Efficacy and Safety of Inosine Pranobex in COVID‐19 Patients: A Multicenter Phase 3 Randomized Double‐Blind, Placebo‐Controlled Trial
et al., Advanced Therapeutics, doi:10.1002/adtp.202200159, CTRI/2021/02/030892, Sep 2022
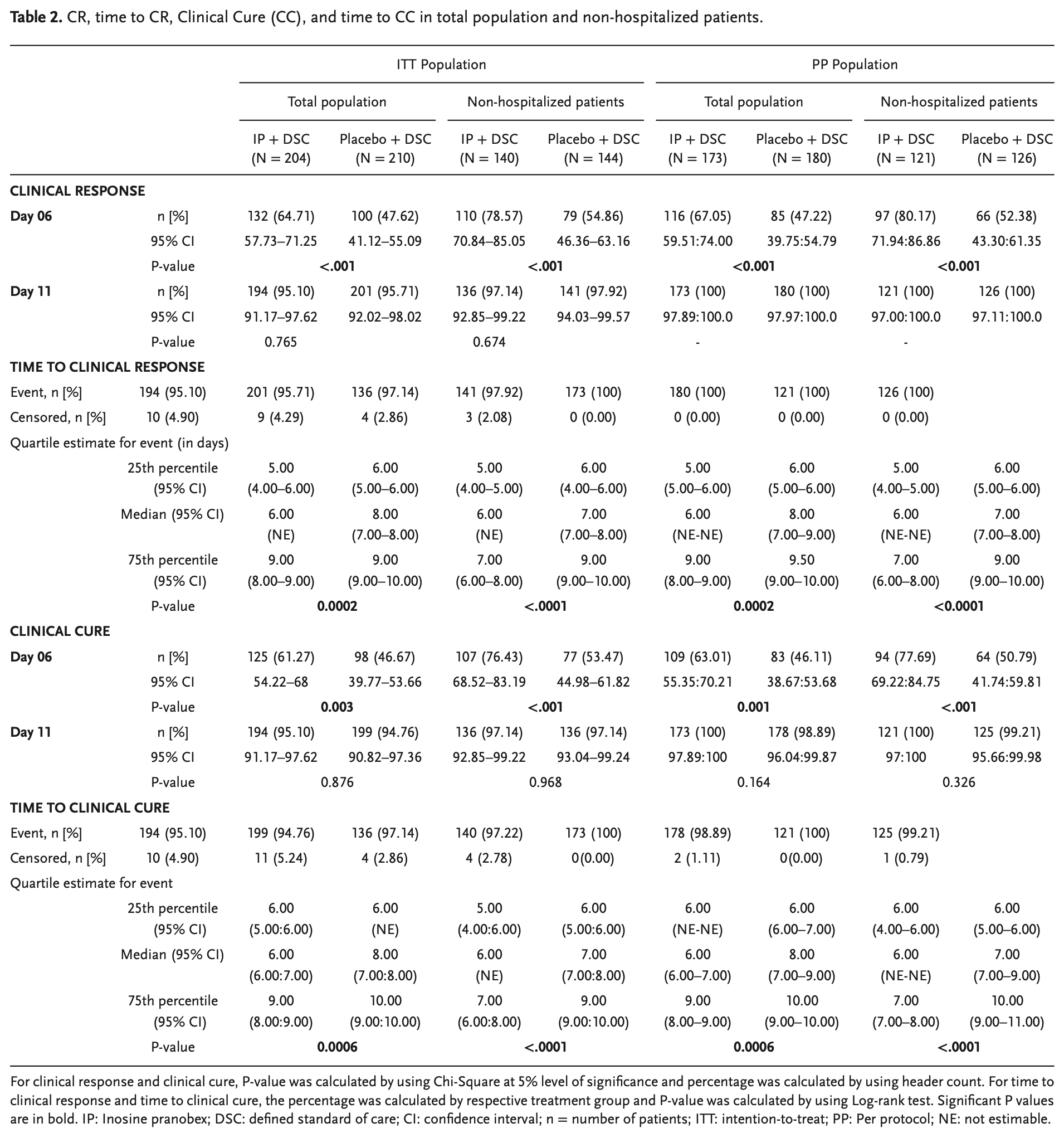
RCT 416 mild-to-moderate COVID-19 patients (both hospitalized and non-hospitalized) showing improved recovery with inosine pranobex.
