The efficacy and safety of hydroxychloroquine at different doses and courses for COVID-19 prevention: a systematic review and network meta-analysis
et al., Expert Review of Anti-infective Therapy, doi:10.1080/14787210.2024.2413419, PROSPERO CRD42024559057, Oct 2024
HCQ for COVID-19
1st treatment shown to reduce risk in
March 2020, now with p < 0.00000000001 from 424 studies, used in 59 countries.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
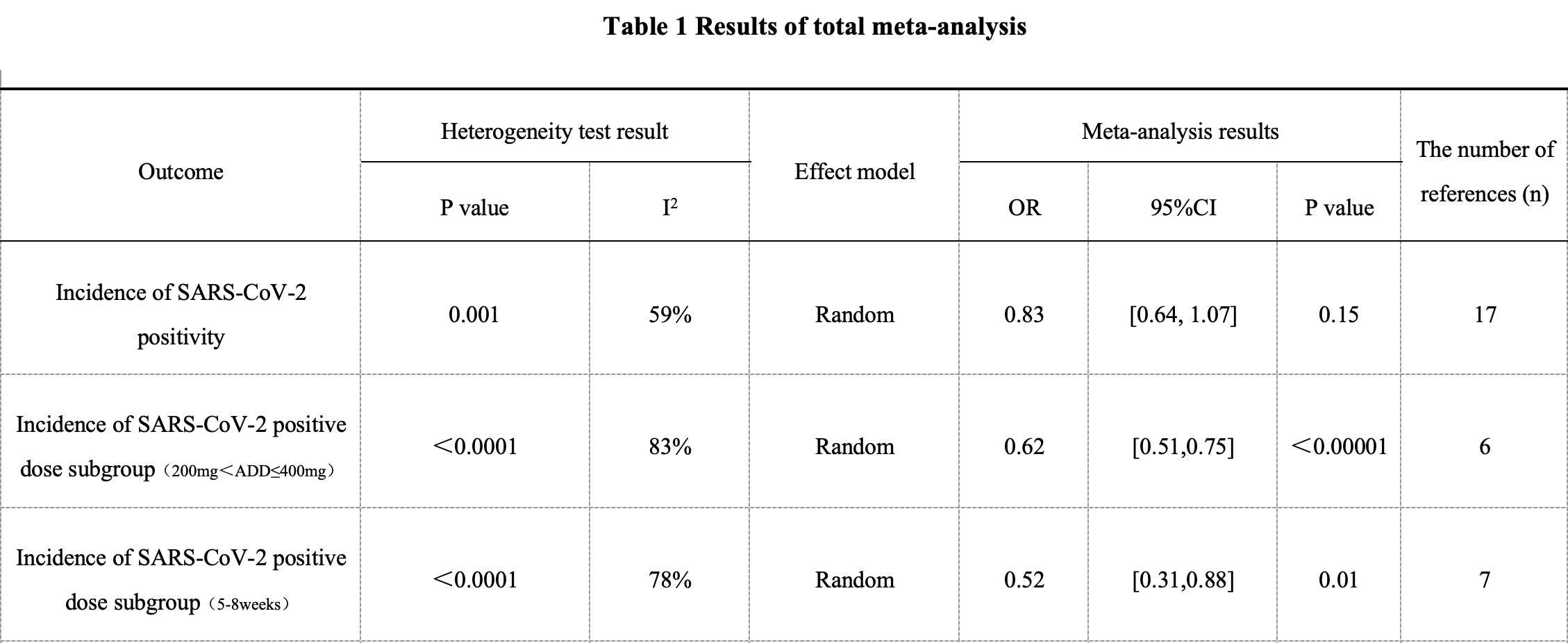
Meta-analysis of 20 RCTs with 12,372 participants showing lower SARS-CoV-2 cases with HCQ without statistical significance overall, but with significance for 200-400mg daily, and for 5-8 weeks duration. Authors note that only 23 of 263 registered trials were published. Note that Shabani et al. has multiple data issues and is a quasi-experimental study.
