Ensitrelvir for Covid-19 Postexposure Prophylaxis in Household Contacts
et al., New England Journal of Medicine, doi:10.1056/NEJMoa2509306 (results released 3/9/2025), SCORPIO-PEP, NCT05897541, Mar 2025
50th treatment shown to reduce risk in
July 2023, now with p = 0.04 from 9 studies, recognized in 2 countries.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
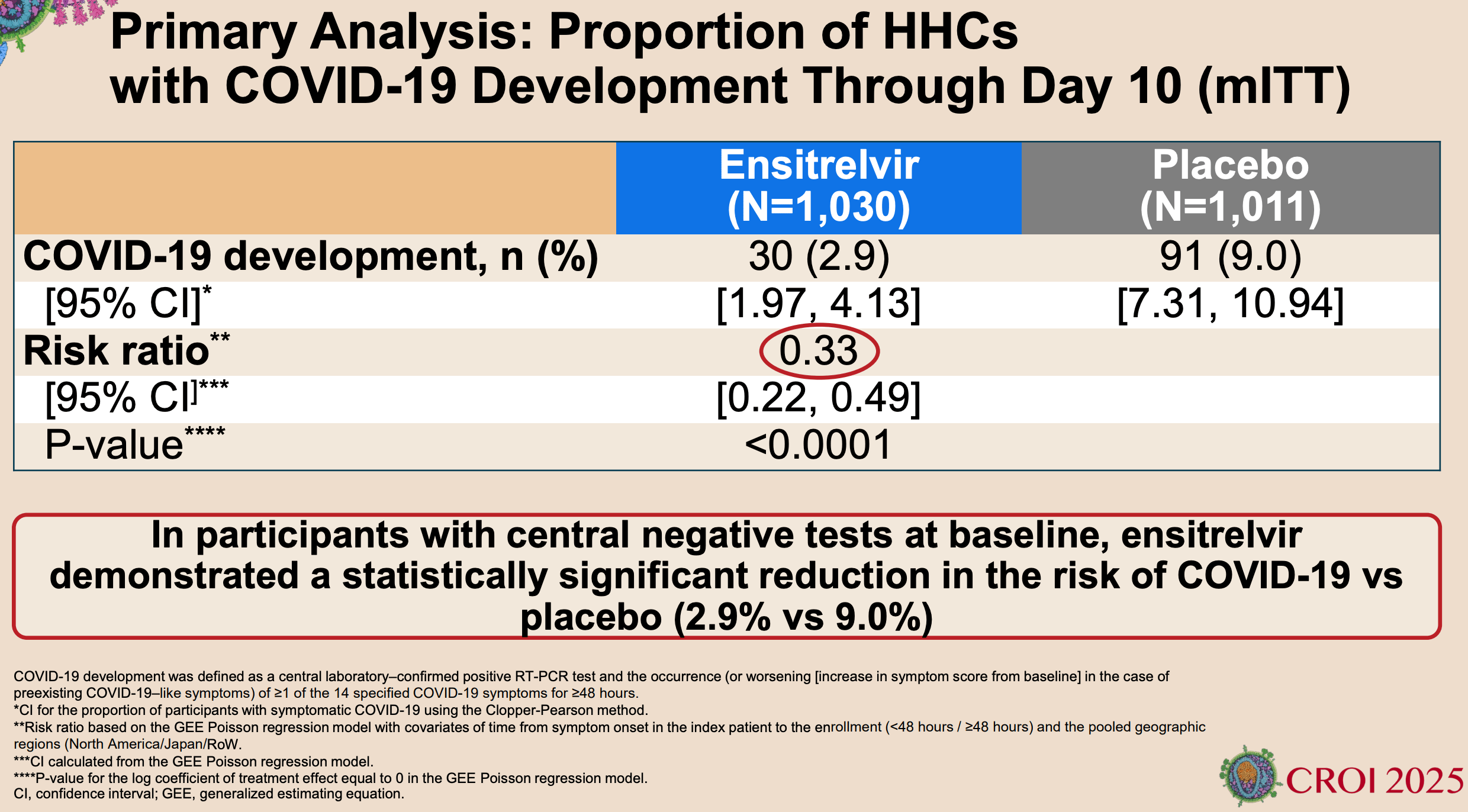
RCT 2,041 household contacts showing significantly lower symptomatic COVID-19 cases with ensitrelvir post-exposure prophylaxis.
