Impact of Antibody Cocktail Therapy Combined with Casirivimab and Imdevimab on Clinical Outcome for Covid-19 patients in A Real-Life Setting: A Single Institute Analysis
et al., International Journal of Infectious Diseases, doi:10.1016/j.ijid.2022.01.067, Nov 2021 (preprint)
18th treatment shown to reduce risk in
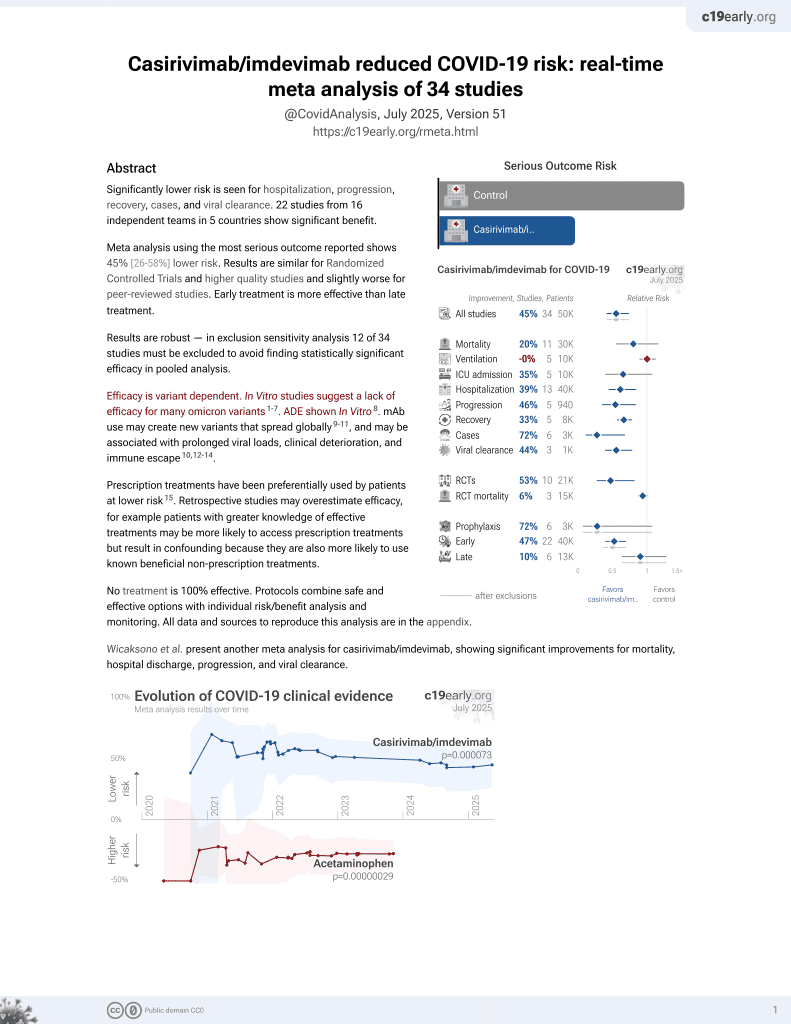
March 2021, now with p = 0.000095 from 34 studies, recognized in 52 countries.
