Treatment with fluvoxamine in nonhospitalized coronavirus disease 2019 patients
et al., The Egyptian Journal of Chest Diseases and Tuberculosis, doi:10.4103/ecdt.ecdt_38_22, Jan 2023
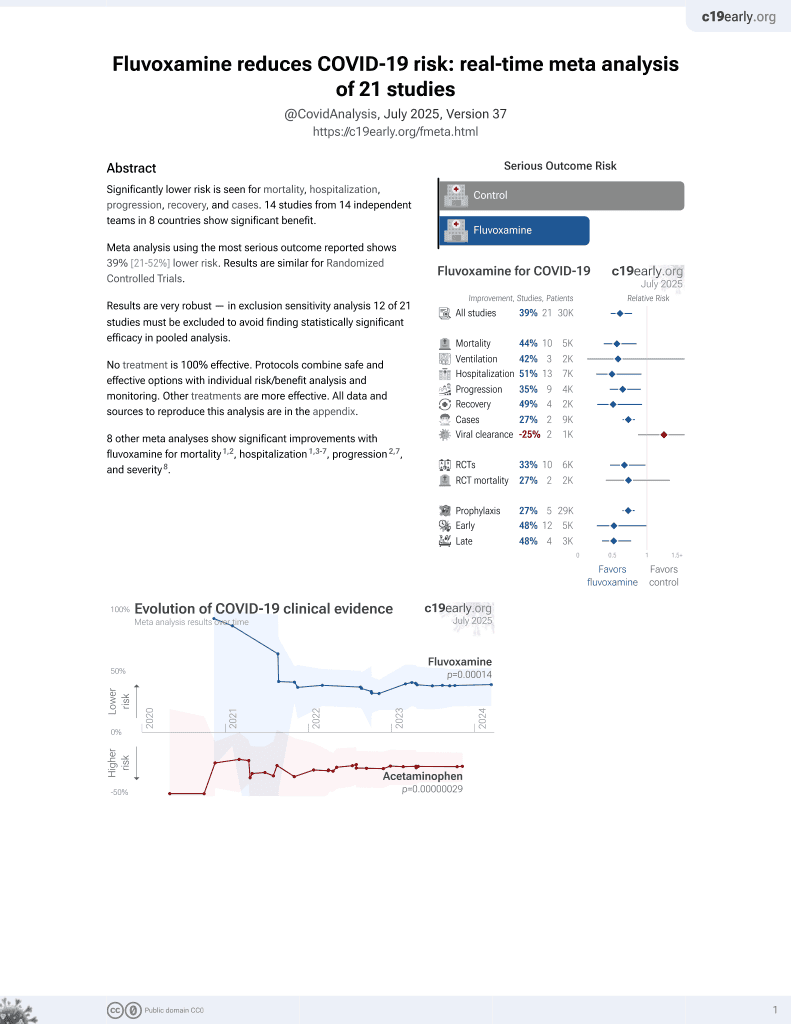
30th treatment shown to reduce risk in
November 2021, now with p = 0.00014 from 21 studies, recognized in 2 countries.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
RCT of 162 nonhospitalized COVID-19 patients (mild and moderate) showing lower hospitalization with treatment.
No multivariable adjustment: despite baseline imbalances in BMI, psychological disease, and other covariates, the primary outcome was analyzed with simple unadjusted group comparisons only.
Unequal group allocation: the moderate group split 51:39 in favor of fluvoxamine, unusual for 1:1 randomization.
No trial registration: no registry number is reported, making it impossible to verify whether the mild/moderate subgrouping and endpoints were pre-specified or introduced after seeing the data.
