Inhaled nitric oxide for the treatment of COVID-19: an open-label, parallel, randomised controlled trial
et al., ERJ Open Research, doi:10.1183/23120541.00006-2024, iNO-COVID-19, RBR-8nfx26, Feb 2025
43rd treatment shown to reduce risk in
June 2022, now with p = 0.013 from 14 studies, recognized in 10 countries.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
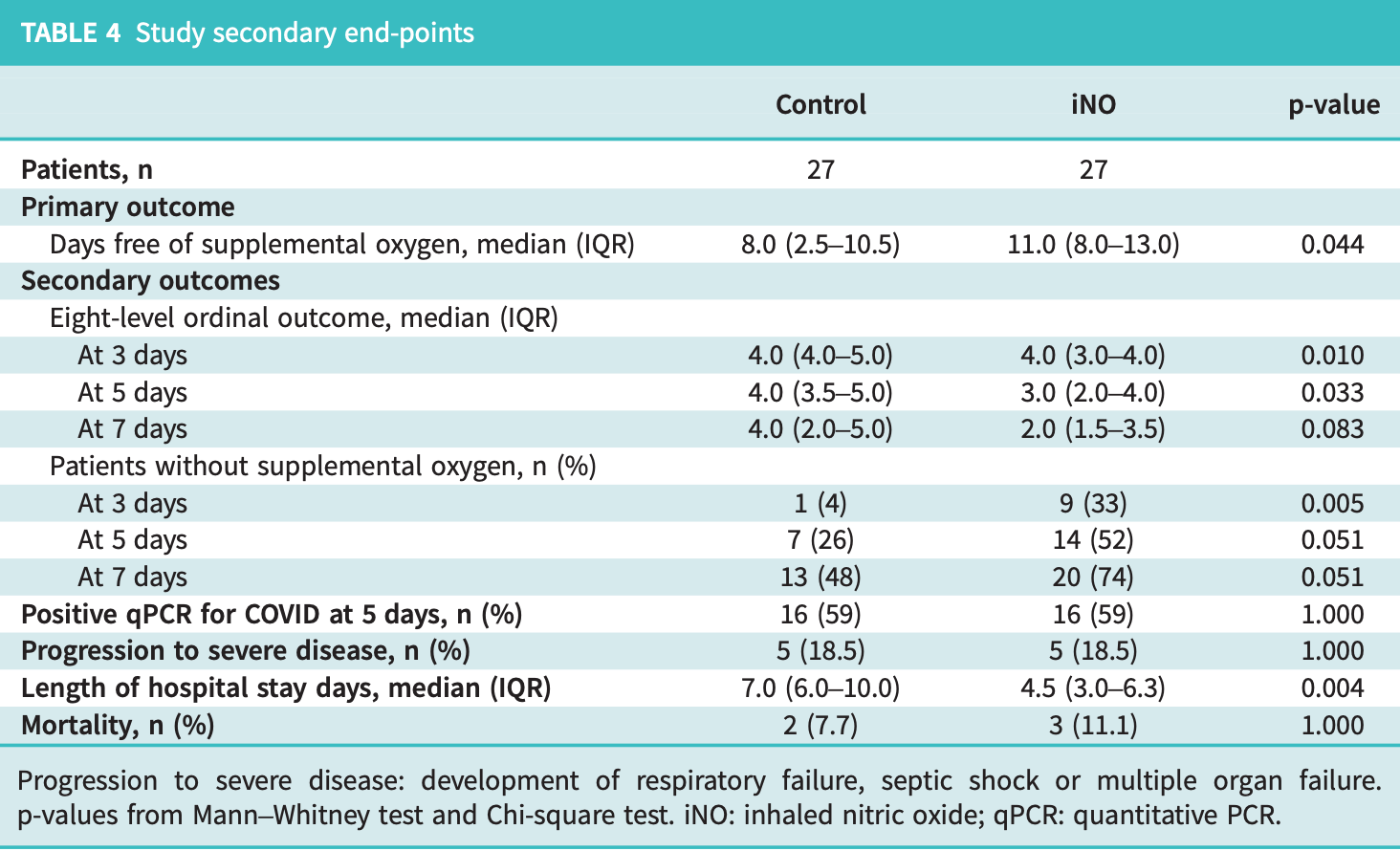
RCT 54 late stage hospitalized COVID-19 patients showing benefit with high-dose inhaled nitric oxide (iNO). Patients receiving 160 ppm iNO for 6 hours had significantly more days free of supplemental oxygen (11 vs 8 days, p=0.044) and shorter hospital stays (4.5 vs 7.0 days, p=0.004) compared to standard care. The iNO group also had better clinical scores at days 3 and 5, and a higher proportion of patients were free of ventilatory support at day 3 (33% vs 4%, p=0.005). There was no significant difference for mortality. Authors hypothesize that the benefit results from iNO's antimicrobial effects, pulmonary vasodilation, and anti-inflammatory properties.
