Early Remdesivir to Prevent Progression to Severe Covid-19 in Outpatients
et al., New England Journal of Medicine, doi:10.1056/NEJMoa2116846, PINETREE, NCT04501952, Dec 2021
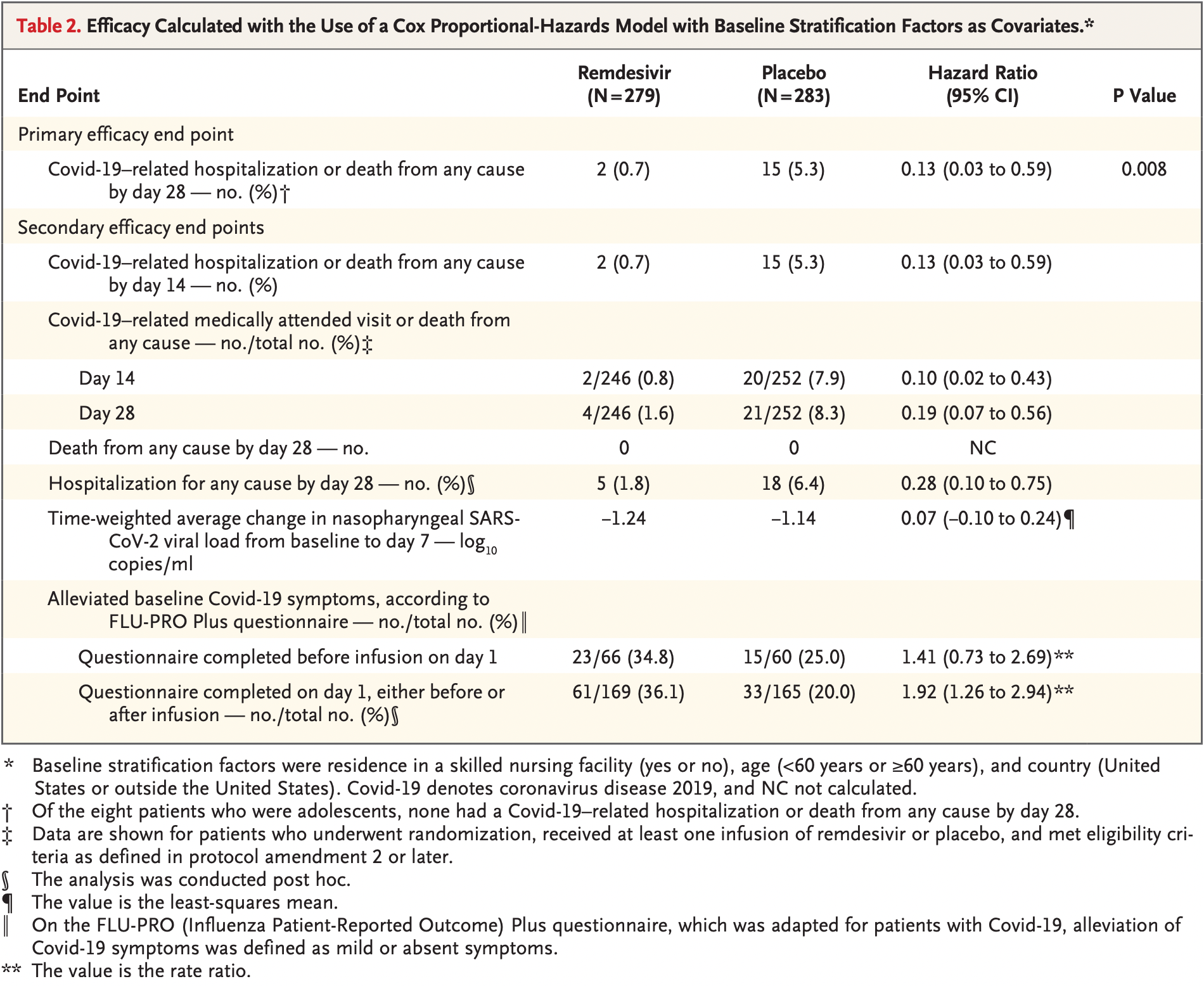
RCT high-risk outpatients, 279 treated with remdesivir and 283 control patients, median 5 days from symptoms, showing significantly lower hospitalization with treatment.
