A Mouthwash with Cetylpyridinium Chloride is reducing salivary SARS-CoV-2 viral load in +COVID-19
et al., Revista Estomatologia, doi:10.25100/re.v31i1.12669, NCT04563689, May 2023
58th treatment shown to reduce risk in
September 2025, now with p = 0.0035 from 4 studies.
Lower risk for recovery and viral clearance.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
RCT 23 patients in Colombia, showing improved viral clearance with cetylpyridinium chloride plus chlorhexidine mouthwash.
Targeted administration to the respiratory tract provides treatment directly
to the typical source of initial SARS-CoV-2 infection and replication, and
allows for rapid onset of action, higher local drug concentration, and reduced systemic side effects (early treatment may be more beneficial).
|
risk of hospitalization, 67.6% lower, RR 0.32, p = 0.48, treatment 0 of 12 (0.0%), control 1 of 11 (9.1%), NNT 11, relative risk is not 0 because of continuity correction due to zero events (with reciprocal of the contrasting arm).
|
|
risk of no viral clearance, 83.3% lower, RR 0.17, p = 0.06, treatment 1 of 12 (8.3%), control 5 of 10 (50.0%), NNT 2.4, day 5.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Rengifo et al., 22 May 2023, Double Blind Randomized Controlled Trial, placebo-controlled, Colombia, peer-reviewed, 13 authors, study period July 2020 - September 2020, average treatment delay 5.5 days, this trial uses multiple treatments in the treatment arm (combined with chlorhexidine) - results of individual treatments may vary, trial NCT04563689 (history).
A Mouthwash with Cetylpyridinium Chloride is reducing salivary SARS-CoV-2 viral load in +COVID-19. Un enjuague con Cloruro de Cetilpiridinio redujo la carga salival del virus SARS CoV-2 en pacientes con Covid-19