Efficacy is variant dependent.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
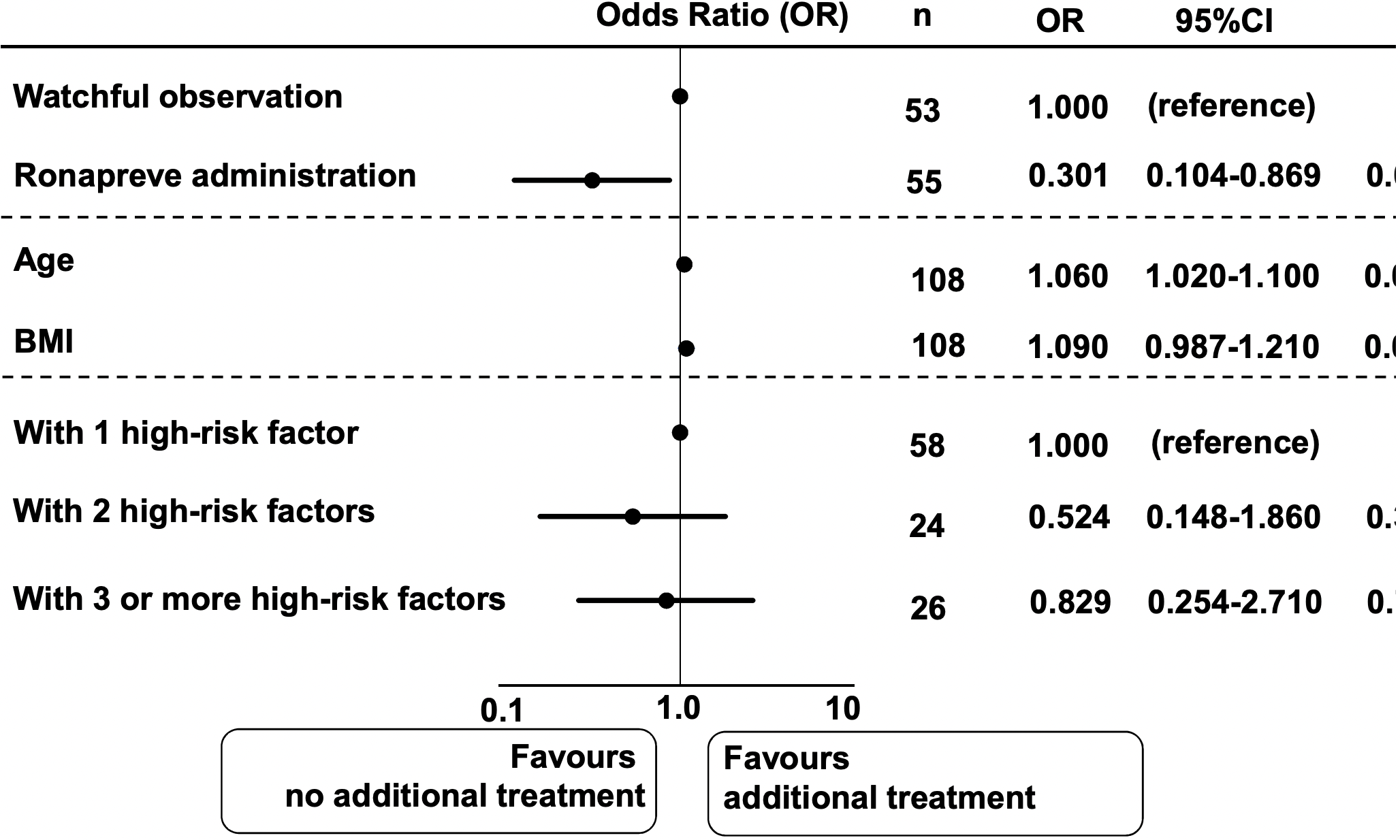
Retrospective 55 patients in Japan treated a median of 3 days from symptom onset with casirivimab/imdevimab, and 53 control patients, showing lower risk of further treatment including oxygen or antivirals.
Efficacy is variant dependent. In Vitro research suggests a lack of efficacy for many omicron variants1-7.
Standard of Care (SOC) for COVID-19 in the study country,
Japan, is very poor with very low average efficacy for approved treatments8.
Only expensive, high-profit treatments were approved for early treatment. Low-cost treatments were excluded, reducing the probability of early treatment due to access and cost barriers, and eliminating complementary and synergistic benefits seen with many low-cost treatments.
|
risk of further treatment including oxygen or antivirals, 57.6% lower, RR 0.42, p = 0.049, treatment 13 of 55 (23.6%), control 22 of 53 (41.5%), NNT 5.6, adjusted per study, odds ratio converted to relative risk, multivariable.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
1.
Liu et al., Striking Antibody Evasion Manifested by the Omicron Variant of SARS-CoV-2, bioRxiv, doi:10.1101/2021.12.14.472719.
2.
