All-Trans Retinoic Acid Exhibits Antiviral Effect against SARS-CoV-2 by Inhibiting 3CLpro Activity
et al., Viruses, doi:10.3390/v13081669, Aug 2021
Vitamin A for COVID-19
49th treatment shown to reduce risk in
May 2023, now with p = 0.004 from 14 studies.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
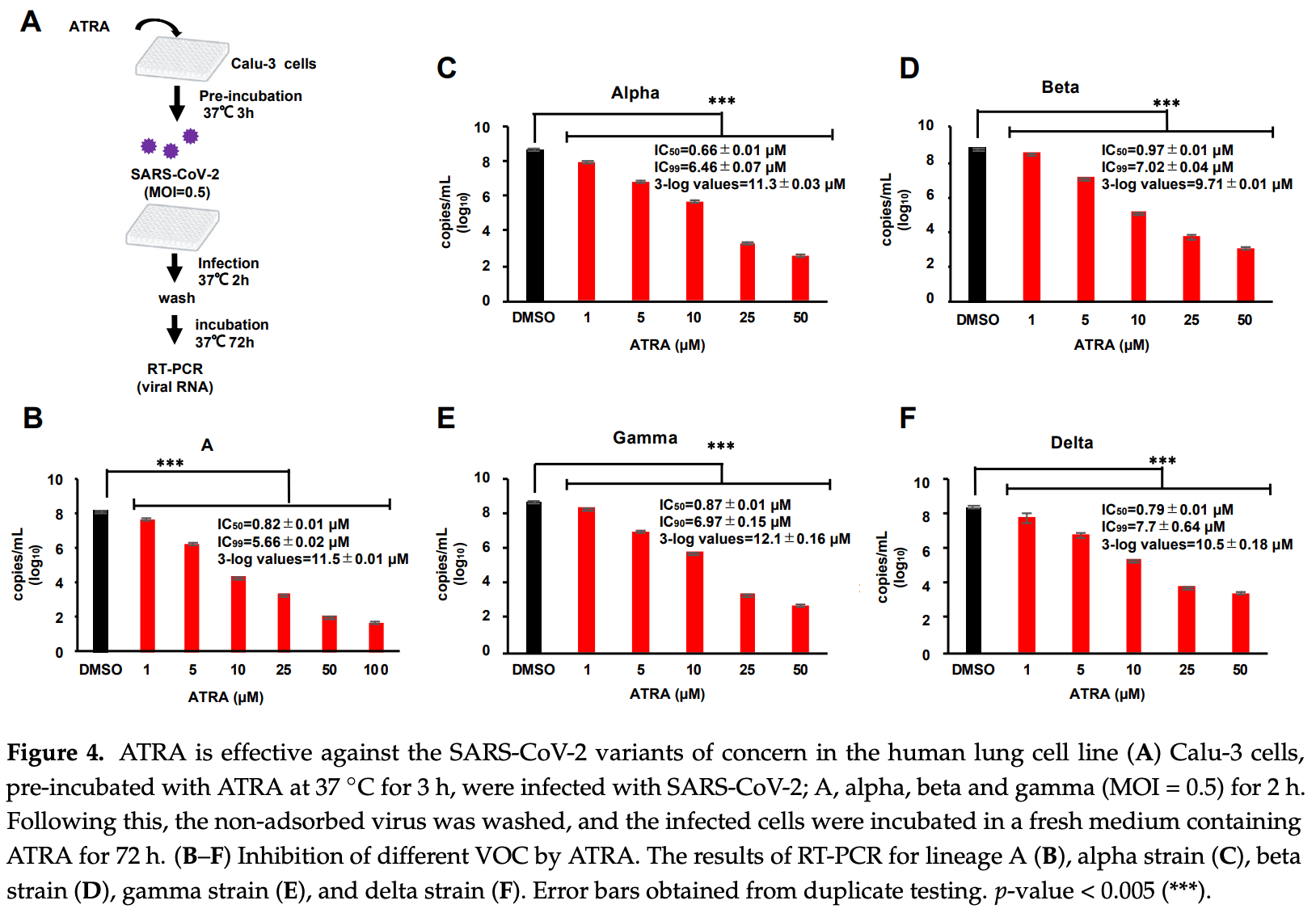
In vitro and in silico study showing that all-trans retinoic acid is a potent SARS-CoV-2 3CLpro inhibitor, inhibits SARS-CoV-2 replication in VeroE6/TMPRSS2 cells, and is effective against alpha, beta, gamma, and delta variants in Calu-3 cells.