doi:10.25100/re.v31i1.12669
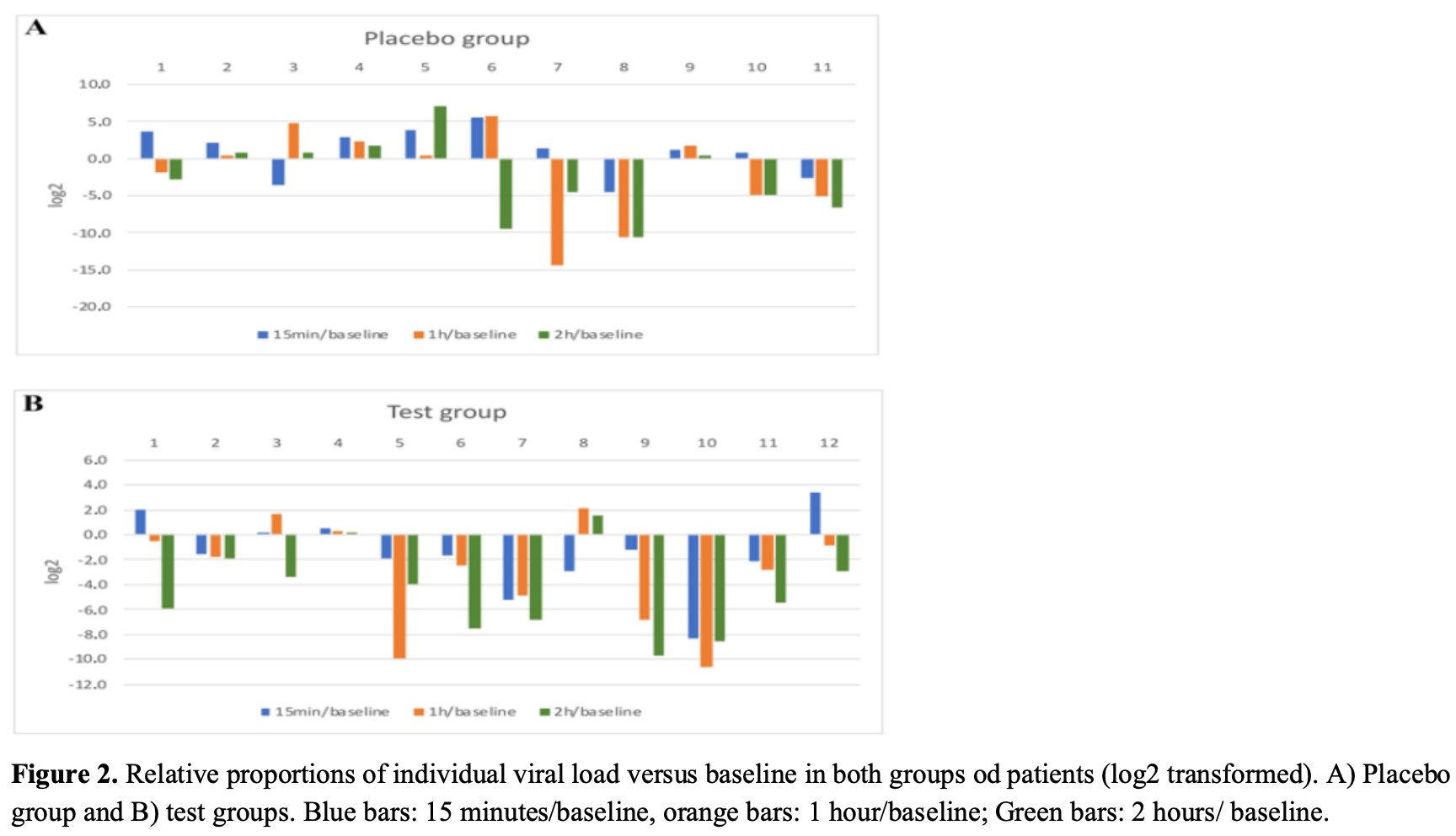
Aim: The aim of this randomised, double-blind, placebo-controlled pilot clinical trial is to evaluate the capacity of a mouthwash to reduce SARS-CoV-2 viral load in the saliva of patients with COVID-19. Methods: Twenty-three symptomatic SARS-CoV-2-positive outpatients were selected and randomised into two groups and registered at NTC 04563689. Both groups rinsed and gargled for one minute with either distilled water (Placebo) or with 0.05% Cetylpyridinium chloride (CPC) plus 0.12% Chlorhexidine (CHX) mouthwash (PERIOAID Intensive Care ). Saliva samples were collected before the use of placebo or mouthwash and after 15 minutes and 1 and 2 hours of either of the above treatment. A saliva sample was also taken five days after regular use of placebo or mouthwash twice daily. The virus was detected by qRT-PCR. Results: A great heterogeneity in the viral load values was observed at baseline in both groups for nasopharyngeal and saliva samples. Most of the patients who used the mouthwash (8/12) had a significant decrease in baseline viral load after 15 min (greater than 99% reduction). This inhibitory effect was maintained for up to two hours in 10 of the 12 patients. At five days, SARS-CoV-2 RNA was detected in only 1 patient from the mouthwash group and in 5 from the placebo group. Conclusions: This study points out that a CPC mouthwash can reduce the viral load in saliva of COVID-positive patients. This finding may be important in transmission control of SARS-CoV-2. Nevertheless, the clinical relevance of CPC mouthwash-reduction on SARS-CoV-2 shedding in saliva requires further study.
KEY WORDS Antiviral mouthwash; SARS COV-2; Covid-19 patients; salivary viral load.