11 meta-analyses show significant improvements with hydroxychloroquine for mortality2-6,
hospitalization2,
recovery7,
combined death/hospitalization/cases8,
cases9-11, and
viral clearance12.
Currently there are 38 HCQ for COVID-19 early treatment studies, showing 76% lower mortality [61‑85%], 67% lower ventilation [-710‑99%], 31% lower ICU admission [1‑53%], and 42% lower hospitalization [29‑52%].
1.
Shabani et al., Evaluation of the Prophylactic Effect of Hydroxychloroquine on People in Close-Contact with Patients with Covid-19, Pulmonary Pharmacology & Therapeutics, doi:10.1016/j.pupt.2021.102069.
2.
Landsteiner de Sampaio Amêndola et al., COVID-19 Infection in Rheumatic Patients on Chronic Antimalarial Drugs: A Systematic Review and Meta-Analysis, Journal of Clinical Medicine, doi:10.3390/jcm11226865.
3.
Risch, H., Early Outpatient Treatment of Symptomatic, High-Risk Covid-19 Patients that Should be Ramped-Up Immediately as Key to the Pandemic Crisis, American Journal of Epidemiology, kwaa093, 27 May 2020, doi:10.1093/aje/kwaa093.
4.
Risch (B), H., Response to: “Early Outpatient Treatment of Symptomatic, High-Risk Covid-19 Patients” and “Re: Early Outpatient Treatment of Symptomatic, High-Risk Covid-19 Patients that Should be Ramped-Up Immediately as Key to the Pandemic Crisis”, American Journal of Epidemiology, July 20, 2020, doi:10.1093/aje/kwaa152.
5.
Stricker et al., Hydroxychloroquine Pre-Exposure Prophylaxis for COVID-19 in Healthcare Workers from India: A Meta-Analysis, Journal of Infection and Public Health, doi:10.1016/j.jiph.2021.08.001.
6.
Amal et al., Exploring the long-term hydroxychloroquine’s effects on COVID-19 outcomes in patients with autoimmune diseases: a systematic review and meta-analysis, European Journal of Clinical Pharmacology, doi:10.1007/s00228-026-04066-y.
7.
Prodromos et al., Hydroxychloroquine is effective, and consistently so used early, for Covid-19: A systematic review, New Microbes and New Infections, doi:10.1016/j.nmni.2020.100776.
8.
Ladapo et al., Randomized Controlled Trials of Early Ambulatory Hydroxychloroquine in the Prevention of COVID-19 Infection, Hospitalization, and Death: Meta-Analysis, medRxiv, doi:10.1101/2020.09.30.20204693.
9.
García-Albéniz et al., Systematic review and meta-analysis of randomized trials of hydroxychloroquine for the prevention of COVID-19, European Journal of Epidemiology, doi:10.1007/s10654-022-00891-4.
10.
Han et al., The efficacy and safety of hydroxychloroquine for COVID-19 prophylaxis and clinical assessment: an updated meta-analysis of randomized trials, Journal of Thoracic Disease, doi:10.21037/jtd-23-1043.
Wang et al., 4 Oct 2024, China, peer-reviewed, 6 authors, trial PROSPERO CRD42024559057.
Contact: zhanzk0424@163.com.
The efficacy and safety of hydroxychloroquine at different doses and courses for COVID-19 prevention: a systematic review and network meta-analysis
Expert Review of Anti-infective Therapy, doi:10.1080/14787210.2024.2413419
Background: The optimal strategy for using hydroxychloroquine to prevent coronavirus disease 2019 in patients, either before or after exposure, remains unclear. We evaluated the safety and efficacy of different doses and treatment durations of hydroxychloroquine for COVID-19 prevention. Method: Databases including PubMed, Web of Science, Cochrane Library, EMBASE, Medline, and ClinicalTrials.gov were systematically searched for randomized controlled trials (RCTs) comparing different doses of hydroxychloroquine for COVID-19 prevention, from their inception to February 2024. Results: A total of 20 RCTs involving 12,372 patients were included. Meta-analysis results showed no significant difference between the hydroxychloroquine and control groups in reducing the incidence of syndrome coronavirus type 2 (SARS-CoV-2) positivity (OR = 0.83, 95% CI = 0.67, 1.03). However, the subgroup receiving a daily dose of 200-400 mg (OR = 0.62, 95% CI = 0.51, 0.75) and a treatment duration of 5-8 weeks (OR = 0.52, 95% CI = 0.31, 0.88) showed statistically significant reductions in SARS-CoV-2 positivity. According to the surface under the cumulative ranking curve (SUCRA), the most effective intervention was a 200-400 mg dose for 5-8 weeks. Conclusions: A hydroxychloroquine dose of 200-400 mg for a duration of 5-8 weeks may moderately reduce the risk of COVID-19 with a relatively low risk of adverse events.
