Effect of low dose acetylsalicylic acid and anticoagulant on clinical outcomes in COVID-19, analytical cross-sectional study
et al., Health Science Reports, doi:10.1002/hsr2.699, Jul 2022
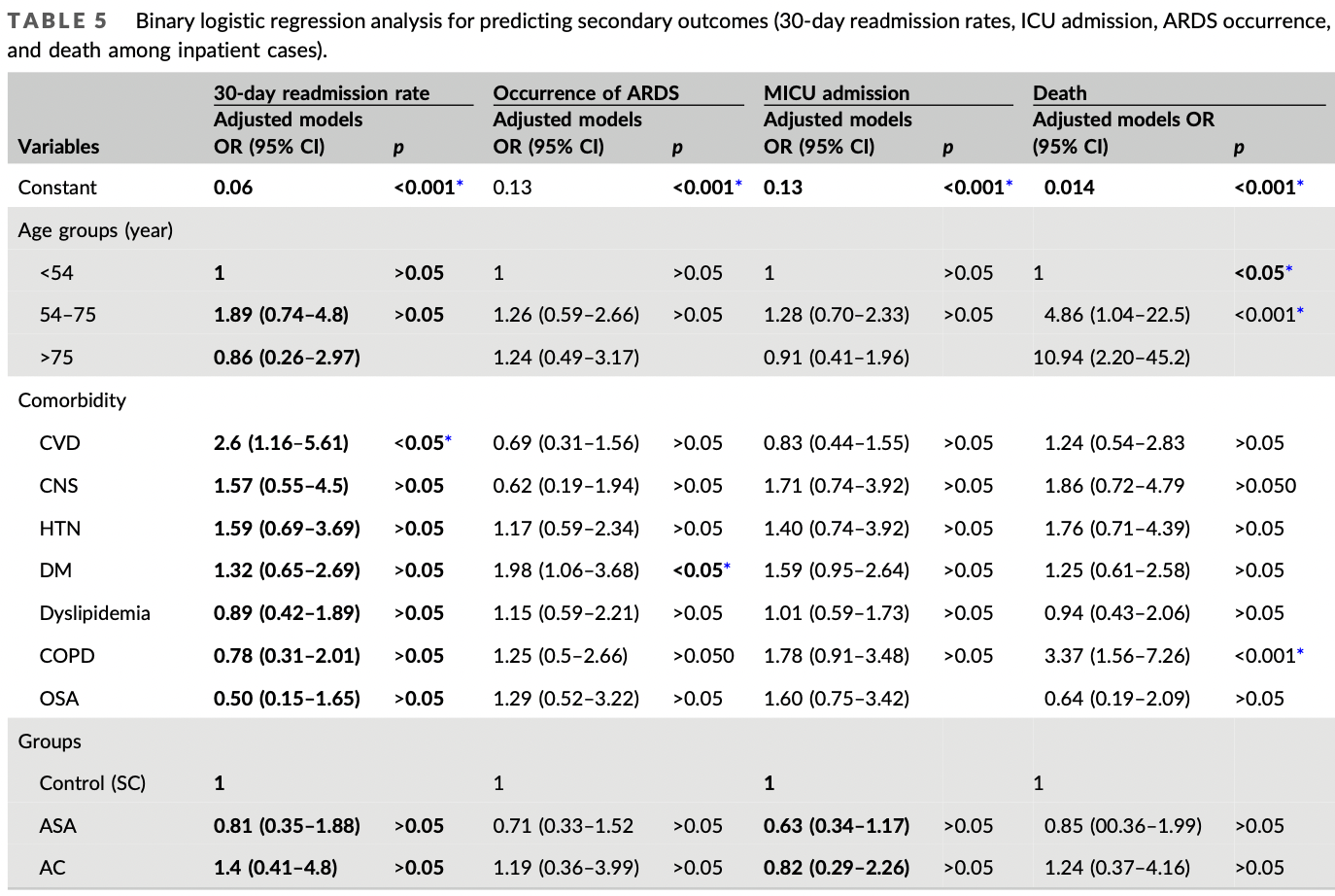
Retrospective 539 patients in the USA, showing lower mortality, ICU admission, and ARDS with aspirin treatment, without statistical significance.
Standard of Care (SOC) for COVID-19 in the study country,
the USA, is very poor with very low average efficacy for approved treatments1.