Gérard, Zhou, Wu, Kamo, Choi, Kim show increased risk of acute kidney injury, Leo, Briciu, Muntean, Petrov, Arch show increased risk of liver injury, Negru, Cheng, Mohammed, Kwok, Zhu show increased risk of cardiac disorders, and Kwok, Merches, Akinci, Tang, Bagheri show increased risk of mitochondrial toxicity with remdesivir.
|
risk of death/hospitalization, 87.0% lower, RR 0.13, p = 0.008, treatment 2 of 279 (0.7%), control 15 of 283 (5.3%), NNT 22, adjusted per study, COVID-19 related hospitalization or death from any cause @day 28, primary outcome.
|
|
risk of hospitalization, 71.8% lower, RR 0.28, p = 0.009, treatment 5 of 279 (1.8%), control 18 of 283 (6.4%), NNT 22.
|
|
risk of no recovery, 29.1% lower, RR 0.71, p = 0.31, treatment 43 of 66 (65.2%), control 45 of 60 (75.0%), adjusted per study, inverted to make RR<1 favor treatment, alleviation of symptoms @day 14.
|
|
risk of no recovery, 47.9% lower, RR 0.52, p = 0.003, treatment 108 of 169 (63.9%), control 132 of 165 (80.0%), NNT 6.2, adjusted per study, inverted to make RR<1 favor treatment, post-hoc alleviation of symptoms @day 14.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
1.
Gérard et al., Remdesivir and Acute Renal Failure: A Potential Safety Signal From Disproportionality Analysis of the WHO Safety Database, Clinical Pharmacology & Therapeutics, doi:10.1002/cpt.2145.
2.
Zhou et al., Acute Kidney Injury and Drugs Prescribed for COVID-19 in Diabetes Patients: A Real-World Disproportionality Analysis, Frontiers in Pharmacology, doi:10.3389/fphar.2022.833679.
3.
Wu et al., Acute Kidney Injury Associated With Remdesivir: A Comprehensive Pharmacovigilance Analysis of COVID-19 Reports in FAERS, Frontiers in Pharmacology, doi:10.3389/fphar.2022.692828.
4.
Kamo et al., Association of Antiviral Drugs for the Treatment of COVID-19 With Acute Renal Failure, In Vivo, doi:10.21873/invivo.13637.
5.
Choi et al., Comparative effectiveness of combination therapy with nirmatrelvir–ritonavir and remdesivir versus monotherapy with remdesivir or nirmatrelvir–ritonavir in patients hospitalised with COVID-19: a target trial emulation study, The Lancet Infectious Diseases, doi:10.1016/S1473-3099(24)00353-0.
6.
Kim et al., Investigating the Safety Profile of Fast‐Track COVID‐19 Drugs Using the FDA Adverse Event Reporting System Database: A Comparative Observational Study, Pharmacoepidemiology and Drug Safety, doi:10.1002/pds.70043.
7.
Leo et al., Hepatocellular liver injury in hospitalized patients affected by COVID-19: Presence of different risk factors at different time points, Digestive and Liver Disease, doi:10.1016/j.dld.2021.12.014.
8.
Briciu et al., Evolving Clinical Manifestations and Outcomes in COVID-19 Patients: A Comparative Analysis of SARS-CoV-2 Variant Waves in a Romanian Hospital Setting, Pathogens, doi:10.3390/pathogens12121453.
9.
Muntean et al., Effects of COVID-19 on the Liver and Mortality in Patients with SARS-CoV-2 Pneumonia Caused by Delta and Non-Delta Variants: An Analysis in a Single Centre, Pharmaceuticals, doi:10.3390/ph17010003.
10.
Petrov et al., The Effect of Potentially Hepatotoxic Medicinal Products on Alanine Transaminase Levels in COVID-19 Patients: A Case–Control Study, Safety and Risk of Pharmacotherapy, doi:10.30895/2312-7821-2025-458.
11.
Arch et al., Evaluation of the effectiveness of remdesivir in treating severe COVID-19 using data from the ISARIC WHO Clinical Characterisation Protocol UK: a prospective, national cohort study, medRxiv, doi:10.1101/2021.06.18.21259072.
12.
Negru et al., Comparative Pharmacovigilance Analysis of Approved and Repurposed Antivirals for COVID-19: Insights from EudraVigilance Data, Biomedicines, doi:10.3390/biomedicines13061387.
13.
Cheng et al., Cardiovascular Safety of COVID-19 Treatments: A Disproportionality Analysis of Adverse Event Reports from the WHO VigiBase, Infectious Diseases and Therapy, doi:10.1007/s40121-025-01225-z.
14.
Mohammed et al., Bradycardia associated with remdesivir treatment in coronavirus disease 2019 patients: A propensity score-matched analysis, Medicine, doi:10.1097/MD.0000000000044501.
15.
Kwok et al., Remdesivir induces persistent mitochondrial and structural damage in human induced pluripotent stem cell-derived cardiomyocytes, Cardiovascular Research, doi:10.1093/cvr/cvab311.
16.