RESMÚMEN Objetivo: El objetivo de este ensayo clínico piloto aleatorizado, doble ciego y controlado con placebo es evaluar la capacidad de un enjuague bucal para reducir la carga viral del SARS-CoV-2 en la saliva de pacientes con COVID-19. Materiales y métodos: Veintitrés pacientes ambulatorios positivos para SARS-CoV-2 sintomáticos fueron seleccionados y aleatorizados en dos grupos y registrados en el NTC 04563689. Ambos grupos se enjuagaron y hicieron gárgaras durante un minuto con agua destilada (placebo) o con cloruro de cetilpiridinio al 0 ,05 % (CPC). ) más enjuague bucal con Clorhexidina (CHX) al 0,12% (PERIOAID Intensive Care ). Se recolectaron muestras de saliva antes del uso de placebo o enjuague bucal y después de 15 minutos y 1 y 2 horas de cualquiera de los tratamientos anteriores. También se tomó una muestra de saliva cinco días después del uso regular de placebo o enjuague bucal dos veces al día. El virus fue detectado por qRT-PCR. Resultados: Se demostró una gran heterogeneidad en los valores de carga viral al inicio del estudio en grupos ambos para muestras de nasofaringe y saliva. La mayoría de los pacientes que usaron el enjuague bucal (8/12) tuvieron una disminución significativa en la carga viral inicial después de 15 minutos (reducción superior al 99 %). Este..
CONFLICT OF INTEREST Authors from Universidad del Valle and from ESE Centro do not have any interest conflict to declare however, the authors associated to Dentaid Research Centre worked within the company that provided the mouthwashes used in this study.
SOURCE OF FUNDING The study was funded by investigators of Universidad del Valle, but mouthwashes and oral hygiene kits for patients were donated by Dentaid company.
References
Alexandersen, Chamings, Bhatta, SARS-CoV-2 genomic and subgenomic RNAs in diagnostic samples are not an indicator of active replication, Nat Commun, doi:10.1038/s41467-020-19883-7
Amirian, Potential fecal transmission of SARS-CoV-2: Current evidence and implications for public health, Int J Infect Dis, doi:10.1016/j.ijid.2020.04.057
Anderson, Sivalingam, Kang, Ananthanarayanan, Arumugam et al., Povidone-Iodine Demonstrates Rapid in vitro Virucidal Activity Against SARS-CoV-2, The Virus Causing COVID-19 Disease, Infect Dis Ther, doi:10.1007/s40121-020-00316-3
Bidra, Pelletier, Westover, Frank, Brown et al., Comparison of in vitro Inactivation of SARS CoV-2 with Hydrogen Peroxide and Povidone-Iodine Oral Antiseptic Rinses, J Prosthodont, doi:10.1111/jopr.13220
Burton, Clarkson, Goulao, Glenny, Mcbain et al., Antimicrobial mouthwashes (gargling) and nasal sprays administered to patients with suspected or confirmed COVID-19 infection to improve patient outcomes and to protect healthcare workers treating them, Cochrane Database Syst Rev
Chau, Lam, Dung, Yen, Minh et al., The natural history and transmission potential of asymptomatic SARS-CoV-2 infection, Clin Infect Dis
Davies, Buczkowski, Welch, Effective in vitro inactivation of SARS-CoV-2 by commercially available mouthwashes, J Gen Virol, doi:10.1099/jgv.0.001578
Ellinger, Bojkova, Zaliani, Cinati, Claussen et al., A SARS-CoV-2 cytopathicity dataset generated by high-content screening of a large drug repurposing collection, Sci Data, doi:10.1038/s41597-021-00848-4
Ferretti, Wymant, Kendall, Quantifying SARS-CoV-2 transmission suggests epidemic control with digital contact tracing, Science, doi:10.1126/science.abb6936
Gottsauner, Michaelides, Schmidt, Scholz, Buchalla et al., A prospective clinical pilot study on the effects of a hydrogen peroxide mouthrinse on the intraoral viral load of SARS-CoV-2, Clin Oral Investig, doi:10.1007/s00784-020-03549-1
