A bidimensional early intervention strategy of standard of care in combination with corticosteroids in elderly patients with mild to moderate COVID-19 (BEAT-COV study): a multicentre, open-label, randomized controlled trial
et al., BMC Infectious Diseases, doi:10.1186/s12879-026-12637-8, NCT05855395, Mar 2026
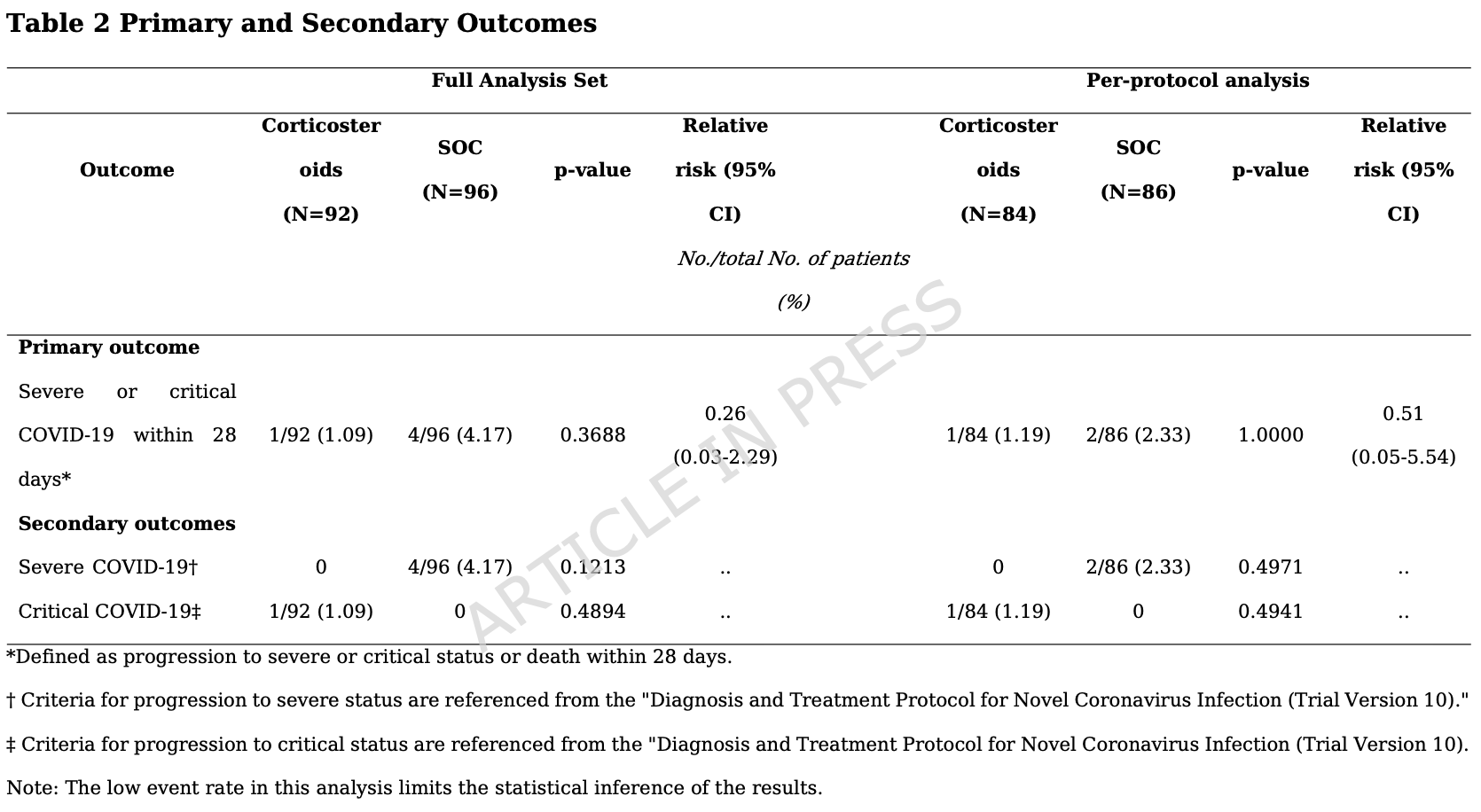
RCT 188 elderly patients with mild to moderate COVID-19 showing no significant reduction in progression to severe or critical COVID-19 with oral corticosteroids (dexamethasone 3 mg, 210 prednisolone 20 mg, or methylprednisolone 16 mg).