Zhu et al., Cardiovascular Risks of COVID-19 Therapeutics: Integrated Analysis of FAERS, Electronic Health Records, and Transcriptomics, Pharmaceuticals, doi:10.3390/ph19040574.
17.
Merches et al., The potential of remdesivir to affect function, metabolism and proliferation of cardiac and kidney cells in vitro, Archives of Toxicology, doi:10.1007/s00204-022-03306-1.
18.
Akinci et al., Elucidation of remdesivir cytotoxicity pathways through genome-wide CRISPR-Cas9 screening and transcriptomics, bioRxiv, doi:10.1101/2020.08.27.270819.
Gottlieb et al., 22 Dec 2021, Double Blind Randomized Controlled Trial, multiple countries, peer-reviewed, 30 authors, study period 18 September, 2020 - 8 April, 2021, average treatment delay 5.0 days, trial NCT04501952 (history) (PINETREE).
Early Remdesivir to Prevent Progression to Severe Covid-19 in Outpatients
New England Journal of Medicine, doi:10.1056/nejmoa2116846
BACKGROUND Remdesivir improves clinical outcomes in patients hospitalized with moderate-tosevere coronavirus disease 2019 (Covid-19). Whether the use of remdesivir in symptomatic, nonhospitalized patients with Covid-19 who are at high risk for disease progression prevents hospitalization is uncertain.
METHODS We conducted a randomized, double-blind, placebo-controlled trial involving nonhospitalized patients with Covid-19 who had symptom onset within the previous 7 days and who had at least one risk factor for disease progression (age ≥60 years, obesity, or certain coexisting medical conditions). Patients were randomly assigned to receive intravenous remdesivir (200 mg on day 1 and 100 mg on days 2 and 3) or placebo. The primary efficacy end point was a composite of Covid-19related hospitalization or death from any cause by day 28. The primary safety end point was any adverse event. A secondary end point was a composite of a Covid-19related medically attended visit or death from any cause by day 28.
RESULTS A total of 562 patients who underwent randomization and received at least one dose of remdesivir or placebo were included in the analyses: 279 patients in the remdesivir group and 283 in the placebo group. The mean age was 50 years, 47.9% of the patients were women, and 41.8% were Hispanic or Latinx. The most common coexisting conditions were diabetes mellitus (61.6%), obesity (55.2%), and hypertension (47.7%). Covid-19-related hospitalization or death from any cause occurred in 2 patients (0.7%) in the remdesivir group and in 15 (5.3%) in the placebo group (hazard ratio, 0.13; 95% confidence interval [CI], 0.03 to 0.59; P = 0.008). A total of 4 of 246 patients (1.6%) in the remdesivir group and 21 of 252 (8.3%) in the placebo group had a Covid-19-related medically attended visit by day 28 (hazard ratio, 0.19; 95% CI, 0.07 to 0.56). No patients had died by day 28. Adverse events occurred in 42.3% of the patients in the remdesivir group and in 46.3% of those in the placebo group.
CONCLUSIONS Among nonhospitalized patients who were at high risk for Covid-19 progression, a 3-day course of remdesivir had an acceptable safety profile and resulted in an 87% lower risk of hospitalization or death than placebo. (Funded by Gilead Sciences; PINETREE ClinicalTrials.gov number, NCT04501952; EudraCT number, 2020 -003510 -12.
Appendix The authors' full names and academic degrees are as follows: Robert L. Gottlieb
References
Ader, Bouscambert-Duchamp, Hites, Remdesivir plus standard of care versus standard of care alone for the treatment of patients admitted to hos-The New England Journal of Medicine nloaded from
Beigel, Tomashek, Dodd, Remdesivir for the treatment of Covid-19 -final report, N Engl J Med
Cevik, Kuppalli, Kindrachuk, Peiris, Virology, transmission, and pathogenesis of SARS-CoV-2, BMJ
Chen, Winkler, Case, In vivo monoclonal antibody efficacy against SARS-CoV-2 variant strains, Nature
Dougan, Nirula, Azizad, Bamlanivimab plus etesevimab in mild or moderate Covid-19, N Engl J Med
Fintzi, Bonnett, Sweeney, Deconstructing the treatment effect of remdesivir in the Adaptive COVID-19
Gallo Marin, Aghagoli, Lavine, Predictors of COVID-19 severity: a literature review, Rev Med Virol
Goldman, Lye, Hui, Remdesivir for 5 or 10 days in patients with severe Covid-19, N Engl J Med
Gottlieb, Nirula, Chen, Effect of bamlanivimab as monotherapy or in combination with etesevimab on viral load in patients with mild to moderate COVID-19: a randomized clinical trial, JAMA