Declaration of interest The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
Author contributions Hang Wang, Qiyuan Yang, and Zhikun Zhan conceptualized and designed the study. Hang Wang, Qiyuan Yang, Yinjun Mao, and Pinfang Huang collected the data. Haibo Ding and Zhikun Zhan performed the data analyses. Hang Wang checked the statistical consistency. Qiyuan Yang and Zhikun Zhan contributed to data interpretation and drafted the manuscript. Yinjun Mao, Haibo Ding, Pinfang Huang, and Zhikun Zhan critically reviewed the manuscript. All authors have agreed on the submission of the final version of the manuscript for publication.
References
Abella, Jolkovsky, Biney, Efficacy and safety of hydroxychloroquine vs placebo for pre-exposure SARS-CoV-2 prophylaxis among health care workers: a randomized clinical trial, JAMA Intern Med, doi:10.1001/jamainternmed.2020.6319
Ader, Peiffer-Smadja, Poissy, An open-label randomized controlled trial of the effect of lopinavir/ritonavir, lopinavir/ritonavir plus ifn-β-1a and hydroxychloroquine in hospitalized patients with COVID-19, Clin Microbiol Infect, doi:10.1016/j.cmi.2021.05.020
Barnabas, Brown, Bershteyn, Efficacy of hydroxychloroquine for post-exposure prophylaxis to prevent severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection among adults exposed to coronavirus disease (COVID-19): a structured summary of a study protocol for a randomised controlled trial, Trials, doi:10.1186/s13063-020-04446-4
Barnabas, Brown, Bershteyn, Hydroxychloroquine as postexposure prophylaxis to prevent severe acute respiratory syndrome coronavirus 2 infection: a randomized trial, Ann Intern Med, doi:10.7326/m20-6519
Boulware, Pullen, Bangdiwala, A randomized trial of hydroxychloroquine as postexposure prophylaxis for covid-19, N Engl J Med, doi:10.1056/NEJMoa2016638
Chi, Ko, Yang, Addition of rapamycin and hydroxychloroquine to metronomic chemotherapy as a second line treatment results in high salvage rates for refractory metastatic solid tumors: a pilot safety and effectiveness analysis in a small patient cohort, Oncotarget, doi:10.18632/oncotarget.3793
De Wit, Van Doremalen, Falzarano, SARS and MERS: recent insights into emerging coronaviruses, Nat Rev Microbiol, doi:10.1038/nrmicro.2016.81
Devaux, Rolain, Colson, New insights on the antiviral effects of chloroquine against coronavirus: what to expect for COVID-19?, Int J Antimicrob Agents, doi:10.1016/j.ijantimicag.2020.105938
Dhibar, Arora, Chaudhary, The 'myth of hydroxychloroquine (HCQ) as post-exposure prophylaxis (PEP) for the prevention of COVID-19' is far from reality, Sci Rep, doi:10.1038/s41598-022-26053-w
García-Albéniz, Amo, Polo, Systematic review and meta-analysis of randomized trials of hydroxychloroquine for the prevention of COVID-19, Eur J Epidemiol, doi:10.1007/s10654-022-00891-4
Grau-Pujol, Camprubí-Ferrer, Marti-Soler, Pre-exposure prophylaxis with hydroxychloroquine for COVID-19: a double-blind, placebo-controlled randomized clinical trial, Trials, doi:10.1186/s13063-021-05758-9
Guan, Ni, Hu, Clinical characteristics of coronavirus disease 2019 in China, N Engl J Med, doi:10.1056/NEJMoa2002032
Hong, Friedland, Hu, Safety and efficacy of hydroxychloroquine as prophylactic against COVID-19 in healthcare workers: a meta-analysis of randomised clinical trials, BMJ Open, doi:10.1136/bmjopen-2022-065305