Ruan et al., 6 Mar 2026, Randomized Controlled Trial, China, peer-reviewed, median age 71.0, 21 authors, trial NCT05855395 (history).
Contact: zhangwenhong@fudan.edu.cn.
A bidimensional early intervention strategy of standard of care in combination with corticosteroids in elderly patients with mild to moderate COVID-19 (BEAT-COV study): a multicentre, open-label, randomized controlled trial
BMC Infectious Diseases, doi:10.1186/s12879-026-12637-8
study): a multicentre, open-label, randomized controlled trial
Declarations
Author contributions W.H.Z., Q.L.R., J.Q.W., and F.S. designed the study and led this study.
Ethics approval and consent to participate The protocol and all amendments were approved by the ethics committees of Huashan Hospital (Study Number 2023-065) and its collaborating hospitals. Written informed consent was obtained from all participants or their legal representatives before screening. All patient data were treated with strict confidentiality, and records were anonymized for analysis. All procedures involving human participants in this study were conducted in accordance with the ethical standards of the institutional and national research committees, and in full compliance with the principles outlined in the 1964 Declaration of Helsinki and its subsequent amendments.
Consent for publication Not applicable.
Competing interests All authors declare that they have no conflicts of interest.
References
Bernal, Da Silva, Musungaie, Kovalchuk, Gonzalez et al., Study Group. Molnupiravir for Oral Treatment of Covid-19 in Nonhospitalized Patients, N Engl J Med, doi:10.1056/NEJMoa2116044
Burki, WHO ends the COVID-19 public health emergency, Lancet Respir Med, doi:10.1016/S2213-2600(23)00217-5
Cao, Wang, Lu, Huang, Yang et al., Oral Simnotrelvir for Adult Patients with Mild-to-Moderate Covid-19, N Engl J Med, doi:10.1056/NEJMoa2301425
Cheong, Sio, Chan, Neng, Sam et al., et SARS-CoV-2 infection during the omicron wave in Hong Kong: an observational study, Lancet, doi:10.1016/S0140-6736(22)01586-0
Driscoll, Bistrian, Cytokine storm associated with severe COVID-19 infections: The potential mitigating role of omega-3 fatty acid triglycerides in the ICU, FASEB J, doi:10.1126/sciimmunol.abm5505
Fan, Dai, Ling, Wu, Tang et al., Oral VV116 versus placebo in patients with mild-to-moderate COVID-19 in China: a multicentre, double-blind, phase 3, randomised controlled study, Lancet Infect Dis, doi:10.1016/S1473-3099(23)00577-7
Galani, Rovina, Lampropoulou, Triantafyllia, Manioudaki et al., Untuned antiviral immunity in COVID-19 revealed by temporal type I/III interferon patterns and flu comparison, Nat Immunol, doi:10.1038/s41590-020-00840-x
Hadjadj, Yatim, Barnabei, Corneau, Boussier et al., Impaired type I interferon activity and inflammatory responses in severe COVID-19 patients, Science, doi:10.1126/science.abc6027
Hossain, Islam, Kamruzzaman, Hossain, Shekhar, Exploring age-related influences on COVID-19 severity and therapeutic approaches for older patients, Aging Health Res, doi:10.1016/j.ahr.2025.100259
Huang, Wang, Li, Ren, Zhao et al., Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China, Lancet, doi:10.1016/S0140-6736(20)30183-5
Lim, Randomization in clinical studies, Korean J Anesthesiol, doi:10.4097/kja.19049
Lu, Ling, Jiang, Tan, Wei et al., Geriatric risk and protective factors for serious COVID-19 outcomes among older adults in Shanghai Omicron wave, Emerg Microbes Infect, doi:10.1080/22221751.2022.2109517
Lu, Ling, Jiang, Tan, Wei et al., Primary assessment of the diversity of Omicron sublineages and the epidemiologic features of autumn/winter 2022 COVID-19 wave in Chinese mainland, Front Med, doi:10.1007/s11684-022-0981-7
