.00; 95% CrI, 0.39-2.32), posterior probability of superiority was 50.0%. Among noncritically ill patients, hospital survival was 84.1% (37/44) for ivermectin and 77 .8% (35/45) for control (adjusted OR, 1.16; 95% CrI, 0.5-3.07), posterior probability of superiority was 63.3%.
CONCLUSIONS: For critically and noncritically ill hospitalized patients with COVID-19, ivermectin was unlikely to improve the primary composite outcome of organ support-free days and hospital survival.
KEYWORDS: adaptive platform trial; antiviral; COVID-19; intensive care; ivermectin; pneumonia
Madiha Hashmi, MD 1
Rashan Haniffa, PhD 2,3
Deva Jayakumar, MS, MSc 4
Abigail Beane, PhD 2,3
Elizabeth Lorenzi, PhD 5
Lindsay R. Berry, PhD 5
Muhammad Nasir Khoso, MBBS 6
Quratul Ain Khan, MD 7
Ashok Kumar, FCPS 8
Aneela Altaf Kidwai, FCPS 9
Thomas E. Hills, MBBS, PhD 10
Djillali Annane, MD, PhD 11
Diptesh Aryal, MD, PhD 12
Carly Au, BSc 13
Kenneth Baillie, MBChB, PhD 14
Richard Beasley, MBChB, MD 10
Janis Best-Lane, BSc 15
Marc Bonten, MD, PhD 16,17
Charlotte A. Bradbury, MD, PhD 18
Copyright © 2026 The Author(s). Published by Wolters Kluwer Health, Inc. on behalf of the Society of Critical Care Medicine and Wolters Kluwer Health, Inc. This is an open-access article distributed under the terms of the Creative Commons AttributionNon Commercial-No Derivatives License 4.0 (CCBY-NC-ND), where it is permissible to download and share the work provided it is properly cited. The work cannot be changed in any way ..
DOI record:
{
"DOI": "10.1097/ccm.0000000000007134",
"ISSN": [
"0090-3493",
"1530-0293"
],
"URL": "http://dx.doi.org/10.1097/CCM.0000000000007134",
"abstract": "<jats:sec>\n <jats:title>Objective:</jats:title>\n <jats:p>To determine whether ivermectin improves outcomes for critically and noncritically ill hospitalized patients with COVID-19.</jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>Design:</jats:title>\n <jats:p>An ongoing international, multifactorial, adaptive platform, randomized, controlled trial.</jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>Setting:</jats:title>\n <jats:p>Hospitals in Pakistan, India, and Ireland between June 11, 2021, and September 9, 2022.</jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>Patients:</jats:title>\n <jats:p>Critically and noncritically ill patients.</jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>Interventions:</jats:title>\n <jats:p>Randomized to ivermectin or no ivermectin (control).</jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>Measurements and Main Results:</jats:title>\n <jats:p>The primary outcome was respiratory and cardiovascular organ support-free days, assessed on an ordinal scale combining in-hospital death (assigned a value of –1) and days free of organ support through day 21 in survivors. Analyses used a Bayesian cumulative logistic model. Enrollment was closed for operational futility, following external evidence suggesting no benefit with ivermectin in nonhospitalized patients with COVID-19. Among 61 critically ill patients, the median number of organ support-free days was –1, indicating death was the most common vital outcome (interquartile range [IQR], –1 to 17), for the ivermectin group and –1 (IQR, –1 to 17.25) for the control group (adjusted proportional odds ratio [OR], 0.94; 95% credible interval [CrI], 0.40–2.07) and the posterior probability of superiority to control was 44.2%. Among 89 noncritically ill patients, the median number of organ support-free days was 22 (IQR, 18.5–22) for ivermectin and 22 (IQR, 16–22) for control (adjusted proportional OR, 1.04; 95% CrI, 0.48–2.34) and the posterior probability of superiority was 53.7%. Among critically ill patients, hospital survival was 35.1% (13/37) for ivermectin and 37.5% (9/24) for control (adjusted OR, 1.00; 95% CrI, 0.39–2.32), posterior probability of superiority was 50.0%. Among noncritically ill patients, hospital survival was 84.1% (37/44) for ivermectin and 77.8% (35/45) for control (adjusted OR, 1.16; 95% CrI, 0.5–3.07), posterior probability of superiority was 63.3%.</jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>Conclusions:</jats:title>\n <jats:p>For critically and noncritically ill hospitalized patients with COVID-19, ivermectin was unlikely to improve the primary composite outcome of organ support-free days and hospital survival.</jats:p>\n </jats:sec>",
"author": [
{
"affiliation": [
{
"name": "Department of Critical Care Medicine, Ziauddin University—Karachi, Sindh, Pakistan."