11 preclinical studies support the efficacy of vitamin A for COVID-19:
Vitamin A has been identified by the European Food Safety Authority (EFSA) as having sufficient evidence for a causal relationship between intake and optimal immune system function11-13.
Vitamin A has potent antiviral activity against SARS-CoV-2 in both human cell lines and human organoids of the lower respiratory tract (active metabolite all-trans retinoic acid, ATRA)8, is predicted to bind critical host and viral proteins for SARS-CoV-2 and may compensate for gene expression changes related to SARS-CoV-22-4, may be beneficial for COVID-19 via antiviral, anti-inflammatory, and immunomodulatory effects according to network pharmacology analysis5, reduces barrier compromise caused by TNF-α in Calu-3 cells7, inhibits mouse coronavirus replication10, may stimulate innate immunity by activating interferon responses in an IRF3-dependent manner (ATRA)10, may reduce excessive inflammation induced by SARS-CoV-22, shows SARS-CoV-2 antiviral activity In Vitro2,6,9 , is effective against multiple SARS-CoV-2 variants in Calu-3 cells9, and inhibits the entry and replication of SARS-CoV-2 via binding to ACE2 / 3CLpro / RdRp / helicase / 3'-to-5' exonuclease2.
1.
Voloudakis et al., A genetically based computational drug repurposing framework for rapid identification of candidate compounds: application to COVID-19, medRxiv, doi:10.1101/2025.01.10.25320348.
2.
Huang et al., All-trans retinoic acid acts as a dual-purpose inhibitor of SARS-CoV-2 infection and inflammation, Computers in Biology and Medicine, doi:10.1016/j.compbiomed.2024.107942.
3.
Chakraborty et al., In-silico screening and in-vitro assay show the antiviral effect of Indomethacin against SARS-CoV-2, Computers in Biology and Medicine, doi:10.1016/j.compbiomed.2022.105788.
4.
Pandya et al., Unravelling Vitamin B12 as a potential inhibitor against SARS-CoV-2: A computational approach, Informatics in Medicine Unlocked, doi:10.1016/j.imu.2022.100951.
5.
Li et al., Revealing the targets and mechanisms of vitamin A in the treatment of COVID-19, Aging, doi:10.18632/aging.103888.
6.
Moatasim et al., Potent Antiviral Activity of Vitamin B12 against Severe Acute Respiratory Syndrome Coronavirus 2, Middle East Respiratory Syndrome Coronavirus, and Human Coronavirus 229E, Microorganisms, doi:10.3390/microorganisms11112777.
7.
DiGuilio et al., The multiphasic TNF-α-induced compromise of Calu-3 airway epithelial barrier function, Experimental Lung Research, doi:10.1080/01902148.2023.2193637.
8.
Tong et al., A Retinol Derivative Inhibits SARS-CoV-2 Infection by Interrupting Spike-Mediated Cellular Entry, mBio, doi:10.1128/mbio.01485-22.
9.
Morita et al., All-Trans Retinoic Acid Exhibits Antiviral Effect against SARS-CoV-2 by Inhibiting 3CLpro Activity, Viruses, doi:10.3390/v13081669.
10.
Franco et al., Retinoic Acid-Mediated Inhibition of Mouse Coronavirus Replication Is Dependent on IRF3 and CaMKK, Viruses, doi:10.3390/v16010140.
11.
Galmés et al., Suboptimal Consumption of Relevant Immune System Micronutrients Is Associated with a Worse Impact of COVID-19 in Spanish Populations, Nutrients, doi:10.3390/nu14112254.
12.