Lucas, Wong, Klein, Castro, Silva et al., Longitudinal analyses reveal immunological misfiring in severe COVID-19, Nature, doi:10.1038/s41586-020-2588-y
Paludan, Mogensen, Innate immunological pathways in COVID-19 pathogenesis, Sci Immunol, doi:10.1126/sciimmunol.abm5505
Rahimi, Bezmin Abadi, Challenges of managing the asymptomatic carriers of SARS-CoV-2, Travel Med Infect Dis, doi:10.1016/j.tmaid.2020.101677
Ramakrishnan, Nicolau, Jr, Langford, Mahdi et al., Inhaled budesonide in the treatment of early COVID-19 (STOIC): a phase 2, open-label, randomised controlled trial, Lancet Respir Med, doi:10.1016/S2213-2600(21)00160-0
Recovery Collaborative Group, Horby, Lim, Emberson, Mafham et al., Dexamethasone in Hospitalized Patients with Covid-19, N Engl J Med, doi:10.1056/NEJMoa2021436
Ren, Fan, Hou, Su, Cai, COVID-19 immune features revealed by a large-scale single-cell transcriptome atlas, Cell, doi:10.1016/j.cell.2021.01.053
Valle, Kim-Schulze, Huang, Beckmann, Nirenberg et al., An inflammatory cytokine signature predicts COVID-19 severity and survival, Nat Med, doi:10.1038/s41591-020-1051-9
Wang, Wu, Zhang, Wu, Yu et al., C-Reactive Protein Level May Predict the Risk of COVID-19 Aggravation, Open Forum Infect Dis, doi:10.1093/ofid/ofaa153
Ye, Wang, Mao, The pathogenesis and treatment of the `Cytokine Storm' in COVID-19, J Infect, doi:10.1016/j.jinf.2020.03.037
Yu, Bafadhel, Dorward, Hayward, Saville et al., Inhaled budesonide for COVID-19 in people at high risk of complications in the community in the UK (PRINCIPLE): a randomised, controlled, open-label, adaptive platform trial, Lancet, doi:10.1016/S0140-6736(21)01744-X
Zhang, Wu, Xu, Asymptomatic carriers of COVID-19 as a concern for disease prevention and control: more testing, more follow-up, Biosci Trends, doi:10.5582/bst.2020.03069
DOI record:
{
"DOI": "10.1186/s12879-026-12637-8",
"ISSN": [
"1471-2334"
],
"URL": "http://dx.doi.org/10.1186/s12879-026-12637-8",
"alternative-id": [
"12637"
],
"assertion": [
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "Received",
"name": "received",
"order": 1,
"value": "1 December 2025"
},
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "Accepted",
"name": "accepted",
"order": 2,
"value": "16 January 2026"
},
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "First Online",
"name": "first_online",
"order": 3,
"value": "6 March 2026"
},
{
"group": {
"label": "Declarations",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 1
},
{
"group": {
"label": "Ethics approval and consent to participate",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 2,
"value": "The protocol and all amendments were approved by the ethics committees of Huashan Hospital (Study Number 2023-065) and its collaborating hospitals. Written informed consent was obtained from all participants or their legal representatives before screening. All patient data were treated with strict confidentiality, and records were anonymized for analysis. All procedures involving human participants in this study were conducted in accordance with the ethical standards of the institutional and national research committees, and in full compliance with the principles outlined in the 1964 Declaration of Helsinki and its subsequent amendments."
},
{
"group": {
"label": "Consent for publication",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 3,
"value": "Not applicable."
},
{
"group": {
"label": "Competing interests",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 4,
"value": "The authors declare no competing interests."