}
],
"family": "Hashmi",
"given": "Madiha",
"sequence": "first"
},
{
"affiliation": [
{
"name": "Mahidol Oxford Tropical Medicine Research Unit, Bangkok, Thailand."
},
{
"name": "Nuffield Department of Clinical Medicine, University of Oxford, Oxford, United Kingdom."
}
],
"family": "Haniffa",
"given": "Rashan",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Critical Care, Dr Kamakshi Memorial Hospital, Chennai, Tamil Nadu, India."
}
],
"family": "Jayakumar",
"given": "Deva",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Mahidol Oxford Tropical Medicine Research Unit, Bangkok, Thailand."
},
{
"name": "Nuffield Department of Clinical Medicine, University of Oxford, Oxford, United Kingdom."
}
],
"family": "Beane",
"given": "Abigail",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Berry Consultants, Austin, TX."
}
],
"family": "Lorenzi",
"given": "Elizabeth",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Berry Consultants, Austin, TX."
}
],
"family": "Berry",
"given": "Lindsay R.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "South City Hospital, Karachi, Pakistan."
}
],
"family": "Nasir Khoso",
"given": "Muhammad",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Critical Care, North Nazimabad Campus, Dr Ziauddin Group of Hospitals, Karachi, Pakistan."
}
],
"family": "Ain Khan",
"given": "Quratul",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Chest Medicine and Critical Care, Ziauddin University, Karachi, Sindh, Pakistan."
}
],
"family": "Kumar",
"given": "Ashok",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Medicine, Abbasi Shaheed Hospital, Nazimabad, Karachi, Pakistan."
}
],
"family": "Altaf Kidwai",
"given": "Aneela",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Medical Research Institute, Wellington, New Zealand."
}
],
"family": "Hills",
"given": "Thomas E.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Service de Réanimation médicale et unité de ventilation à domicile, centre de référence neuromusculaire GNHM, CHU Raymond Poincaré, APHP, Université de Versailles Saint Quentin en Yvelines, Garches, France."
}
],
"family": "Annane",
"given": "Djillali",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Nepal Intensive Care Research Foundation, Kathmandu, Nepal."
}
],
"family": "Aryal",
"given": "Diptesh",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Clinical Trials Unit, Intensive Care National Audit & Research Centre, London, United Kingdom."
}
],
"family": "Au",
"given": "Carly",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Centre for Inflammation Research, Deanery of Clinical Sciences, University of Edinburgh, Midlothian, United Kingdom."
}
],
"family": "Baillie",
"given": "Kenneth",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Medical Research Institute, Wellington, New Zealand."
}
],
"family": "Beasley",
"given": "Richard",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Division of Anaesthetics, Pain Medicine, and Intensive Care, Imperial College London, London, United Kingdom."
}
],
"family": "Best-Lane",
"given": "Janis",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Medical Microbiology, University Medical Center, Utrecht, The Netherlands."
},
{
"name": "Julius Center for Health Sciences and Primary Care, University Medical Center, Utrecht, The Netherlands."
}
],
"family": "Bonten",
"given": "Marc",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "University of Bristol, Bristol, United Kingdom."
}
],
"family": "Bradbury",
"given": "Charlotte A.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Jena University Hospital, Jena, Germany."
}
],
"family": "Brunkhorst",
"given": "Frank M.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Australian and New Zealand Intensive Care Research Centre, School of Public Health and Preventive Medicine, Monash University, Melbourne, VIC, Australia."
},
{
"name": "Department of Intensive Care and Hyperbaric Medicine, The Alfred, Melbourne, VIC, Australia."
}
],
"family": "Burrell",
"given": "Aidan",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Global Coalition for Adaptive Research, Larkspur, CA."
}
],
"family": "Buxton",
"given": "Meredith",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Anesthesiology and Intensive Care, IRCCS Humanitas Research Hospital, Milan, Italy."