Enrollment number inconsistencies: the abstract reports 27+27 randomized with 1 excluded; the results text says 28 were allocated to iNO; the flowchart shows 54 randomized as 27+27.
Percentage calculation errors: vaccination rates (70.8% and 66.7%) correspond to a denominator of 24, not the reported 27 per group, suggesting unreported missing data.
|
risk of death, 50.0% higher, RR 1.50, p = 1.00, treatment 3 of 27 (11.1%), control 2 of 27 (7.4%).
|
|
risk of oxygen therapy, 50.0% lower, RR 0.50, p = 0.09, treatment 7 of 27 (25.9%), control 14 of 27 (51.9%), NNT 3.9, day 7.
|
|
risk of oxygen therapy, 35.0% lower, RR 0.65, p = 0.09, treatment 13 of 27 (48.1%), control 20 of 27 (74.1%), NNT 3.9, day 5.
|
|
risk of oxygen therapy, 30.8% lower, RR 0.69, p = 0.01, treatment 18 of 27 (66.7%), control 26 of 27 (96.3%), NNT 3.4, day 3.
|
|
risk of progression, no change, RR 1.00, p = 1.00, treatment 5 of 27 (18.5%), control 5 of 27 (18.5%).
|
|
hospitalization time, 35.7% lower, relative time 0.64, p = 0.001, treatment median 4.5 IQR 3.3 n=27, control median 7.0 IQR 4.0 n=27.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Friedrich et al., 6 Feb 2025, Randomized Controlled Trial, Brazil, peer-reviewed, median age 55.0, 20 authors, study period November 2020 - March 2022, trial RBR-8nfx26 (iNO-COVID-19).
Contact: mhjones@pucrs.br.
Inhaled nitric oxide for the treatment of COVID-19: an open-label, parallel, randomised controlled trial
doi:10.1183/23120541.00006-2024].
