Vitamin C may increase the recovery rate of outpatient cases of SARS-CoV-2 infection by 70%: reanalysis of the COVID A to Z Randomized Clinical Trial
et al., Research Square, doi:10.21203/rs.3.rs-289381/v1, Mar 2021
Vitamin C for COVID-19
6th treatment shown to reduce risk in
September 2020, now with p = 0.000000069 from 72 studies, recognized in 22 countries.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
Reanalysis of Thomas et al. showing that vitamin C increased the recovery rate by 70%, p = 0.025.
7 meta-analyses show significant improvements with vitamin C for mortality1-5,
progression6,
severity1,5, and
cases7.
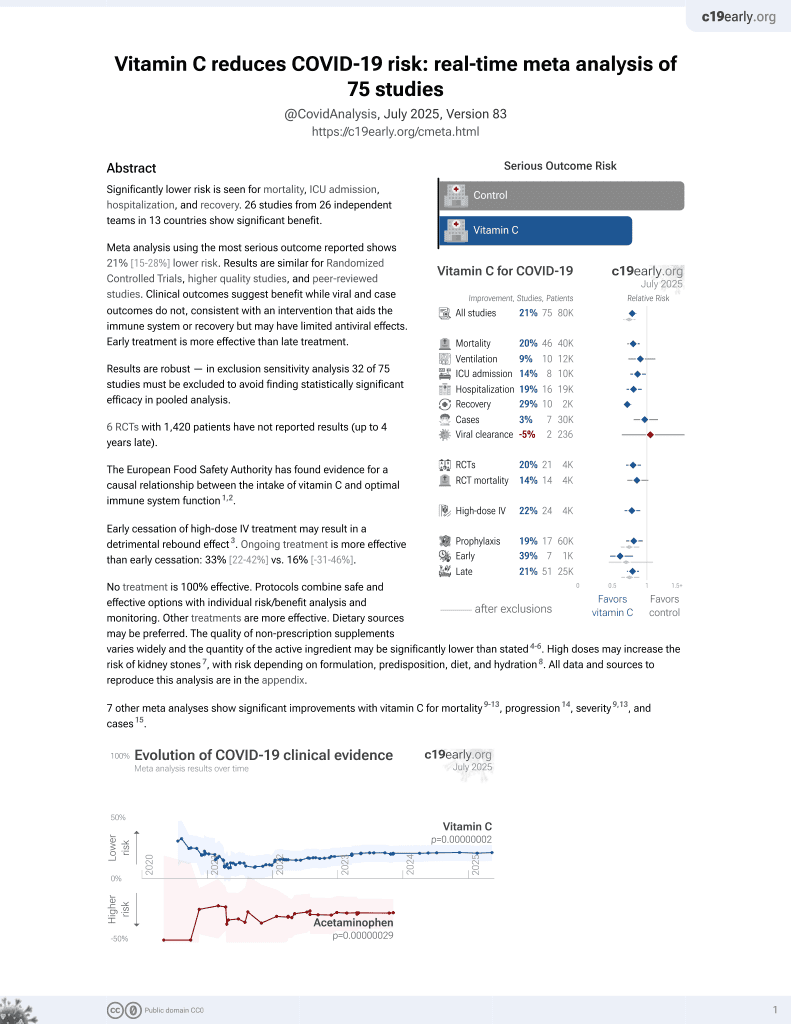
Currently there are 72 vitamin C for COVID-19 studies, showing 19% lower mortality [9‑27%], 9% lower ventilation [-12‑27%], 23% lower ICU admission [10‑34%], 19% lower hospitalization [7‑30%], and 3% fewer cases [-16‑19%].
1.
Bhowmik et al., Impact of high-dose vitamin C on the mortality, severity, and duration of hospital stay in COVID-19 patients: A meta-analysis, Health Science Reports, doi:10.1002/hsr2.762.
2.