Galmés (B) et al., Current State of Evidence: Influence of Nutritional and Nutrigenetic Factors on Immunity in the COVID-19 Pandemic Framework, Nutrients, doi:10.3390/nu12092738.
13.
EFSA, Scientific Opinion on the substantiation of health claims related to vitamin A and cell differentiation (ID 14), function of the immune system (ID 14), maintenance of skin and mucous membranes (ID 15, 17), maintenance of vision (ID 16), maintenance of bone (ID 13, 17), maintenance of teeth (ID 13, 17), maintenance of hair (ID 17), maintenance of nails (ID 17), metabolism of iron (ID 206), and protection of DNA, proteins and lipids from oxidative damage (ID 209) pursuant to Article 13(1) of Regulation (EC) No 1924/2006, EFSA Journal, doi:10.2903/j.efsa.2009.1221.
Morita et al., 23 Aug 2021, peer-reviewed, 9 authors.
In vitro studies are an important part of preclinical research, however results may be very different in vivo.
All-Trans Retinoic Acid Exhibits Antiviral Effect against SARS-CoV-2 by Inhibiting 3CLpro Activity
Viruses, doi:10.3390/v13081669
The pandemic of COVID-19 caused by SARS-CoV-2 continues to spread despite the global efforts taken to control it. The 3C-like protease (3CLpro), the major protease of SARS-CoV-2, is one of the most interesting targets for antiviral drug development because it is highly conserved among SARS-CoVs and plays an important role in viral replication. Herein, we developed high throughput screening for SARS-CoV-2 3CLpro inhibitor based on AlphaScreen. We screened 91 natural product compounds and found that all-trans retinoic acid (ATRA), an FDA-approved drug, inhibited 3CLpro activity. The 3CLpro inhibitory effect of ATRA was confirmed in vitro by both immunoblotting and AlphaScreen with a 50% inhibition concentration (IC 50 ) of 24.7 ± 1.65 µM. ATRA inhibited the replication of SARS-CoV-2 in VeroE6/TMPRSS2 and Calu-3 cells, with IC 50 = 2.69 ± 0.09 µM in the former and 0.82 ± 0.01 µM in the latter. Further, we showed the anti-SARS-CoV-2 effect of ATRA on the currently circulating variants of concern (VOC); alpha, beta, gamma, and delta. These results suggest that ATRA may be considered as a potential therapeutic agent against SARS-CoV-2.
Supplementary Materials: The following are available online at https://www.mdpi.com/article/ 10.3390/v13081669/s1, Figure S1 : The comparison of FRET and AlphaScreen, Figure S2 : Graphical determination of the type pf inhibition, Figure S3 : Cytotoxicity of ATRA, Figure S4 : Protein expression in ATRA treated Calu-3 cells during SARS-CoV-2 infection, Figure S5 : Amino acid sequence alignment of 3CLpro, Table S1 : The list of compounds screened by the enzyme assay.
Conflicts of Interest: The authors declare no competing financial interest. Y.Y. is a current employee of Kanto Chemical Co., Inc. T.K. and J.Y. are a current employee of TOKIWA Phytochemical Co., Ltd.