}
],
"author": [
{
"affiliation": [],
"family": "Ruan",
"given": "Qiaoling",
"sequence": "first"
},
{
"affiliation": [],
"family": "Zhang",
"given": "Yanliang",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Zhao",
"given": "Tingting",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Zhu",
"given": "Feng",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Liu",
"given": "Xi",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Cao",
"given": "Ran",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Li",
"given": "Yongzhong",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Zhang",
"given": "Xiaoqiang",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Song",
"given": "Wei",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Geng",
"given": "Jiawei",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Zou",
"given": "Deming",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Li",
"given": "Yuanyuan",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Zhang",
"given": "Liaoyun",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Wu",
"given": "Chao",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Lan",
"given": "Yuanbo",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ding",
"given": "Xiangchun",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Huang",
"given": "Fuli",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Wu",
"given": "Jiqin",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Sun",
"given": "Feng",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Shao",
"given": "Lingyun",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Zhang",
"given": "Wenhong",
"sequence": "additional"
}
],
"container-title": "BMC Infectious Diseases",
"container-title-short": "BMC Infect Dis",
"content-domain": {
"crossmark-restriction": false,
"domain": [
"link.springer.com"
]
},
"created": {
"date-parts": [
[
2026,
3,
6
]
],
"date-time": "2026-03-06T15:35:57Z",
"timestamp": 1772811357000
},
"deposited": {
"date-parts": [
[
2026,
3,
6
]
],
"date-time": "2026-03-06T15:36:03Z",
"timestamp": 1772811363000
},
"funder": [
{
"award": [
"2023YFC3043400"
],
"award-info": [
{
"award-number": [
"2023YFC3043400"
]
}
],
"name": "National Key Research and Development Program of China"
},
{
"award": [
"SSIII-202402"
],
"award-info": [
{
"award-number": [
"SSIII-202402"
]
}
],
"name": "Shanghai Sci-Tech Inno Center for Infection & Immunity"
}
],
"indexed": {
"date-parts": [
[
2026,
3,
7
]
],
"date-time": "2026-03-07T05:41:15Z",
"timestamp": 1772862075618,
"version": "3.50.1"
},
"is-referenced-by-count": 0,
"issued": {
"date-parts": [
[
2026,
3,
6
]
]
},
"language": "en",
"license": [
{
"URL": "https://creativecommons.org/licenses/by-nc-nd/4.0",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2026,
3,
6
]
],
"date-time": "2026-03-06T00:00:00Z",
"timestamp": 1772755200000
}
},
{
"URL": "https://creativecommons.org/licenses/by-nc-nd/4.0",
"content-version": "am",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2026,
3,
6
]
],
"date-time": "2026-03-06T00:00:00Z",
"timestamp": 1772755200000
}
}
],
"link": [
{
"URL": "https://link.springer.com/article/10.1186/s12879-026-12637-8",
"content-type": "text/html",
"content-version": "vor",
"intended-application": "text-mining"
}
],
"member": "297",
"original-title": [],
"prefix": "10.1186",
"published": {
"date-parts": [
[
2026,
3,
6
]
]
},
"published-online": {
"date-parts": [
[
2026,
3,
6
]
]
},
"publisher": "Springer Science and Business Media LLC",
"reference": [
{
"DOI": "10.1016/S2213-2600(23)00217-5",
"author": "T Burki",
"doi-asserted-by": "publisher",
"first-page": "588",
"issue": "7",
"journal-title": "Lancet Respir Med",
"key": "12637_CR1",
"unstructured": "Burki T. WHO ends the COVID-19 public health emergency. Lancet Respir Med. 2023;11(7):588. https://doi.org/10.1016/S2213-2600(23)00217-5.",
"volume": "11",
"year": "2023"
},
{
"key": "12637_CR2",
"unstructured": "Centers for Disease Control and Prevention. COVID-NET (COVID-19-Associated Hospitalization Surveillance Network). COVID Data Tracker [Internet]. Available from: https://covid.cdc.gov/covid-data-tracker/#covidnet-hospitalization-network. Accessed 1 December 2024."
},
{
"DOI": "10.1007/s11684-022-0981-7",
"author": "G Lu",
"doi-asserted-by": "publisher",
"first-page": "758",
"issue": "4",
"journal-title": "Front Med",
"key": "12637_CR3",
"unstructured": "Lu G, Ling Y, Jiang M, Tan Y, Wei D, Jiang L, et al. Primary assessment of the diversity of Omicron sublineages and the epidemiologic features of autumn/winter 2022 COVID-19 wave in Chinese Mainland. Front Med. 2023;17(4):758–67. https://doi.org/10.1007/s11684-022-0981-7.",
"volume": "17",
"year": "2023"
},
{
"DOI": "10.1016/j.tmaid.2020.101677",
"author": "F Rahimi",
"doi-asserted-by": "publisher",
"first-page": "101677",
"journal-title": "Travel Med Infect Dis",
"key": "12637_CR4",
"unstructured": "Rahimi F, Talebi Bezmin Abadi A. Challenges of managing the asymptomatic carriers of SARS-CoV-2. Travel Med Infect Dis. 2020;37:101677. https://doi.org/10.1016/j.tmaid.2020.101677.",
"volume": "37",
"year": "2020"
},
{
"DOI": "10.5582/bst.2020.03069",
"author": "J Zhang",
"doi-asserted-by": "publisher",
"first-page": "206",
"issue": "3",
"journal-title": "Biosci Trends",
"key": "12637_CR5",
"unstructured": "Zhang J, Wu S, Xu L. Asymptomatic carriers of COVID-19 as a concern for disease prevention and control: more testing, more follow-up. Biosci Trends. 2020;14(3):206–8. https://doi.org/10.5582/bst.2020.03069.",
"volume": "14",
"year": "2020"
},
{
"DOI": "10.1016/j.ahr.2025.100259",
"doi-asserted-by": "publisher",
"key": "12637_CR6",
"unstructured": "Shafaat Hossain S, Islam M, Kamruzzaman. Hossain Uddin Shekhar. Exploring age-related influences on COVID-19 severity and therapeutic approaches for older patients. Aging Health Res. 2025;5(100259). https://doi.org/10.1016/j.ahr.2025.100259."