}
],
"family": "Cecconi",
"given": "Maurizio",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Epidemiology and Preventive Medicine, Monash University, Melbourne, VIC, Australia."
},
{
"name": "Infectious Diseases Unit, Alfred Hospital, Melbourne, VIC, Australia."
}
],
"family": "Cheng",
"given": "Allen C.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "National University of Singapore, Singapore."
}
],
"family": "Cove",
"given": "Matthew E.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Medical Microbiology, University Medical Center, Utrecht, The Netherlands."
}
],
"family": "de Jong",
"given": "Menno",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Berry Consultants, Austin, TX."
}
],
"family": "Detry",
"given": "Michelle A.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Auckland City Hospital, Auckland, New Zealand."
}
],
"family": "Duffy",
"given": "Eamon",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "University of Oxford, Oxford, United Kingdom."
}
],
"family": "Estcourt",
"given": "Lise J.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Berry Consultants, Austin, TX."
}
],
"family": "Fitzgerald",
"given": "Mark",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Sunnybrook Research Institute, Sunnybrook Health Sciences Centre, Toronto, ON, Canada."
},
{
"name": "Department of Medicine, University of Toronto, Toronto, ON, Canada."
}
],
"family": "Fowler",
"given": "Rob",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Medical Microbiology, Vaccine & Infectious Diseases Institute (VAXINFECTIO), University of Antwerp, Antwerp, Belgium."
}
],
"family": "Goossens",
"given": "Herman",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Monash University, Melbourne, VIC, Australia."
}
],
"family": "Green",
"given": "Cameron",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "University College Dublin Clinical Research Centre, St Vincent’s University Hospital, Dublin, Ireland."
}
],
"family": "Hays",
"given": "Leanne M. C.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Australian and New Zealand Intensive Care Research Centre, School of Public Health and Preventive Medicine, Monash University, Melbourne, VIC, Australia."
}
],
"family": "Higgins",
"given": "Alisa M.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "University of Pittsburgh, Pittsburgh, PA."
}
],
"family": "Huang",
"given": "David T.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "The University of Tokyo, Bunkyo-ku, Tokyo, Japan."
}
],
"family": "Ichihara",
"given": "Nao",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Pulmonary, Critical Care and Sleep Medicine, Hospital for Advanced Medicine and Surgery, Kathmandu, Nepal."
}
],
"family": "Koirala",
"given": "Sabin",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Universite de Sherbrooke, Sherbrooke, QC, Canada."
}
],
"family": "Lamontagne",
"given": "François",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "McGill University Faculty of Medicine, Montreal, QC, Canada."
}
],
"family": "Lawler",
"given": "Patrick R.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Berry Consultants, Austin, TX."
}
],
"family": "Lewis",
"given": "Roger J.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "The University of Western Australia, Perth, WA, Australia."
}
],
"family": "Litton",
"given": "Edward",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "University College Dublin Clinical Research Centre, St Vincent’s University Hospital, Dublin, Ireland."
}
],
"family": "Mahon",
"given": "Niamh",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "University of Toronto, Toronto, Ontario, Canada."
}
],
"family": "Marshall",
"given": "John C.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "ICU, QUB, Belfast, United Kingdom."
},
{
"name": "Centre for Experimental Medicine, Queen’s University Belfast, Belfast, United Kingdom."
}
],
"family": "McAuley",
"given": "Daniel F.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Berry Consultants, Austin, TX."
}
],
"family": "McGlothlin",
"given": "Anna",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Auckland City Hospital, Auckland, New Zealand."
}
],
"family": "McGuinness",
"given": "Shay",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Monash University, Melbourne, VIC, Australia."
}
],
"family": "McQuilten",
"given": "Zoe K.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Pulmonary Allergy and Critical Care Medicine, University of Pittsburgh Medical Center Health System, Pittsburgh, PA."
}
],
"family": "McVerry",
"given": "Bryan J.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Clinical Trials Unit, Intensive Care National Audit & Research Centre, London, United Kingdom."
}
],
"family": "Mouncey",
"given": "Paul R.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Middlemore Hospital, Auckland, New Zealand."
}
],
"family": "Morpeth",
"given": "Susan",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Radboud Universitair Medisch Centrum, Nijmegen, The Netherlands."