Hutton, Salanti, Caldwell, The PRISMA extension statement for reporting of systematic reviews incorporating network meta-analyses of health care interventions: checklist and explanations, Ann Intern Med, doi:10.7326/m14-2385
Khan, Adil, Alkhathlan, COVID-19: a global challenge with old history, epidemiology and progress so far, Molecules, doi:10.3390/molecules26010039
Lewis, Chaudhuri, Alshamsi, The efficacy and safety of hydroxychloroquine for COVID-19 prophylaxis: a systematic review and meta-analysis of randomized trials, PLOS ONE, doi:10.1371/journal.pone.0244778
Li, Wang, Tian, COVID-19 vaccine development: milestones, lessons and prospects, Signal Transduct Target Ther, doi:10.1038/s41392-022-00996-y
Liu, Luo, Lv, Effect of hydroxychloroquine blood concentration on the efficacy and ocular toxicity of systemic lupus erythematosus, Sci Rep, doi:10.1038/s41598-024-58391-2
Llanos-Cuentas, Schwalb, Quintana, Hydroxychloroquine to prevent SARS-CoV-2 infection among healthcare workers: early termination of a phase 3, randomised, open-label, controlled clinical trial, BMC Res Notes, doi:10.1186/s13104-023-06281-7
Lother, Abassi, Agostinis, Post-exposure prophylaxis or pre-emptive therapy for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2): study protocol for a pragmatic randomized-controlled trial, Can J Anaesth, doi:10.1007/s12630-020-01684-7
Mckinnon, Wang, Zervos, Safety and tolerability of hydroxychloroquine in health care workers and first responders for the prevention of COVID-19: WHIP COVID-19 study, Int J Infect Dis, doi:10.1016/j.ijid.2021.12.343
Mitjà, Corbacho-Monné, Ubals, A cluster-randomized trial of hydroxychloroquine for prevention of covid-19, N Engl J Med, doi:10.1056/NEJMoa2021801
Naggie, Milstone, Castro, Hydroxychloroquine for pre-exposure prophylaxis of COVID-19 in health care workers: a randomized, multicenter, placebo-controlled trial healthcare worker exposure response and outcomes of Hydroxychloroquine (HERO-HCQ), Int J Infect Dis, doi:10.1016/j.ijid.2023.01.019
Nct, The PATCH trial (prevention and treatment of COVID-19 with hydroxychloroquine)
Polo, García-Albéniz, Terán, Daily tenofovir disoproxil fumarate/emtricitabine and hydroxychloroquine for pre-exposure prophylaxis of COVID-19: a double-blind placebo-controlled randomized trial in healthcare workers, Clin Microbiol Infect, doi:10.1016/j.cmi.2022.07.006
Rajasingham, Bangdiwala, Nicol, Hydroxychloroquine as pre-exposure prophylaxis for coronavirus disease 2019 (COVID-19) in healthcare workers: a randomized trial, Clin Infect Dis, doi:10.1093/cid/ciaa1571
Ramatillah, Gan, Novarticia, Side effects of CoronaVac ® COVID-19 vaccination: investigation in North Jakarta district public health center communities in Indonesia, Heliyon, doi:10.1016/j.heliyon.2024.e30087
Rojas-Serrano, Portillo-Vásquez, Thirion-Romero, Hydroxychloroquine for prophylaxis of COVID-19 in health workers: a randomized clinical trial, PLOS ONE, doi:10.1371/journal.pone.0261980
Sabnis, Novel papain-like protease inhibitors for treating viral infections, in particular, coronavirus infections, ACS Med Chem Lett, doi:10.1021/acsmedchemlett.4c00331
