Metformin at the time of Covid-19 infection and risk of Long Covid: A Target Trial Emulation Study
et al., Authorea Inc., doi:10.22541/au.176418398.81170401/v1, Nov 2025
Metformin for COVID-19
3rd treatment shown to reduce risk in
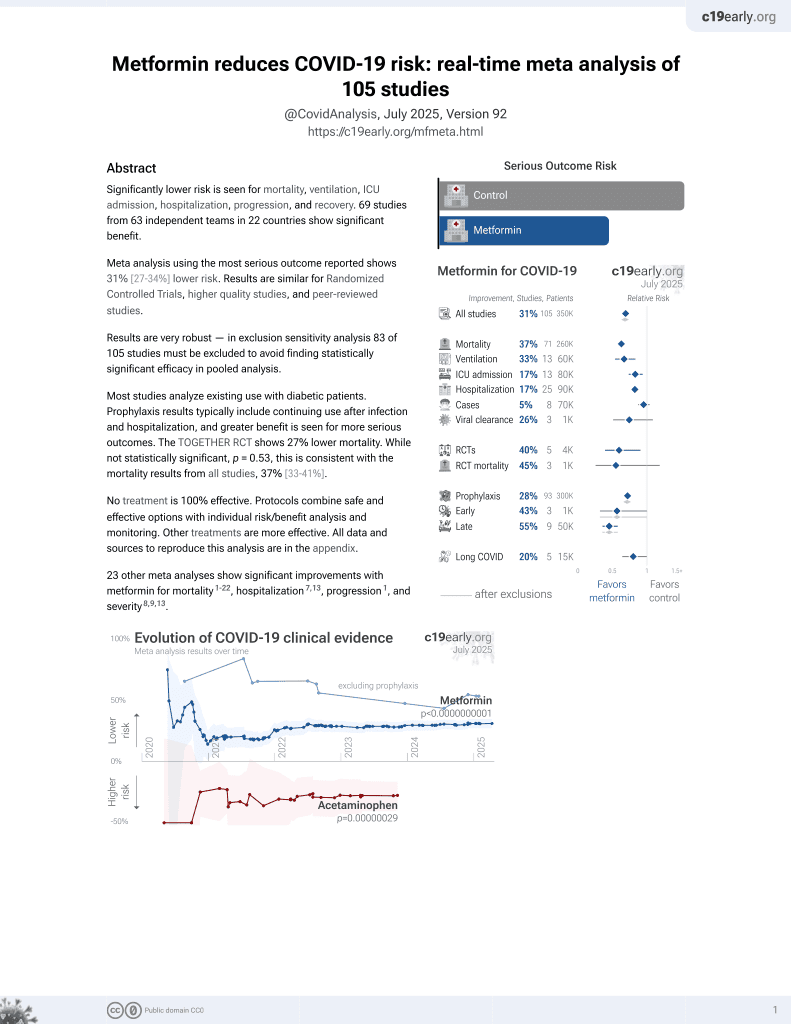
July 2020, now with p < 0.00000000001 from 112 studies.
Lower risk for mortality, ventilation, ICU, hospitalization, progression, recovery, and viral clearance.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
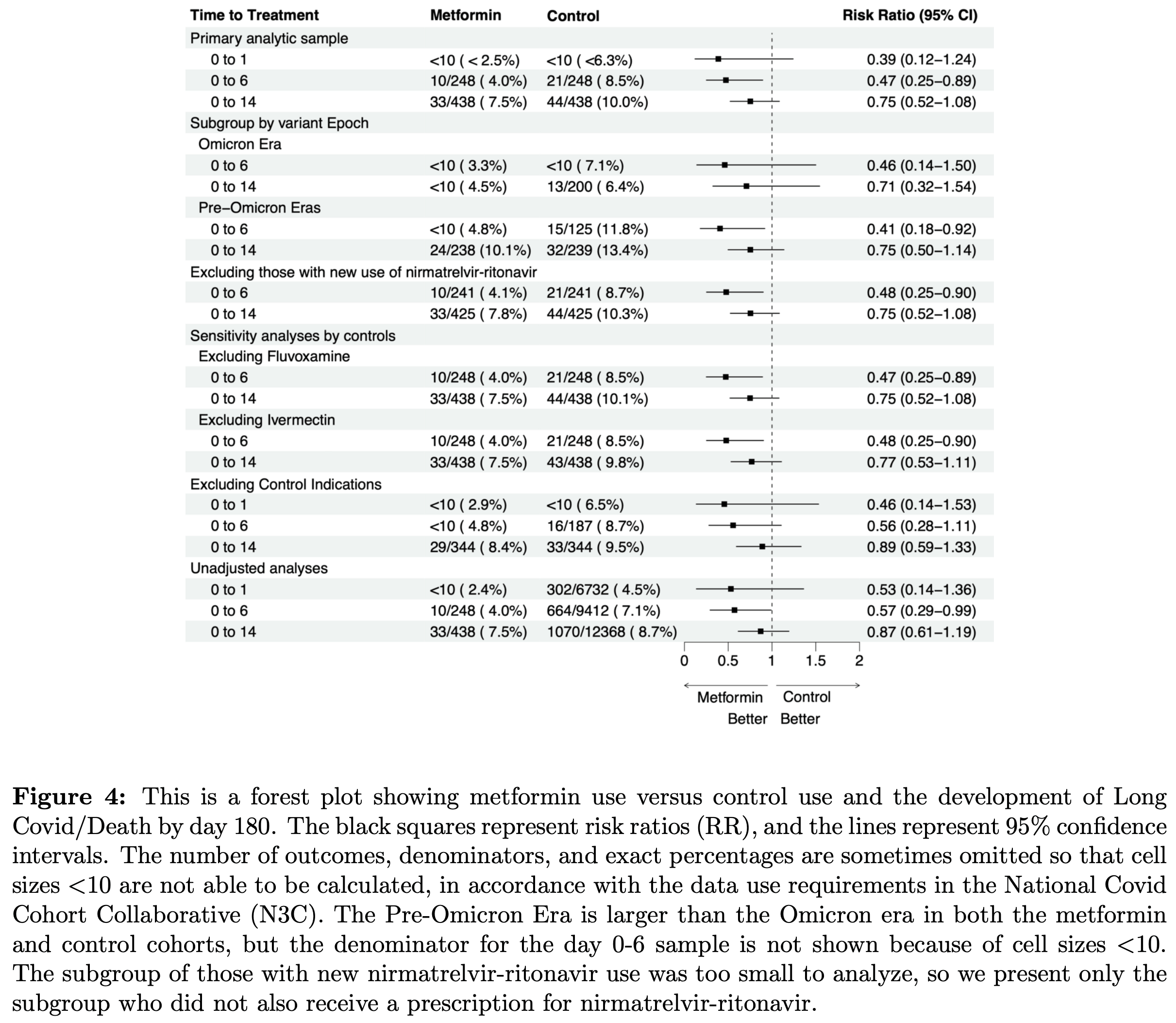
Target trial emulation of 496 outpatients in the N3C nationwide EHR database, showing lower risk of long COVID or death with metformin prescribed within 6 days of documented SARS-CoV-2 infection.
Low event count: the primary result depends on just 10 vs. 21 events across 248 weighted patients per group. The estimate is statistically fragile and cannot stably support the many covariates used in entropy balancing, meaning even minor misclassification of a few patients could substantially shift the result.
Healthy-user bias: people who obtained a new metformin prescription for acute COVID without having diabetes represent a specific population - likely well-informed, engaged with emerging evidence, and proactive about health behaviors. The active comparator design helps but cannot fully equalize these unmeasured behavioral differences.
No control medication details: authors do not specify the number of control patients for each of the control medications - data which greatly affects interpretation of the results.
Differential BMI missingness: BMI was missing in 47% of the metformin group vs. 28% of controls. A missingness indicator was included in weighting, but this only works if missingness is random conditional on observed variables. If not, the adjustment is inadequate for a variable strongly associated with long COVID risk.
Implausible results: the nirmatrelvir-excluded subgroup reports a CI of (0.25-0.60) in the text, implausibly narrow compared to the nearly identical primary analysis CI of (0.25-0.89). This is likely a typo for (0.25-0.90) as shown in the forest plot.
Indication asymmetry in exclusions: diabetes was excluded from both groups but asthma and allergic rhinitis (indications for control drugs) were not. Control patients with asthma may have worse respiratory outcomes generally, inflating long COVID rates in the comparator group.
Composite endpoint conflation: the LC/death composite mixes two very different outcomes. If metformin reduced deaths but not long COVID specifically, the composite would still show benefit. LC-only results are not clearly separated.