Only expensive, high-profit treatments were approved for early treatment. Low-cost treatments were excluded, reducing the probability of early treatment due to access and cost barriers, and eliminating complementary and synergistic benefits seen with many low-cost treatments.
|
risk of death, 13.6% lower, RR 0.86, p = 0.72, treatment 15 of 87 (17.2%), control 24 of 223 (10.8%), adjusted per study, odds ratio converted to relative risk, multivariable.
|
|
risk of ICU admission, 27.8% lower, RR 0.72, p = 0.17, treatment 28 of 87 (32.2%), control 77 of 223 (34.5%), adjusted per study, odds ratio converted to relative risk, multivariable.
|
|
risk of ARDS, 25.1% lower, RR 0.75, p = 0.39, treatment 13 of 87 (14.9%), control 40 of 223 (17.9%), NNT 33, adjusted per study, odds ratio converted to relative risk, multivariable.
|
|
risk of hospitalization, 2.4% lower, OR 0.98, p = 0.94, treatment 25, control 176, adjusted per study, multivariable, RR approximated with OR.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Malik et al., 11 Jul 2022, retrospective, USA, peer-reviewed, 16 authors, study period 1 March, 2020 - 1 December, 2020.
Contact: muhammadbmalik@gmail.com, dr_samar11@yahoo.com.
Effect of low dose acetylsalicylic acid and anticoagulant on clinical outcomes in COVID‐19, analytical cross‐sectional study
Health Science Reports, doi:10.1002/hsr2.699
Background and aims: The therapeutic strategy for the treatment of known sequelae of COVID-19 has shifted from reactive to preventative. In this study, we aim to evaluate the effects of acetylsalicylic acid (ASA), and anticoagulants on COVID-19 related morbidity and mortality. Methods: This record-based analytical cross-sectional study targeted 539 COVID-19 patients in a single United States medical center between March and December 2020. Through a random stratified sample, we recruited outpatient (n = 206) and inpatient (n = 333) cases from three management protocols, including standard care (SC) (n = 399), low-dose ASA only (ASA) (n = 112), and anticoagulation only (AC) (n = 28). Collected data included demographics, comorbidities, and clinical outcomes. The primary outcome measure was inpatient admission. Exploratory secondary outcome measures included length of stay, 30-day readmission rates, medical intensive care unit (MICU) admission, need for mechanical ventilation, the occurrence of acute respiratory distress syndrome (ARDS), bleeding events, clotting events, and mortality. The collected data were coded and analyzed using standard tests. Results: Age, mean number of comorbidities, and all individual comorbidities except for asthma, and malignancy were significantly lower in the SC compared to ASA and AC. After adjusting for age and comorbidity via binary logistic regression models, no statistical differences were found between groups for the studied outcomes. When compared to the SC group, ASA had lower 30-day readmission rates (odds ration
from March until December, and the many outcomes were studied in detail. Every case fulfilling the selection criteria has an equal chance of being recruited into the sample (so it is a random sample) to find the association and risk assessment.
| CONCLUSIONS Low-dose ASA is a promising, effective, protective medication in potentially improving the outcomes in COVID-19 cases, through reducing mortality, and morbidity (hospital readmission rates, the occurrence of ARDS, and ICU admission) but not statistically significant due to the higher risk patients in these groups. further prospective research is still needed.
| RECOMMENDATION
CONFLICT OF INTEREST The authors declare no conflict of interest.
TRANSPARENCY STATEMENT I affirm that this manuscript is an honest, accurate, and transparent account of the study being reported; that no important aspects of the study have been omitted; and that any discrepancies from the study as planned (and, if relevant, registered) have been explained.
ETHICS STATEMENT This study was reviewed by the institutional review board (IRB) at Upstate Medical University and was considered exempt according to the #4(iii) exemption category in federal regulations. The requirement for written informed consent was waived by the IRB and the study was conducted in accordance with ethical principles.
