HCQ for COVID-19
1st treatment shown to reduce risk in
March 2020, now with p < 0.00000000001 from 424 studies, used in 59 countries.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
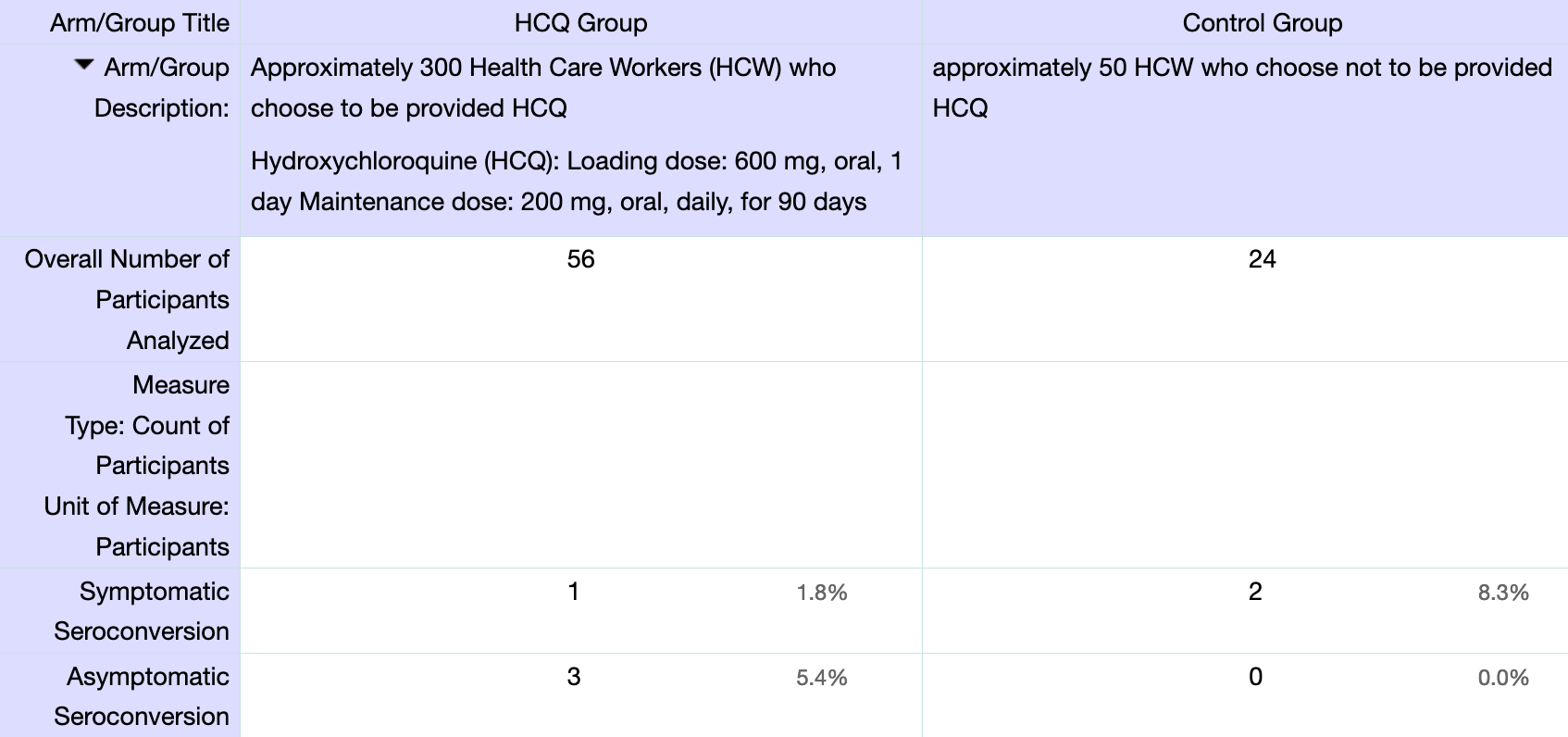
Prospective study of HCQ prophylaxis in the USA, with 56 HCQ patients and 24 control patients, showing no significant differences. NCT04354870 (history)
Standard of Care (SOC) for COVID-19 in the study country,
the USA, is very poor with very low average efficacy for approved treatments1.
Only expensive, high-profit treatments were approved for early treatment. Low-cost treatments were excluded, reducing the probability of early treatment due to access and cost barriers, and eliminating complementary and synergistic benefits seen with many low-cost treatments.
|
risk of symptomatic case, 78.6% lower, RR 0.21, p = 0.21, treatment 1 of 56 (1.8%), control 2 of 24 (8.3%), NNT 15.
|
|
risk of case, 14.3% lower, RR 0.86, p = 1.00, treatment 4 of 56 (7.1%), control 2 of 24 (8.3%), NNT 84.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Belmont et al., 6 Oct 2021, prospective, USA, preprint, 1 author, trial NCT04354870 (history).