Concerns with this trial include:
| Severity | Issue |
| CRITICAL | 1. Potential imputation bias |
| CRITICAL | 2. HHC/patient mismatch |
| CRITICAL | 3. HDL adverse event ascertainment |
| MAJOR | 4. Efficacy in Japan not shown in the US |
| MAJOR | 5. Efficacy for <48 hours from onset |
Responses: authors have not responded to any of these issues.
CRITICALPotential imputation bias.
The protocol states that “missing data will not be imputed” but then specifies imputation in Table 9-1: “For participants with intercurrent events ‘b or c’, the participant will be assumed to have been infected with SARS-CoV-2” - withdrawals “before occurrence of the event for the primary endpoint” are treated as positive COVID-19 events. Imputing dropouts as positive may be reasonable in certain trials but does not make sense for a COVID-19 prophylaxis trial - e.g., consider if there were 5 events and 50 dropouts. Imputation could significantly alter the result. In this trial there were 22 treatment and 35 placebo dropouts. We do not know how many led to imputation, if any (the protocol reports both the imputation and that no missing data were imputed).
CRITICALHHC/patient mismatch.
The distribution of Randomized Household Contacts per Index Patient appears inconsistent. Table S1 shows that out of 1,319 Index Patients, 546 had ≥4 randomized HHCs, 275 had 3, 273 had 2, and 225 had 1. This indicates a minimum of (546*4) + (275*3) + (273*2) + (225*1) = 3,780 randomized HHCs. However, the CONSORT diagram shows only 2,387 randomized HHCs.
CRITICALHDL adverse event ascertainment.
Previous trials reported HDL decreases as the most common TEAE (>30% of patients). In this study, authors acknowledge a 27% median reduction in HDL on day 6 in the treatment group. The protocol specified that HDL-C results would be withheld from study sites/blinded personnel until unblinding because HDL-C decreases could reveal treatment assignment. Authors state that the changes are transient and are not a safety concern. We do not see evidence that HDL values were reported after unblinding and captured as adverse events. Therefore, the headline AE table may not fully account for treatment adverse effects.
MAJOREfficacy in Japan not shown in the US.
Figure 3 shows efficacy in Japan but only a non-significant trend in the US.
MAJOREfficacy for <48 hours from onset.
Figure 3 shows efficacy for <48 hours from onset, but no significant effect for ≥48 hours. In some countries, utility may be reduced by delays involved in accessing prescription treatment.
c19early.org
Ensitrelvir - the best COVID-19 treatment?
May 2026 news portrays ensitrelvir as a new amazing COVID-19 treatment, despite the SCORPIO-PEP study having released results in 2024, and ensitrelvir being approved in Japan in 2022. Data and safety are favorable compared with paxlovid, and ensitrelvir should have been approved in the US. Here we compare the recovery and prophylaxis results for different treatments seen in key trials.
| Paxlovid | Ensitrelvir | Ivermectin | HCQ | |
|---|---|---|---|---|
| Prophylaxis | RR 0.73, p < 0.131 | RR 0.32, p < 0.0012 | HR 0.28, p < 0.0013 |
HR 0.43, p < 0.0014 withheld 820 days
|
| Recovery |
HR 0.83, p < 0.0025 best case use
|
RR 0.95, p = 0.146 best case use
|
HR 0.83, p < 0.0017 worst case: delay, regimen, population withheld 600 days
|
HR 0.76, p = 0.038 late treatment withheld 5 years
|
| Side effects | Significant DDI risks, black-box warning | Significant DDI risks | Relatively low (not zero) | Moderate DDI, off-target risks |
| Cost | $1,3909 | $50010 | <$111 | <$112 |
| Revenue | $28.5B | ~$1.0B | Many low-cost manufacturers | Many low-cost manufacturers |
|
risk of symptomatic case, 67.6% lower, RR 0.32, p < 0.001, treatment 30 of 1,030 (2.9%), control 91 of 1,011 (9.0%), NNT 16.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
1.