References
Aghajani, Moradi, Amini, Decreased in-hospital mortality associated with aspirin administration in hospitalized patients due to severe COVID-19, J Med Virol, doi:10.1002/jmv.27053
Ahmed, Merrell, Ismail, Rationales and uncertainties for aspirin use in COVID-19: a narrative review, Family Med Commun Health, doi:10.1136/fmch-2020-000741
Chow, Khanna, Kethireddy, Aspirin use is associated with decreased mechanical ventilation, intensive care unit admission, and in-hospital mortality in hospitalized patients with coronavirus disease 2019, Anesth Analg, doi:10.1213/ANE.0000000000005292
Formiga, Rubio-Rivas, Mora-Luján, Does admission acetylsalicylic acid uptake in hospitalized COVID-19 patients have a protective role? Data from the spanish SEMI-COVID-19 registry, Intern Emerg Med, doi:10.1007/s11739-021-02870-1
Glatthaar-Saalmüller, Mair, Saalmüller, Antiviral activity of aspirin against RNA viruses of the respiratory tract-an in vitro study, Influenza Other Respir Viruses, doi:10.1111/irv.12421
Griffin, Jensen, Khan, Arterial thromboembolic complications in COVID-19 in low-risk patients despite prophylaxis, Br J Haematol, doi:10.1111/bjh.16792
Liu, Huang, Li, Effect of low-dose aspirin on mortality and viral duration of the hospitalized adults with COVID-19, Medicine, doi:10.1097/MD.0000000000024544
Martha, Pranata, Lim, Wibowo, Akbar, Active prescription of low-dose aspirin during or prior to hospitalization and mortality in COVID-19: a systematic review and meta-analysis of adjusted effect estimates, Int J Infect Dis, doi:10.1016/j.ijid.2021.05.016
Mazloomzadeh, Khaleghparast, Ghadrdoost, Effect of intermediate-dose vs standard-dose prophylactic anticoagulation on thrombotic events, extracorporeal membrane oxygenation treatment, or mortality among patients with COVID-19 admitted tothe intensive care unit, JAMA, doi:10.1001/jama.2021.4152
Merzon, Green, Vinker, The use of aspirin for primary prevention of cardiovascular disease is associated with a lower likelihood of COVID-19 infection, FEBS J, doi:10.1111/febs.15784
Pranata, Huang, Lim, Wahjoepramono, July, Impact of cerebrovascular and cardiovascular diseases on mortality and severity of COVID-19-systematic review, meta-analysis, and meta-regression, J Stroke Cerebrovasc Dis, doi:10.1016/j.jstrokecerebrovasdis.2020.104949
Salah, Mehta, Meta-analysis of the effect of aspirin on mortality in COVID-19, Am J Cardiol, doi:10.1016/j.amjcard.2020.12.073
Samar, Albeladi, Elshabrawy, Role of neutrophil to lymphocyte ratio as a prognostic indicator for COVID-19, Health Sci Rep, doi:10.1002/hsr2.442
Wang, Li, Gu, Wang, Liu et al., Effect of antiplatelet therapy on acute respiratory distress syndrome and mortality in critically ill patients: a meta-analysis, PLoS One, doi:10.1371/journal.pone.0154754
Wen, Hsiao, Chan, Lin, Shen et al., Acute respiratory infection and use of nonsteroidal anti-Inflammatory drugs on risk of acute myocardial infarction: a nationwide casecrossover study, J Infect Dis, doi:10.1093/infdis/jiw603
Yuan, Chen, Li, Chen, Wang et al., Mortality and prehospitalization use of low-dose aspirin in COVID-19 patients with coronary artery disease, J Cell Mol Med, doi:10.1111/jcmm.16198
Zhou, Li, Yang, Antiplatelet therapy after percutaneous coronary intervention in patients with COVID-19, Circulation, doi:10.1161/CIRCULATIONAHA.120.046988
DOI record:
{
"DOI": "10.1002/hsr2.699",
"ISSN": [
"2398-8835",
"2398-8835"
],
"URL": "http://dx.doi.org/10.1002/hsr2.699",
"alternative-id": [
"10.1002/hsr2.699"
],
"assertion": [
{
"group": {
"label": "Publication History",
"name": "publication_history"
},
"label": "Received",
"name": "received",
"order": 0,
"value": "2021-10-30"
},
{
"group": {
"label": "Publication History",
"name": "publication_history"
},
"label": "Accepted",
"name": "accepted",
"order": 1,
"value": "2022-05-26"
},
{
"group": {
"label": "Publication History",
"name": "publication_history"
},
"label": "Published",
"name": "published",
"order": 2,
"value": "2022-07-11"
}
],
"author": [
{
"ORCID": "http://orcid.org/0000-0002-9493-6226",