In summary, we report a successful novel strategy to treat SARS-CoV-2 respiratory infection with inhaled nitric oxide in high concentration (>160 ppm) for 6 h, and this approach could potentially be applied to other pulmonary infections https://bit.ly/40ztJE9
Author contributions: F. Friedrich, M. Cypel, V.S. Michaelsen
References
Abou, Huette, Debouvries, Inhaled nitric oxide for critically ill Covid-19 patients: a prospective study, Crit Care
Akaberi, Krambrich, Ling, Mitigation of the replication of SARS-CoV-2 by nitric oxide in vitro, Redox Biol
Akerström, Gunalan, Keng, Dual effect of nitric oxide on SARS-CoV replication: viral RNA production and palmitoylation of the S protein are affected, Virology
Bartley, Gardner, Spina, High-dose inhaled nitric oxide as adjunct therapy in cystic fibrosis targeting Burkholderia multivorans, Case Rep Pediatr
Bath, Coleman, Gordon, Nitric oxide for the prevention and treatment of viral, bacterial, protozoal and fungal infections, Res
Bentur, Gur, Ashkenazi, Pilot study to test inhaled nitric oxide in cystic fibrosis patients with refractory Mycobacterium abscessus lung infection, J Cyst Fibros, doi:10.1183/23120541.00006-202410
Bogdanovski, Chau, Robinson, Antibacterial activity of high-dose nitric oxide against pulmonary Mycobacterium abscessus disease, Access Microbiol
Bonaventura, Vecchié, Dagna, Endothelial dysfunction and immunothrombosis as key pathogenic mechanisms in COVID-19, Nat Rev Immunol
Chen, Liu, Gao, Inhalation of nitric oxide in the treatment of severe acute respiratory syndrome: a rescue trial in Beijing, Clin Infect Dis
Chen, Ostermann, Review of anti-inflammatory and antiviral therapeutics for hospitalized patients infected with severe acute respiratory syndrome coronavirus 2, Crit Care Clin
Croen, Evidence for antiviral effect of nitric oxide. Inhibition of herpes simplex virus type 1 replication, J Clin Invest
Dhont, Derom, Van, The pathophysiology of 'happy' hypoxemia in COVID-19, Respir Res
Fakhr, Fenza, Gianni, Inhaled high dose nitric oxide is a safe and effective respiratory treatment in spontaneous breathing hospitalized patients with COVID-19 pneumonia, Nitric Oxide
Fenza, Shetty, Gianni, High-dose inhaled nitric oxide in acute hypoxemic respiratory failure due to COVID-19: a multicenter phase II trial, Am J Respir Crit Care Med
Ferrari, Santini, Protti, Inhaled nitric oxide in mechanically ventilated patients with COVID-19, J Crit Care
Garfield, Fadyen, Briar, Potential for personalised application of inhaled nitric oxide in COVID-19 pneumonia, Br J Anaesth
Garren, Ashcraft, Qian, Nitric oxide and viral infection: recent developments in antiviral therapies and platforms, Appl Mater Today
Goldbart, Golan, Pillar, Inhaled nitric oxide therapy in acute bronchiolitis: a multicenter randomized clinical trial, Sci Rep
Hermann, Idziorek, Kusnierz, Role of nitric oxide in the regulation of lymphocyte apoptosis and HIV-1 replication, Int J Immunopharmacol
Ichinose, Roberts, Zapol, Inhaled nitric oxide: a selective pulmonary vasodilator: current uses and therapeutic potential, Circulation
Kamenshchikov, Berra, Carroll, Therapeutic effects of inhaled nitric oxide therapy in COVID-19 patients, Biomedicines
Kamenshchikov, Safaee, Kravchenko, Assessment of continuous low-dose and high-dose burst of inhaled nitric oxide in spontaneously breathing COVID-19 patients: a randomized controlled trial, Nitric Oxide Biol Chem, doi:10.1183/23120541.00006-2024
Keyaerts, Vijgen, Chen, Inhibition of SARS-coronavirus infection in vitro by S-nitroso-N-acetylpenicillamine, a nitric oxide donor compound, Int J Infect Dis
