Nitric Oxide Gas Inhalation Therapy in Spontaneous Breathing Patients With Mild/Moderate COVID-19: a Randomized Clinical Trial
et al., NCT04305457, NoCovid, NCT04305457, Mar 2026
43rd treatment shown to reduce risk in
June 2022, now with p = 0.013 from 14 studies, recognized in 10 countries.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
Early terminated RCT 62 hosptialized patients, showing no significant difference with nitric oxide treatment.
Standard of Care (SOC) for COVID-19 in the study country,
the USA, is very poor with very low average efficacy for approved treatments1.
Only expensive, high-profit treatments were approved for early treatment. Low-cost treatments were excluded, reducing the probability of early treatment due to access and cost barriers, and eliminating complementary and synergistic benefits seen with many low-cost treatments.
|
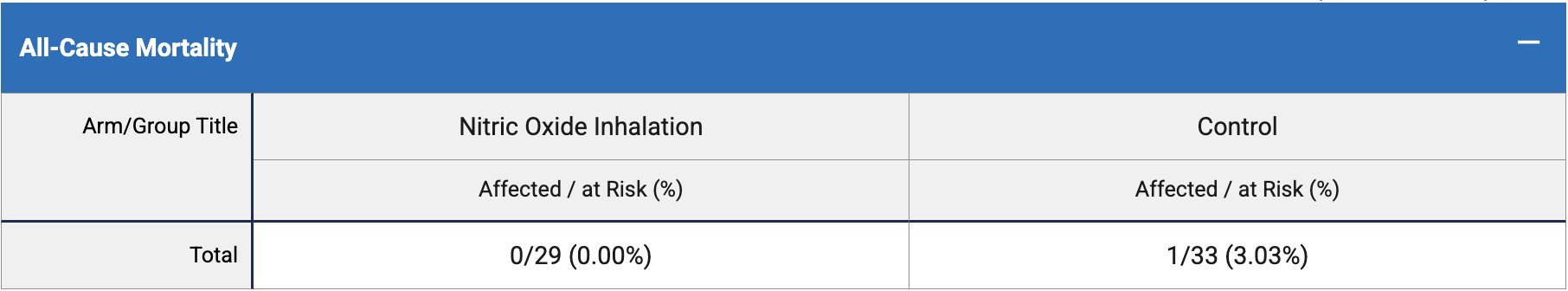
risk of death, 65.3% lower, RR 0.35, p = 1.00, treatment 0 of 29 (0.0%), control 1 of 33 (3.0%), NNT 33, relative risk is not 0 because of continuity correction due to zero events (with reciprocal of the contrasting arm).
|
|
risk of mechanical ventilation, 13.8% higher, RR 1.14, p = 1.00, treatment 1 of 29 (3.4%), control 1 of 33 (3.0%).
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Berra et al., 23 Mar 2026, Randomized Controlled Trial, USA, preprint, 1 author, trial NCT04305457 (history) (NoCovid).
Contact: lberra@mgh.harvard.edu.