Gupta, Gonzalez-Rojas, Juarez, Early treatment for Covid-19 with SARS-CoV-2 neutralizing antibody sotrovimab, N Engl J Med
Huang, Wang, Li, Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China, Lancet
Mozaffari, Chandak, Zhang, Remdesivir treatment in hospitalized patients with COVID-19: a comparative analysis of in-hospital all-cause mortality in a large multi-center observational cohort, Clin Infect Dis
Mulangu, Dodd, Rt, A randomized, controlled trial of Ebola virus disease therapeutics, N Engl J Med
Muthuri, Venkatesan, Myles, Effectiveness of neuraminidase inhibitors in reducing mortality in patients admitted to hospital with influenza A H1N1pdm09 virus infection: a meta-analysis of individual participant data, Lancet Respir Med
Nicholson, Aoki, Osterhaus, Efficacy and safety of oseltamivir in treatment of acute influenza: a randomised controlled trial, Lancet
Pitts, Lu, Pont, Remdesivir retains potent antiviral activity against the SARS-CoV-2 delta variant and other variants of concern
Pizzorno, Padey, Julien, Characterization and treatment of SARS-CoV-2 in nasal and bronchial human airway epithelia, Cell Rep Med
Schäfer, Martinez, Won, Therapeutic efficacy of an oral nucleoside analog of remdesivir against SARS-CoV-2 pathogenesis in mice, doi:10.1101/2021.09.13.460111v3
Sheahan, Sims, Graham, Broad-spectrum antiviral GS-5734 inhibits both epidemic and zoonotic coronaviruses, Sci Transl Med
Siddiqi, Mehra, COVID-19 illness in native and immunosuppressed states: a clinical-therapeutic staging proposal, J Heart Lung Transplant
Spinner, Gottlieb, Criner, Effect of remdesivir vs standard care on clinical status at 11 days in patients with moderate COVID-19: a randomized clinical trial, JAMA
Stokes, Zambrano, Anderson, Coronavirus disease 2019 case surveillance -United States, January 22, MMWR Morb Mortal Wkly Rep
The, Group, Initiation of antiretroviral therapy in early asymptomatic HIV infection, N Engl J Med
Wang, Hu, Hu, Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China, JAMA
Weinreich, Sivapalasingam, Norton, REGN-COV2, a neutralizing antibody cocktail, in outpatients with Covid-19, N Engl J Med
Williamson, Feldmann, Schwarz, Clinical benefit of remdesivir in rhesus macaques infected with SARS-CoV-2, Nature
DOI record:
{
"DOI": "10.1056/nejmoa2116846",
"ISSN": [
"0028-4793",
"1533-4406"
],
"URL": "http://dx.doi.org/10.1056/nejmoa2116846",
"alternative-id": [
"10.1056/NEJMoa2116846"
],
"author": [
{
"ORCID": "http://orcid.org/0000-0001-8376-8709",
"affiliation": [
{
"name": "From Baylor University Medical Center and Baylor Scott and White Research Institute, Dallas (R.L.G.), and Care United Research, Forney (S.M.) — all in Texas; the Nuren Medical and Research Center, Miami (C.E.V.), Evolution Clinical Trials, Hialeah Gardens (G.P.), the Midland Florida Clinical Research Center, DeLand (G.O.), Luminous Clinical Research–South Florida Urgent Care, Pembroke Pines (A.H.), and Triple O Research Institute Professional Association, West Palm Beach (O.O.) — all in Florida; Hospital..."
}
],
"authenticated-orcid": false,
"family": "Gottlieb",
"given": "Robert L.",
"sequence": "first"
},
{
"affiliation": [
{
"name": "From Baylor University Medical Center and Baylor Scott and White Research Institute, Dallas (R.L.G.), and Care United Research, Forney (S.M.) — all in Texas; the Nuren Medical and Research Center, Miami (C.E.V.), Evolution Clinical Trials, Hialeah Gardens (G.P.), the Midland Florida Clinical Research Center, DeLand (G.O.), Luminous Clinical Research–South Florida Urgent Care, Pembroke Pines (A.H.), and Triple O Research Institute Professional Association, West Palm Beach (O.O.) — all in Florida; Hospital..."
}
],
"family": "Vaca",
"given": "Carlos E.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Baylor University Medical Center and Baylor Scott and White Research Institute, Dallas (R.L.G.), and Care United Research, Forney (S.M.) — all in Texas; the Nuren Medical and Research Center, Miami (C.E.V.), Evolution Clinical Trials, Hialeah Gardens (G.P.), the Midland Florida Clinical Research Center, DeLand (G.O.), Luminous Clinical Research–South Florida Urgent Care, Pembroke Pines (A.H.), and Triple O Research Institute Professional Association, West Palm Beach (O.O.) — all in Florida; Hospital..."