|
risk of no recovery, 27.4% lower, RR 0.73, p = 0.003, treatment 79 of 204 (38.7%), control 112 of 210 (53.3%), NNT 6.8, mid-recovery, day 6.
|
|
risk of no recovery, 6.4% lower, RR 0.94, p = 1.00, treatment 10 of 204 (4.9%), control 11 of 210 (5.2%), NNT 297, day 11.
|
|
risk of no improvement, 32.6% lower, RR 0.67, p < 0.001, treatment 72 of 204 (35.3%), control 110 of 210 (52.4%), NNT 5.9, mid-recovery, day 6.
|
|
risk of no improvement, 14.4% higher, RR 1.14, p = 0.82, treatment 10 of 204 (4.9%), control 9 of 210 (4.3%), day 11.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Jayanthi et al., 16 Sep 2022, Double Blind Randomized Controlled Trial, placebo-controlled, India, peer-reviewed, 13 authors, study period 8 February, 2021 - 16 June, 2021, trial CTRI/2021/02/030892.
Contact: ashok.swain@themismedicare.com.
Inosine pranobex is an orally administered small-molecule immunomodulator with broad antiviral activity. It enhances natural killer cell cytotoxicity and T-lymphocyte function and promotes Th1-type cytokine responses, thereby augmenting host antiviral defenses.
Abstract:
Efficacy and Safety of Inosine Pranobex in COVID-19 Patients: A Multicenter Phase 3 Randomized Double-Blind, Placebo-Controlled Trial
Jayanthi C R, Ashok K Swain,* Ranganath T Ganga, Dnyaneshwar Halnor, Ajit Avhad, Mohd. Saif Khan, Ayan Ghosh, Sumer Sanjiv Choudhary, Anand Namdevrao Yannawar, Shubhangi Despande, Manish Patel, Krishna Prasad Anne, and Yogesh Bangar
Inosine pranobex (IP), an immunomodulatory agent, is used in the treatment of various viral infections. The results of a phase 3 randomized controlled trial are reported, evaluating the efficacy and safety of IP in the treatment of mild to moderate COVID-19. It includes 416 symptomatic patients with confirmed SARS-CoV-2 infection. In addition to a defined standard of care, patients randomly (1:1) receive either IP 500 mg tablet (IP group) or a matching placebo (placebo group) at 50 mg kg -1 body weight/day rounded to the nearest 500 mg dose (maximum 4 g day -1 ) administered in 3-4 divided doses for 10 days. Compared to the placebo group, IP group shows significantly higher rates of clinical response (CR) and clinical cure (CC) on Day-6 for both non-hospitalized patients and the total population. IP group shows significantly earlier CR and CC with fewer adverse events and no mortality. Based on these findings and the fact that IP increases natural killer cell-mediated cytotoxicity of virus-infected cells as an early immune response to viral infection and enhances NKG2D ligand expression, it is concluded that IP should be started early to maximize the benefit in mild to moderate COVID-19 patients. (Trial registration number: CTRI/2021/02/030892).
J. C R
Faculty of Medicine and Department of Pharmacology Victoria Hospital, Bangalore Medical College and Research Institute KR Road Fort, Bangalore, Karnataka 560002, India
A. K Swain
Medical Services
Themis Medicare Ltd.