Pfizer, Pfizer Shares Top-Line Results from Phase 2/3 EPIC-PEP Study of PAXLOVID™ for Post-Exposure Prophylactic Use, Press Release, www.pfizer.com/news/press-release/press-release-detail/pfizer-shares-top-line-results-phase-23-epic-pep-study.
2.
Hayden et al., Ensitrelvir for Covid-19 Postexposure Prophylaxis in Household Contacts, New England Journal of Medicine, doi:10.1056/NEJMoa2509306.
3.
Desort-Henin et al., The SAIVE Trial, Post-Exposure use of ivermectin in Covid-19 prevention: Efficacy and Safety Results, ECCMID 2023 (results released 1/5/2023), www.medincell.com/wp-content/uploads/2024/03/Poster-SAIVE-April2023-OK3.pdf.
4.
Schilling et al., Evaluation of hydroxychloroquine or chloroquine for the prevention of COVID-19 (COPCOV): A double-blind, randomised, placebo-controlled trial, PLOS Medicine, doi:10.1371/journal.pmed.1004428.
5.
Hammond et al., Oral Nirmatrelvir for High-Risk, Nonhospitalized Adults with Covid-19, New England Journal of Medicine, doi:10.1056/NEJMoa2118542.
6.
Luetkemeyer et al., Ensitrelvir for the Treatment of Nonhospitalized Adults with COVID-19: Results from the SCORPIO-HR, Phase 3, Randomized, Double-blind, Placebo-Controlled Trial, Clinical Infectious Diseases, doi:10.1093/cid/ciaf029.
7.
Hayward et al., Ivermectin for COVID-19 in adults in the community (PRINCIPLE): an open, randomised, controlled, adaptive platform trial of short- and longer-term outcomes, Journal of Infection, doi:10.1016/j.jinf.2024.106130.
8.
Hobbs et al., The PRINCIPLE randomised controlled open label platform trial of hydroxychloroquine for treating COVID19 in community based patients at high risk, Scientific Reports, doi:10.1038/s41598-025-09275-6.
Hayden et al., 9 Mar 2025, Double Blind Randomized Controlled Trial, placebo-controlled, multiple countries, peer-reviewed, 16 authors, trial NCT05897541 (history) (SCORPIO-PEP).
Abstract: ## BACKGROUND
Ensitrelvir, an oral inhibitor of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) 3C-like protease, is approved in Japan for the treatment of mild-tomoderate coronavirus disease 2019 (Covid-19). Previously, no antiviral agents were approved for postexposure prophylaxis in household contacts of patients with Covid-19.
METHODS
In this double-blind, randomized, placebo-controlled trial, we randomly assigned persons who were SARS-CoV-2-negative on local diagnostic testing but were household contacts of a patient with Covid-19 (the index patient) to receive either ensitrelvir (375 mg on day 1 and 125 mg daily on days 2 through 5) or placebo within 72 hours after symptom onset in the index patient. The primary end point was Covid-19 (defined by a central laboratory-confirmed positive reverse-transcriptase-polymerase-chain-reaction assay and the presence of ≥1 of 14 prespecified Covid-19 symptoms lasting ≥48 hours) by day 10 in a household contact in the modified intention-to-treat population (all the participants who underwent randomization, had a central laboratory-confirmed negative RT-PCR test for SARS-CoV-2 at baseline, and received at least one dose of the trial drug or placebo).
RESULTS
The modified intention-to-treat population included 1030 participants in the ensitrelvir group and 1011 in the placebo group. The mean age of the participants was 42.4 years; 71.1% had undergone randomization within 48 hours after symptom onset in the index patient, and 37.0% had at least one risk factor for severe Covid-19. The incidence of Covid-19 was lower in the ensitrelvir group than in the placebo group (2.9% vs. 9.0%; risk ratio, 0.33; 95% confidence interval [CI], 0.22 to 0.49; P<0.001). The incidence of adverse events during the trial was similar in the two groups (15.1% in the ensitrelvir group and 15.5% in the placebo group), as was the incidence of serious adverse events (0.2% in each group). No Covid-19-related hospitalizations or deaths were reported.