}
],
"family": "Netea",
"given": "Mihai",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Fiona Stanley Hospital, Murdoch, WA, Australia."
}
],
"family": "Orr",
"given": "Katrina",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "The University of Auckland, Auckland, New Zealand."
}
],
"family": "Parke",
"given": "Rachael L.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Monash University, Melbourne, VIC, Australia."
}
],
"family": "Parker",
"given": "Jane C.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Auckland City Hospital, Auckland, New Zealand."
},
{
"name": "Royal Prince Alfred Hospital, Camperdown, NSW, Australia."
}
],
"family": "Patanwala",
"given": "Asad",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "European Clinical Research Alliance on Infectious Diseases (Ecraid), Utrecht, The Netherlands."
}
],
"family": "Peters",
"given": "Svenja",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Universidad de La Sabana, Chia, Colombia."
}
],
"family": "Reyes",
"given": "Luis Felipe",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Clinical Trials Unit, Intensive Care National Audit & Research Centre, London, United Kingdom."
}
],
"family": "Rowan",
"given": "Kathryn M.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "St Marianna University School of Medicine, Kawasaki, Kanagawa, Japan."
}
],
"family": "Saito",
"given": "Hiroki",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Berry Consultants, Austin, TX."
}
],
"family": "Saunders",
"given": "Christina T.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Unity Health Toronto, Toronto, ON, Canada."
}
],
"family": "Santos",
"given": "Marlene",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "University of Pittsburgh, Pittsburgh, PA."
}
],
"family": "Seymour",
"given": "Christopher W.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Centre for Inflammation Research, The University of Edinburgh College of Medicine and Veterinary Medicine, Edinburgh, United Kingdom."
}
],
"family": "Shankar-Hari",
"given": "Manu",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Australian and New Zealand Intensive Care Research Centre, School of Public Health and Preventive Medicine, Monash University, Melbourne, VIC, Australia."
}
],
"family": "Singh",
"given": "Vanessa",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Faculty of Medicine, Dentistry and Health Sciences, University of Melbourne, Melbourne, VIC, Australia."
}
],
"family": "Slater",
"given": "Matthew",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Medicine, Yong Loo Lin School of Medicine, National University of Singapore, Singapore."
}
],
"family": "Tambyah",
"given": "Paul A.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "The Peter Doherty Institute for Infection and Immunity, Melbourne, VIC, Australia."
}
],
"family": "Tong",
"given": "Steven Y. C.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Universite Laval, Quebec, QC, Canada."
}
],
"family": "Turgeon",
"given": "Alexis F.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Medical Research Institute, Wellington, New Zealand."
}
],
"family": "Turner",
"given": "Anne M.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Internal Medicine, Radboud University Medical Center, Nijmegen, The Netherlands."
}
],
"family": "van de Veerdonk",
"given": "Frank",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Institute for Infectious Disease and Infection Control, Jena University Hospital, Friedrich-Schiller-University, Jena, Germany."
},
{
"name": "Department of Anesthesiology and Intensive Care Medicine, Jena University Hospital, Friedrich-Schiller-University, Jena, Germany."
},
{
"name": "Leibniz Institute for Natural Product Research and Infection Biology, Hans Knöll Institute (HKI), Jena, Germany."
}
],
"family": "Weis",
"given": "Sebastian",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "University of Manitoba, Winnipeg, MB, Canada."
}
],
"family": "Zarychanski",
"given": "Ryan",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Auckland City Hospital, Auckland, New Zealand."
}
],
"family": "McArthur",
"given": "Colin J.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "University of Pittsburgh, Pittsburgh, PA."
}
],
"family": "Angus",
"given": "Derek C.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Berry Consultants, Austin, TX."
}
],
"family": "Berry",
"given": "Scott M.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Imperial College London, London, United Kingdom."
}
],
"family": "Gordon",
"given": "Anthony C.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Intensive Care Center, University Medical Centre, Utrecht, The Netherlands."
}
],
"family": "Derde",
"given": "Lennie P. G.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Monash University, Clayton, VIC, Australia."
}
],
"family": "Webb",
"given": "Steve A.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "The University of British Columbia, Vancouver, BC, Canada."
