Phase I/II clinical trial of efficacy and safety of EGCG oxygen nebulization inhalation in the treatment of COVID-19 pneumonia patients with cancer
et al., BMC Cancer, doi:10.1186/s12885-024-12228-3, NCT05758571, Apr 2024
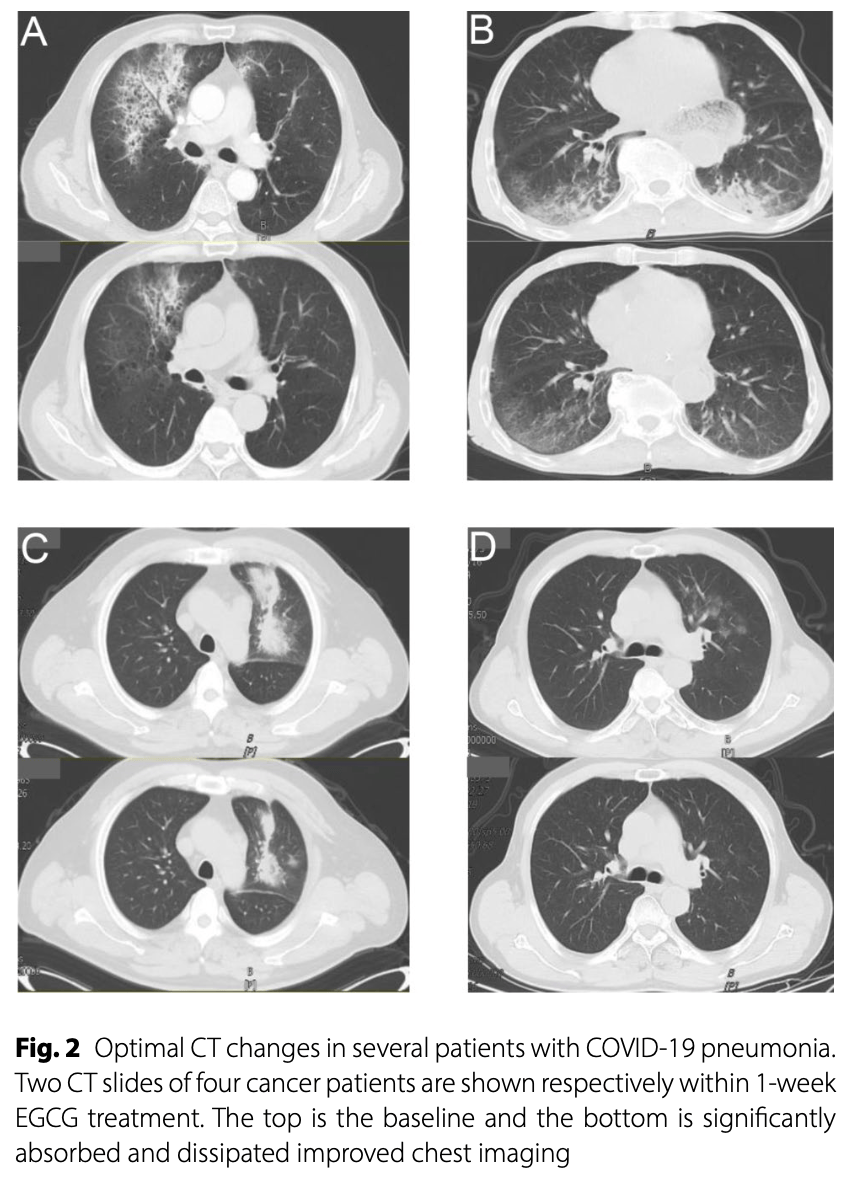
Phase I/II trial of EGCG nebulization inhalation in 54 COVID-19 pneumonia patients with cancer showing good tolerability and improvement in CT scans in 56% of patients after 7 days, with non-progression of pneumonia in 82% of patients. There was no significant difference in inflammation-related laboratory parameters before and after treatment. The maximum tolerated dose was 5878 μmol/L.