He, Zhao, Dong, Zhuang, Song et al., Effects of severe acute respiratory syndrome (SARS) coronavirus infection on peripheral blood lymphocytes and their subsets, Int J Infect Dis
Herrera, Serrano, Roldán, Sanz, Is the oral cavity relevant in SARS-CoV-2 pandemic?, Clin Oral Investig, doi:10.1007/s00784-020-03413-2
Huang, Li, Dunn, He, Taking account of asymptomatic infections: A modeling study of the COVID-19 outbreak on the Diamond Princess cruise ship, PLoS One, doi:10.1371/journal.pone.0248273
Jain, Grover, Singh, Sharma, Das et al., Chlorhexidine: An effective anticovid mouth rinse, J Indian Soc Periodontol, doi:10.4103/jisp.jisp_824_20
Koch-Heier, Hoffmann, Schindler, Lussi, Planz, Inactivation of SARS-CoV-2 through Treatment with the Mouth Rinsing Solutions ViruProX® and BacterX® Pro, Microorganisms
Komine, Yamaguchi, Okamoto, Yamamoto, Virucidal activity of oral care products against SARS-CoV-2 in vitro, J Oral Maxillofac Surg Med Pathol, doi:10.1016/j.ajoms.2021.02.002
Lu, Wang, Sakthivel, Whitaker, Murray et al., Randomized, double-blind, placebo-controlled clinical trial to assess the safety and effectiveness of a novel dual-action oral topical formulation against upper respiratory infections, BMC Infect Dis, doi:10.1186/s12879-016-2177-8
Martínez-Lamas, Diz, Pérez, Campo, Cabrera et al., Is povidone iodine mouthwash effective against SARS-CoV-2? First in vivo tests, Oral Dis, doi:10.1111/odi.13526
Meister, Brüggemann, Todt, Conzelmann, Müller et al., Virucidal Efficacy of Different Oral Rinses Against Severe Acute Respiratory Syndrome Coronavirus 2, J Infect Dis, doi:10.1093/infdis/jiaa471
O'donnell, Thomas, Stanton, Maillard, Murphy et al., Potential Role of Oral Rinses Targeting the Viral Lipid Envelope in SARS-CoV-2 Infection. Function (Oxf), doi:10.1093/function/zqaa002
Popkin, Zilka, Dimaano, Fujioka, Rackley et al., Cetylpyridinium Chloride (CPC) Exhibits Potent, Rapid Activity Against Influenza Viruses in vitro and in vivo, Pathog Immun, doi:10.20411/pai.v2i2.200
Seneviratne, Balan, Ko, Udawatte, Lai et al., Efficacy of commercial mouth-rinses on SARS-CoV-2 viral load in saliva: randomized control trial in Singapore, Infection, doi:10.1007/s15010-020-01563-9
Shen, Niu, Wang, Huang, Wang et al., High-Throughput Screening and Identification of Potent Broad-Spectrum Inhibitors of Coronaviruses, J Virol, doi:10.1128/JVI.00023-19
To, Tsang, Yip, Chan, Wu et al., Consistent detection of 2019 novel coronavirus in saliva, Clin Infect Dis, doi:10.1093/cid/ciaa149
Walsh, Jordan, Clyne, Rohde, Drummond et al., SARS-CoV-2 detection, viral load and infectivity over the course of an infection, J Infect, doi:10.1016/j.jinf.2020.06.067
Wang, Li, Guo, Zhen, Kong et al., Concentration and detection of SARS coronavirus in sewage from Xiao Tang Shan Hospital and the 309th Hospital of the Chinese People's Liberation Army, Water Sci Technol, doi:10.2166/wst.2005.0266
Who, Coronavirus disease (COVID-19) situation reports
Wölfel, Corman, Guggemos, Seilmaier, Zange et al., Virological assessment of hospitalized patients with COVID-2019, Nature, doi:10.1038/s41586-020-2196-x
Zhu, Guo, Xu, Chen, Viral dynamics of SARS-CoV-2 in saliva from infected patients, J Infect, doi:10.1016/j.jinf.2020.06.059
DOI record:
{
"DOI": "10.25100/re.v31i1.12669",
"ISSN": [
"2248-7220"
],
"URL": "http://dx.doi.org/10.25100/re.v31i1.12669",
"abstract": "<jats:p>Aim: The aim of this randomised, double-blind, placebo-controlled pilot clinical trial is to evaluate the capacity of a mouthwash to reduce SARS-CoV-2 viral load in the saliva of patients with COVID-19.