},
{
"DOI": "10.1080/22221751.2022.2109517",
"author": "G Lu",
"doi-asserted-by": "publisher",
"first-page": "2045",
"issue": "1",
"journal-title": "Emerg Microbes Infect",
"key": "12637_CR7",
"unstructured": "Lu G, Ling Y, Jiang M, Tan Y, Wei D, Jiang L, et al. Geriatric risk and protective factors for serious COVID-19 outcomes among older adults in Shanghai Omicron wave. Emerg Microbes Infect. 2022;11(1):2045–54. https://doi.org/10.1080/22221751.2022.2109517.",
"volume": "11",
"year": "2022"
},
{
"key": "12637_CR8",
"unstructured": "National Bureau of Statistics of China. Seventh National Population Census Bulletin [第七次全国人口普查公报] [Internet]. 2021 May 11. Available from: https://www.gov.cn/guoqing/2021-05/13/content_5606149.htm. Accessed 10 March 2024."
},
{
"DOI": "10.1016/j.jinf.2020.03.037",
"author": "Q Ye",
"doi-asserted-by": "publisher",
"first-page": "607",
"issue": "6",
"journal-title": "J Infect",
"key": "12637_CR9",
"unstructured": "Ye Q, Wang B, Mao J. The pathogenesis and treatment of the `Cytokine storm’ in COVID-19. J Infect. 2020;80(6):607–13. https://doi.org/10.1016/j.jinf.2020.03.037.",
"volume": "80",
"year": "2020"
},
{
"DOI": "10.1016/S0140-6736(20)30183-5",
"author": "C Huang",
"doi-asserted-by": "publisher",
"first-page": "497",
"issue": "10223",
"journal-title": "Lancet",
"key": "12637_CR10",
"unstructured": "Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395(10223):497–506. https://doi.org/10.1016/S0140-6736(20)30183-5.",
"volume": "395",
"year": "2020"
},
{
"DOI": "10.1038/s41590-020-00840-x",
"author": "IE Galani",
"doi-asserted-by": "publisher",
"first-page": "32",
"issue": "1",
"journal-title": "Nat Immunol",
"key": "12637_CR11",
"unstructured": "Galani IE, Rovina N, Lampropoulou V, Triantafyllia V, Manioudaki M, Pavlos E, et al. Untuned antiviral immunity in COVID-19 revealed by Temporal type I/III interferon patterns and flu comparison. Nat Immunol. 2021;22(1):32–40. https://doi.org/10.1038/s41590-020-00840-x.",
"volume": "22",
"year": "2021"
},
{
"DOI": "10.1126/science.abc6027",
"author": "J Hadjadj",
"doi-asserted-by": "publisher",
"first-page": "718",
"issue": "6504",
"journal-title": "Science",
"key": "12637_CR12",
"unstructured": "Hadjadj J, Yatim N, Barnabei L, Corneau A, Boussier J, Smith N, et al. Impaired type I interferon activity and inflammatory responses in severe COVID-19 patients. Science. 2020;369(6504):718–24. https://doi.org/10.1126/science.abc6027.",
"volume": "369",
"year": "2020"
},
{
"DOI": "10.1038/s41591-020-1051-9",
"author": "DM Del Valle",
"doi-asserted-by": "publisher",
"first-page": "1636",
"issue": "10",
"journal-title": "Nat Med",
"key": "12637_CR13",
"unstructured": "Del Valle DM, Kim-Schulze S, Huang HH, Beckmann ND, Nirenberg S, Wang B, et al. An inflammatory cytokine signature predicts COVID-19 severity and survival. Nat Med. 2020;26(10):1636–43. https://doi.org/10.1038/s41591-020-1051-9.",
"volume": "26",
"year": "2020"
},
{
"DOI": "10.1038/s41586-020-2588-y",
"author": "C Lucas",
"doi-asserted-by": "publisher",
"first-page": "463",
"issue": "7821",