Yin et al., 17 Apr 2024, prospective, China, peer-reviewed, median age 68.0, 15 authors, study period 5 January, 2023 - 31 March, 2023, trial NCT05758571 (history).
Contact: mengxiangjiao@sina.com, zhx87520052@163.com.
EGCG (epigallocatechin gallate) is a small molecule polyphenol derived from Camellia sinensis that exhibits antiviral activity against SARS-CoV-2 via dual mechanisms: blocking viral entry by interfering with the Spike-ACE2 receptor interaction and inhibiting the viral 3CL protease (Mpro). Additionally, EGCG provides anti-inflammatory effects by modulating cytokine production, potentially mitigating the cytokine storm associated with severe COVID-19.
Phase I/II clinical trial of efficacy and safety of EGCG oxygen nebulization inhalation in the treatment of COVID-19 pneumonia patients with cancer
BMC Cancer, doi:10.1186/s12885-024-12228-3
Background The antiviral drug Nirmatrelvir was found to be a key drug in controlling the progression of pneumonia during the infectious phase of COVID-19. However, there are very few options for effective treatment for cancer patients who have viral pneumonia. Glucocorticoids is one of the effective means to control pneumonia, but there are many adverse events. EGCG is a natural low toxic compound with anti-inflammatory function. Thus, this study was designed to investigate the safety and efficacy of epigallocatechin-3-gallate (EGCG) aerosol to control COVID-19 pneumonia in cancer populations.
Methods The study was designed as a prospective, single-arm, open-label phase I/II trial at Shandong Cancer Hospital and Institute, between January 5, 2023 to March 31,2023 with viral pneumonia on radiographic signs after confirmed novel coronavirus infection. These patients were treated with EGCG nebulization 10 ml three times daily for at least seven days. EGCG concentrations were increased from 1760-8817umol/L to 4 levels with dose escalation following a standard Phase I design of 3-6 patients per level. Any grade adverse event caused by EGCG was considered a dose-limiting toxicity (DLT). The maximum tolerated dose (MTD) is defined as the highest dose with less than one-third of patients experiencing dose limiting toxicity (DLT) due to EGCG. The primary end points were the toxicity of EGCG and CT findings, and the former was graded by Common Terminology Criteria for Adverse Events (CTCAE) v. 5.0. The secondary end point was the laboratory parameters before and after treatment. Result A total of 60 patients with high risk factors for severe COVID-19 pneumonia (factors such as old age, smoking and combined complications)were included in this phase I-II study. The 54 patients in the final analysis were pathologically confirmed to have tumor burden and completed the whole course of treatment. A patient with bucking at a level of 1760 umol/L and no acute toxicity associated with EGCG has been reported at the second or third dose gradients. At dose escalation to 8817umol/L, Grade 1 adverse events of nausea and stomach discomfort
Supplementary Information The online version contains supplementary material available at https://doi. org/10.1186/s12885-024-12228-3 .
Supplementary Material 1 Author contributions Dr Meng had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. Concept and design: Hanxi.
Consent for publication Not applicable.
Competing interests The authors declare no competing interests.