11/12, Udyog Nagar, S.V. Road, Goregaon (W), Mumbai, Maharashtra 400104, India
[E-mail: ashok.swain@themismedicare.com](mailto:ashok.swain@themismedicare.com)
R. T Ganga
Department of Pulmonary Medicine
All India Institute of Medical Sciences
Gate No, 1, Great Eastern Rd, opposite Gurudwara, AIIMS Campus,
Tatibandh, Raipur, Chhattisgarh 492099, India
The ORCID identification number(s) for the author(s) of this article can be found under https://doi.org/10.1002/adtp.202200159
DOI: 10.1002/adtp.202200159
D. Halnor
Department of Medicine
Vijay Vallabh Hospital And Medical Research Centre
423, Tirupati Nagar, Phase 1, Virar (West), Dist. Palghar, Bolinj, Maharashtra 401303, India
A. Avhad
Department of Medicine Family Care Hospitals
P.K. Road Opposite Seven Square Academy, Mira Road (East), Thane, Maharashtra 401107, India
M. S. Khan
Department of Critical Care, Trauma and Emergency Medicine Rajendra Institute of Medical Sciences (RIMS)
Bariatu, Ranchi, Jharkhand 834009, India
A. Ghosh
Department of Community Medicine College of Medicine and JNM College Nadia, Kalyani, West Bengal 741235, India
S. S. Choudhary
Department of Pulmonary Medicine
Datta Meghe Medical College and Shalinitai Meghe Hospital and Research Centre, Off campus college of DMIMS deemed University Wanadongri, Hingna, Nagpur, Maharashtra 441110, India
DOI record:
{
"DOI": "10.1002/adtp.202200159",
"ISSN": [
"2366-3987",
"2366-3987"
],
"URL": "http://dx.doi.org/10.1002/adtp.202200159",
"alternative-id": [
"10.1002/adtp.202200159"
],
"assertion": [
{
"group": {
"label": "Publication History",
"name": "publication_history"
},
"label": "Received",
"name": "received",
"order": 0,
"value": "2022-07-22"
},
{
"group": {
"label": "Publication History",
"name": "publication_history"
},
"label": "Published",
"name": "published",
"order": 2,
"value": "2022-09-16"
}
],
"author": [
{
"affiliation": [
{
"name": "Faculty of Medicine and Department of Pharmacology Victoria Hospital, Bangalore Medical College and Research Institute KR Road Fort Bangalore Karnataka 560002 India"
}
],
"family": "C R",
"given": "Jayanthi",
"sequence": "first"
},
{
"ORCID": "http://orcid.org/0000-0001-9874-9869",
"affiliation": [
{
"name": "Medical Services Themis Medicare Ltd. 11/12, Udyog Nagar, S.V. Road, Goregaon (W) Mumbai Maharashtra 400104 India"
}
],
"authenticated-orcid": false,
"family": "Swain",
"given": "Ashok K",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Pulmonary Medicine All India Institute of Medical Sciences Gate No, 1, Great Eastern Rd, opposite Gurudwara, AIIMS Campus, Tatibandh Raipur Chhattisgarh 492099 India"
}
],
"family": "Ganga",
"given": "Ranganath T",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Medicine Vijay Vallabh Hospital And Medical Research Centre 423, Tirupati Nagar, Phase 1, Virar (West), Dist. Palghar Bolinj Maharashtra 401303 India"
}
],
"family": "Halnor",
"given": "Dnyaneshwar",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Medicine Family Care Hospitals P.K. Road Opposite Seven Square Academy, Mira Road (East) Thane Maharashtra 401107 India"
}
],
"family": "Avhad",
"given": "Ajit",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Critical Care, Trauma and Emergency Medicine Rajendra Institute of Medical Sciences (RIMS) Bariatu Ranchi Jharkhand 834009 India"
}
],
"family": "Khan",
"given": "Mohd. Saif",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Community Medicine College of Medicine and JNM College Nadia Kalyani West Bengal 741235 India"
}
],
"family": "Ghosh",