\nMethods: Twenty-three symptomatic SARS-CoV-2-positive outpatients were selected and randomised into two groups and registered at NTC 04563689. Both groups rinsed and gargled for one minute with either distilled water (Placebo) or with 0.05% Cetylpyridinium chloride (CPC) plus 0.12% Chlorhexidine (CHX) mouthwash (PERIOAID Intensive CareÒ). Saliva samples were collected before the use of placebo or mouthwash and after 15 minutes and 1 and 2 hours of either of the above treatment. A saliva sample was also taken five days after regular use of placebo or mouthwash twice daily. The virus was detected by qRT-PCR.
\nResults: A great heterogeneity in the viral load values was observed at baseline in both groups for nasopharyngeal and saliva samples. Most of the patients who used the mouthwash (8/12) had a significant decrease in baseline viral load after 15 min (greater than 99% reduction). This inhibitory effect was maintained for up to two hours in 10 of the 12 patients. At five days, SARS-CoV-2 RNA was detected in only 1 patient from the mouthwash group and in 5 from the placebo group.
\nConclusions: This study points out that a CPC mouthwash can reduce the viral load in saliva of COVID-positive patients. This finding may be important in transmission control of SARS-CoV-2. Nevertheless, the clinical relevance of CPC mouthwash-reduction on SARS-CoV-2 shedding in saliva requires further study</jats:p>",
"author": [
{
"affiliation": [],
"family": "Contreras Renjifo",
"given": "Adolfo",
"sequence": "first"
},
{
"ORCID": "http://orcid.org/0000-0001-6400-4906",
"affiliation": [],
"authenticated-orcid": false,
"family": "León Berrios",
"given": "Rubén",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0001-9006-6721",
"affiliation": [],
"authenticated-orcid": false,
"family": "Castillo Giraldo",
"given": "Andrés",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Bahamon",
"given": "José Luis",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Giraldo",
"given": "Yurani",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0003-0196-1957",
"affiliation": [],
"authenticated-orcid": false,
"family": "Libreros Zúñiga",
"given": "Gerardo Andrés",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-6530-3593",
"affiliation": [],
"authenticated-orcid": false,
"family": "Contreras Parra",
"given": "Alejandro",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-2830-8053",
"affiliation": [],
"authenticated-orcid": false,
"family": "Gispert",
"given": "Juan",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-0138-9199",
"affiliation": [],
"authenticated-orcid": false,
"family": "Balanta- Melo",
"given": "Julián",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Alban",
"given": "Fausto Andrés",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-5371-7449",
"affiliation": [],
"authenticated-orcid": false,
"family": "Parra",
"given": "Beatriz",
"sequence": "additional"
}
],
"container-title": "Revista Estomatología",
"container-title-short": "Rev Estomatol",
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2023,
7,
27
]
],
"date-time": "2023-07-27T19:32:52Z",
"timestamp": 1690486372000
},
"deposited": {
"date-parts": [
[
2023,
7,
27
]
],
"date-time": "2023-07-27T19:33:02Z",
"timestamp": 1690486382000
},
"indexed": {
"date-parts": [
[
2023,
7,
28
]
],
"date-time": "2023-07-28T04:26:48Z",
"timestamp": 1690518408470
},
"is-referenced-by-count": 0,
"issue": "1",
"issued": {
"date-parts": [
[
2023,
6,
5
]
]
},
"journal-issue": {
"issue": "1",
"published-online": {
"date-parts": [
[
2023,
1,
23
]
]
}
},
"license": [
{
"URL": "http://creativecommons.org/licenses/by-nc-nd/4.0",
"content-version": "unspecified",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2023,
6,
5
]
],
"date-time": "2023-06-05T00:00:00Z",
"timestamp": 1685923200000
}
}
],
"link": [
{
"URL": "https://estomatologia.univalle.edu.co/index.php/revista_estomatologia/article/download/12669/15872",
"content-type": "application/pdf",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://estomatologia.univalle.edu.co/index.php/revista_estomatologia/article/download/12669/15873",
"content-type": "text/xml",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://estomatologia.univalle.edu.co/index.php/revista_estomatologia/article/download/12669/15872",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "10728",
"original-title": [],
"prefix": "10.25100",
"published": {
"date-parts": [
[
2023,
6,
5
]
]
},
"published-online": {
"date-parts": [
[
2023,
6,
5
]
]
},
"publisher": "Universidad del Valle",
"reference": [
{
"key": "182094",
"unstructured": "WHO. Coronavirus disease (COVID‐19) situation reports. (2019). https://www.who.int/emergencies/diseases/novel-coronavirus-2019/situation-reports."