Sheward et al., Variable loss of antibody potency against SARS-CoV-2 B.1.1.529 (Omicron), bioRxiv, doi:10.1101/2021.12.19.473354.
3.
VanBlargan et al., An infectious SARS-CoV-2 B.1.1.529 Omicron virus escapes neutralization by several therapeutic monoclonal antibodies, bioRxiv, doi:10.1101/2021.12.15.472828.
4.
Tatham et al., Lack of Ronapreve (REGN-CoV; casirivimab and imdevimab) virological efficacy against the SARS-CoV 2 Omicron variant (B.1.1.529) in K18-hACE2 mice, bioRxiv, doi:10.1101/2022.01.23.477397.
5.
Pochtovyi et al., In Vitro Efficacy of Antivirals and Monoclonal Antibodies against SARS-CoV-2 Omicron Lineages XBB.1.9.1, XBB.1.9.3, XBB.1.5, XBB.1.16, XBB.2.4, BQ.1.1.45, CH.1.1, and CL.1, Vaccines, doi:10.3390/vaccines11101533.
6.
Haars et al., Prevalence of SARS-CoV-2 Omicron Sublineages and Spike Protein Mutations Conferring Resistance against Monoclonal Antibodies in a Swedish Cohort during 2022–2023, Microorganisms, doi:10.3390/microorganisms11102417.
Kakinoki et al., 4 Nov 2021, retrospective, Japan, peer-reviewed, 16 authors, average treatment delay 3.0 days.
Impact of Antibody Cocktail Therapy Combined with Casirivimab and Imdevimab on Clinical Outcome for patients with COVID-19 in A Real-Life Setting: A Single Institute Analysis
International Journal of Infectious Diseases, doi:10.1016/j.ijid.2022.01.067
This is a PDF file of an article that has undergone enhancements after acceptance, such as the addition of a cover page and metadata, and formatting for readability, but it is not yet the definitive version of record. This version will undergo additional copyediting, typesetting and review before it is published in its final form, but we are providing this version to give early visibility of the article. Please note that, during the production process, errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
AUTHOR CONTRIBUTIONS: Design of research studies: YK and KY. Data acquisition: TK, KY, GA, and NS. Data analysis: YK and KY. Writing the manuscript: YK. All authors worked hard to take care of patients with Covid-19.
References
Baum, Fulton, Wloga, Antibody cocktail to SARS-CoV-2 spike protein prevents rapid mutational escape seen with individual antibodies, Science
Copin, Baum, Wloga, The monoclonal antibody combination REGEN-COV protects against SARS-C0V-2 mutational escape in preclinical and human studies, Cell
Hansen, Baum, Pascal, Studies in humanized mice and convalescent humans yield a SARS-CoV-2 antibody cocktail, Science
Ikemura, Hoshino, Higuchi, SARS-CoV-2 Omicron variant escapes neutralization by vaccinated and convalescent sera and therapeutic monoclonal antibodies, doi:10.1101/2021.12.13.21267761
Kanda, Investigation of the freely-available easy-to-use software"EZR" (Easy R) for medical statistics, Bone Marrow Transplant, doi:10.1038/bmt.2012.244
Wang, Nair, Liu, Antibody resistance of SARS-CoV-2 variants B.1.351 and B.1.1.7, Nature
Weinreich, Sivapalasingam, Norton, REGEN-COV antibody cocktail clinical outcomes study in Covid-19 outpatients, doi:10.1101/2021.05.19.21257469v2
Weinreich, Sivapalasingam, Norton, REGN-COV2, a neutralizing antibody cocktail, in outpatients with Covid-19, N Engl J Med
Welfare, Brien, Forleo-Neto, Musser, Approval for Ronapreve TM (casirivimab and imdevimab) for the treatment of patients with mild to moderate Covid-19, N Engl J Med
Wilhelm, Widera, Grikscheit, Reduced neutralization of SARS-CoV-2 omicron variant by vaccine sera and monoclonal antibodies, doi:10.1101/2021.12.07.21267432
Wu, Zhao, Yu, A new coronavirus associated with human respiratory disease in China, Nature
Zhu, Zhang, Wang, A novel coronavirus from patients with pneumonia in China, 2019, N Engl J Med
DOI record:
{
"DOI": "10.1016/j.ijid.2022.01.067",
"ISSN": [
"1201-9712"
],
"URL": "http://dx.doi.org/10.1016/j.ijid.2022.01.067",