Seet, Quek, Ooi, Positive impact of oral hydroxychloroquine and povidone-iodine throat spray for COVID-19 prophylaxis: an open-label randomized trial, Int J Infect Dis, doi:10.1016/j.ijid.2021.04.035
Shabani, Totonchi, Rezaeimirghaed, Evaluation of the prophylactic effect of hydroxychloroquine on people in close-contact with patients with COVID-19, Pulm Pharmacol Ther, doi:10.1016/j.pupt.2021.102069
Shahbazi, Moradi, Mollasalehi, Unravelling the diagnostic methodologies for SARS-CoV-2; the indispensable need for developing point-of-care testing, Talanta, doi:10.1016/j.talanta.2024.126139
Shi, Zhou, Wang, Potential effectiveness and safety of antiviral agents in children with coronavirus disease 2019: a rapid review and meta-analysis, Ann Transl Med, doi:10.21037/atm-20-3301
Singh, Ryan, Kredo, Chloroquine or hydroxychloroquine for prevention and treatment of COVID-19, Cochrane Database Syst Rev, doi:10.1002/14651858.CD013587.pub2
Siordia, Bernaba, Yoshino, Systematic and statistical review of coronavirus disease 19 treatment trials, SN Compr Clin Med, doi:10.1007/s42399-020-00399-6
Skarzynska, Matusiak, Skarzynski, Adverse audio-vestibular effects of drugs and vaccines used in the treatment and prevention of COVID-19: a review, Audiol Res, doi:10.3390/audiolres12030025
Song, Deng, Wang, Naphthoquine: a potent broad-spectrum anti-coronavirus drug in vitro, Molecules, doi:10.3390/molecules27030712
Vijayaraghavan, Jha, Rajbhandari, Hydroxychloroquine plus personal protective equipment versus personal protective equipment alone for the prevention of laboratory-confirmed COVID-19 infections among healthcare workers: a multicentre, parallel-group randomised controlled trial from India, BMJ Open, doi:10.1136/bmjopen-2021-059540
Walbi, Alqhtani, Alqahtani, The relationship between hydroxychloroquine plasma concentration and COVID-19 outcomes in rheumatoid arthritis patients in Saudi Arabia, Saudi Pharm J, doi:10.1016/j.jsps.2022.05.006
Wang, Yang, The therapeutic potential of natural dietary flavonoids against SARS-CoV-2 infection, Nutrients, doi:10.3390/nu15153443
Worldmeter, COVID live update
Yang, Deng, Liu, Population pharmacokinetics of long-term hydroxychloroquine therapy in patients with rheumatic diseases, Br J Clin Pharmacol, doi:10.1111/bcp.16257
Zhai, Ji, Cai, Physiologically-based pharmacokinetics modeling for Hydroxychloroquine as a treatment for malaria and optimized dosing regimens for different populations, J Pers Med, doi:10.3390/jpm12050796
Zhou, Verweij, Bijlsma, Repurposed drug studies on the primary prevention of SARS-CoV-2 infection during the pandemic: systematic review and meta-analysis, BMJ Open Respir Res, doi:10.1136/bmjresp-2023-001674
Zohra, Nassima, Hydroxychloroquine serum concentration in coronavirus disease 2019 (COVID-19) patients: a retrospective study, Forensic Sci Res, doi:10.1080/20961790.2021.1936896
DOI record:
{
"DOI": "10.1080/14787210.2024.2413419",
"ISSN": [
"1478-7210",
"1744-8336"
],
"URL": "http://dx.doi.org/10.1080/14787210.2024.2413419",
"alternative-id": [
"10.1080/14787210.2024.2413419"
],
"assertion": [
{
"label": "Peer Review Statement",
"name": "peerreview_statement",
"order": 1,
"value": "The publishing and review policy for this title is described in its Aims & Scope."