Olczak-Pruc et al., Vitamin C Supplementation for the Treatment of COVID-19: A Systematic Review and Meta-Analysis, Nutrients, doi:10.3390/nu14194217.
3.
Kow et al., The effect of vitamin C on the risk of mortality in patients with COVID-19: a systematic review and meta-analysis of randomized controlled trials, Inflammopharmacology, doi:10.1007/s10787-023-01200-5.
4.
Kow (B) et al., Impact of uricosurics on mortality outcomes in patients with COVID-19: a systematic review and meta-analysis of randomized controlled trials, International Journal of Pharmacy Practice, doi:10.1093/ijpp/riae003.
5.
Qin et al., Effects of Vitamin C Supplements on Clinical Outcomes and Hospitalization Duration for Patients with Coronavirus Disease 2019 (COVID-19): A Systematic Review and Meta-Analysis, Nutrition Reviews, doi:10.1093/nutrit/nuae154.
Hemilä et al., 2 Mar 2021, preprint, 3 authors.
Vitamin C may increase the recovery rate of outpatient cases of SARS-CoV-2 infection by 70%: reanalysis of the COVID A to Z Randomized Clinical Trial
doi:10.21203/rs.3.rs-289381/v1
The COVID A to Z trial (JAMA Netw Open. 2021;4:e210369) is important as it focused speci cally on SARS-CoV-2 coronavirus patients and examined a high dose of vitamin C which was previously predicted to reduce the duration of respiratory virus infections by about 20%. Unfortunately, there are several limitations in the trial methods. The COVID A to Z trial was "stopped early for futility". In the sample size calculation, the authors assumed a 1.0 day shorter symptoms by intervention. Duration of symptoms was reduced by 1.2 days in the vitamin C arm compared with the usual care arm. Given that the observed vitamin C effect was 20% greater than the expected effect (1.2 vs. 1.0), it is illogical to have stopped the trial early because of "futility". In this reanalysis we calculated the rate ratio of recovery between the vitamin C and usual care arms and found that vitamin C increased the rate of recovery by 70% (95% CI 6.8% to 170%, P = 0.025). Furthermore, we calculated quantile treatment effect of vitamin C. At the 60th percentile level of symptom distribution, duration was 9 days in the usual care arm, and 6 days in the vitamin C arm, which corresponds to reduction in symptom duration by 3 days (95% CI 3 to 4.6 days; P < 0.001). The analysis of the quantile treatment effect indicates that there may be around 30% reduction in symptom duration in patients with the longest symptoms. Our reanalysis indicates a need for methodologically sound trials with larger numbers of patients to investigate the treatment effects of vitamin C against SARS-CoV-2.
Declarations Author Contributions HH originated the study, carried out the statistical analyses and wrote the draft. AC and EC participated in the revision of the manuscript.
Con ict of Interest The authors declare that the research was conducted in the absence of any commercial or nancial relationships that could be construed as a potential con ict of interes
Ethics approval Not applicable; this is a secondary analysis of a trial already published. the usual care group were censored, ie, did not recover by the end of the follow-up. Our calculations and the redrawn recovery curve are shown in the supplementary le.