CONCLUSIONS
Ensitrelvir administered to household contacts of a patient with Covid-19 within 72 hours after symptom onset in the index patient was effective in preventing Covid-19 in the contacts. (Funded by Shionogi; SCORPIO-PEP Japan Registry for Clinical Trials number, jRCT2031230124; ClinicalTrials.gov number, NCT05897541.)
w
l
Original Article
Ensitrelvir for Covid-19 Postexposure Prophylaxis in Household Contacts
F.G. Hayden, 1 M. Shinkai, 2 T.W. Clark, 3 A.F. Luetkemeyer, 4 P.E. Sax, 5 W.P. Hanage, 6 K.A. Gebo, 7 H. Ikematsu, 8 K. Izumikawa, 9 A. Fukushi, 10 S. Kezbor, 11 H. Sakaguchi, 10 S. Lacey, 12 G. Ichihashi, 10 N. Ohmagari, 13 and T. Uehara, 10 for the SCORPIO-PEP Study Team*
ABSTRACT
d
i
c
i
n
e
The authors' full names, academic degrees, and affiliations are listed at the end of the article. Frederick G. Hayden can be contacted at fgh@ virginia . edu or at the Division of Infectious Diseases and International Health, P.O. Box 801342, University of Virginia Health System, Charlottesville, VA 22908.
*The SCORPIO-PEP study team members are listed in the Supplementary Appendix, available at NEJM.org.
This article was updated on May 14, 2026, at NEJM.org.
N Engl J Med 2026;394:1905-15. DOI: 10.1056/NEJMoa2509306 Copyright © 2026 Massachusetts Medical Society.
A Quick Take is available at NEJM.org
w
l
D espite improved outcomes that have resulted from..
DOI record:
{
"DOI": "10.1056/nejmoa2509306",
"ISSN": [
"0028-4793",
"1533-4406"
],
"URL": "http://dx.doi.org/10.1056/NEJMoa2509306",
"alternative-id": [
"10.1056/NEJMoa2509306"
],
"author": [
{
"affiliation": [
{
"name": "Division of Infectious Diseases and International Health, Department of Medicine, University of Virginia School of Medicine, Charlottesville"
}
],
"family": "Hayden",
"given": "Frederick G.",
"sequence": "first"
},
{
"affiliation": [
{
"name": "Department of Respiratory Medicine, Tokyo Shinagawa Hospital, Tokyo"
}
],
"family": "Shinkai",
"given": "Masaharu",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "School of Clinical and Experimental Sciences, Faculty of Medicine, University of Southampton, Southampton, United Kingdom"
}
],
"family": "Clark",
"given": "Tristan W.",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0003-0911-1578",
"affiliation": [
{
"name": "Division of HIV, Infectious Diseases and Global Medicine, Zuckerberg San Francisco General, University of California, San Francisco, San Francisco"
}
],
"authenticated-orcid": false,
"family": "Luetkemeyer",
"given": "Anne F.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Harvard Medical School and Brigham and Women’s Hospital, Boston"
}
],
"family": "Sax",
"given": "Paul E.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Center for Communicable Disease Dynamics, Harvard T.H. Chan School of Public Health, Boston"
}
],
"family": "Hanage",
"given": "William P.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Medicine, Johns Hopkins University School of Medicine, Baltimore"
}
],
"family": "Gebo",
"given": "Kelly A.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Ricerca Clinica, Fukuoka, Japan"
}
],
"family": "Ikematsu",
"given": "Hideyuki",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Infectious Diseases, Nagasaki University Graduate School of Biomedical Sciences, Nagasaki, Japan"