}
],
"family": "Paredes",
"given": "Roger",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Baylor University Medical Center and Baylor Scott and White Research Institute, Dallas (R.L.G.), and Care United Research, Forney (S.M.) — all in Texas; the Nuren Medical and Research Center, Miami (C.E.V.), Evolution Clinical Trials, Hialeah Gardens (G.P.), the Midland Florida Clinical Research Center, DeLand (G.O.), Luminous Clinical Research–South Florida Urgent Care, Pembroke Pines (A.H.), and Triple O Research Institute Professional Association, West Palm Beach (O.O.) — all in Florida; Hospital..."
}
],
"family": "Mera",
"given": "Jorge",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Baylor University Medical Center and Baylor Scott and White Research Institute, Dallas (R.L.G.), and Care United Research, Forney (S.M.) — all in Texas; the Nuren Medical and Research Center, Miami (C.E.V.), Evolution Clinical Trials, Hialeah Gardens (G.P.), the Midland Florida Clinical Research Center, DeLand (G.O.), Luminous Clinical Research–South Florida Urgent Care, Pembroke Pines (A.H.), and Triple O Research Institute Professional Association, West Palm Beach (O.O.) — all in Florida; Hospital..."
}
],
"family": "Webb",
"given": "Brandon J.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Baylor University Medical Center and Baylor Scott and White Research Institute, Dallas (R.L.G.), and Care United Research, Forney (S.M.) — all in Texas; the Nuren Medical and Research Center, Miami (C.E.V.), Evolution Clinical Trials, Hialeah Gardens (G.P.), the Midland Florida Clinical Research Center, DeLand (G.O.), Luminous Clinical Research–South Florida Urgent Care, Pembroke Pines (A.H.), and Triple O Research Institute Professional Association, West Palm Beach (O.O.) — all in Florida; Hospital..."
}
],
"family": "Perez",
"given": "Gilberto",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Baylor University Medical Center and Baylor Scott and White Research Institute, Dallas (R.L.G.), and Care United Research, Forney (S.M.) — all in Texas; the Nuren Medical and Research Center, Miami (C.E.V.), Evolution Clinical Trials, Hialeah Gardens (G.P.), the Midland Florida Clinical Research Center, DeLand (G.O.), Luminous Clinical Research–South Florida Urgent Care, Pembroke Pines (A.H.), and Triple O Research Institute Professional Association, West Palm Beach (O.O.) — all in Florida; Hospital..."
}
],
"family": "Oguchi",
"given": "Godson",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Baylor University Medical Center and Baylor Scott and White Research Institute, Dallas (R.L.G.), and Care United Research, Forney (S.M.) — all in Texas; the Nuren Medical and Research Center, Miami (C.E.V.), Evolution Clinical Trials, Hialeah Gardens (G.P.), the Midland Florida Clinical Research Center, DeLand (G.O.), Luminous Clinical Research–South Florida Urgent Care, Pembroke Pines (A.H.), and Triple O Research Institute Professional Association, West Palm Beach (O.O.) — all in Florida; Hospital..."
}
],
"family": "Ryan",
"given": "Pablo",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Baylor University Medical Center and Baylor Scott and White Research Institute, Dallas (R.L.G.), and Care United Research, Forney (S.M.) — all in Texas; the Nuren Medical and Research Center, Miami (C.E.V.), Evolution Clinical Trials, Hialeah Gardens (G.P.), the Midland Florida Clinical Research Center, DeLand (G.O.), Luminous Clinical Research–South Florida Urgent Care, Pembroke Pines (A.H.), and Triple O Research Institute Professional Association, West Palm Beach (O.O.) — all in Florida; Hospital..."
}
],
"family": "Nielsen",
"given": "Bibi U.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Baylor University Medical Center and Baylor Scott and White Research Institute, Dallas (R.L.G.), and Care United Research, Forney (S.M.) — all in Texas; the Nuren Medical and Research Center, Miami (C.E.V.), Evolution Clinical Trials, Hialeah Gardens (G.P.), the Midland Florida Clinical Research Center, DeLand (G.O.), Luminous Clinical Research–South Florida Urgent Care, Pembroke Pines (A.H.), and Triple O Research Institute Professional Association, West Palm Beach (O.O.) — all in Florida; Hospital..."
}
],
"family": "Brown",
"given": "Michael",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Baylor University Medical Center and Baylor Scott and White Research Institute, Dallas (R.L.G.), and Care United Research, Forney (S.M.) — all in Texas; the Nuren Medical and Research Center, Miami (C.E.V.), Evolution Clinical Trials, Hialeah Gardens (G.P.), the Midland Florida Clinical Research Center, DeLand (G.O.), Luminous Clinical Research–South Florida Urgent Care, Pembroke Pines (A.H.), and Triple O Research Institute Professional Association, West Palm Beach (O.O.) — all in Florida; Hospital..."