References
Adamson, All-trans-retinoic acid pharmacology and its impact on the treatment of acute promyelocytic leukemia, Oncologist, doi:10.1634/theoncologist.1-5-305
Anand, Ziebuhr, Wadhwani, Mesters, Hilgenfeld, Coronavirus Main Proteinase (3CL Pro) Structure: Basis for Design of Anti-SARS Drugs, Science, doi:10.1126/science.1085658
Caly, Druce, Catton, Jans, Wagstaff, The FDA-approved drug ivermectin inhibits the replication of SARS-CoV-2 in vitro, Antivir. Res, doi:10.1016/j.antiviral.2020.104787
Chelbi-Alix, Pelicano, Retinoic Acid and Interferon Signaling cross Talk in Normal and RA-Resistant APL Cells, Leukemia, doi:10.1038/sj.leu.2401469
Dollé, Developmental expression of retinoic acid receptors (RARs), Nucl. Recept. Signal, doi:10.1621/nrs.07006
Du, Cooper, Chen, Lee, Rong et al., Discovery of Chebulagic Acid and Punicalagin as Novel Allosteric Inhibitors of SARS-CoV-2 3CLpro
El-Baba, Lutomski, Kantsadi, Malla, John et al., Allosteric inhibition of the SARS-CoV-2 main protease: Insights from mass spectrometry based assays, Angew. Chemie-Int. Ed, doi:10.1002/anie.202010316
Ghosh, Chapsal, Weber, Mitsuya, Design of HIV protease inhibitors targeting protein backbone: An effective strategy for combating drug resistance, Acc. Chem. Res, doi:10.1021/ar7001232
Glickman, Wu, Mercuri, Illy, Bowen et al., A Comparison of ALPHAScreen, TR-FRET, and TRF as Assay Methods for FXR Nuclear Receptors, J. Biomol. Screen, doi:10.1177/108705710200700102
Gudas, Retinoids and vertebrate development, J. Biol. Chem, doi:10.1016/S0021-9258(17)40689-2
Hamamoto, Fukuda, Ishimura, Rumi, Kazumori et al., 9-cis retinoic acid enhances the antiviral effect of interferon on hepatitis C virus replication through increased expression of type I interferon receptor, J. Lab. Clin. Med, doi:10.1067/mlc.2003.8
Hoffmann, Kleine-Weber, Schroeder, Krüger, Herrler et al., SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor
Horby, Mafham, Bell, Linsell, Staplin et al., Lopinavir-ritonavir in patients admitted to hospital with COVID-19 (RECOVERY): A randomised, controlled, open-label, platform trial, Lancet, doi:10.1016/S0140-6736(20)32013-4
Huang, Ye, Chen, Chai, Lu et al., Use of All-Trans Retinoic Acid in the Treatment of Acute Promyelocytic Leukemia
Jin, Du, Xu, Deng, Liu et al., Structure of M pro from SARS-CoV-2 and discovery of its inhibitors, Nature, doi:10.1038/s41586-020-2223-y
Luo, Ross, Retinoic acid exerts dual regulatory actions on the expression and nuclear localization of interferon regulatory factor-1, Exp. Biol. Med, doi:10.1177/153537020623100517
Maeda, Yamaguchi, Hijikata, Morita, Tanaka et al., All-trans retinoic acid attacks reverse transcriptase resulting in inhibition of HIV-1 replication, Hematology, doi:10.1080/10245330701255130
Mark, Ghyselinck, Chambon, Function of retinoic acid receptors during embryonic development, Nucl. Recept. Signal, doi:10.1621/nrs.07002
Matsunaga, Masaoka, Sawasaki, Morishita, Iwatani et al., A cell-free enzymatic activity assay for the evaluation of HIV-1 drug resistance to protease inhibitors, Front. Microbiol
Matsuyama, Nao, Shirato, Kawase, Saito et al., Enhanced isolation of SARS-CoV-2 by TMPRSS2-expressing cells, doi:10.1073/pnas.2002589117
Mucida, Park, Kim, Turovskaya, Scott et al., Reciprocal TH17 and regulatory T cell differentiation mediated by retinoic acid, Science, doi:10.1126/science.1145697