Non-standardized primary endpoint: hospitalization decisions were made during remote phone follow-up without protocol-defined criteria, and whether decision-makers were blinded is unclear.
|
risk of hospitalization, 59.9% lower, RR 0.40, p = 0.03, treatment 88, control 74, all patients.
|
|
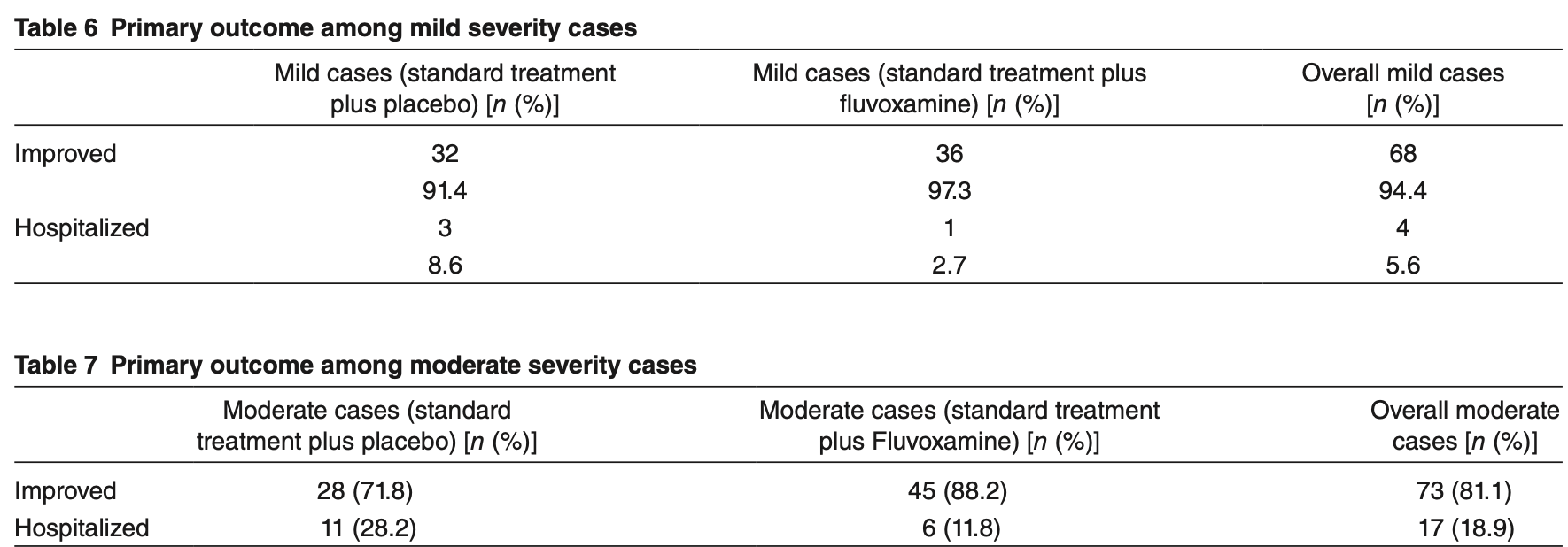
risk of hospitalization, 58.3% lower, RR 0.42, p = 0.06, treatment 6 of 51 (11.8%), control 11 of 39 (28.2%), NNT 6.1, moderate cases.
|
|
risk of hospitalization, 68.5% lower, RR 0.32, p = 0.35, treatment 1 of 37 (2.7%), control 3 of 35 (8.6%), NNT 17, mild cases.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Ibrahim et al., 31 Jan 2023, Randomized Controlled Trial, placebo-controlled, Egypt, peer-reviewed, 2 authors.
Contact: mohammedmansour.md@gmail.com.
Abstract: ## Treatment with fluvoxamine in nonhospitalized coronavirus disease 2019 patients
Mohammed A. Ibrahim, Mohammed Shehta
Department of Chest Medicine, Mansoura University, Mansoura, Egypt
Correspondence to Mohammed A. Ibrahim, MD, Department of Chest Medicine, Mansoura University, Dakahlia Governorate, Mansoura 35511, Egypt.