"journal-title": "Nature",
"key": "12637_CR14",
"unstructured": "Lucas C, Wong P, Klein J, Castro TBR, Silva J, Sundaram M, et al. Longitudinal analyses reveal immunological misfiring in severe COVID-19. Nature. 2020;584(7821):463–9. https://doi.org/10.1038/s41586-020-2588-y.",
"volume": "584",
"year": "2020"
},
{
"DOI": "10.1016/j.cell.2021.01.053",
"author": "X Ren",
"doi-asserted-by": "publisher",
"first-page": "1895",
"issue": "7",
"journal-title": "Cell",
"key": "12637_CR15",
"unstructured": "Ren X, Wen W, Fan X, Hou W, Su B, Cai P, et al. COVID-19 immune features revealed by a large-scale single-cell transcriptome atlas. Cell. 2021;184(7):1895–e91319. https://doi.org/10.1016/j.cell.2021.01.053.",
"volume": "184",
"year": "2021"
},
{
"DOI": "10.1126/sciimmunol.abm5505",
"author": "SR Paludan",
"doi-asserted-by": "publisher",
"first-page": "eabm5505",
"issue": "67",
"journal-title": "Sci Immunol",
"key": "12637_CR16",
"unstructured": "Paludan SR, Mogensen TH. Innate immunological pathways in COVID-19 pathogenesis. Sci Immunol. 2022;7(67):eabm5505. https://doi.org/10.1126/sciimmunol.abm5505.",
"volume": "7",
"year": "2022"
},
{
"DOI": "10.1126/sciimmunol.abm5505",
"author": "DF Driscoll",
"doi-asserted-by": "publisher",
"first-page": "e23066",
"issue": "8",
"journal-title": "FASEB J",
"key": "12637_CR17",
"unstructured": "Driscoll DF, Bistrian BR. Cytokine storm associated with severe COVID-19 infections: the potential mitigating role of omega-3 fatty acid triglycerides in the ICU. FASEB J. 2023;37(8):e23066. https://doi.org/10.1126/sciimmunol.abm5505.",
"volume": "37",
"year": "2023"
},
{
"key": "12637_CR18",
"unstructured": "COVID-19 Treatment Guidelines Panel. Coronavirus Disease 2019 (COVID-19) Treatment Guidelines. National Institutes of Health. 2024. Available at https://www.covid19treatmentguidelines.nih.gov/. Accessed 24 November, 2024."
},
{
"DOI": "10.3760/cma.j.cn114798-20230108-00040",
"author": "Chinese Medical Association General Practice Branch, Chinese Association of Chinese Medicine General Practice Branch, Chinese Preventive Medicine Association Respiratory Disease Prevention and Control Professional Committee",
"doi-asserted-by": "publisher",
"first-page": "115",
"issue": "2",
"journal-title": "Chin J Gen Pract",
"key": "12637_CR19",
"unstructured": "Chinese Medical Association General Practice Branch, Chinese Association of Chinese Medicine General Practice Branch, Chinese Preventive Medicine Association Respiratory Disease Prevention and Control Professional Committee. Guidelines for primary diagnosis, treatment, and services of novel coronavirus infection (First Edition) [J]. Chin J Gen Pract. 2023;22(2):115–37. https://doi.org/10.3760/cma.j.cn114798-20230108-00040.",
"volume": "22",
"year": "2023"
},
{
"key": "12637_CR20",
"unstructured": "National Health Commission of the People’s Republic of China. Diagnosis and treatment protocol for novel coronavirus infection (trial version 10) [Internet]. 2023. Available from: https://www.gov.cn/zhengce/zhengceku/2023-01/06/content_5735343.htm. Accessed 10 March 2024."