}
],
"family": "Hidalgo",
"given": "Ausberto",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Baylor University Medical Center and Baylor Scott and White Research Institute, Dallas (R.L.G.), and Care United Research, Forney (S.M.) — all in Texas; the Nuren Medical and Research Center, Miami (C.E.V.), Evolution Clinical Trials, Hialeah Gardens (G.P.), the Midland Florida Clinical Research Center, DeLand (G.O.), Luminous Clinical Research–South Florida Urgent Care, Pembroke Pines (A.H.), and Triple O Research Institute Professional Association, West Palm Beach (O.O.) — all in Florida; Hospital..."
}
],
"family": "Sachdeva",
"given": "Yessica",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Baylor University Medical Center and Baylor Scott and White Research Institute, Dallas (R.L.G.), and Care United Research, Forney (S.M.) — all in Texas; the Nuren Medical and Research Center, Miami (C.E.V.), Evolution Clinical Trials, Hialeah Gardens (G.P.), the Midland Florida Clinical Research Center, DeLand (G.O.), Luminous Clinical Research–South Florida Urgent Care, Pembroke Pines (A.H.), and Triple O Research Institute Professional Association, West Palm Beach (O.O.) — all in Florida; Hospital..."
}
],
"family": "Mittal",
"given": "Shilpi",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Baylor University Medical Center and Baylor Scott and White Research Institute, Dallas (R.L.G.), and Care United Research, Forney (S.M.) — all in Texas; the Nuren Medical and Research Center, Miami (C.E.V.), Evolution Clinical Trials, Hialeah Gardens (G.P.), the Midland Florida Clinical Research Center, DeLand (G.O.), Luminous Clinical Research–South Florida Urgent Care, Pembroke Pines (A.H.), and Triple O Research Institute Professional Association, West Palm Beach (O.O.) — all in Florida; Hospital..."
}
],
"family": "Osiyemi",
"given": "Olayemi",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Baylor University Medical Center and Baylor Scott and White Research Institute, Dallas (R.L.G.), and Care United Research, Forney (S.M.) — all in Texas; the Nuren Medical and Research Center, Miami (C.E.V.), Evolution Clinical Trials, Hialeah Gardens (G.P.), the Midland Florida Clinical Research Center, DeLand (G.O.), Luminous Clinical Research–South Florida Urgent Care, Pembroke Pines (A.H.), and Triple O Research Institute Professional Association, West Palm Beach (O.O.) — all in Florida; Hospital..."
}
],
"family": "Skarbinski",
"given": "Jacek",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Baylor University Medical Center and Baylor Scott and White Research Institute, Dallas (R.L.G.), and Care United Research, Forney (S.M.) — all in Texas; the Nuren Medical and Research Center, Miami (C.E.V.), Evolution Clinical Trials, Hialeah Gardens (G.P.), the Midland Florida Clinical Research Center, DeLand (G.O.), Luminous Clinical Research–South Florida Urgent Care, Pembroke Pines (A.H.), and Triple O Research Institute Professional Association, West Palm Beach (O.O.) — all in Florida; Hospital..."
}
],
"family": "Juneja",
"given": "Kavita",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Baylor University Medical Center and Baylor Scott and White Research Institute, Dallas (R.L.G.), and Care United Research, Forney (S.M.) — all in Texas; the Nuren Medical and Research Center, Miami (C.E.V.), Evolution Clinical Trials, Hialeah Gardens (G.P.), the Midland Florida Clinical Research Center, DeLand (G.O.), Luminous Clinical Research–South Florida Urgent Care, Pembroke Pines (A.H.), and Triple O Research Institute Professional Association, West Palm Beach (O.O.) — all in Florida; Hospital..."
}
],
"family": "Hyland",
"given": "Robert H.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Baylor University Medical Center and Baylor Scott and White Research Institute, Dallas (R.L.G.), and Care United Research, Forney (S.M.) — all in Texas; the Nuren Medical and Research Center, Miami (C.E.V.), Evolution Clinical Trials, Hialeah Gardens (G.P.), the Midland Florida Clinical Research Center, DeLand (G.O.), Luminous Clinical Research–South Florida Urgent Care, Pembroke Pines (A.H.), and Triple O Research Institute Professional Association, West Palm Beach (O.O.) — all in Florida; Hospital..."