"affiliation": [
{
"name": "Department of Medicine SUNY Upstate Medical University Syracuse New York USA"
}
],
"authenticated-orcid": false,
"family": "Malik",
"given": "Muhammad B.",
"sequence": "first"
},
{
"ORCID": "http://orcid.org/0000-0002-9475-6372",
"affiliation": [
{
"name": "Department of Public Health and Community Medicine Zagazig Medical University Zagazig Egypt"
}
],
"authenticated-orcid": false,
"family": "Amer",
"given": "Samar A.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Medicine SUNY Upstate Medical University Syracuse New York USA"
}
],
"family": "Merrell",
"given": "Eric",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Medicine SUNY Upstate Medical University Syracuse New York USA"
}
],
"family": "Russo",
"given": "Ronald",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Cardiovascular Perfusion College of Health Professions SUNY Upstate Medical University Syracuse New York USA"
}
],
"family": "Riley",
"given": "Jeffrey B.",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0003-4238-6231",
"affiliation": [
{
"name": "SUNY Upstate Medical University Syracuse New York USA"
}
],
"authenticated-orcid": false,
"family": "Scro",
"given": "Austin",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "SUNY Upstate Medical University Syracuse New York USA"
}
],
"family": "James",
"given": "Elizabeth",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Medicine SUNY Upstate Medical University Syracuse New York USA"
}
],
"family": "Anuforo",
"given": "Anderson",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Medicine SUNY Upstate Medical University Syracuse New York USA"
}
],
"family": "Adhikari",
"given": "Soumya",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "SUNY Upstate Medical University Syracuse New York USA"
}
],
"family": "Siciliano",
"given": "Rosalie",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Medicine SUNY Upstate Medical University Syracuse New York USA"
}
],
"family": "Chebaya",
"given": "Philip",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Cardiovascular Perfusion College of Health Professions SUNY Upstate Medical University Syracuse New York USA"
}
],
"family": "Darling",
"given": "Edward",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Medicine SUNY Upstate Medical University Syracuse New York USA"
}
],
"family": "Kuhn",
"given": "Michael",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Surgery SUNY Upstate Medical University Syracuse New York USA"
}
],
"family": "Nieman",
"given": "Gary",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Pulmonary and Critical Care SUNY Upstate Medical University Syracuse New York USA"
}
],
"family": "Shawkat",
"given": "Ahmed",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Medicine SUNY Upstate Medical University Syracuse New York USA"
},
{
"name": "Cardiovascular Perfusion College of Health Professions SUNY Upstate Medical University Syracuse New York USA"
},
{
"name": "Department of Surgery SUNY Upstate Medical University Syracuse New York USA"
},
{
"name": "Department of Family Medicine Suez Canal University Egypt"
}
],
"family": "Aiash",
"given": "Hani",
"sequence": "additional"
}
],
"container-title": "Health Science Reports",
"container-title-short": "Health Science Reports",
"content-domain": {
"crossmark-restriction": true,
"domain": [
"onlinelibrary.wiley.com"
]
},
"created": {
"date-parts": [
[
2022,
7,
12
]
],
"date-time": "2022-07-12T05:33:50Z",
"timestamp": 1657604030000
},
"deposited": {
"date-parts": [
[
2022,
7,
12
]
],
"date-time": "2022-07-12T05:33:57Z",
"timestamp": 1657604037000
},
"indexed": {
"date-parts": [
[
2022,
7,
12
]
],
"date-time": "2022-07-12T06:13:18Z",
"timestamp": 1657606398896
},
"is-referenced-by-count": 0,
"issue": "4",
"issued": {
"date-parts": [
[
2022,
7
]
]
},
"journal-issue": {
"issue": "4",
"published-print": {
"date-parts": [
[
2022,
7
]
]
}
},
"language": "en",
"license": [
{
"URL": "http://creativecommons.org/licenses/by/4.0/",
"content-version": "vor",
"delay-in-days": 10,
"start": {
"date-parts": [
[
2022,
7,
11
]
],