Reporting inconsistencies: the absolute risk reduction is stated as 4.5% in the results and 4.4% in the discussion. The event composition (1.7% death + 5.5% LC = 7.2%) does not match the reported 7.0% overall incidence.
Overall the major confounding issues could potentially account for the entire observed effect.
Standard of Care (SOC) for COVID-19 in the study country,
the USA, is very poor with very low average efficacy for approved treatments1.
Only expensive, high-profit treatments were approved for early treatment. Low-cost treatments were excluded, reducing the probability of early treatment due to access and cost barriers, and eliminating complementary and synergistic benefits seen with many low-cost treatments.
This study is excluded in the after exclusion results of meta-analysis:
significant unadjusted confounding possible.
|
long COVID or death, 53.0% lower, RR 0.47, p = 0.02, treatment 10 of 248 (4.0%), control 21 of 248 (8.5%), NNT 23, 0 to 6 days from diagnosis.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Bramante et al., 26 Nov 2025, retrospective, USA, preprint, 15 authors.
Abstract: ## Metformin at the time of Covid-19 infection and risk of Long Covid: A Target Trial Emulation Study
Carolyn Bramante 1 , John B Buse 2 , Jared D. Huling 3 , John Buse B 4 , Christopher Lindsell 5 ,
Thomas Stewart 6 , Russell L. Rothman 7 , David Sahner 8 , Sarah E. Dunsmore 8 , Eric Topol 9 , Talia D. Wiggen 10 , Steve Makkar 8 , Andrew Toler 8 , Taylor Estepp 8 , and Steven Johnson 10
1 University of Minnesota Medical School
2 The University of North Carolina at Chapel Hill Gillings School of Global Public Health
3 University of Minnesota Twin Cities Division of Biostatistics
4 The University of North Carolina at Chapel Hill School of Medicine
5 Duke University Department of Biostatistics and Bioinformatics
6 University of Virginia
7 Vanderbilt Institute for Medicine and Public Health
8 National Center for Advancing Translational Sciences
9 The Scripps Research Institute Skaggs Graduate School of Chemical and Biological Sciences
10 University of Minnesota Twin Cities Institute for Health Informatics
November 26, 2025
Abstract
Background : Our objective was to evaluate metformin prescribed at the time of SARS-CoV-2 infection on the risk of developing Long Covid (LC) in electronic health record data. Methods : Weconducted a new user analysis of metformin prescribed within 6 days of documented infection with severe acute respiratory coronavirus syndrome 2 (SARS-CoV- 2) versus experimental control: prescription for fluvoxamine, fluticasone, ivermectin, or montelukast. Inclusion criteria: a clinic visit in the 0- 6 months and the 6-12 months before infection. Exclusion criteria: metformin or control within 12 months. Primary outcome: LC or death (LC/D), to address death as a competing risk, among patients prescribed drug within Days 0-6 of infection. LC was defined by diagnosis code or computable phenotype. We used entropy balancing to estimate the average treatment effect with a weighted log linear model. Results : After weighting, there were 248 in the metformin and control groups; the average age was 53 (16); 16% were Black; and 16% were Hispanic. In the primary analysis, 10/248 (4.0%) in the metformin group developed LC/D vs. 21/248 (8.5%) in the control group, adjusted risk ratio (aRR) 0.47 (95% CI 0.25 to 0.89). For prescriptions on Days 0-1 relative to infection, aRR was 0.39 (95% CI 0.12-1.24); for prescriptions on Days 0-14 the aRR was 0.75 (95% CI 0.52-1.08). Conclusions : In this observational analysis, metformin prescribed within a week of documented SARS-CoV-2 infection was associated with a 53% lower risk of LC over 6 months than comparator medications. Any risk reduction between 75% to 11% is highly compatible with our data. This analysis of electronic health record diagnoses is important for the reproducibility of clinical trial results that ascertained the same outcome but via participant-report.
Metformin at the time of Covid-19 infection and risk of Long Covid: A Target Trial Emulation Study
Carolyn Bramante, MD, MPH 1 ; Til St¨ urmer, MD, PhD 2 ; Jared D. Huling, PhD 3 ; John B Buse, MD, PhD 4 ; Christopher Lindsell, PhD 5 ; Thomas Stewart, PhD 6 ; Russell L. Rothman, MD, MPP, 7 ; David Sah-
ner, MD 8,11 , Sarah E. Dunsmore, PhD 8 , Eric Topol, MD, 9 Talia D. Wiggen, MPH 10 ; Steve Makkar, PhD 8,11 , Andrew Toler, MS 8,11 , Taylor Estepp, PhD 8,11 , and Steven G. Johnson, PhD 10 on behalf of the N3C Consortium.