},
{
"key": "182095",
"unstructured": "Chau NVV, Thanh Lam V, Thanh Dung N, Yen LM, Minh NNQ, Hung LM, et al. The natural history and transmission potential of asymptomatic SARS-CoV-2 infection. Clin Infect Dis. 2020; 71(10), 2679–2687."
},
{
"DOI": "10.1093/cid/ciaa149",
"doi-asserted-by": "crossref",
"key": "182096",
"unstructured": "To K, Tsang O, Yip C, Chan K, Wu T, Chan J, et al. Consistent detection of 2019 novel coronavirus in saliva. Clin Infect Dis. 2020; 71(15), 841–843."
},
{
"DOI": "10.1016/j.jinf.2020.06.067",
"doi-asserted-by": "crossref",
"key": "182097",
"unstructured": "Walsh KA, Jordan K, Clyne B, Rohde D, Drummond L, Byrne P, et al. SARS-CoV-2 detection, viral load and infectivity over the course of an infection. J Infect. 2020; 81(3): 357–371."
},
{
"DOI": "10.1016/j.jinf.2020.06.059",
"doi-asserted-by": "crossref",
"key": "182098",
"unstructured": "Zhu J, Guo J, Xu Y, Chen X. Viral dynamics of SARS-CoV-2 in saliva from infected patients. J Infect. 2020; 81(3): e48-e50."
},
{
"DOI": "10.1016/j.ijid.2004.07.014",
"doi-asserted-by": "crossref",
"key": "182099",
"unstructured": "He Z, Zhao C, Dong Q, Zhuang H, Song S, Peng G, Dwyer DE. Effects of severe acute respiratory syndrome (SARS) coronavirus infection on peripheral blood lymphocytes and their subsets. Int J Infect Dis. 2005; 9(6): 323-30."
},
{
"DOI": "10.1371/journal.pone.0248273",
"doi-asserted-by": "crossref",
"key": "182100",
"unstructured": "Huang LS, Li L, Dunn L, He M. Taking account of asymptomatic infections: A modeling study of the COVID-19 outbreak on the Diamond Princess cruise ship. PLoS One. 2021; 16(3): e0248273."
},
{
"DOI": "10.1126/science.abb6936",
"doi-asserted-by": "crossref",
"key": "182101",
"unstructured": "Ferretti L, Wymant C, Kendall M, et al. Quantifying SARS-CoV-2 transmission suggests epidemic control with digital contact tracing. Science. 2020; 368(6491): eabb6936."
},
{
"DOI": "10.1002/14651858.CD013627.pub2",
"doi-asserted-by": "crossref",
"key": "182102",
"unstructured": "Burton M, Clarkson J, Goulao B, Glenny AM, McBain A, Schilder A, et al. Antimicrobial mouthwashes (gargling) and nasal sprays administered to patients with suspected or confirmed COVID-19 infection to improve patient outcomes and to protect healthcare workers treating them. Cochrane Database Syst Rev. 2020; 9:CD013627."
},
{
"DOI": "10.1007/s00784-020-03413-2",
"doi-asserted-by": "crossref",
"key": "182103",
"unstructured": "Herrera D, Serrano J, Roldán S, Sanz M. Is the oral cavity relevant in SARS-CoV-2 pandemic? Clin Oral Investig. 2020; 24(8): 2925-2930."
},
{
"DOI": "10.1093/function/zqaa002",
"doi-asserted-by": "crossref",
"key": "182104",
"unstructured": "O'Donnell VB, Thomas D, Stanton R, Maillard J-Y, Murphy R, Jones S, et al. Potential Role of Oral Rinses Targeting the Viral Lipid Envelope in SARS-CoV-2 Infection. Function (Oxf). 2020; 1(1): zqaa002."