},
{
"DOI": "10.1093/ofid/ofaa153",
"author": "G Wang",
"doi-asserted-by": "publisher",
"first-page": "ofaa153",
"issue": "5",
"journal-title": "Open Forum Infect Dis",
"key": "12637_CR21",
"unstructured": "Wang G, Wu C, Zhang Q, Wu F, Yu B, Lv J, et al. C-Reactive protein level May predict the risk of COVID-19 aggravation. Open Forum Infect Dis. 2020;7(5):ofaa153. https://doi.org/10.1093/ofid/ofaa153.",
"volume": "7",
"year": "2020"
},
{
"DOI": "10.4097/kja.19049",
"author": "CY Lim",
"doi-asserted-by": "publisher",
"first-page": "221",
"issue": "3",
"journal-title": "Korean J Anesthesiol",
"key": "12637_CR22",
"unstructured": "Lim CY, In J. Randomization in clinical studies. Korean J Anesthesiol. 2019;72(3):221–32. https://doi.org/10.4097/kja.19049.",
"volume": "72",
"year": "2019"
},
{
"DOI": "10.1002/hsr2.1361",
"author": "HH Cheong",
"doi-asserted-by": "publisher",
"first-page": "e1361",
"issue": "7",
"journal-title": "Health Sci Rep",
"key": "12637_CR23",
"unstructured": "Cheong HH, Sio FI, Chan CC, Neng SI, Sam IP, Cheang T, et al. Clinical characteristics of COVID-19 patients infected by the Omicron variants in Macao, china: A cross-sectional study. Health Sci Rep. 2023;6(7):e1361. https://doi.org/10.1002/hsr2.1361.",
"volume": "6",
"year": "2023"
},
{
"DOI": "10.3389/fmed.2022.912367",
"author": "J Zhang",
"doi-asserted-by": "publisher",
"first-page": "912367",
"journal-title": "Front Med (Lausanne)",
"key": "12637_CR24",
"unstructured": "Zhang J, Chen N, Zhao D, Zhang J, Hu Z, Tao Z. Clinical characteristics of COVID-19 patients infected by the Omicron variant of SARS-CoV-2. Front Med (Lausanne). 2022;9:912367. https://doi.org/10.3389/fmed.2022.912367.",
"volume": "9",
"year": "2022"
},
{
"DOI": "10.1016/j.ceh.2023.10.002",
"author": "DW Yang",
"doi-asserted-by": "publisher",
"first-page": "138",
"journal-title": "Clin eHealth",
"key": "12637_CR25",
"unstructured": "Yang DW, Li J, Feng L, Weng HF, Ju MJ, Wang H, et al. Clinical characteristics and outcome of patients with SARS-CoV-2 Omicron variant in shanghai: A single center, retrospective, observational study. Clin eHealth. 2023;6:138–43. https://doi.org/10.1016/j.ceh.2023.10.002.",
"volume": "6",
"year": "2023"
},
{
"DOI": "10.1503/cmaj.220471",
"author": "T Pitre",
"doi-asserted-by": "publisher",
"first-page": "E969",
"issue": "28",
"journal-title": "CMAJ",
"key": "12637_CR26",
"unstructured": "Pitre T, Van Alstine R, Chick G, Leung G, Mikhail D, Cusano E, et al. Antiviral drug treatment for nonsevere COVID-19: a systematic review and network meta-analysis. CMAJ. 2022;194(28):E969–80. https://doi.org/10.1503/cmaj.220471.",
"volume": "194",
"year": "2022"
},
{
"DOI": "10.1056/NEJMoa2118542",
"author": "J Hammond",
"doi-asserted-by": "publisher",
"first-page": "1397",
"issue": "15",
"journal-title": "N Engl J Med",
"key": "12637_CR27",
"unstructured": "Hammond J, Leister-Tebbe H, Gardner A, Abreu P, Bao W, Wisemandle W, et al. Oral nirmatrelvir for High-Risk, nonhospitalized adults with Covid-19. N Engl J Med. 2022;386(15):1397–408. https://doi.org/10.1056/NEJMoa2118542.",
"volume": "386",
"year": "2022"
},
{
"DOI": "10.1016/S0140-6736(22)01586-0",
"author": "CKH Wong",
"doi-asserted-by": "publisher",
"first-page": "1213",
"issue": "10359",
"journal-title": "Lancet",
"key": "12637_CR28",
"unstructured": "Wong CKH, Au ICH, Lau KTK, Lau EHY, Cowling BJ, Leung GM. Real-world effectiveness of molnupiravir and nirmatrelvir plus Ritonavir against mortality, hospitalisation, and in-hospital outcomes among community-dwelling, ambulatory patients with confirmed SARS-CoV-2 infection during the Omicron wave in Hong kong: an observational study. Lancet. 2022;400(10359):1213–22. https://doi.org/10.1016/S0140-6736(22)01586-0.",
"volume": "400",
"year": "2022"
},