"alternative-id": [
"S1201971222000765"
],
"author": [
{
"ORCID": "http://orcid.org/0000-0002-9992-0557",
"affiliation": [],
"authenticated-orcid": false,
"family": "Kakinoki",
"given": "Yasutaka",
"sequence": "first"
},
{
"ORCID": "http://orcid.org/0000-0001-8263-0011",
"affiliation": [],
"authenticated-orcid": false,
"family": "Yamada",
"given": "Kazuki",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-8537-5519",
"affiliation": [],
"authenticated-orcid": false,
"family": "Tanino",
"given": "Yoko",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-4285-0658",
"affiliation": [],
"authenticated-orcid": false,
"family": "Suzuki",
"given": "Keiko",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ichikawa",
"given": "Takaya",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Suzuki",
"given": "Naoki",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Asari",
"given": "Go",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Nakamura",
"given": "Ai",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Kukita",
"given": "Shin",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Uehara",
"given": "Akito",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Saito",
"given": "Seisuke",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Kuroda",
"given": "Shohei",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Sakagami",
"given": "Hidemitsu",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Nagashima",
"given": "Yuuki",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Takahashi",
"given": "Kae",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0001-7622-8288",
"affiliation": [],
"authenticated-orcid": false,
"family": "Suzuki",
"given": "Satoshi",
"sequence": "additional"
}
],
"container-title": [
"International Journal of Infectious Diseases"
],
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2022,
2,
15
]
],
"date-time": "2022-02-15T03:25:12Z",
"timestamp": 1644895512000
},
"deposited": {
"date-parts": [
[
2022,
2,
15
]
],
"date-time": "2022-02-15T03:25:12Z",
"timestamp": 1644895512000
},
"indexed": {
"date-parts": [
[
2022,
2,
15
]
],
"date-time": "2022-02-15T03:41:21Z",
"timestamp": 1644896481216
},
"is-referenced-by-count": 0,
"issn-type": [
{
"type": "print",
"value": "1201-9712"
}
],
"issued": {
"date-parts": [
[
2022,
2
]
]
},
"language": "en",
"license": [
{
"URL": "https://www.elsevier.com/tdm/userlicense/1.0/",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2022,
2,
1
]
],
"date-time": "2022-02-01T00:00:00Z",
"timestamp": 1643673600000
}
},
{
"URL": "http://creativecommons.org/licenses/by-nc-nd/4.0/",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2022,
1,
31
]
],
"date-time": "2022-01-31T00:00:00Z",
"timestamp": 1643587200000
}
}
],
"link": [
{
"URL": "https://api.elsevier.com/content/article/PII:S1201971222000765?httpAccept=text/xml",
"content-type": "text/xml",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://api.elsevier.com/content/article/PII:S1201971222000765?httpAccept=text/plain",
"content-type": "text/plain",
"content-version": "vor",
"intended-application": "text-mining"
}
],
"member": "78",
"original-title": [],
"prefix": "10.1016",
"published": {
"date-parts": [
[
2022,
2
]
]
},
"published-print": {
"date-parts": [
[
2022,
2
]
]
},
"publisher": "Elsevier BV",
"reference-count": 0,
"references-count": 0,
"relation": {},
"score": 1,
"short-container-title": [
"International Journal of Infectious Diseases"
],
"short-title": [],
"source": "Crossref",
"subject": [
"Infectious Diseases",
"Microbiology (medical)",
"General Medicine"
],
"subtitle": [],
"title": [
"Impact of Antibody Cocktail Therapy Combined with Casirivimab and Imdevimab on Clinical Outcome for Covid-19 patients in A Real-Life Setting: A Single Institute Analysis"
],
"type": "journal-article"
}