Klingström, Åkerström, Hardestam, Nitric oxide and peroxynitrite have different antiviral effects against hantavirus replication and free mature virions, Eur J Immunol
Longobardo, Montanari, Shulman, Inhaled nitric oxide minimally improves oxygenation in COVID-19 related acute respiratory distress syndrome, Br J Anaesth
Marshall, Murthy, Diaz, A minimal common outcome measure set for COVID-19 clinical research, Lancet Infect Dis
Michaelsen, Ribeiro, Ali, Safety of continuous 12-hour delivery of antimicrobial doses of inhaled nitric oxide during ex vivo lung perfusion, J Thorac Cardiovasc Surg
Michaelsen, Ribeiro, Brambate, A novel pre-clinical strategy to deliver antimicrobial doses of inhaled nitric oxide, PLoS ONE
Miller, Mullin, Ghaffari, Gaseous nitric oxide bactericidal activity retained during intermittent high-dose short duration exposure, Nitric Oxide
Pieretti, Rubilar, Weller, Nitric oxide (NO) and nanoparticles: potential small tools for the war against COVID-19 and other human coronavirus infections, Virus Res
Safaee, Wiegand, Pinciroli, High concentrations of nitric oxide inhalation therapy in pregnant patients with severe coronavirus disease 2019 (COVID-19), Obstet Gynecol
Schairer, Chouake, Nosanchuk, The potential of nitric oxide releasing therapies as antimicrobial agents, Virulence
Sorbo, Michaelsen, Ali, High doses of inhaled nitric oxide as an innovative antimicrobial strategy for lung infections, Biomed
Tal, Greenberg, Gay, Nitric oxide inhalations in bronchiolitis: a pilot, randomized, double-blinded, controlled trial, Pediatr Pulmonol
Valsecchi, Winterton, Safaee, High-dose inhaled nitric oxide for the treatment of spontaneously breathing pregnant patients with severe coronavirus disease 2019 (COVID-19) pneumonia, Obstet Gynecol
Wiegand, Safaee, Carroll, Rescue treatment with high-dose gaseous nitric oxide in spontaneously breathing patients with severe coronavirus disease 2019, Crit Care Explor
Wolak, Dicker, Shifer, A safety evaluation of intermittent high-dose inhaled nitric oxide in viral pneumonia due to COVID-19: a randomised clinical study, Sci Rep
Young, Dyar, Xiong, Methaemoglobin production in normal adults inhaling low concentrations of nitric oxide, Intensive Care Med
Young, Sear, Valvini, Kinetics of methaemoglobin and serum nitrogen oxide production during inhalation of nitric oxide in volunteers, Br J Anaesth
DOI record:
{
"DOI": "10.1183/23120541.00006-2024",
"ISSN": [
"2312-0541"
],
"URL": "http://dx.doi.org/10.1183/23120541.00006-2024",
"abstract": "<jats:sec>\n <jats:title>Background</jats:title>\n <jats:p>Inhaled nitric oxide (iNO) in high concentration inhibits SARS-CoV-2 replication in epithelial cells and may prevent severe disease in hospitalised patients. The aim of this study was to evaluate safety and efficacy of iNO 160 ppm on supplemental oxygen in patients hospitalised with COVID-19.</jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>Methods</jats:title>\n <jats:p>We conducted an open-label, randomised clinical trial in hospitalised patients with COVID-19 receiving supplemental oxygen. Patients were randomly assigned to receive iNO for 6 h in addition to standard of care. The primary safety end-point was assessed by incidence of adverse events. The secondary efficacy end-point was the number of days free of supplemental oxygen within 15 days after randomisation.</jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>Results</jats:title>\n <jats:p>55 patients were enrolled, 27 in the iNO group and 27 in the control group, and one patient was excluded. No adverse events occurred with the inhalation of nitric oxide. Median (IQR) number of days free of supplemental oxygen was 11 (8.0–13.0) in the iNO group and 8 (2.5–10.5) in the control group (p=0.044). The iNO group had a shorter length of hospital stay (4.5 days (3.0–6.3) compared to 7.0 days (6.0–10.0) in the control group; p=0.004). Clinical score was lower in the iNO group on days 3 and 5 (p=0.010 and p=0.033). The number of patients weaned from ventilatory support on day 3 was higher in the iNO group (n=9 (33%)) when compared to controls (n=1 (4%)); p=0.005. Respiratory failure and mortality did not differ between the groups.</jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>Conclusion</jats:title>\n <jats:p>The iNO treatment was well tolerated and safe. Among adults hospitalised for COVID-19, iNO 160 ppm for 6 h increased the number of days free from supplemental oxygen, shortened the days of ventilatory support and the length of hospital stay, and improved the clinical score.</jats:p>\n </jats:sec>",
"accepted": {
"date-parts": [
[
2025,
1,
14
]
]
},
"alternative-id": [
"10.1183/23120541.00006-2024"
],
"author": [
{
"affiliation": [],
"family": "Friedrich",
"given": "Frederico",
"sequence": "first"
},
{
"affiliation": [],
"family": "Cypel",
"given": "Marcelo",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Michaelsen",
"given": "Vinicius Schenk",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Brum",
"given": "Marcos Otávio Antunes",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ramos",
"given": "Fabiano",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Marco",
"given": "Roberta",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Chatkin",
"given": "Gustavo",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Grun",
"given": "Lucas Kich",
"sequence": "additional"
},
{
"affiliation": [],
"family": "dos Santos",
"given": "Letícya Simone Melo",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Santos",
"given": "Amanda Paz",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Fernandes",
"given": "Morgana Thaís Carollo",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Paungartner",
"given": "Luciana Medeiros",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Maranghello",
"given": "Marcelo Scheffer",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Lantmann",
"given": "Caroline",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Eggers",
"given": "Gustavo",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Sanches",
"given": "Gustavo Duenhas",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0002-1146-0745",
"affiliation": [],
"authenticated-orcid": false,
"family": "Marinowic",
"given": "Daniel",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Rosa",
"given": "Regis Goulart",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Barbé-Tuana",
"given": "Florencia María",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Jones",
"given": "Marcus Herbert",
"sequence": "additional"
}
],
"container-title": "ERJ Open Research",
"container-title-short": "ERJ Open Res",
"content-domain": {
"crossmark-restriction": true,
"domain": [
"publications.ersnet.org"
]
},
"created": {
"date-parts": [
[
2025,
2,
6
]
],
"date-time": "2025-02-06T13:11:09Z",
"timestamp": 1738847469000
},
"deposited": {
"date-parts": [
[
2025,
8,
26
]
],
"date-time": "2025-08-26T10:50:17Z",
"timestamp": 1756205417000
},
"funder": [
{
"name": "University Health Network and The University of Toronto"
},
{
"award": [
"Code 001"
],
"name": "Coordenação de Aperfeiçoamento de Pessoal de Nivel Superior – Brasil"
},
{
"award": [
"No. 07/2020"
],
"name": "MCTIC/CNPq/FNDCT/MS/SCITIE/Decit"
}
],
"indexed": {
"date-parts": [
[
2025,
8,
27
]
],
"date-time": "2025-08-27T00:12:46Z",
"timestamp": 1756253566951,
"version": "3.44.0"
},