Tel: +01009400754;
e-mail: mohammedmansour.md@gmail.com
Received:
05 March 2022
Revised:
22 March 2022
Accepted:
29 March 2022
Published:
07 February 2023
The Egyptian Journal of Chest Diseases and Tuberculosis 2023, 72:40-45
DOI record:
{
"DOI": "10.4103/ecdt.ecdt_38_22",
"ISSN": [
"0422-7638"
],
"URL": "http://dx.doi.org/10.4103/ecdt.ecdt_38_22",
"abstract": "<jats:sec>\n <jats:title>Context</jats:title>\n <jats:p>Fluvoxamine may have a potential immune-regulatory action and a therapeutic role in severe acute respiratory syndrome-coronavirus (SARS-CoV-2) infection that may prevent progression and/or hospitalization.</jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>Aims</jats:title>\n <jats:p>Trial that compared fluvoxamine versus placebo in nonhospitalized adults with confirmed SARS-CoV-2 infection (mild and moderate coronavirus disease 2019 cases).</jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>Settings and design</jats:title>\n <jats:p>This is a double-blinded, randomized clinical trial.</jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>Patients and methods</jats:title>\n <jats:p>The study enrolled 162 cases with positive PCR assay for SARS-CoV-2 infection and who were symptomatic within 7 days of the first dose of study medication.</jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>Statistical analysis</jats:title>\n <jats:p>The demographic, clinical, and laboratory data gathered together will be tabulated and statistically analyzed. The statistical analysis of data was carried out using Excel and the SPSS programs statistical package for AQ8 Social Sciences, version 17. Quantitative data were described as median (minimum–maximum). An analysis of the data was carried out to test statistically significant differences between groups. Quantitative data were presented as mean±SD and the Student’s <jats:italic toggle=\"yes\">t</jats:italic> test was used to compare between two groups.</jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>Results</jats:title>\n <jats:p>In all, 162 patients completed the study; 72 patients were of mild severity; 90 patients were moderate cases and each group was randomized to receive fluvoxamine or placebo besides standard care. In the mild group, no significant difference was recorded while slight significance exists in the moderate severity group.</jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>Conclusions</jats:title>\n <jats:p>Fluvoxamine may have an added value besides the current standard care in reducing the need for hospitalization in outpatient cases, especially pneumonic ones; however, more larger studies are needed.</jats:p>\n </jats:sec>",
"author": [
{
"affiliation": [],
"family": "Ibrahim",
"given": "Mohammed A.",
"sequence": "first"
},
{
"affiliation": [],
"family": "Shehta",
"given": "Mohammed",
"sequence": "additional"
}
],
"container-title": "The Egyptian Journal of Chest Diseases and Tuberculosis",
"content-domain": {
"crossmark-restriction": true,
"domain": [
"lww.com",
"ovid.com"
]
},
"created": {
"date-parts": [
[
2023,
2,
7
]
],
"date-time": "2023-02-07T13:42:37Z",
"timestamp": 1675777357000
},
"deposited": {
"date-parts": [
[
2024,
9,
2
]
],
"date-time": "2024-09-02T05:07:01Z",
"timestamp": 1725253621000
},
"indexed": {
"date-parts": [
[
2026,
1,
15
]
],
"date-time": "2026-01-15T22:10:01Z",
"timestamp": 1768515001400,
"version": "3.49.0"
},
"is-referenced-by-count": 2,
"issue": "1",
"issued": {
"date-parts": [
[
2023,
1
]
]
},
"journal-issue": {
"issue": "1",
"published-print": {
"date-parts": [
[
2023
]
]
}
},
"language": "en",
"link": [
{
"URL": "https://journals.lww.com/10.4103/ecdt.ecdt_38_22",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "2581",
"original-title": [],
"page": "40-45",
"prefix": "10.4103",
"published": {
"date-parts": [
[
2023,
1
]
]
},
"published-print": {
"date-parts": [
[
2023,
1
]
]
},
"publisher": "Medknow",
"reference": [
{
"DOI": "10.1038/cddis.2014.301",
"article-title": "Fluvoxamine alleviates ER stress via induction of sigma-1 receptor",
"author": "Omi",
"doi-asserted-by": "crossref",
"first-page": "e1332",