"date-time": "2022-07-11T00:00:00Z",
"timestamp": 1657497600000
}
},
{
"URL": "http://doi.wiley.com/10.1002/tdm_license_1.1",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2022,
7,
1
]
],
"date-time": "2022-07-01T00:00:00Z",
"timestamp": 1656633600000
}
}
],
"link": [
{
"URL": "https://onlinelibrary.wiley.com/doi/pdf/10.1002/hsr2.699",
"content-type": "application/pdf",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://onlinelibrary.wiley.com/doi/full-xml/10.1002/hsr2.699",
"content-type": "application/xml",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://onlinelibrary.wiley.com/doi/pdf/10.1002/hsr2.699",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "311",
"original-title": [],
"prefix": "10.1002",
"published": {
"date-parts": [
[
2022,
7
]
]
},
"published-online": {
"date-parts": [
[
2022,
7,
11
]
]
},
"published-print": {
"date-parts": [
[
2022,
7
]
]
},
"publisher": "Wiley",
"reference": [
{
"key": "e_1_2_14_2_1",
"unstructured": "Center for Disease Control and Prevention. COVID‐19 cases in the U.S. 2021. Accessed May 24 2021.https://www.cdc.gov/coronavirus/2019-ncov/index.html"
},
{
"key": "e_1_2_14_3_1",
"unstructured": "Center for Disease Control and Prevention.Critical Care Guidance. 2021. Accessed May 242021.https://www.cdc.gov/coronavirus/2019-ncov/hcp/clinical-guidance-management-patients.html"
},
{
"key": "e_1_2_14_4_1",
"unstructured": "National Institute of Health. Outpatient management of acute COVID‐19. 2021. Accessed May 24 2021.https://www.covid19treatmentguidelines.nih.gov/management/clinical-management/nonhospitalized-patients--general-management/"
},
{
"DOI": "10.1111/irv.12421",
"doi-asserted-by": "publisher",
"key": "e_1_2_14_5_1"
},
{
"DOI": "10.1371/journal.pone.0154754",
"doi-asserted-by": "publisher",
"key": "e_1_2_14_6_1"
},
{
"DOI": "10.1016/j.ijid.2021.05.016",
"doi-asserted-by": "publisher",
"key": "e_1_2_14_7_1"
},
{
"DOI": "10.1016/j.amjcard.2020.12.073",
"doi-asserted-by": "publisher",
"key": "e_1_2_14_8_1"
},
{
"DOI": "10.1136/fmch-2020-000741",
"doi-asserted-by": "crossref",
"key": "e_1_2_14_9_1",
"unstructured": "Sayed AhmedHA MerrellE IsmailM et al.Rationales and uncertainties for aspirin use in COVID‐19: a narrative review.Family Med Commun Health.2021;9(2).doi:10.1136/fmch-2020-000741"
},
{
"DOI": "10.1016/j.jstrokecerebrovasdis.2020.104949",
"doi-asserted-by": "publisher",
"key": "e_1_2_14_10_1"
},
{
"DOI": "10.1111/bjh.16792",
"doi-asserted-by": "publisher",
"key": "e_1_2_14_11_1"
},
{
"DOI": "10.1213/ANE.0000000000005292",
"doi-asserted-by": "publisher",
"key": "e_1_2_14_12_1"
},
{
"DOI": "10.1093/infdis/jiw603",
"doi-asserted-by": "publisher",
"key": "e_1_2_14_13_1"
},
{
"DOI": "10.1001/jama.2021.4152",
"doi-asserted-by": "publisher",
"key": "e_1_2_14_14_1"
},
{
"DOI": "10.1111/febs.15784",
"doi-asserted-by": "publisher",
"key": "e_1_2_14_15_1"
},
{
"DOI": "10.1111/jcmm.16198",
"doi-asserted-by": "publisher",
"key": "e_1_2_14_16_1"
},
{
"DOI": "10.1002/jmv.27053",
"doi-asserted-by": "publisher",
"key": "e_1_2_14_17_1"
},
{
"DOI": "10.1161/CIRCULATIONAHA.120.046988",
"doi-asserted-by": "publisher",
"key": "e_1_2_14_18_1"
},
{
"DOI": "10.1097/MD.0000000000024544",
"doi-asserted-by": "publisher",
"key": "e_1_2_14_19_1"
},
{
"DOI": "10.1007/s11739-021-02870-1",
"doi-asserted-by": "publisher",
"key": "e_1_2_14_20_1"
},
{
"article-title": "Role of neutrophil to lymphocyte ratio as a prognostic indicator for COVID‐19",
"author": "Samar AA",
"first-page": "e442",
"issue": "4",
"journal-title": "Health Sci Rep",
"key": "e_1_2_14_21_1",
"volume": "4",
"year": "2021"
}
],
"reference-count": 20,
"references-count": 20,
"relation": {},
"resource": {
"primary": {
"URL": "https://onlinelibrary.wiley.com/doi/10.1002/hsr2.699"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [
"General Medicine"
],
"subtitle": [],
"title": "Effect of low dose acetylsalicylic acid and anticoagulant on clinical outcomes in COVID‐19, analytical cross‐sectional study",
"type": "journal-article",
"update-policy": "http://dx.doi.org/10.1002/crossmark_policy",
"volume": "5"
}