}
],
"family": "Murthy",
"given": "Srinivas",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Critical Care, King Saud Bin Abdulaziz University for Health Sciences, Riyadh, Saudi Arabia."
}
],
"family": "Arabi",
"given": "Yaseen",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Australian and New Zealand Intensive Care Research Centre, School of Public Health and Preventive Medicine, Monash University, Melbourne, VIC, Australia."
},
{
"name": "University College Dublin Clinical Research Centre, St Vincent’s University Hospital, Dublin, Ireland."
}
],
"family": "Nichol",
"given": "Alistair D.",
"sequence": "additional"
},
{
"affiliation": [],
"name": "and the Randomized, Embedded, Multifactorial Adaptive Platform Trial for Community-Acquired Pneumonia (REMAP-CAP) Investigators",
"sequence": "additional"
}
],
"container-title": "Critical Care Medicine",
"content-domain": {
"crossmark-restriction": true,
"domain": [
"lww.com",
"ovid.com"
]
},
"created": {
"date-parts": [
[
2026,
5,
8
]
],
"date-time": "2026-05-08T14:03:20Z",
"timestamp": 1778249000000
},
"deposited": {
"date-parts": [
[
2026,
5,
8
]
],
"date-time": "2026-05-08T14:03:23Z",
"timestamp": 1778249003000
},
"indexed": {
"date-parts": [
[
2026,
5,
8
]
],
"date-time": "2026-05-08T14:18:17Z",
"timestamp": 1778249897265,
"version": "3.51.4"
},
"is-referenced-by-count": 0,
"issued": {
"date-parts": [
[
2026,
5,
8
]
]
},
"language": "en",
"license": [
{
"URL": "http://creativecommons.org/licenses/by-nc-nd/4.0/",
"content-version": "unspecified",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2026,
5,
8
]
],
"date-time": "2026-05-08T00:00:00Z",
"timestamp": 1778198400000
}
}
],
"link": [
{
"URL": "https://journals.lww.com/10.1097/CCM.0000000000007134",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "276",
"original-title": [],
"prefix": "10.1097",
"published": {
"date-parts": [
[
2026,
5,
8
]
]
},
"published-online": {
"date-parts": [
[
2026,
5,
8
]
]
},
"publisher": "Ovid Technologies (Wolters Kluwer Health)",
"reference": [
{
"DOI": "10.1016/S0140-6736(22)00372-5",
"article-title": "The global COVID-19 treatment divide.",
"author": "Usher",
"doi-asserted-by": "crossref",
"first-page": "779",
"journal-title": "Lancet",
"key": "R2-20260508",
"volume": "399",
"year": "2022"
},
{
"DOI": "10.1007/s00134-021-06448-5",
"article-title": "Lopinavir-ritonavir and hydroxychloroquine for critically ill patients with COVID-19: REMAP-CAP randomized controlled trial.",
"author": "Arabi",
"doi-asserted-by": "crossref",
"first-page": "867",
"journal-title": "Intensive Care Med",
"key": "R3-20260508",
"volume": "47",
"year": "2021"
},
{
"DOI": "10.1007/s00011-008-8007-8",
"article-title": "Ivermectin inhibits LPS-induced production of inflammatory cytokines and improves LPS-induced survival in mice.",
"author": "Zhang",
"doi-asserted-by": "crossref",
"first-page": "524",
"journal-title": "Inflamm Res",
"key": "R4-20260508",
"volume": "57",
"year": "2008"
},
{
"DOI": "10.1016/j.intimp.2008.12.016",
"article-title": "Inhibitory effects of ivermectin on nitric oxide and prostaglandin E2 production in LPS-stimulated RAW 264.7 macrophages.",
"author": "Zhang",
"doi-asserted-by": "crossref",
"first-page": "354",
"journal-title": "Int Immunopharmacol",
"key": "R5-20260508",
"volume": "9",
"year": "2009"
},
{
"article-title": "Over 25 years of clinical experience with ivermectin: An overview of safety for an increasing number of indications.",
"author": "Kircik",
"first-page": "325",
"journal-title": "J Drugs Dermatol",
"key": "R6-20260508",
"volume": "15",
"year": "2016"
},
{