"given": "Ayan",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Pulmonary Medicine Datta Meghe Medical College and Shalinitai Meghe Hospital and Research Centre, Off campus college of DMIMS deemed University Wanadongri, Hingna Nagpur Maharashtra 441110 India"
}
],
"family": "Choudhary",
"given": "Sumer Sanjiv",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Pulmonary Medicine Sonali Memorial Hospitals Jai Hind Nagar, Thergaon Pune Maharashtra 411033 India"
}
],
"family": "Yannawar",
"given": "Anand Namdevrao",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Medicine GMERS Medical College and General Hospital Gotri Road, Gotri Vadodara Gujarat 390021 India"
}
],
"family": "Despande",
"given": "Shubhangi",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Medicine V.S. General Hospital and Sardar Vallabhbhai Patel Institute of Medical Sciences and Research Madalpur Gam, Paldi Ahmedabad Gujarat 380006 India"
}
],
"family": "Patel",
"given": "Manish",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Medicine Pranaam Hospital 1–56/6/40& 41, Mythri Nagar, Madeenaguda Hyderabad Telangana 500050 India"
}
],
"family": "Anne",
"given": "Krishna Prasad",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Medical Services Themis Medicare Ltd. 11/12, Udyog Nagar, S.V. Road, Goregaon (W) Mumbai Maharashtra 400104 India"
}
],
"family": "Bangar",
"given": "Yogesh",
"sequence": "additional"
}
],
"container-title": "Advanced Therapeutics",
"container-title-short": "Advanced Therapeutics",
"content-domain": {
"crossmark-restriction": true,
"domain": [
"onlinelibrary.wiley.com"
]
},
"created": {
"date-parts": [
[
2022,
9,
12
]
],
"date-time": "2022-09-12T07:19:37Z",
"timestamp": 1662967177000
},
"deposited": {
"date-parts": [
[
2022,
12,
15
]
],
"date-time": "2022-12-15T02:08:25Z",
"timestamp": 1671070105000
},
"indexed": {
"date-parts": [
[
2022,
12,
15
]
],
"date-time": "2022-12-15T06:02:22Z",
"timestamp": 1671084142711
},
"is-referenced-by-count": 0,
"issue": "12",
"issued": {
"date-parts": [
[
2022,
9,
16
]
]
},
"journal-issue": {
"issue": "12",
"published-print": {
"date-parts": [
[
2022,
12
]
]
}
},
"language": "en",
"license": [
{
"URL": "http://onlinelibrary.wiley.com/termsAndConditions#vor",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2022,
9,
16
]
],
"date-time": "2022-09-16T00:00:00Z",
"timestamp": 1663286400000
}
},
{
"URL": "http://doi.wiley.com/10.1002/tdm_license_1.1",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2022,
9,
16
]
],
"date-time": "2022-09-16T00:00:00Z",
"timestamp": 1663286400000
}
}
],
"link": [
{
"URL": "https://onlinelibrary.wiley.com/doi/pdf/10.1002/adtp.202200159",
"content-type": "application/pdf",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://onlinelibrary.wiley.com/doi/full-xml/10.1002/adtp.202200159",
"content-type": "application/xml",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://onlinelibrary.wiley.com/doi/pdf/10.1002/adtp.202200159",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "311",
"original-title": [],
"page": "2200159",
"prefix": "10.1002",
"published": {
"date-parts": [
[
2022,
9,
16
]
]
},
"published-online": {
"date-parts": [
[
2022,
9,
16
]
]
},
"published-print": {
"date-parts": [
[
2022,
12
]
]
},
"publisher": "Wiley",
"reference": [
{
"DOI": "10.1007/s12325-019-00995-6",
"doi-asserted-by": "publisher",
"key": "e_1_2_9_1_1"
},
{
"DOI": "10.3390/pathogens9121055",
"doi-asserted-by": "publisher",
"key": "e_1_2_9_2_1"
},
{
"key": "e_1_2_9_3_1",
"unstructured": "Central Drugs Standard Control Organization List of new drugs approved in the year 2022 till date https://cdsco.gov.in/opencms/resources/UploadCDSCOWeb/2018/UploadApprovalNewDrugs/aprilnew%20drugs%20approval%202022.pdf(accessed: April 2022)."