"is-referenced-by-count": 0,
"issue": "4",
"issued": {
"date-parts": [
[
2025,
2,
6
]
]
},
"journal-issue": {
"issue": "4",
"published-online": {
"date-parts": [
[
2025,
8,
26
]
]
},
"published-print": {
"date-parts": [
[
2025,
7
]
]
}
},
"language": "en",
"license": [
{
"URL": "http://creativecommons.org/licenses/by-nc/4.0/",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2025,
2,
6
]
],
"date-time": "2025-02-06T00:00:00Z",
"timestamp": 1738800000000
}
}
],
"link": [
{
"URL": "https://syndication.highwire.org/content/doi/10.1183/23120541.00006-2024",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "81",
"original-title": [],
"page": "00006-2024",
"prefix": "10.1183",
"published": {
"date-parts": [
[
2025,
2,
6
]
]
},
"published-online": {
"date-parts": [
[
2025,
2,
6
]
]
},
"published-print": {
"date-parts": [
[
2025,
7
]
]
},
"publisher": "European Respiratory Society (ERS)",
"reference": [
{
"DOI": "10.1038/s41577-021-00536-9",
"doi-asserted-by": "publisher",
"key": "2025082603500522000_11.4.00006-2024.1"
},
{
"DOI": "10.1016/j.ccc.2022.02.002",
"article-title": "Review of anti-inflammatory and antiviral therapeutics for hospitalized patients infected with severe acute respiratory syndrome coronavirus 2",
"author": "Chen",
"doi-asserted-by": "crossref",
"first-page": "587",
"journal-title": "Crit Care Clin",
"key": "2025082603500522000_11.4.00006-2024.2",
"volume": "38",
"year": "2022"
},
{
"DOI": "10.1016/j.virusres.2020.198202",
"doi-asserted-by": "publisher",
"key": "2025082603500522000_11.4.00006-2024.3"
},
{
"DOI": "10.3390/biomedicines10020369",
"article-title": "Therapeutic effects of inhaled nitric oxide therapy in COVID-19 patients",
"author": "Kamenshchikov",
"doi-asserted-by": "crossref",
"first-page": "369",
"journal-title": "Biomedicines",
"key": "2025082603500522000_11.4.00006-2024.4",
"volume": "10",
"year": "2022"
},
{
"article-title": "High doses of inhaled nitric oxide as an innovative antimicrobial strategy for lung infections",
"author": "Sorbo",
"first-page": "1525",
"journal-title": "Biomed",
"key": "2025082603500522000_11.4.00006-2024.5",
"volume": "10",
"year": "2022"
},
{
"DOI": "10.1086/425357",
"doi-asserted-by": "publisher",
"key": "2025082603500522000_11.4.00006-2024.6"
},
{
"DOI": "10.1186/s13054-020-03371-x",
"doi-asserted-by": "publisher",
"key": "2025082603500522000_11.4.00006-2024.7"
},
{
"DOI": "10.1016/j.bja.2020.11.006",
"doi-asserted-by": "publisher",
"key": "2025082603500522000_11.4.00006-2024.8"
},
{
"DOI": "10.1016/j.bja.2020.10.011",
"article-title": "Inhaled nitric oxide minimally improves oxygenation in COVID-19 related acute respiratory distress syndrome",
"author": "Longobardo",
"doi-asserted-by": "crossref",
"first-page": "e44",
"journal-title": "Br J Anaesth",
"key": "2025082603500522000_11.4.00006-2024.9",
"volume": "126",
"year": "2021"
},
{
"DOI": "10.12688/f1000research.51270.2",
"article-title": "Nitric oxide for the prevention and treatment of viral, bacterial, protozoal and fungal infections",
"author": "Bath",
"doi-asserted-by": "crossref",
"first-page": "536",
"journal-title": "F1000Res",
"key": "2025082603500522000_11.4.00006-2024.10",
"volume": "10",
"year": "2021"
},
{
"DOI": "10.1016/j.apmt.2020.100887",
"article-title": "Nitric oxide and viral infection: recent developments in antiviral therapies and platforms",
"author": "Garren",
"doi-asserted-by": "crossref",
"first-page": "100887",
"journal-title": "Appl Mater Today",
"key": "2025082603500522000_11.4.00006-2024.11",
"volume": "22",
"year": "2021"
},
{
"DOI": "10.1016/j.ijid.2004.04.012",
"doi-asserted-by": "publisher",
"key": "2025082603500522000_11.4.00006-2024.12"
},
{