},
{
"URL": "http://www.tandfonline.com/action/journalInformation?show=aimsScope&journalCode=ierz20",
"label": "Aim & Scope",
"name": "aims_and_scope_url",
"order": 2,
"value": "http://www.tandfonline.com/action/journalInformation?show=aimsScope&journalCode=ierz20"
},
{
"group": {
"label": "Publication History",
"name": "publication_history"
},
"label": "Received",
"name": "received",
"order": 0,
"value": "2024-06-21"
},
{
"group": {
"label": "Publication History",
"name": "publication_history"
},
"label": "Revised",
"name": "revised",
"order": 1,
"value": "2024-09-30"
},
{
"group": {
"label": "Publication History",
"name": "publication_history"
},
"label": "Accepted",
"name": "accepted",
"order": 2,
"value": "2024-10-03"
},
{
"group": {
"label": "Publication History",
"name": "publication_history"
},
"label": "Published",
"name": "published",
"order": 3,
"value": "2024-10-04"
}
],
"author": [
{
"affiliation": [
{
"name": "Department of Pharmacy, the First Affiliated Hospital, Fujian Medical University, Fuzhou, China"
},
{
"name": "Department of Pharmacy, National Regional Medical Center, Binhai Campus of the First Affiliated Hospital, Fujian Medical University, Fuzhou, China"
}
],
"family": "Hang",
"given": "Wang",
"sequence": "first"
},
{
"affiliation": [
{
"name": "Department of Pharmacy, the First Affiliated Hospital, Fujian Medical University, Fuzhou, China"
},
{
"name": "Department of Pharmacy, National Regional Medical Center, Binhai Campus of the First Affiliated Hospital, Fujian Medical University, Fuzhou, China"
}
],
"family": "Qiyuan",
"given": "Yang",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Pharmacy, the First Affiliated Hospital, Fujian Medical University, Fuzhou, China"
},
{
"name": "Department of Pharmacy, National Regional Medical Center, Binhai Campus of the First Affiliated Hospital, Fujian Medical University, Fuzhou, China"
}
],
"family": "Yinjun",
"given": "Mao",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Respiratory and Critical Care Medicine, the First Affiliated Hospital, Fujian Medical University, Fuzhou, China"
}
],
"family": "Haibo",
"given": "Ding",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Pharmacy, the First Affiliated Hospital, Fujian Medical University, Fuzhou, China"
},
{
"name": "Department of Pharmacy, National Regional Medical Center, Binhai Campus of the First Affiliated Hospital, Fujian Medical University, Fuzhou, China"
}
],
"family": "Pinfang",
"given": "Huang",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Pharmacy, the First Affiliated Hospital, Fujian Medical University, Fuzhou, China"
},
{
"name": "Department of Pharmacy, National Regional Medical Center, Binhai Campus of the First Affiliated Hospital, Fujian Medical University, Fuzhou, China"
}
],
"family": "Zhikun",
"given": "Zhan",
"sequence": "additional"
}
],
"container-title": "Expert Review of Anti-infective Therapy",
"container-title-short": "Expert Review of Anti-infective Therapy",
"content-domain": {
"crossmark-restriction": true,
"domain": [
"www.tandfonline.com"
]
},
"created": {
"date-parts": [
[
2024,
10,
4
]
],
"date-time": "2024-10-04T16:16:21Z",
"timestamp": 1728058581000
},
"deposited": {
"date-parts": [