Supplementary Files This is a list of supplementary les associated with this preprint. Click to download. COVIDSupplement31.pdf
References
Amrhein, Greenland, Mcshane, Scientists rise up against statistical signi cance, Nature, doi:10.1038/d41586-019-00857-9
Anderson, Suranyi, Beaton, The effect on winter illness of large doses of vitamin C, Can Med Assoc J
Carfì, Bernabei, Landi, Gemelli, Against COVID-19 Post-Acute Care Study Group. Persistent Symptoms in Patients After Acute COVID-19, JAMA, doi:10.1101/2020.12.24.20248802v2
Carr, Maggini, Vitamin C and immune function, Nutrients, doi:10.3390/nu9111211
Carr, Rowe, The emerging role of vitamin C in the prevention and treatment of COVID-19, Nutrients, doi:10.3390/nu12113286
Davis, Assaf, Mccorkell, Wei, Low et al., Characterizing Long COVID in an international cohort: 7 months of symptoms and their impact, MedRxiv, doi:10.1101/2020.12.24.20248802v2
Hemilä, Chalker, Vitamin C Can Shorten the Length of Stay in the ICU: A Meta-Analysis, Nutrients, doi:10.3390/nu11040708
Hemilä, Chalker, Vitamin C for preventing and treating the common cold, Cochrane Database Syst Rev, doi:10.1002/14651858.CD000980.pub4
Hemilä, Chalker, Vitamin C may reduce the duration of mechanical ventilation in critically ill patients: a meta-regression analysis, J Intensive Care, doi:10.1186/s40560-020-0432-y
Hemilä, De Man, Vitamin C and COVID-19, Front. Med, doi:10.3389/fmed.2020.559811
Hemilä, Duration of the common cold and similar continuous outcomes should be analyzed on the relative scale: a case study of two zinc lozenge trials, BMC Med Res Methodol, doi:10.1186/s12874-017-0356-y
Hemilä, Vitamin C and infections, Nutrients, doi:10.3390/nu9040339
Hemilä, Vitamin C may alleviate exercise-induced bronchoconstriction: a meta-analysis, BMJ Open, doi:10.1136/bmjopen-2012-002416
Hemilä, Vitamin C supplementation and common cold symptoms: factors affecting the magnitude of the bene t, Med Hypotheses, doi:10.1054/mehy.1997.0639
Hemilä, Vitamin, the placebo effect, and the common cold: a case study of how preconceptions in uence the analysis of results, comments in
Hong, Christiani, Li, Quantile regression for survival data in modern cancer research: expanding statistical tools for precision medicine, Precis Clin Med, doi:10.1093/pcmedi/pbz007
Karlowski, Chalmers, Frenkel, Kapikian, Lewis et al., Ascorbic acid for the common cold. A prophylactic and therapeutic trial, JAMA, doi:10.1001/jama.1975.03240220018013
Koenker, Quantile treatment effects, p
Koenker, quantreg: Quantile Regression
Markwell, Vitamin C in the prevention of colds, Med J Aust, doi:10.5694/j.1326-5377.1947.tb74386.x
Padayatty, Sun, Wang, Riordan, Hewitt et al., Vitamin C pharmacokinetics: implications for oral and intravenous use, Ann Intern Med, doi:10.7326/0003-4819-140-7-200404060-00010
Schmidt, Rothman, Mistaken inference caused by reliance on and misinterpretation of a signi cance test, Int J Cardiol, doi:10.1016/j.ijcard.2014.09.205
Thomas, Patel, Bittel, Wolski, Wang et al., Effect of High-Dose Zinc and Ascorbic Acid Supplementation vs Usual Care on Symptom Length and Reduction Among Ambulatory Patients With SARS-CoV-2 Infection: The COVID A to Z Randomized Clinical Trial, JAMA Netw Open, doi:10.1001/jamanetworkopen.2021.0369
Zhang, Rao, Li, Zhu, Liu et al., Pilot trial of high-dose vitamin C in critically ill COVID-19 patients, Ann Intensive Care, doi:10.1186/s13613-020-00792-3
DOI record:
{
"DOI": "10.21203/rs.3.rs-289381/v1",
"URL": "http://dx.doi.org/10.21203/rs.3.rs-289381/v1",