}
],
"family": "Osinusi",
"given": "Anu",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Baylor University Medical Center and Baylor Scott and White Research Institute, Dallas (R.L.G.), and Care United Research, Forney (S.M.) — all in Texas; the Nuren Medical and Research Center, Miami (C.E.V.), Evolution Clinical Trials, Hialeah Gardens (G.P.), the Midland Florida Clinical Research Center, DeLand (G.O.), Luminous Clinical Research–South Florida Urgent Care, Pembroke Pines (A.H.), and Triple O Research Institute Professional Association, West Palm Beach (O.O.) — all in Florida; Hospital..."
}
],
"family": "Chen",
"given": "Shuguang",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Baylor University Medical Center and Baylor Scott and White Research Institute, Dallas (R.L.G.), and Care United Research, Forney (S.M.) — all in Texas; the Nuren Medical and Research Center, Miami (C.E.V.), Evolution Clinical Trials, Hialeah Gardens (G.P.), the Midland Florida Clinical Research Center, DeLand (G.O.), Luminous Clinical Research–South Florida Urgent Care, Pembroke Pines (A.H.), and Triple O Research Institute Professional Association, West Palm Beach (O.O.) — all in Florida; Hospital..."
}
],
"family": "Camus",
"given": "Gregory",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Baylor University Medical Center and Baylor Scott and White Research Institute, Dallas (R.L.G.), and Care United Research, Forney (S.M.) — all in Texas; the Nuren Medical and Research Center, Miami (C.E.V.), Evolution Clinical Trials, Hialeah Gardens (G.P.), the Midland Florida Clinical Research Center, DeLand (G.O.), Luminous Clinical Research–South Florida Urgent Care, Pembroke Pines (A.H.), and Triple O Research Institute Professional Association, West Palm Beach (O.O.) — all in Florida; Hospital..."
}
],
"family": "Abdelghany",
"given": "Mazin",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Baylor University Medical Center and Baylor Scott and White Research Institute, Dallas (R.L.G.), and Care United Research, Forney (S.M.) — all in Texas; the Nuren Medical and Research Center, Miami (C.E.V.), Evolution Clinical Trials, Hialeah Gardens (G.P.), the Midland Florida Clinical Research Center, DeLand (G.O.), Luminous Clinical Research–South Florida Urgent Care, Pembroke Pines (A.H.), and Triple O Research Institute Professional Association, West Palm Beach (O.O.) — all in Florida; Hospital..."
}
],
"family": "Davies",
"given": "Santosh",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Baylor University Medical Center and Baylor Scott and White Research Institute, Dallas (R.L.G.), and Care United Research, Forney (S.M.) — all in Texas; the Nuren Medical and Research Center, Miami (C.E.V.), Evolution Clinical Trials, Hialeah Gardens (G.P.), the Midland Florida Clinical Research Center, DeLand (G.O.), Luminous Clinical Research–South Florida Urgent Care, Pembroke Pines (A.H.), and Triple O Research Institute Professional Association, West Palm Beach (O.O.) — all in Florida; Hospital..."
}
],
"family": "Behenna-Renton",
"given": "Nicole",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Baylor University Medical Center and Baylor Scott and White Research Institute, Dallas (R.L.G.), and Care United Research, Forney (S.M.) — all in Texas; the Nuren Medical and Research Center, Miami (C.E.V.), Evolution Clinical Trials, Hialeah Gardens (G.P.), the Midland Florida Clinical Research Center, DeLand (G.O.), Luminous Clinical Research–South Florida Urgent Care, Pembroke Pines (A.H.), and Triple O Research Institute Professional Association, West Palm Beach (O.O.) — all in Florida; Hospital..."
}
],
"family": "Duff",
"given": "Frank",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-3708-8734",
"affiliation": [
{
"name": "From Baylor University Medical Center and Baylor Scott and White Research Institute, Dallas (R.L.G.), and Care United Research, Forney (S.M.) — all in Texas; the Nuren Medical and Research Center, Miami (C.E.V.), Evolution Clinical Trials, Hialeah Gardens (G.P.), the Midland Florida Clinical Research Center, DeLand (G.O.), Luminous Clinical Research–South Florida Urgent Care, Pembroke Pines (A.H.), and Triple O Research Institute Professional Association, West Palm Beach (O.O.) — all in Florida; Hospital..."
}
],
"authenticated-orcid": false,
"family": "Marty",
"given": "Francisco M.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Baylor University Medical Center and Baylor Scott and White Research Institute, Dallas (R.L.G.), and Care United Research, Forney (S.M.) — all in Texas; the Nuren Medical and Research Center, Miami (C.E.V.), Evolution Clinical Trials, Hialeah Gardens (G.P.), the Midland Florida Clinical Research Center, DeLand (G.O.), Luminous Clinical Research–South Florida Urgent Care, Pembroke Pines (A.H.), and Triple O Research Institute Professional Association, West Palm Beach (O.O.) — all in Florida; Hospital..."