Naoki, Arihiro, Toshiyuki, Noriko, Fumio et al., The genome landscape of the African Green Monkey kidney-derived vero cell line, DNA Res, doi:10.1093/dnares/dsu029
Pillaiyar, Manickam, Namasivayam, Hayashi, Jung, An overview of severe acute respiratory syndromecoronavirus (SARS-CoV) 3CL protease inhibitors: Peptidomimetics and small molecule chemotherapy, J. Med. Chem, doi:10.1021/acs.jmedchem.5b01461
Sanders, Monogue, Jodlowski, Cutrell, Pharmacologic treatments for coronavirus disease 2019 (COVID-19): A review, JAMA J. Am. Med. Assoc, doi:10.1001/jama.2020.6019
Soye, Trottier, Di Lenardo, Restori, Reichman et al., In vitro inhibition of mumps virus by retinoids, Virol. J, doi:10.1186/1743-422X-10-337
Soye, Trottier, Richardson, Ward, Miller, RIG-I is required for the inhibition of measles virus by retinoids, PLoS ONE, doi:10.1371/journal.pone.0022323
Trott, Olson, Vina, Improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading, J. Comput. Chem, doi:10.1002/jcc.21334
Vuong, Khan, Fischer, Arutyunova, Lamer et al., Feline coronavirus drug inhibits the main protease of SARS-CoV-2 and blocks virus replication, Nat. Commun, doi:10.1038/s41467-020-18096-2
Wang, Cao, Zhang, Yang, Liu et al., Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro, Cell Res, doi:10.1038/s41422-020-0282-0
Wang, Nair, Liu, Iketani, Luo et al., Antibody resistance of SARS-CoV-2 variants B.1.351 and B.1.1.7, Nature, doi:10.1038/s41586-021-03398-2
Yamada, Sato, Sotoyama, Orba, Sawa et al., RIG-I triggers a signaling-abortive anti-SARS-CoV-2 defense in human lung cells, Nat. Immunol, doi:10.1038/s41590-021-00942-0
Yamaguchi, Maeda, Ueda, Hijikata, Morita et al., Dichotomy of all-trans retinoic acid inducing signals for adult T-cell leukemia, Leukemia, doi:10.1038/sj.leu.2403760
Yamaoka, Matsunaga, Jeremiah, Nishi, Miyakawa et al., Zika virus protease induces caspase-independent pyroptotic cell death by directly cleaving gasdermin D, Biochem. Biophys. Res. Commun, doi:10.1016/j.bbrc.2020.11.023
Yamaoka, Matsuyama, Fukushi, Matsunaga, Matsushima et al., Development of monoclonal antibody and diagnostic test for Middle East respiratory syndrome coronavirus using cell-free synthesized nucleocapsid antigen, Front. Microbiol, doi:10.3389/fmicb.2016.00509
Zhang, Lin, Sun, Curth, Drosten et al., Crystal structure of SARS-CoV-2 main protease provides a basis for design of improved a-ketoamide inhibitors, Science, doi:10.1126/science.abb3405
Zhu, Xu, Chen, Guo, Shen et al., Identification of SARS-CoV-2 3CL protease inhibitors by a quantitative high-throughput screening, ACS Pharmacol. Transl. Sci
Zhu, Zhang, Wang, Li, Yang et al., A novel coronavirus from patients with pneumonia in China, 2019, N. Engl. J. Med, doi:10.1056/NEJMoa2001017
DOI record:
{
"DOI": "10.3390/v13081669",
"ISSN": [
"1999-4915"
],
"URL": "http://dx.doi.org/10.3390/v13081669",
"abstract": "<jats:p>The pandemic of COVID-19 caused by SARS-CoV-2 continues to spread despite the global efforts taken to control it. The 3C-like protease (3CLpro), the major protease of SARS-CoV-2, is one of the most interesting targets for antiviral drug development because it is highly conserved among SARS-CoVs and plays an important role in viral replication. Herein, we developed high throughput screening for SARS-CoV-2 3CLpro inhibitor based on AlphaScreen. We screened 91 natural product compounds and found that all-trans retinoic acid (ATRA), an FDA-approved drug, inhibited 3CLpro activity. The 3CLpro inhibitory effect of ATRA was confirmed in vitro by both immunoblotting and AlphaScreen with a 50% inhibition concentration (IC50) of 24.7 ± 1.65 µM. ATRA inhibited the replication of SARS-CoV-2 in VeroE6/TMPRSS2 and Calu-3 cells, with IC50 = 2.69 ± 0.09 µM in the former and 0.82 ± 0.01 µM in the latter. Further, we showed the anti-SARS-CoV-2 effect of ATRA on the currently circulating variants of concern (VOC); alpha, beta, gamma, and delta. These results suggest that ATRA may be considered as a potential therapeutic agent against SARS-CoV-2.