[
2024,
10,
4
]
],
"date-time": "2024-10-04T16:16:26Z",
"timestamp": 1728058586000
},
"funder": [
{
"DOI": "10.13039/501100001809",
"award": [
"82304604"
],
"doi-asserted-by": "publisher",
"id": [
{
"asserted-by": "publisher",
"id": "10.13039/501100001809",
"id-type": "DOI"
}
],
"name": "National Natural Science Foundation of China"
},
{
"award": [
"2022GGB002"
],
"name": "the Fujian Provincial Health Technology Project"
},
{
"award": [
"2023Y9034"
],
"name": "the Joint Funds for the Innovation of Science and Technology"
}
],
"indexed": {
"date-parts": [
[
2024,
10,
5
]
],
"date-time": "2024-10-05T04:18:42Z",
"timestamp": 1728101922440
},
"is-referenced-by-count": 0,
"issued": {
"date-parts": [
[
2024,
10,
4
]
]
},
"language": "en",
"link": [
{
"URL": "https://www.tandfonline.com/doi/pdf/10.1080/14787210.2024.2413419",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "301",
"original-title": [],
"prefix": "10.1080",
"published": {
"date-parts": [
[
2024,
10,
4
]
]
},
"published-online": {
"date-parts": [
[
2024,
10,
4
]
]
},
"publisher": "Informa UK Limited",
"reference": [
{
"DOI": "10.3390/nu15153443",
"doi-asserted-by": "publisher",
"key": "e_1_3_6_2_1"
},
{
"DOI": "10.1056/NEJMoa2002032",
"doi-asserted-by": "publisher",
"key": "e_1_3_6_3_1"
},
{
"DOI": "10.1038/nrmicro.2016.81",
"doi-asserted-by": "publisher",
"key": "e_1_3_6_4_1"
},
{
"DOI": "10.3390/molecules26010039",
"doi-asserted-by": "publisher",
"key": "e_1_3_6_5_1"
},
{
"key": "e_1_3_6_6_1",
"unstructured": "WORLDMETER. COVID live update [EB/OL]. (2024-05-12). https://www. worldometers. info/coronavirus/"
},
{
"DOI": "10.1016/j.talanta.2024.126139",
"doi-asserted-by": "publisher",
"key": "e_1_3_6_7_1"
},
{
"DOI": "10.1016/j.heliyon.2024.e30087",
"doi-asserted-by": "publisher",
"key": "e_1_3_6_8_1"
},
{
"DOI": "10.1038/s41392-022-00996-y",
"doi-asserted-by": "publisher",
"key": "e_1_3_6_9_1"
},
{
"DOI": "10.1021/acsmedchemlett.4c00331",
"doi-asserted-by": "publisher",
"key": "e_1_3_6_10_1"
},
{
"DOI": "10.3390/molecules27030712",
"doi-asserted-by": "publisher",
"key": "e_1_3_6_11_1"
},
{
"DOI": "10.3390/audiolres12030025",
"doi-asserted-by": "publisher",
"key": "e_1_3_6_12_1"
},
{
"DOI": "10.21037/atm-20-3301",
"doi-asserted-by": "publisher",
"key": "e_1_3_6_13_1"
},
{
"DOI": "10.1007/s10654-022-00891-4",
"doi-asserted-by": "publisher",
"key": "e_1_3_6_14_1"
},
{
"DOI": "10.1136/bmjopen-2022-065305",
"doi-asserted-by": "publisher",
"key": "e_1_3_6_15_1"
},
{
"DOI": "10.1371/journal.pone.0244778",
"doi-asserted-by": "publisher",
"key": "e_1_3_6_16_1"
},
{
"DOI": "10.1002/14651858.CD013587.pub2",
"doi-asserted-by": "publisher",
"key": "e_1_3_6_17_1"
},
{
"DOI": "10.1136/bmjresp-2023-001674",
"doi-asserted-by": "publisher",
"key": "e_1_3_6_18_1"
},
{
"DOI": "10.7326/m14-2385",
"doi-asserted-by": "publisher",
"key": "e_1_3_6_19_1"
},
{
"DOI": "10.1001/jamainternmed.2020.6319",
"doi-asserted-by": "publisher",
"key": "e_1_3_6_20_1"
},
{
"DOI": "10.1016/j.cmi.2021.05.020",
"doi-asserted-by": "publisher",
"key": "e_1_3_6_21_1"
},
{
"DOI": "10.7326/m20-6519",
"doi-asserted-by": "publisher",
"key": "e_1_3_6_22_1"
},
{
"DOI": "10.1056/NEJMoa2016638",
"doi-asserted-by": "publisher",
"key": "e_1_3_6_23_1"