}
],
"family": "Katz",
"given": "Morgan J.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Baylor University Medical Center and Baylor Scott and White Research Institute, Dallas (R.L.G.), and Care United Research, Forney (S.M.) — all in Texas; the Nuren Medical and Research Center, Miami (C.E.V.), Evolution Clinical Trials, Hialeah Gardens (G.P.), the Midland Florida Clinical Research Center, DeLand (G.O.), Luminous Clinical Research–South Florida Urgent Care, Pembroke Pines (A.H.), and Triple O Research Institute Professional Association, West Palm Beach (O.O.) — all in Florida; Hospital..."
}
],
"family": "Ginde",
"given": "Adit A.",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0003-1206-6261",
"affiliation": [
{
"name": "From Baylor University Medical Center and Baylor Scott and White Research Institute, Dallas (R.L.G.), and Care United Research, Forney (S.M.) — all in Texas; the Nuren Medical and Research Center, Miami (C.E.V.), Evolution Clinical Trials, Hialeah Gardens (G.P.), the Midland Florida Clinical Research Center, DeLand (G.O.), Luminous Clinical Research–South Florida Urgent Care, Pembroke Pines (A.H.), and Triple O Research Institute Professional Association, West Palm Beach (O.O.) — all in Florida; Hospital..."
}
],
"authenticated-orcid": false,
"family": "Brown",
"given": "Samuel M.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Baylor University Medical Center and Baylor Scott and White Research Institute, Dallas (R.L.G.), and Care United Research, Forney (S.M.) — all in Texas; the Nuren Medical and Research Center, Miami (C.E.V.), Evolution Clinical Trials, Hialeah Gardens (G.P.), the Midland Florida Clinical Research Center, DeLand (G.O.), Luminous Clinical Research–South Florida Urgent Care, Pembroke Pines (A.H.), and Triple O Research Institute Professional Association, West Palm Beach (O.O.) — all in Florida; Hospital..."
}
],
"family": "Schiffer",
"given": "Joshua T.",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-7665-7100",
"affiliation": [
{
"name": "From Baylor University Medical Center and Baylor Scott and White Research Institute, Dallas (R.L.G.), and Care United Research, Forney (S.M.) — all in Texas; the Nuren Medical and Research Center, Miami (C.E.V.), Evolution Clinical Trials, Hialeah Gardens (G.P.), the Midland Florida Clinical Research Center, DeLand (G.O.), Luminous Clinical Research–South Florida Urgent Care, Pembroke Pines (A.H.), and Triple O Research Institute Professional Association, West Palm Beach (O.O.) — all in Florida; Hospital..."
}
],
"authenticated-orcid": false,
"family": "Hill",
"given": "Joshua A.",
"sequence": "additional"
}
],
"container-title": [
"New England Journal of Medicine"
],
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2021,
12,
22
]
],
"date-time": "2021-12-22T22:01:34Z",
"timestamp": 1640210494000
},
"deposited": {
"date-parts": [
[
2021,
12,
22
]
],
"date-time": "2021-12-22T22:01:38Z",
"timestamp": 1640210498000
},
"funder": [
{
"DOI": "10.13039/100005564",
"award": [
"N/A"
],
"doi-asserted-by": "publisher",
"name": "Gilead Sciences"
}
],
"indexed": {
"date-parts": [
[
2021,
12,
23
]
],
"date-time": "2021-12-23T05:42:10Z",
"timestamp": 1640238130808
},
"is-referenced-by-count": 0,
"issn-type": [
{
"type": "print",
"value": "0028-4793"
},
{
"type": "electronic",
"value": "1533-4406"
}
],
"issued": {
"date-parts": [
[
2021,
12,
22
]
]
},
"language": "en",
"license": [
{
"URL": "http://www.nejmgroup.org/legal/terms-of-use.htm",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2021,
12,
22
]
],
"date-time": "2021-12-22T00:00:00Z",
"timestamp": 1640131200000
}
}
],
"link": [
{
"URL": "http://www.nejm.org/doi/pdf/10.1056/NEJMoa2116846",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "150",
"original-title": [],
"prefix": "10.1056",
"published": {
"date-parts": [
[
2021,
12,
22
]
]
},
"published-online": {
"date-parts": [
[
2021,
12,
22
]
]
},
"publisher": "Massachusetts Medical Society",
"reference-count": 25,
"references-count": 25,
"relation": {},
"score": 1,
"short-container-title": [
"N Engl J Med"
],
"short-title": [],
"source": "Crossref",
"subject": [
"General Medicine"
],
"subtitle": [],
"title": [
"Early Remdesivir to Prevent Progression to Severe Covid-19 in Outpatients"
],
"type": "journal-article"
}