"journal-title": "Cell Death Dis",
"key": "R1-6-20240902",
"volume": "5",
"year": "2014"
},
{
"DOI": "10.1016/j.jphs.2014.11.010",
"article-title": "Activation of sigma-1 receptor chaperone in the treatment of neuropsychiatric diseases and its clinical implication",
"author": "Hashimoto",
"doi-asserted-by": "crossref",
"first-page": "6",
"journal-title": "J Pharmacol Sci",
"key": "R2-6-20240902",
"volume": "127",
"year": "2015"
},
{
"article-title": "Safety and efficacy of fluvoxamine in COVID-19 ICU patients: An open label, prospective cohort trial with matched controls",
"author": "Calusic",
"first-page": "1",
"journal-title": "Br J Clin Pharmacol",
"key": "R3-6-20240902",
"volume": "1",
"year": "2021"
},
{
"DOI": "10.1001/jama.2020.22760",
"article-title": "Fluvoxamine vs placebo and clinical deterioration in outpatients with symptomatic COVID-19: a randomized clinical trial",
"author": "Lenze",
"doi-asserted-by": "crossref",
"first-page": "2292",
"journal-title": "JAMA",
"key": "R4-6-20240902",
"volume": "324",
"year": "2020"
},
{
"DOI": "10.1126/scitranslmed.aau5266",
"article-title": "Modulation of the sigma-1 receptor-IRE1 pathway is beneficial in preclinical models of inflammation and sepsis",
"author": "Rosen",
"doi-asserted-by": "crossref",
"first-page": "eaau5266",
"journal-title": "Sci Transl Med",
"key": "R5-6-20240902",
"volume": "11",
"year": "2019"
},
{
"article-title": "Evaluation of the effect of antidepressant drug, fluvoxamine, on cyclooxygenase-2 protein expression in lipopolysaccharide-stimulated macrophages",
"author": "Esfahani",
"first-page": "1",
"journal-title": "Adv Biomed Res",
"key": "R6-6-20240902",
"volume": "8",
"year": "2019"
},
{
"DOI": "10.1016/j.ijantimicag.2020.106044",
"article-title": "Widely available lysosome targeting agents should be considered as potential therapy for COVID-19",
"author": "Homolak",
"doi-asserted-by": "crossref",
"first-page": "106044",
"journal-title": "Int J Antimicrob Agents",
"key": "R7-6-20240902",
"volume": "56",
"year": "2020"
},
{
"DOI": "10.3389/fphar.2021.652688",
"article-title": "Fluvoxamine: a review of its mechanism of action and its role in COVID-19",
"author": "Sukhatme",
"doi-asserted-by": "crossref",
"first-page": "1",
"journal-title": "Front Pharmacol",
"key": "R8-6-20240902",
"volume": "12",
"year": "2021"
},
{
"DOI": "10.1016/j.jocn.2021.03.010",
"article-title": "Drug repurposing of selective serotonin reuptake inhibitors: could these drugs help fight COVID-19 and save lives?",
"author": "Pashaei",
"doi-asserted-by": "crossref",
"first-page": "163",
"journal-title": "J Clin Neurosci",
"key": "R9-6-20240902",
"volume": "88",
"year": "2021"
},
{
"DOI": "10.3389/fphar.2021.652688",
"article-title": "Fluvoxamine: a review of its mechanism of action and its role in COVID-19",
"author": "Sukhatme",
"doi-asserted-by": "crossref",
"first-page": "652688",
"journal-title": "Front Pharmacol",
"key": "R10-6-20240902",
"volume": "12",
"year": "2021"
},
{
"DOI": "10.1681/ASN.2018101032",
"article-title": "Comparative cardiac safety of selective serotonin reuptake inhibitors among individuals receiving maintenance hemodialysis",
"author": "Assimon",
"doi-asserted-by": "crossref",
"first-page": "611",
"journal-title": "J Am Soc Nephrol",
"key": "R11-6-20240902",
"volume": "30",
"year": "2019"
},
{
"DOI": "10.1016/S2214-109X(21)00448-4",
"article-title": "Effect of early treatment with fluvoxamine on risk of emergency care and hospitalisation among patients with COVID-19: the TOGETHER randomised, platform clinical trial",
"author": "Reis",
"doi-asserted-by": "crossref",
"first-page": "e42",
"journal-title": "Lancet Glob Health",
"key": "R12-6-20240902",
"volume": "10",
"year": "2022"
}
],
"reference-count": 12,
"references-count": 12,
"relation": {},
"resource": {
"primary": {
"URL": "https://journals.lww.com/10.4103/ecdt.ecdt_38_22"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "Treatment with fluvoxamine in nonhospitalized coronavirus disease 2019 patients",
"type": "journal-article",
"update-policy": "https://doi.org/10.1097/lww.0000000000001000",
"volume": "72"
}