"DOI": "10.1016/j.redox.2020.101734",
"article-title": "Mitigation of the replication of SARS-CoV-2 by nitric oxide in vitro",
"author": "Akaberi",
"doi-asserted-by": "crossref",
"first-page": "101734",
"journal-title": "Redox Biol",
"key": "2025082603500522000_11.4.00006-2024.13",
"volume": "37",
"year": "2020"
},
{
"article-title": "Rescue treatment with high-dose gaseous nitric oxide in spontaneously breathing patients with severe coronavirus disease 2019",
"author": "Wiegand",
"first-page": "e0277",
"journal-title": "Crit Care Explor",
"key": "2025082603500522000_11.4.00006-2024.14",
"volume": "2",
"year": "2020"
},
{
"DOI": "10.1016/j.niox.2021.08.003",
"article-title": "Inhaled high dose nitric oxide is a safe and effective respiratory treatment in spontaneous breathing hospitalized patients with COVID-19 pneumonia",
"author": "Safaee Fakhr",
"doi-asserted-by": "crossref",
"first-page": "7",
"journal-title": "Nitric Oxide",
"key": "2025082603500522000_11.4.00006-2024.15",
"volume": "116",
"year": "2021"
},
{
"DOI": "10.1016/j.jcrc.2020.08.007",
"doi-asserted-by": "publisher",
"key": "2025082603500522000_11.4.00006-2024.16"
},
{
"key": "2025082603500522000_11.4.00006-2024.17",
"unstructured": "Brazilian Registry of Clinical Trials – REBEC. Date last accessed: 10 October 2024. https://ensaiosclinicos.gov.br/"
},
{
"DOI": "10.1016/S1473-3099(20)30483-7",
"doi-asserted-by": "publisher",
"key": "2025082603500522000_11.4.00006-2024.18"
},
{
"DOI": "10.1371/journal.pone.0258368",
"article-title": "A novel pre-clinical strategy to deliver antimicrobial doses of inhaled nitric oxide.",
"author": "Michaelsen",
"doi-asserted-by": "crossref",
"first-page": "e0258368",
"journal-title": "PLoS ONE",
"key": "2025082603500522000_11.4.00006-2024.19",
"volume": "16",
"year": "2021"
},
{
"DOI": "10.1007/BF01705726",
"doi-asserted-by": "publisher",
"key": "2025082603500522000_11.4.00006-2024.20"
},
{
"DOI": "10.1093/bja/76.5.652",
"doi-asserted-by": "publisher",
"key": "2025082603500522000_11.4.00006-2024.21"
},
{
"DOI": "10.1016/j.jcf.2019.05.002",
"article-title": "Pilot study to test inhaled nitric oxide in cystic fibrosis patients with refractory Mycobacterium abscessus lung infection",
"author": "Bentur",
"doi-asserted-by": "crossref",
"first-page": "225",
"journal-title": "J Cyst Fibros",
"key": "2025082603500522000_11.4.00006-2024.22",
"volume": "19",
"year": "2020"
},
{
"DOI": "10.1002/ppul.23905",
"article-title": "Nitric oxide inhalations in bronchiolitis: a pilot, randomized, double-blinded, controlled trial",
"author": "Tal",
"doi-asserted-by": "crossref",
"first-page": "95",
"journal-title": "Pediatr Pulmonol",
"key": "2025082603500522000_11.4.00006-2024.23",
"volume": "53",
"year": "2018"
},
{
"DOI": "10.1038/s41598-020-66433-8",
"article-title": "Inhaled nitric oxide therapy in acute bronchiolitis: a multicenter randomized clinical trial",
"author": "Goldbart",
"doi-asserted-by": "crossref",
"first-page": "9605",
"journal-title": "Sci Rep",
"key": "2025082603500522000_11.4.00006-2024.24",
"volume": "10",
"year": "2020"
},
{
"DOI": "10.1016/S0192-0561(97)00060-X",
"doi-asserted-by": "publisher",
"key": "2025082603500522000_11.4.00006-2024.25"
},
{
"DOI": "10.1172/JCI116479",
"doi-asserted-by": "publisher",
"key": "2025082603500522000_11.4.00006-2024.26"
},
{
"DOI": "10.1002/eji.200535587",
"doi-asserted-by": "publisher",
"key": "2025082603500522000_11.4.00006-2024.27"
},
{
"DOI": "10.1016/j.virol.2009.09.007",
"doi-asserted-by": "publisher",
"key": "2025082603500522000_11.4.00006-2024.28"
},
{
"author": "Bartley",
"first-page": "1536714",
"journal-title": "Case Rep Pediatr",
"key": "2025082603500522000_11.4.00006-2024.29",