},
{
"DOI": "10.20411/pai.v2i2.200",
"doi-asserted-by": "crossref",
"key": "182105",
"unstructured": "Popkin D, Zilka S, Dimaano M, Fujioka H, Rackley C, Salata R, et al. Cetylpyridinium Chloride (CPC) Exhibits Potent, Rapid Activity Against Influenza Viruses in vitro and in vivo. Pathog Immun. 2017; 2(2): 252-269."
},
{
"DOI": "10.1128/JVI.00023-19",
"doi-asserted-by": "crossref",
"key": "182106",
"unstructured": "Shen L, Niu J, Wang C, Huang B, Wang W, Zhu N, et al. High-Throughput Screening and Identification of Potent Broad-Spectrum Inhibitors of Coronaviruses. J Virol. 2019; 93(12):e00023-19."
},
{
"DOI": "10.3201/eid2608.201246",
"doi-asserted-by": "crossref",
"key": "182107",
"unstructured": "Lu X, Wang L, Sakthivel S, Whitaker B, Murray J, Kamili S et al. US CDC Real-Time Reverse Transcription PCR Panel for Detection of Severe Acute Respiratory Syndrome Coronavirus 2. Emerg Infect Dis. 2020; 26(8):1654–1665."
},
{
"DOI": "10.1186/s12879-016-2177-8",
"doi-asserted-by": "crossref",
"key": "182108",
"unstructured": "Mukherjee PK, Esper F, Buchheit K, Arters K, Adkins I, Ghannoum M, et al. Randomized, double-blind, placebo-controlled clinical trial to assess the safety and effectiveness of a novel dual-action oral topical formulation against upper respiratory infections. BMC Infect Dis. 2017; 17(1): 74."
},
{
"DOI": "10.1038/s41597-021-00848-4",
"doi-asserted-by": "crossref",
"key": "182109",
"unstructured": "Ellinger B, Bojkova D, Zaliani A, Cinati J, Claussen C, Westhaus S, et al. A SARS-CoV-2 cytopathicity dataset generated by high-content screening of a large drug repurposing collection. Sci Data. 2021; 8(1): 70."
},
{
"DOI": "10.1016/j.ajoms.2021.02.002",
"doi-asserted-by": "crossref",
"key": "182110",
"unstructured": "Komine A, Yamaguchi E, Okamoto N, Yamamoto K. Virucidal activity of oral care products against SARS-CoV-2 in vitro. J Oral Maxillofac Surg Med Pathol. 2021; 10.1016/j.ajoms.2021.02.002."
},
{
"DOI": "10.1093/infdis/jiaa471",
"doi-asserted-by": "crossref",
"key": "182111",
"unstructured": "Meister T, Brüggemann Y, Todt D, Conzelmann C, Müller J, Groß R, et al. Virucidal Efficacy of Different Oral Rinses Against Severe Acute Respiratory Syndrome Coronavirus 2. J Infect Dis. 2020; 222(8):1289-1292."
},
{
"DOI": "10.3390/microorganisms9030521",
"doi-asserted-by": "crossref",
"key": "182112",
"unstructured": "Koch-Heier J, Hoffmann H, Schindler M, Lussi A, Planz O. Inactivation of SARS-CoV-2 through Treatment with the Mouth Rinsing Solutions ViruProX® and BacterX® Pro. Microorganisms. 2021; 9(3):521."
},
{
"DOI": "10.1099/jgv.0.001578",
"doi-asserted-by": "crossref",
"key": "182113",
"unstructured": "Davies K, Buczkowski H, Welch SR, et al. Effective in vitro inactivation of SARS-CoV-2 by commercially available mouthwashes. J Gen Virol. 2021;102(4):10. 1099/jgv.0.001578."
},
{
"DOI": "10.4103/jisp.jisp_824_20",
"doi-asserted-by": "crossref",
"key": "182114",
"unstructured": "Jain A, Grover V, Singh C, Sharma A, Das DK, Singh P, et al. Chlorhexidine: An effective anticovid mouth rinse. J Indian Soc Periodontol. 2021; 25(1):86-88."