}
],
"family": "Izumikawa",
"given": "Koichi",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Drug Development and Regulatory Science Division, Shionogi, Osaka, Japan"
}
],
"family": "Fukushi",
"given": "Akimasa",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Clinical Development, Shionogi, Florham Park, NJ"
}
],
"family": "Kezbor",
"given": "Safwan",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Drug Development and Regulatory Science Division, Shionogi, Osaka, Japan"
}
],
"family": "Sakaguchi",
"given": "Hiroki",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Biostatistics, Shionogi, London"
}
],
"family": "Lacey",
"given": "Stuart",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Drug Development and Regulatory Science Division, Shionogi, Osaka, Japan"
}
],
"family": "Ichihashi",
"given": "Genki",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Disease Control and Prevention Center, National Center for Global Health, Tokyo"
}
],
"family": "Ohmagari",
"given": "Norio",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Drug Development and Regulatory Science Division, Shionogi, Osaka, Japan"
}
],
"family": "Uehara",
"given": "Takeki",
"sequence": "additional"
}
],
"container-title": "New England Journal of Medicine",
"container-title-short": "N Engl J Med",
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2026,
5,
13
]
],
"date-time": "2026-05-13T21:00:43Z",
"timestamp": 1778706043000
},
"deposited": {
"date-parts": [
[
2026,
5,
13
]
],
"date-time": "2026-05-13T21:01:02Z",
"timestamp": 1778706062000
},
"funder": [
{
"DOI": "10.13039/501100005612",
"doi-asserted-by": "publisher",
"id": [
{
"asserted-by": "publisher",
"id": "10.13039/501100005612",
"id-type": "DOI"
}
],
"name": "Shionogi"
}
],
"indexed": {
"date-parts": [
[
2026,
5,
13
]
],
"date-time": "2026-05-13T21:15:01Z",
"timestamp": 1778706901286,
"version": "3.51.4"
},
"is-referenced-by-count": 0,
"issue": "19",
"issued": {
"date-parts": [
[
2026,
5,
14
]
]
},
"journal-issue": {
"issue": "19",
"published-print": {
"date-parts": [
[
2026,
5,
14
]
]
}
},
"language": "en",
"license": [
{
"URL": "http://www.nejmgroup.org/legal/terms-of-use.htm",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2026,
5,
14
]
],
"date-time": "2026-05-14T00:00:00Z",
"timestamp": 1778716800000
}
}
],
"link": [
{
"URL": "http://www.nejm.org/doi/pdf/10.1056/NEJMoa2509306",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "150",
"original-title": [],
"page": "1905-1915",
"prefix": "10.1056",
"published": {
"date-parts": [
[
2026,
5,
14
]
]
},
"published-print": {
"date-parts": [
[
2026,
5,
14
]
]
},
"publisher": "Massachusetts Medical Society",
"reference": [
{
"DOI": "10.1016/j.jtbi.2022.111368",
"doi-asserted-by": "publisher",
"key": "e_1_3_5_2_2"
},
{
"DOI": "10.15585/mmwr.mm7112e2",
"doi-asserted-by": "publisher",
"key": "e_1_3_5_3_2"
},
{
"DOI": "10.1007/s12016-022-08921-5",
"doi-asserted-by": "publisher",
"key": "e_1_3_5_4_2"
},
{
"DOI": "10.1038/s41586-024-08511-9",
"doi-asserted-by": "publisher",
"key": "e_1_3_5_5_2"
},
{
"DOI": "10.1001/jamanetworkopen.2022.9317",
"doi-asserted-by": "publisher",
"key": "e_1_3_5_6_2"
},
{
"DOI": "10.1186/s12879-025-10610-5",
"doi-asserted-by": "publisher",