</jats:p>",
"alternative-id": [
"v13081669"
],
"author": [
{
"ORCID": "http://orcid.org/0000-0001-5109-2003",
"affiliation": [],
"authenticated-orcid": false,
"family": "Morita",
"given": "Takeshi",
"sequence": "first"
},
{
"affiliation": [],
"family": "Miyakawa",
"given": "Kei",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Jeremiah",
"given": "Sundararaj Stanleyraj",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Yamaoka",
"given": "Yutaro",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Sada",
"given": "Mitsuru",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Kuniyoshi",
"given": "Tomoko",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Yang",
"given": "Jinwei",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Kimura",
"given": "Hirokazu",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ryo",
"given": "Akihide",
"sequence": "additional"
}
],
"container-title": "Viruses",
"container-title-short": "Viruses",
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2021,
8,
24
]
],
"date-time": "2021-08-24T03:19:33Z",
"timestamp": 1629775173000
},
"deposited": {
"date-parts": [
[
2021,
8,
25
]
],
"date-time": "2021-08-25T09:34:19Z",
"timestamp": 1629884059000
},
"funder": [
{
"DOI": "10.13039/100009619",
"award": [
"JP19fk0108110",
"JP20he0522001"
],
"doi-asserted-by": "publisher",
"name": "Japan Agency for Medical Research and Development"
}
],
"indexed": {
"date-parts": [
[
2024,
4,
1
]
],
"date-time": "2024-04-01T15:54:42Z",
"timestamp": 1711986882817
},
"is-referenced-by-count": 18,
"issue": "8",
"issued": {
"date-parts": [
[
2021,
8,
23
]
]
},
"journal-issue": {
"issue": "8",
"published-online": {
"date-parts": [
[
2021,
8
]
]
}
},
"language": "en",
"license": [
{
"URL": "https://creativecommons.org/licenses/by/4.0/",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2021,
8,
23
]
],
"date-time": "2021-08-23T00:00:00Z",
"timestamp": 1629676800000
}
}
],
"link": [
{
"URL": "https://www.mdpi.com/1999-4915/13/8/1669/pdf",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "1968",
"original-title": [],
"page": "1669",
"prefix": "10.3390",
"published": {
"date-parts": [
[
2021,
8,
23
]
]
},
"published-online": {
"date-parts": [
[
2021,
8,
23
]
]
},
"publisher": "MDPI AG",
"reference": [
{
"DOI": "10.1056/NEJMoa2001017",
"doi-asserted-by": "publisher",
"key": "ref1"
},
{
"DOI": "10.1038/s41586-021-03398-2",
"doi-asserted-by": "publisher",
"key": "ref2"
},
{
"DOI": "10.1001/jama.2020.6019",
"doi-asserted-by": "publisher",
"key": "ref3"
},
{
"DOI": "10.1126/science.abb3405",
"doi-asserted-by": "publisher",
"key": "ref4"
},
{
"DOI": "10.1126/science.1085658",
"doi-asserted-by": "publisher",
"key": "ref5"
},
{
"DOI": "10.1021/acs.jmedchem.5b01461",
"doi-asserted-by": "publisher",
"key": "ref6"
},
{
"DOI": "10.1038/s41586-020-2223-y",
"doi-asserted-by": "publisher",
"key": "ref7"
},
{
"DOI": "10.1038/s41467-020-18096-2",
"doi-asserted-by": "publisher",
"key": "ref8"