"abstract": "<jats:title>Abstract</jats:title>\n <jats:p>The COVID A to Z trial (JAMA Netw Open. 2021;4:e210369) is important as it focused specifically on SARS-CoV-2 coronavirus patients and examined a high dose of vitamin C which was previously predicted to reduce the duration of respiratory virus infections by about 20%. Unfortunately, there are several limitations in the trial methods. The COVID A to Z trial was “stopped early for futility”. In the sample size calculation, the authors assumed a 1.0 day shorter symptoms by intervention. Duration of symptoms was reduced by 1.2 days in the vitamin C arm compared with the usual care arm. Given that the observed vitamin C effect was 20% greater than the expected effect (1.2 vs. 1.0), it is illogical to have stopped the trial early because of “futility”. In this reanalysis we calculated the rate ratio of recovery between the vitamin C and usual care arms and found that vitamin C increased the rate of recovery by 70% (95% CI 6.8% to 170%, P = 0.025). Furthermore, we calculated quantile treatment effect of vitamin C. At the 60th percentile level of symptom distribution, duration was 9 days in the usual care arm, and 6 days in the vitamin C arm, which corresponds to reduction in symptom duration by 3 days (95% CI 3 to 4.6 days; P < 0.001). The analysis of the quantile treatment effect indicates that there may be around 30% reduction in symptom duration in patients with the longest symptoms. Our reanalysis indicates a need for methodologically sound trials with larger numbers of patients to investigate the treatment effects of vitamin C against SARS-CoV-2.</jats:p>",
"accepted": {
"date-parts": [
[
2021,
3,
1
]
]
},
"author": [
{
"ORCID": "http://orcid.org/0000-0002-4710-307X",
"affiliation": [
{
"name": "University of Helsinki, Helsinki, Finland"
}
],
"authenticated-orcid": false,
"family": "Hemilä",
"given": "Harri",
"sequence": "first"
},
{
"ORCID": "http://orcid.org/0000-0002-5890-2977",
"affiliation": [
{
"name": "University of Otago, Christchurch, New Zealand"
}
],
"authenticated-orcid": false,
"family": "Carr",
"given": "Anitra",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0003-1593-3770",
"affiliation": [
{
"name": "University of Sydney, Sydney, Australia"
}
],
"authenticated-orcid": false,
"family": "Chalker",
"given": "Elizabeth",
"sequence": "additional"
}
],
"container-title": [],
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2021,
3,
1
]
],
"date-time": "2021-03-01T23:27:31Z",
"timestamp": 1614641251000
},
"deposited": {
"date-parts": [
[
2022,
7,
29
]
],
"date-time": "2022-07-29T01:27:24Z",
"timestamp": 1659058044000
},
"group-title": "In Review",
"indexed": {
"date-parts": [
[
2024,
3,
3
]
],
"date-time": "2024-03-03T13:26:47Z",
"timestamp": 1709472407652
},
"institution": [
{
"name": "Research Square"
}
],
"is-referenced-by-count": 1,
"issued": {
"date-parts": [
[
2021,
3,
1
]
]
},
"license": [
{
"URL": "https://creativecommons.org/licenses/by/4.0/",
"content-version": "unspecified",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2021,
3,
1
]
],
"date-time": "2021-03-01T00:00:00Z",
"timestamp": 1614556800000
}
}
],
"link": [
{
"URL": "https://www.researchsquare.com/article/rs-289381/v1",
"content-type": "text/html",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://www.researchsquare.com/article/rs-289381/v1.html",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "8761",
"original-title": [],
"posted": {
"date-parts": [
[
2021,
3,
1
]
]
},
"prefix": "10.21203",
"published": {
"date-parts": [
[
2021,
3,
1
]
]
},
"publisher": "Research Square Platform LLC",
"reference-count": 0,
"references-count": 0,
"relation": {
"is-preprint-of": [
{
"asserted-by": "subject",
"id": "10.3389/fimmu.2021.674681",
"id-type": "doi"
}
]
},
"resource": {
"primary": {
"URL": "https://www.researchsquare.com/article/rs-289381/v1"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"subtype": "preprint",
"title": "Vitamin C may increase the recovery rate of outpatient cases of SARS-CoV-2 infection by 70%: reanalysis of the COVID A to Z Randomized Clinical Trial",
"type": "posted-content"
}