{
"DOI": "10.1016/S2213-2600(21)00160-0",
"author": "S Ramakrishnan",
"doi-asserted-by": "publisher",
"first-page": "763",
"issue": "7",
"journal-title": "Lancet Respir Med",
"key": "12637_CR29",
"unstructured": "Ramakrishnan S, Nicolau DV Jr, Langford B, Mahdi M, Jeffers H, Mwasuku C, et al. Inhaled Budesonide in the treatment of early COVID-19 (STOIC): a phase 2, open-label, randomised controlled trial. Lancet Respir Med. 2021;9(7):763–72. https://doi.org/10.1016/S2213-2600(21)00160-0.",
"volume": "9",
"year": "2021"
},
{
"DOI": "10.1016/S0140-6736(21)01744-X",
"author": "LM Yu",
"doi-asserted-by": "publisher",
"first-page": "843",
"issue": "10303",
"journal-title": "Lancet",
"key": "12637_CR30",
"unstructured": "Yu LM, Bafadhel M, Dorward J, Hayward G, Saville BR, Gbinigie O, et al. Inhaled Budesonide for COVID-19 in people at high risk of complications in the community in the UK (PRINCIPLE): a randomised, controlled, open-label, adaptive platform trial. Lancet. 2021;398(10303):843–55. https://doi.org/10.1016/S0140-6736(21)01744-X.",
"volume": "398",
"year": "2021"
},
{
"DOI": "10.1056/NEJMoa2021436",
"author": "P Horby",
"doi-asserted-by": "publisher",
"first-page": "693",
"issue": "8",
"journal-title": "N Engl J Med",
"key": "12637_CR31",
"unstructured": "RECOVERY Collaborative Group, Horby P, Lim WS, Emberson JR, Mafham M, Bell JL, et al. Dexamethasone in hospitalized patients with Covid-19. N Engl J Med. 2021;384(8):693–704. https://doi.org/10.1056/NEJMoa2021436.",
"volume": "384",
"year": "2021"
},
{
"key": "12637_CR32",
"unstructured": "Centers for Disease Control and Prevention. Trends in COVID-19 Deaths. COVID Data Tracker [Internet]. Available from: https://covid.cdc.gov/covid-data-tracker/#trends_weeklydeaths_weeklydeathrateaa_00. Accessed 1 December 2024."
},
{
"DOI": "10.1016/S1473-3099(23)00577-7",
"author": "X Fan",
"doi-asserted-by": "publisher",
"first-page": "129",
"issue": "2",
"journal-title": "Lancet Infect Dis",
"key": "12637_CR33",
"unstructured": "Fan X, Dai X, Ling Y, Wu L, Tang L, Peng C, et al. Oral VV116 versus placebo in patients with mild-to-moderate COVID-19 in china: a multicentre, double-blind, phase 3, randomised controlled study. Lancet Infect Dis. 2024;24(2):129–39. https://doi.org/10.1016/S1473-3099(23)00577-7.",
"volume": "24",
"year": "2024"
},
{
"DOI": "10.1056/NEJMoa2301425",
"author": "B Cao",
"doi-asserted-by": "publisher",
"first-page": "230",
"issue": "3",
"journal-title": "N Engl J Med",
"key": "12637_CR34",
"unstructured": "Cao B, Wang Y, Lu H, Huang C, Yang Y, Shang L, et al. Oral Simnotrelvir for adult patients with Mild-to-Moderate Covid-19. N Engl J Med. 2024;390(3):230–41. https://doi.org/10.1056/NEJMoa2301425.",
"volume": "390",
"year": "2024"
},
{
"DOI": "10.1056/NEJMoa2116044",
"author": "A Jayk Bernal",
"doi-asserted-by": "publisher",
"first-page": "509",
"issue": "6",
"journal-title": "N Engl J Med",
"key": "12637_CR35",
"unstructured": "Jayk Bernal A, Gomes da Silva MM, Musungaie DB, Kovalchuk E, Gonzalez A, Delos Reyes V, et al. MOVe-OUT study Group. Molnupiravir for oral treatment of Covid-19 in nonhospitalized patients. N Engl J Med. 2022;386(6):509–20. https://doi.org/10.1056/NEJMoa2116044.",
"volume": "386",
"year": "2022"
}
],
"reference-count": 35,
"references-count": 35,
"relation": {},
"resource": {
"primary": {
"URL": "https://link.springer.com/10.1186/s12879-026-12637-8"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "A bidimensional early intervention strategy of standard of care in combination with corticosteroids in elderly patients with mild to moderate COVID-19 (BEAT-COV study): a multicentre, open-label, randomized controlled trial",
"type": "journal-article",
"update-policy": "https://doi.org/10.1007/springer_crossmark_policy"
}