"key": "e_1_3_5_7_2"
},
{
"DOI": "10.1371/journal.pone.0313680",
"doi-asserted-by": "publisher",
"key": "e_1_3_5_8_2"
},
{
"DOI": "10.1038/s41467-024-44973-1",
"doi-asserted-by": "publisher",
"key": "e_1_3_5_9_2"
},
{
"DOI": "10.15585/mmwr.mm7109e1",
"doi-asserted-by": "publisher",
"key": "e_1_3_5_10_2"
},
{
"DOI": "10.1056/NEJMoa1915341",
"doi-asserted-by": "publisher",
"key": "e_1_3_5_11_2"
},
{
"DOI": "10.1001/jama.285.6.748",
"doi-asserted-by": "publisher",
"key": "e_1_3_5_12_2"
},
{
"DOI": "10.1086/381128",
"doi-asserted-by": "publisher",
"key": "e_1_3_5_13_2"
},
{
"DOI": "10.1056/NEJMoa2309002",
"doi-asserted-by": "publisher",
"key": "e_1_3_5_14_2"
},
{
"DOI": "10.1016/j.jinf.2023.08.016",
"doi-asserted-by": "publisher",
"key": "e_1_3_5_15_2"
},
{
"DOI": "10.1016/j.bbrc.2023.01.040",
"doi-asserted-by": "publisher",
"key": "e_1_3_5_16_2"
},
{
"DOI": "10.1093/jac/dkad027",
"doi-asserted-by": "publisher",
"key": "e_1_3_5_17_2"
},
{
"DOI": "10.1016/j.antiviral.2024.105852",
"doi-asserted-by": "publisher",
"key": "e_1_3_5_18_2"
},
{
"key": "e_1_3_5_19_2",
"unstructured": "Shionogi announces approval in Japan of a supplemental indication for Xocova (ensitrelvir fumaric acid) for the post-exposure prophylaxis of Covid-19. News release of Shionogi Osaka Japan March 23 2026 (https://www.shionogi.com/content/dam/shionogi/seu/news/pdf/2026/NP-EU-ENS-0142_%20PEP%20approval%20in%20Japan.pdf)."
},
{
"DOI": "10.1128/aac.00697-22",
"doi-asserted-by": "publisher",
"key": "e_1_3_5_20_2"
},
{
"DOI": "10.1093/cid/ciac933",
"doi-asserted-by": "publisher",
"key": "e_1_3_5_21_2"
},
{
"DOI": "10.1001/jamanetworkopen.2023.54991",
"doi-asserted-by": "publisher",
"key": "e_1_3_5_22_2"
},
{
"DOI": "10.1093/cid/ciaf029",
"doi-asserted-by": "publisher",
"key": "e_1_3_5_23_2"
},
{
"key": "e_1_3_5_24_2",
"unstructured": "CovSPECTRUM. SARS-CoV-2 sequencing data (https://cov-spectrum.org/explore/Japan/AllSamples/Past6M/variants?nextcladePangoLineage=JN.1*&)."
},
{
"DOI": "10.1093/cid/ciab701",
"doi-asserted-by": "publisher",
"key": "e_1_3_5_25_2"
},
{
"DOI": "10.1016/j.jiac.2022.08.010",
"doi-asserted-by": "publisher",
"key": "e_1_3_5_26_2"
},
{
"key": "e_1_3_5_27_2",
"unstructured": "The 70th meeting of the advisory board for countermeasures to SARS-cov-2. Tokyo: Ministry of Health Labour and Welfare 2022."
},
{
"DOI": "10.1016/j.antiviral.2025.106097",
"doi-asserted-by": "publisher",
"key": "e_1_3_5_28_2"
},
{
"key": "e_1_3_5_29_2",
"unstructured": "Global Initiative on Sharing All Influenza Data (GISAID). hCoV-19 (COVID-19) dashboard. 2025 (https://gisaid.org/)."
},
{
"key": "e_1_3_5_30_2",
"unstructured": "National Institute of Infectious Diseases. Amino acid substitutions due to viral genome mutations that may affect the efficacy of therapeutic drugs for the new coronavirus (SARS-CoV-2). 7th ed. 2025. (In Japanese) (https://id-info.jihs.go.jp/relevant-information/covid-19/past-variants/20250131_SARS-CoV-2_mutation_7.pdf)."
}
],
"reference-count": 29,
"references-count": 29,
"relation": {},
"resource": {
"primary": {
"URL": "http://www.nejm.org/doi/10.1056/NEJMoa2509306"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "Ensitrelvir for Covid-19 Postexposure Prophylaxis in Household Contacts",
"type": "journal-article",
"volume": "394"
}