},
{
"DOI": "10.1038/sj.leu.2401469",
"doi-asserted-by": "publisher",
"key": "ref9"
},
{
"DOI": "10.1067/mlc.2003.8",
"doi-asserted-by": "publisher",
"key": "ref10"
},
{
"DOI": "10.1371/journal.pone.0022323",
"doi-asserted-by": "publisher",
"key": "ref11"
},
{
"DOI": "10.1186/1743-422X-10-337",
"doi-asserted-by": "publisher",
"key": "ref12"
},
{
"DOI": "10.1080/10245330701255130",
"doi-asserted-by": "publisher",
"key": "ref13"
},
{
"DOI": "10.1038/sj.leu.2403760",
"doi-asserted-by": "publisher",
"key": "ref14"
},
{
"DOI": "10.1177/108705710200700102",
"doi-asserted-by": "publisher",
"key": "ref15"
},
{
"DOI": "10.1073/pnas.2002589117",
"doi-asserted-by": "publisher",
"key": "ref16"
},
{
"DOI": "10.3389/fmicb.2015.01220",
"doi-asserted-by": "publisher",
"key": "ref17"
},
{
"DOI": "10.1016/j.bbrc.2020.11.023",
"doi-asserted-by": "publisher",
"key": "ref18"
},
{
"DOI": "10.3389/fmicb.2016.00509",
"doi-asserted-by": "publisher",
"key": "ref19"
},
{
"DOI": "10.1002/jcc.21334",
"doi-asserted-by": "publisher",
"key": "ref20"
},
{
"key": "ref21",
"unstructured": "Discovery of Chebulagic Acid and Punicalagin as Novel Allosteric Inhibitors of SARS-CoV-2 3CLprohttps://reader.elsevier.com/reader/sd/pii/S0166354221000656?token=067A519D50C243D8DB69226FA5814A6B5F535267D684F0327D013091EF027785765816A014815C1A69403D353EB45C3F&originRegion=us-east-1&originCreation=20210729065020"
},
{
"DOI": "10.1021/acsptsci.0c00108",
"doi-asserted-by": "publisher",
"key": "ref22"
},
{
"DOI": "10.1177/153537020623100517",
"doi-asserted-by": "publisher",
"key": "ref23"
},
{
"DOI": "10.1093/dnares/dsu029",
"doi-asserted-by": "publisher",
"key": "ref24"
},
{
"DOI": "10.1038/s41422-020-0282-0",
"doi-asserted-by": "publisher",
"key": "ref25"
},
{
"key": "ref26",
"unstructured": "SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitorhttps://reader.elsevier.com/reader/sd/pii/S0092867420302294?token=9EBCF58606E2B75994845A858932AF32E87A4870844A01D4C9A8810584C7FC69E3D6BBAC9349F8716F2A547B5A3E5055&originRegion=us-east-1&originCreation=20210518035149"
},
{
"DOI": "10.1038/s41590-021-00942-0",
"doi-asserted-by": "publisher",
"key": "ref27"
},
{
"key": "ref28"
},
{
"author": "Huang",
"key": "ref29",
"series-title": "Use of All-Trans Retinoic Acid in the Treatment of Acute Promyelocytic Leukemia",
"volume": "Volume 32",
"year": "1989"
},
{
"DOI": "10.1002/anie.202010316",
"doi-asserted-by": "publisher",
"key": "ref30"
},
{
"DOI": "10.1021/ar7001232",
"doi-asserted-by": "publisher",
"key": "ref31"
},
{
"DOI": "10.1621/nrs.07006",
"doi-asserted-by": "publisher",
"key": "ref32"
},
{
"DOI": "10.1016/S0021-9258(17)40689-2",
"doi-asserted-by": "publisher",
"key": "ref33"
},
{
"DOI": "10.1621/nrs.07002",
"doi-asserted-by": "publisher",
"key": "ref34"
},
{
"DOI": "10.1126/science.1145697",
"doi-asserted-by": "publisher",
"key": "ref35"
},
{
"DOI": "10.1016/j.antiviral.2020.104787",
"doi-asserted-by": "publisher",
"key": "ref36"
},
{
"DOI": "10.1634/theoncologist.1-5-305",
"doi-asserted-by": "publisher",
"key": "ref37"
},
{
"DOI": "10.1016/S0140-6736(20)32013-4",
"doi-asserted-by": "publisher",
"key": "ref38"
}
],
"reference-count": 38,
"references-count": 38,
"relation": {},
"resource": {
"primary": {
"URL": "https://www.mdpi.com/1999-4915/13/8/1669"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "All-Trans Retinoic Acid Exhibits Antiviral Effect against SARS-CoV-2 by Inhibiting 3CLpro Activity",
"type": "journal-article",
"volume": "13"
}