"DOI": "10.1016/j.antiviral.2020.104787",
"article-title": "The FDA-approved drug ivermectin inhibits the replication of SARS-CoV-2 in vitro.",
"author": "Caly",
"doi-asserted-by": "crossref",
"first-page": "104787",
"journal-title": "Antiviral Res",
"key": "R7-20260508",
"volume": "178",
"year": "2020"
},
{
"DOI": "10.3389/fimmu.2021.663586",
"article-title": "Repurposing ivermectin for COVID-19: Molecular aspects and therapeutic possibilities.",
"author": "Wehbe",
"doi-asserted-by": "crossref",
"first-page": "663586",
"journal-title": "Front Immunol",
"key": "R8-20260508",
"volume": "12",
"year": "2021"
},
{
"DOI": "10.1038/s41598-021-86679-0",
"article-title": "Ivermectin reduces in vivo coronavirus infection in a mouse experimental model.",
"author": "Arévalo",
"doi-asserted-by": "crossref",
"first-page": "7132",
"journal-title": "Sci Rep",
"key": "R9-20260508",
"volume": "11",
"year": "2021"
},
{
"DOI": "10.15252/emmm.202114122",
"article-title": "Attenuation of clinical and immunological outcomes during SARS-CoV-2 infection by ivermectin.",
"author": "de Melo",
"doi-asserted-by": "crossref",
"first-page": "e14122",
"journal-title": "EMBO Mol Med",
"key": "R10-20260508",
"volume": "13",
"year": "2021"
},
{
"DOI": "10.1002/cpt.1889",
"article-title": "The approved dose of ivermectin alone is not the ideal dose for the treatment of COVID-19.",
"author": "Schmith",
"doi-asserted-by": "crossref",
"first-page": "762",
"journal-title": "Clin Pharmacol Ther",
"key": "R11-20260508",
"volume": "108",
"year": "2020"
},
{
"DOI": "10.1056/NEJMoa2201662",
"article-title": "Randomized trial of metformin, ivermectin, and fluvoxamine for Covid-19.",
"author": "Bramante",
"doi-asserted-by": "crossref",
"first-page": "599",
"journal-title": "N Engl J Med",
"key": "R12-20260508",
"volume": "387",
"year": "2022"
},
{
"DOI": "10.1001/jamainternmed.2022.0189",
"article-title": "Efficacy of ivermectin treatment on disease progression among adults with mild to moderate COVID-19 and comorbidities: The I-TECH randomized clinical trial.",
"author": "Lim",
"doi-asserted-by": "crossref",
"first-page": "426",
"journal-title": "JAMA Intern Med",
"key": "R13-20260508",
"volume": "182",
"year": "2022"
},
{
"DOI": "10.1001/jama.2021.3071",
"article-title": "Effect of ivermectin on time to resolution of symptoms among adults with mild COVID-19: A randomized clinical trial.",
"author": "López-Medina",
"doi-asserted-by": "crossref",
"first-page": "1426",
"journal-title": "JAMA",
"key": "R14-20260508",
"volume": "325",
"year": "2021"
},
{
"DOI": "10.1001/jama.2022.18590",
"article-title": "Effect of ivermectin vs placebo on time to sustained recovery in outpatients with mild to moderate COVID-19: A randomized clinical trial.",
"author": "Naggie",
"doi-asserted-by": "crossref",
"first-page": "1595",
"journal-title": "JAMA",
"key": "R15-20260508",
"volume": "328",
"year": "2022"
},
{
"DOI": "10.1001/jama.2023.1650",
"article-title": "Effect of higher-dose ivermectin for 6 days vs placebo on time to sustained recovery in outpatients with COVID-19: A randomized clinical trial.",
"author": "Naggie",
"doi-asserted-by": "crossref",
"first-page": "888",
"journal-title": "JAMA",
"key": "R16-20260508",
"volume": "329",
"year": "2023"
},
{
"DOI": "10.1056/NEJMoa2115869",
"article-title": "Effect of early treatment with ivermectin among patients with Covid-19.",
"author": "Reis",
"doi-asserted-by": "crossref",
"first-page": "1721",
"journal-title": "N Engl J Med",
"key": "R17-20260508",
"volume": "386",
"year": "2022"
},
{
"DOI": "10.1186/s12879-021-06348-5",