1. Division of General Internal Medicine, Department of Medicine, University of MN Medical..
DOI record:
{
"DOI": "10.22541/au.176418398.81170401/v1",
"URL": "http://dx.doi.org/10.22541/au.176418398.81170401/v1",
"accepted": {
"date-parts": [
[
2025,
11,
26
]
]
},
"author": [
{
"ORCID": "https://orcid.org/0000-0001-5858-2080",
"affiliation": [
{
"name": "University of Minnesota Medical School"
}
],
"authenticated-orcid": true,
"family": "Bramante",
"given": "Carolyn",
"sequence": "first"
},
{
"ORCID": "https://orcid.org/0000-0002-9204-7177",
"affiliation": [
{
"name": "The University of North Carolina at Chapel Hill Gillings School of Global Public Health"
}
],
"authenticated-orcid": true,
"family": "Buse",
"given": "John B",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "University of Minnesota Twin Cities Division of Biostatistics"
}
],
"family": "Huling",
"given": "Jared D.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "The University of North Carolina at Chapel Hill School of Medicine"
}
],
"family": "B",
"given": "John Buse",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Duke University Department of Biostatistics and Bioinformatics"
}
],
"family": "Lindsell",
"given": "Christopher",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0002-5138-0758",
"affiliation": [
{
"name": "University of Virginia"
}
],
"authenticated-orcid": true,
"family": "Stewart",
"given": "Thomas",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Vanderbilt Institute for Medicine and Public Health"
}
],
"family": "Rothman",
"given": "Russell L.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "National Center for Advancing Translational Sciences"
}
],
"family": "Sahner",
"given": "David",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "National Center for Advancing Translational Sciences"
}
],
"family": "Dunsmore",
"given": "Sarah E.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "The Scripps Research Institute Skaggs Graduate School of Chemical and Biological Sciences"
}
],
"family": "Topol",
"given": "Eric",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "University of Minnesota Twin Cities Institute for Health Informatics"
}
],
"family": "Wiggen",
"given": "Talia D.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "National Center for Advancing Translational Sciences"
}
],
"family": "Makkar",
"given": "Steve",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "National Center for Advancing Translational Sciences"
}
],
"family": "Toler",
"given": "Andrew",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "National Center for Advancing Translational Sciences"
}
],
"family": "Estepp",
"given": "Taylor",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "University of Minnesota Twin Cities Institute for Health Informatics"
}
],
"family": "Johnson",
"given": "Steven",
"sequence": "additional"
}
],
"container-title": [],
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2025,
11,
26
]
],
"date-time": "2025-11-26T19:06:28Z",
"timestamp": 1764183988000
},
"deposited": {
"date-parts": [
[
2025,
11,
26
]
],
"date-time": "2025-11-26T19:06:28Z",
"timestamp": 1764183988000
},
"group-title": "Preprints",
"indexed": {
"date-parts": [
[
2026,
3,
31
]
],
"date-time": "2026-03-31T22:34:50Z",
"timestamp": 1774996490836,
"version": "3.50.1"
},
"institution": [
{
"name": "Authorea Inc."
}
],
"is-referenced-by-count": 1,
"issued": {
"date-parts": [
[
2025,
11,
26
]
]
},
"member": "311",
"original-title": [],
"posted": {
"date-parts": [
[
2025,
11,
26
]
]
},
"prefix": "10.22541",
"published": {
"date-parts": [
[
2025,
11,
26
]
]
},
"publisher": "Wiley",
"reference-count": 0,
"references-count": 0,
"relation": {},
"resource": {
"primary": {
"URL": "https://www.authorea.com/users/1004233/articles/1364487-metformin-at-the-time-of-covid-19-infection-and-risk-of-long-covid-a-target-trial-emulation-study?commit=311a767f291ba7474f4d7f75d943f42b9328652b"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"subtype": "preprint",
"title": "Metformin at the time of Covid-19 infection and risk of Long Covid: A Target Trial Emulation Study",
"type": "posted-content"
}