},
{
"DOI": "10.1038/s41598-022-26053-w",
"doi-asserted-by": "publisher",
"key": "e_1_3_6_24_1"
},
{
"DOI": "10.1186/s13063-021-05758-9",
"doi-asserted-by": "publisher",
"key": "e_1_3_6_25_1"
},
{
"key": "e_1_3_6_26_1",
"unstructured": "Baylor Research Institute. Hydroxychloroquine in the Prevention of COVID-19 Infection in Healthcare Workers [EB/OL]. (2024-05-12). https://classic.clinicaltrials.gov/ct2/show/NCT04333225"
},
{
"DOI": "10.1186/s13063-020-04446-4",
"doi-asserted-by": "publisher",
"key": "e_1_3_6_27_1"
},
{
"DOI": "10.1186/s13104-023-06281-7",
"doi-asserted-by": "publisher",
"key": "e_1_3_6_28_1"
},
{
"DOI": "10.1007/s12630-020-01684-7",
"doi-asserted-by": "publisher",
"key": "e_1_3_6_29_1"
},
{
"DOI": "10.1016/j.ijid.2021.12.343",
"doi-asserted-by": "publisher",
"key": "e_1_3_6_30_1"
},
{
"DOI": "10.1056/NEJMoa2021801",
"doi-asserted-by": "publisher",
"key": "e_1_3_6_31_1"
},
{
"DOI": "10.1016/j.ijid.2023.01.019",
"doi-asserted-by": "publisher",
"key": "e_1_3_6_32_1"
},
{
"key": "e_1_3_6_33_1",
"unstructured": "NCT. The PATCH Trial (Prevention And Treatment of COVID-19 With Hydroxychloroquine) [EB/OL]. (2024-05-14). https://classic.clinicaltrials.gov/ct2/show/NCT04329923"
},
{
"DOI": "10.1016/j.cmi.2022.07.006",
"doi-asserted-by": "publisher",
"key": "e_1_3_6_34_1"
},
{
"DOI": "10.1093/cid/ciaa1571",
"doi-asserted-by": "publisher",
"key": "e_1_3_6_35_1"
},
{
"DOI": "10.1371/journal.pone.0261980",
"doi-asserted-by": "publisher",
"key": "e_1_3_6_36_1"
},
{
"DOI": "10.1016/j.ijid.2021.04.035",
"doi-asserted-by": "publisher",
"key": "e_1_3_6_37_1"
},
{
"DOI": "10.1016/j.pupt.2021.102069",
"doi-asserted-by": "publisher",
"key": "e_1_3_6_38_1"
},
{
"DOI": "10.1136/bmjopen-2021-059540",
"doi-asserted-by": "publisher",
"key": "e_1_3_6_39_1"
},
{
"key": "e_1_3_6_40_1",
"unstructured": "Our World in Data. Ohns Hopkins University CSSE COVID-19 data. [EB/OL]. (2024-05-12). https://ourworldindata.org/explorers/coronavirus-data-explorer"
},
{
"DOI": "10.1016/j.ijantimicag.2020.105938",
"doi-asserted-by": "publisher",
"key": "e_1_3_6_41_1"
},
{
"DOI": "10.18632/oncotarget.3793",
"doi-asserted-by": "publisher",
"key": "e_1_3_6_42_1"
},
{
"DOI": "10.1038/s41598-024-58391-2",
"doi-asserted-by": "publisher",
"key": "e_1_3_6_43_1"
},
{
"DOI": "10.3390/jpm12050796",
"doi-asserted-by": "publisher",
"key": "e_1_3_6_44_1"
},
{
"DOI": "10.1111/bcp.16257",
"doi-asserted-by": "publisher",
"key": "e_1_3_6_45_1"
},
{
"DOI": "10.1080/20961790.2021.1936896",
"doi-asserted-by": "publisher",
"key": "e_1_3_6_46_1"
},
{
"DOI": "10.1016/j.jsps.2022.05.006",
"doi-asserted-by": "publisher",
"key": "e_1_3_6_47_1"
},
{
"DOI": "10.1007/s42399-020-00399-6",
"doi-asserted-by": "publisher",
"key": "e_1_3_6_48_1"
}
],
"reference-count": 47,
"references-count": 47,
"relation": {},
"resource": {
"primary": {
"URL": "https://www.tandfonline.com/doi/full/10.1080/14787210.2024.2413419"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "The efficacy and safety of hydroxychloroquine at different doses and courses for COVID-19 prevention: a systematic review and network meta-analysis",
"type": "journal-article",
"update-policy": "http://dx.doi.org/10.1080/tandf_crossmark_01"
}