"volume": "2020",
"year": "2020"
},
{
"DOI": "10.1097/AOG.0000000000004128",
"article-title": "High concentrations of nitric oxide inhalation therapy in pregnant patients with severe coronavirus disease 2019 (COVID-19)",
"author": "Safaee",
"doi-asserted-by": "crossref",
"first-page": "1109",
"journal-title": "Obstet Gynecol",
"key": "2025082603500522000_11.4.00006-2024.30",
"volume": "136",
"year": "2020"
},
{
"DOI": "10.1097/AOG.0000000000004847",
"article-title": "High-dose inhaled nitric oxide for the treatment of spontaneously breathing pregnant patients with severe coronavirus disease 2019 (COVID-19) pneumonia",
"author": "Valsecchi",
"doi-asserted-by": "crossref",
"first-page": "195",
"journal-title": "Obstet Gynecol",
"key": "2025082603500522000_11.4.00006-2024.31",
"volume": "140",
"year": "2022"
},
{
"DOI": "10.1164/rccm.202304-0637OC",
"article-title": "High-dose inhaled nitric oxide in acute hypoxemic respiratory failure due to COVID-19: a multicenter phase II trial",
"author": "Fenza",
"doi-asserted-by": "crossref",
"first-page": "1293",
"journal-title": "Am J Respir Crit Care Med",
"key": "2025082603500522000_11.4.00006-2024.32",
"volume": "208",
"year": "2023"
},
{
"DOI": "10.4161/viru.20328",
"doi-asserted-by": "publisher",
"key": "2025082603500522000_11.4.00006-2024.33"
},
{
"DOI": "10.1016/j.niox.2008.08.002",
"doi-asserted-by": "publisher",
"key": "2025082603500522000_11.4.00006-2024.34"
},
{
"DOI": "10.1099/acmi.0.000154",
"article-title": "Antibacterial activity of high-dose nitric oxide against pulmonary Mycobacterium abscessus disease",
"author": "Bogdanovski",
"doi-asserted-by": "crossref",
"first-page": "acmi000154",
"journal-title": "Access Microbiol",
"key": "2025082603500522000_11.4.00006-2024.35",
"volume": "2",
"year": "2020"
},
{
"DOI": "10.1186/s12931-020-01462-5",
"doi-asserted-by": "publisher",
"key": "2025082603500522000_11.4.00006-2024.36"
},
{
"DOI": "10.1016/j.jtcvs.2020.11.150",
"article-title": "Safety of continuous 12-hour delivery of antimicrobial doses of inhaled nitric oxide during ex vivo lung perfusion",
"author": "Michaelsen",
"doi-asserted-by": "crossref",
"first-page": "841",
"journal-title": "J Thorac Cardiovasc Surg",
"key": "2025082603500522000_11.4.00006-2024.37",
"volume": "163",
"year": "2022"
},
{
"DOI": "10.1161/01.CIR.0000134595.80170.62",
"doi-asserted-by": "publisher",
"key": "2025082603500522000_11.4.00006-2024.38"
},
{
"DOI": "10.1038/s41598-024-68055-w",
"article-title": "A safety evaluation of intermittent high-dose inhaled nitric oxide in viral pneumonia due to COVID-19: a randomised clinical study",
"author": "Wolak",
"doi-asserted-by": "crossref",
"first-page": "17201",
"journal-title": "Sci Rep",
"key": "2025082603500522000_11.4.00006-2024.39",
"volume": "14",
"year": "2024"
},
{
"DOI": "10.1016/j.niox.2024.06.003",
"article-title": "Assessment of continuous low-dose and high-dose burst of inhaled nitric oxide in spontaneously breathing COVID-19 patients: a randomized controlled trial",
"author": "Kamenshchikov",
"doi-asserted-by": "crossref",
"first-page": "41",
"journal-title": "Nitric Oxide Biol Chem",
"key": "2025082603500522000_11.4.00006-2024.40",
"volume": "149",
"year": "2024"
}
],
"reference-count": 40,
"references-count": 40,
"relation": {},
"resource": {
"primary": {
"URL": "https://publications.ersnet.org/lookup/doi/10.1183/23120541.00006-2024"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "Inhaled nitric oxide for the treatment of COVID-19: an open-label, parallel, randomised controlled trial",
"type": "journal-article",
"update-policy": "https://doi.org/10.1183/ers-crossmark-policy",
"volume": "11"
}