},
{
"DOI": "10.1186/s12879-016-1965-5",
"doi-asserted-by": "publisher",
"key": "e_1_2_9_4_1"
},
{
"DOI": "10.1016/j.intimp.2016.11.023",
"doi-asserted-by": "publisher",
"key": "e_1_2_9_5_1"
},
{
"DOI": "10.1002/eji.201847948",
"doi-asserted-by": "publisher",
"key": "e_1_2_9_6_1"
},
{
"key": "e_1_2_9_7_1",
"unstructured": "Clinical Trials Registry – India (CTRI) An Open‐Label Prospective Randomized Comparative Parallel Group Multi‐Center Proof of Concept Study to Assess the Efficacy and Safety of Inosine Pranobex Added to Current Standard of Care (CSC) in COVID‐19 Patients http://ctri.nic.in/Clinicaltrials/showallp.php?mid1=46240&EncHid=&userName=inosine%20pranobex(accessed: October 2021)."
},
{
"author": "Jing Y.",
"first-page": "bbaa234",
"journal-title": "Briefings Bioinf.",
"key": "e_1_2_9_8_1",
"year": "2020"
},
{
"key": "e_1_2_9_9_1",
"unstructured": "ASM.org COVID‐19‐Associated Mucormycosis: Triple Threat of the Pandemic https://asm.org/Articles/2021/July/COVID‐19‐Associated‐Mucormycosis‐Triple‐Threat‐of(accessed: September 2021)."
},
{
"DOI": "10.1016/j.micinf.2010.08.009",
"doi-asserted-by": "publisher",
"key": "e_1_2_9_10_1"
},
{
"key": "e_1_2_9_11_1",
"unstructured": "T.Thacker The Economic Times https://economictimes.indiatimes.com/news/india/hcq‐another‐drug‐dropped‐from‐covid‐treatment‐protocol/articleshow/86469107.cms(accessed: April 2022)."
},
{
"key": "e_1_2_9_12_1",
"unstructured": "Neosine tablets ‐ OTC‐ Aflofarm https://www.aflofarm.com.pl/en/products/otc/neosine‐tablets/(accessed: September 2021)."
},
{
"key": "e_1_2_9_13_1",
"unstructured": "Guidelines on Clinical Management of COVID – 19 Ministry of Health & Family Welfare Directorate General of Health Services (EMR Division) https://www.mohfw.gov.in/pdf/GuidelinesonClinicalManagementofCOVID1912020.pdf(accessed: October 2020)."
},
{
"key": "e_1_2_9_14_1",
"unstructured": "COVID‐19 Therapeutic Trial Synopsis World Health Organization R&D Blueprint https://www.who.int/blueprint/priority‐diseases/key‐action/COVID19_Treatment_Trial_Design_Master_Protocol_synopsis_Final_18022020.pdf(accessed: September 2021)."
},
{
"DOI": "10.1093/occmed/kqx086",
"doi-asserted-by": "publisher",
"key": "e_1_2_9_15_1"
},
{
"DOI": "10.1155/2014/171053",
"doi-asserted-by": "publisher",
"key": "e_1_2_9_16_1"
},
{
"key": "e_1_2_9_17_1",
"unstructured": "www.MeDitorial.cz Inosine pranobex in the prevention and treatment of acute respiratory viral infections including COVID‐19 Answers to Frequently Asked Questions from Healthcare Professionals https://www.prolekare.cz/tema/hpv‐condylomata‐ari/detail/inosine‐pranobex‐in‐the‐prevention‐and‐treatment‐of‐acute‐respiratory‐viral‐infections‐including‐covid‐19‐answers‐to‐frequently‐asked‐questions‐from‐healthcare‐professionals‐121758(accessed: August 2021)."
}
],
"reference-count": 17,
"references-count": 17,
"relation": {},
"resource": {
"primary": {
"URL": "https://onlinelibrary.wiley.com/doi/10.1002/adtp.202200159"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [
"Pharmacology (medical)",
"Biochemistry (medical)",
"Genetics (clinical)",
"Pharmaceutical Science",
"Pharmacology",
"Medicine (miscellaneous)"
],
"subtitle": [],
"title": "Efficacy and Safety of Inosine Pranobex in COVID‐19 Patients: A Multicenter Phase 3 Randomized Double‐Blind, Placebo‐Controlled Trial",
"type": "journal-article",
"update-policy": "http://dx.doi.org/10.1002/crossmark_policy",
"volume": "5"
}