"article-title": "Ivermectin to prevent hospitalizations in patients with COVID-19 (IVERCOR-COVID19) a randomized, double-blind, placebo-controlled trial.",
"author": "Vallejos",
"doi-asserted-by": "crossref",
"first-page": "635",
"journal-title": "BMC Infect Dis",
"key": "R18-20260508",
"volume": "21",
"year": "2021"
},
{
"DOI": "10.1016/j.ijid.2020.11.191",
"article-title": "A five-day course of ivermectin for the treatment of COVID-19 may reduce the duration of illness.",
"author": "Ahmed",
"doi-asserted-by": "crossref",
"first-page": "214",
"journal-title": "Int J Infect Dis",
"key": "R19-20260508",
"volume": "103",
"year": "2021"
},
{
"DOI": "10.1016/j.jiac.2021.08.021",
"article-title": "Single-dose oral ivermectin in mild and moderate COVID-19 (RIVET-COV): A single-centre randomized, placebo-controlled trial.",
"author": "Mohan",
"doi-asserted-by": "crossref",
"first-page": "1743",
"journal-title": "J Infect Chemother",
"key": "R20-20260508",
"volume": "27",
"year": "2021"
},
{
"DOI": "10.1016/j.clinthera.2021.04.007",
"article-title": "Effects of ivermectin in patients with COVID-19: A multicenter, double-blind, randomized, controlled clinical trial.",
"author": "Shahbaznejad",
"doi-asserted-by": "crossref",
"first-page": "1007",
"journal-title": "Clin Ther",
"key": "R21-20260508",
"volume": "43",
"year": "2021"
},
{
"DOI": "10.1513/AnnalsATS.202003-192SD",
"article-title": "The REMAP-CAP (Randomized Embedded Multifactorial Adaptive Platform for Community-Acquired Pneumonia) study. Rationale and design.",
"author": "Angus",
"doi-asserted-by": "crossref",
"first-page": "879",
"journal-title": "Ann Am Thorac Soc",
"key": "R22-20260508",
"volume": "17",
"year": "2020"
},
{
"DOI": "10.1001/jama.2022.23257",
"article-title": "Long-term (180-day) outcomes in critically ill patients with COVID-19 in the REMAP-CAP randomized clinical trial.",
"author": "Higgins",
"doi-asserted-by": "crossref",
"first-page": "39",
"journal-title": "JAMA",
"key": "R23-20260508",
"volume": "329",
"year": "2023"
},
{
"DOI": "10.1016/S1473-3099(20)30483-7",
"article-title": "A minimal common outcome measure set for COVID-19 clinical research.",
"author": "Marshall",
"doi-asserted-by": "crossref",
"first-page": "e192",
"journal-title": "Lancet Infect Dis",
"key": "R24-20260508",
"volume": "20",
"year": "2020"
},
{
"DOI": "10.1056/EVIDoa2200293",
"article-title": "Anticoagulation strategies in non-critically ill patients with Covid-19.",
"author": "McQuilten",
"doi-asserted-by": "crossref",
"first-page": "EVIDoa2200293",
"journal-title": "NEJM Evid",
"key": "R25-20260508",
"volume": "2",
"year": "2023"
},
{
"DOI": "10.1136/bmj.m3939",
"article-title": "Convalescent plasma in the management of moderate Covid-19 in adults in India: Open label phase II multicentre randomised controlled trial (PLACID trial).",
"author": "Agarwal",
"doi-asserted-by": "crossref",
"first-page": "m3939",
"journal-title": "BMJ",
"key": "R26-20260508",
"volume": "371",
"year": "2020"
},
{
"article-title": "PRICE COVID19 Data Report December 2021 Pakistan Registry of Intensive Care.",
"author": "Farooq",
"journal-title": "medRxiv",
"key": "R27-20260508"
}
],
"reference-count": 26,
"references-count": 26,
"relation": {},
"resource": {
"primary": {
"URL": "https://journals.lww.com/10.1097/CCM.0000000000007134"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "Ivermectin for Critically and Noncritically Ill Hospitalized Patients With COVID-19: Randomized, Embedded, Multifactorial Adaptive Platform Trial for Community-Acquired Pneumonia (REMAP-CAP)",
"type": "journal-article",
"update-policy": "https://doi.org/10.1097/lww.0000000000001000"
}