},
{
"DOI": "10.1007/s40121-020-00316-3",
"doi-asserted-by": "crossref",
"key": "182115",
"unstructured": "Anderson DE, Sivalingam V, Kang AEZ, Ananthanarayanan A, Arumugam H, Jenkins TM, et al. Povidone-Iodine Demonstrates Rapid in vitro Virucidal Activity Against SARS-CoV-2, The Virus Causing COVID-19 Disease. Infect Dis Ther. 2020; 9(3): 669-675."
},
{
"DOI": "10.1111/jopr.13220",
"doi-asserted-by": "crossref",
"key": "182116",
"unstructured": "Bidra AS, Pelletier JS, Westover JB, Frank S, Brown SM, Tessema B. Comparison of in vitro Inactivation of SARS CoV-2 with Hydrogen Peroxide and Povidone-Iodine Oral Antiseptic Rinses. J Prosthodont. 2020; 29(7): 599-603."
},
{
"DOI": "10.1007/s00784-020-03549-1",
"doi-asserted-by": "crossref",
"key": "182117",
"unstructured": "Gottsauner MJ, Michaelides I, Schmidt B, Scholz K, Buchalla W, Widbiller M, et al. A prospective clinical pilot study on the effects of a hydrogen peroxide mouthrinse on the intraoral viral load of SARS-CoV-2. Clin Oral Investig. 2020; 24(10): 3707-3713."
},
{
"DOI": "10.1111/odi.13526",
"doi-asserted-by": "crossref",
"key": "182118",
"unstructured": "Martínez-Lamas L, Diz P, Pérez MT, Del Campo V, Cabrera JJ, López AM, et al. Is povidone iodine mouthwash effective against SARS-CoV-2? First in vivo tests. Oral Dis. 2020; 10.1111/odi.13526."
},
{
"DOI": "10.1007/s15010-020-01563-9",
"doi-asserted-by": "crossref",
"key": "182119",
"unstructured": "Seneviratne C J, Balan P, Ko K, Udawatte N S, Lai D, Ng D, et al. Efficacy of commercial mouth-rinses on SARS-CoV-2 viral load in saliva: randomized control trial in Singapore. Infection. 2021; 49(2): 305-311."
},
{
"DOI": "10.1016/j.ijid.2020.04.057",
"doi-asserted-by": "crossref",
"key": "182120",
"unstructured": "Amirian ES. Potential fecal transmission of SARS-CoV-2: Current evidence and implications for public health. Int J Infect Dis. 2020; 95:363-370."
},
{
"DOI": "10.1038/s41586-020-2196-x",
"doi-asserted-by": "crossref",
"key": "182121",
"unstructured": "Wölfel R, Corman VM, Guggemos W, Seilmaier M, Zange S, Müller MA, et al. Virological assessment of hospitalized patients with COVID-2019. Nature. 2020; 581(7809): 465-469."
},
{
"DOI": "10.2166/wst.2005.0266",
"doi-asserted-by": "crossref",
"key": "182122",
"unstructured": "Wang XW, Li J, Guo T, Zhen B, Kong Q, Yi B, et al. Concentration and detection of SARS coronavirus in sewage from Xiao Tang Shan Hospital and the 309th Hospital of the Chinese People's Liberation Army. Water Sci Technol. 2005;52(8):213-221."
},
{
"DOI": "10.1038/s41467-020-19883-7",
"doi-asserted-by": "crossref",
"key": "182123",
"unstructured": "Alexandersen S, Chamings A, Bhatta TR. SARS-CoV-2 genomic and subgenomic RNAs in diagnostic samples are not an indicator of active replication. Nat Commun. 2020;11(1):6059."
}
],
"reference-count": 30,
"references-count": 30,
"relation": {},
"resource": {
"primary": {
"URL": "https://estomatologia.univalle.edu.co/index.php/revista_estomatologia/article/view/12669"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subtitle": [],
"title": "A Mouthwash with Cetylpyridinium Chloride Is Reducing Salivary SARS-CoV-2 Viral Load in +COVID-19.",
"type": "journal-article",
"volume": "31"
}