Publisher's Note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Aktas, Prozorovski, Smorodchenko, Green tea epigallocatechin-3-gallate mediates T cellular NF-kappa B inhibition and exerts neuroprotection in autoimmune encephalomyelitis, J Immunol Nov, doi:10.4049/jimmunol.173.9.5794
Andrianopoulos, Papathanasiou, Papathanakos, Chaidos, Koulouras, Tocilizumab's efficacy in patients with Coronavirus Disease 2019 (COVID-19) is determined by the presence of cytokine storm, J Med Virol, doi:10.1002/jmv.26209
Berlin, Gulick, Martinez, Severe Covid-19, N Engl J Med Dec, doi:10.1056/NEJMcp2009575
Bettuzzi, Gabba, Cataldo, Efficacy of a polyphenolic, standardized Green Tea Extract for the treatment of COVID-19 syndrome: a proof-of-Principle Study, COVID
Bian, Zheng, Wei, Safety and efficacy of meplazumab in healthy volunteers and COVID-19 patients: a randomized phase 1 and an exploratory phase 2 trial, Signal Transduct Target Ther May, doi:10.1038/s41392-021-00603-6
Cai, Li, Liang, Bioavailability of tea catechins and its improvement, Molecules Sep, doi:10.3390/molecules23092346
Chakrawarti, Agrawal, Dang, Gupta, Gabrani, Therapeutic effects of EGCG: a patent review, Expert Opin Ther Pat, doi:10.1080/13543776.2016.1203419
Colotta, Allavena, Sica, Garlanda, Mantovani, Cancer-related inflammation, the seventh hallmark of cancer: links to genetic instability, Carcinog Jul, doi:10.1093/carcin/bgp127
Cortiula, Pettke, Bartoletti, Puglisi, Helleday, Managing COVID-19 in the oncology clinic and avoiding the distraction effect, Ann Oncol, doi:10.1016/j.annonc.2020.03.286
D' Agnillo, Walters, Xiao, Lung epithelial and endothelial damage, loss of tissue repair, inhibition of fibrinolysis, and cellular senescence in fatal COVID-19, Sci Transl Med Nov, doi:10.1126/scitranslmed.abj7790
Dai, Liu, Liu, Patients with Cancer Appear more vulnerable to SARS-CoV-2: a Multicenter Study during the COVID-19 outbreak, Cancer Discov Jun, doi:10.1158/2159-8290.Cd-20-0422
Dorward, Russell, Um, Tissue-specific immunopathology in fatal COVID-19, Am J Respir Crit Care Med Jan, doi:10.1164/rccm.202008-3265OC
Dube, Nicolazzo, Larson, Chitosan nanoparticles enhance the intestinal absorption of the green tea catechins (+)-catechin and (-)-epigallocatechin gallate, Eur J Pharm Sci, doi:10.1016/j.ejps.2010.06.010
Elinav, Nowarski, Thaiss, Hu, Flavell, Inflammation-induced cancer: crosstalk between tumours, immune cells and microorganisms, Nat Rev Cancer Nov, doi:10.1038/nrc3611
Gallus, Bosetti, Gorini, The Association of Tobacco Smoking, second-hand smoke, and Novel Tobacco products with COVID-19 severity and mortality in Italy: results from the COSMO-IT Study, J Epidemiol, doi:10.2188/jea.JE20220321
Gharibi, Babaloo, Hosseini, Targeting STAT3 in cancer and autoimmune diseases, Eur J Pharmacol Jul, doi:10.1016/j.ejphar.2020.173107
Hammond, Leister-Tebbe, Gardner, Oral nirmatrelvir for High-Risk, nonhospitalized adults with Covid-19, N Engl J Med, doi:10.1056/NEJMoa2118542
Hu, Webster, Cao, Shao, The safety of green tea and green tea extract consumption in adults -results of a systematic review, Regul Toxicol Pharmacol, doi:10.1016/j.yrtph.2018.03.019
Kada, Kaneko, Matsuzaki, Matsuzaki, Hara, Detection and chemical identification of natural bio-antimutagens. A case of the green tea factor, Mutat Res, doi:10.1016/0027-5107(85)90109-5
Kim, Garg, 'halloran, Risk factors for Intensive Care Unit Admission and In-hospital mortality among hospitalized adults identified through the US Coronavirus Disease 2019 (COVID-19)-Associated Hospitalization Surveillance Network (COVID-NET), Clin Infect Dis, doi:10.1093/cid/ciaa1012
Lamers, Haagmans, SARS-CoV-2 pathogenesis, Nat Rev Microbiol, doi:10.1038/s41579-022-00713-0
Li, Liu, Zhang, SARS-CoV-2 and viral sepsis: observations and hypotheses, Lancet May, doi:10.1016/s0140-6736(20)30920-x
Li, Xing, Zhang, Phase II trial of Epigallocatechin-3-Gallate in Acute Radiation-Induced Esophagitis for Esophagus Cancer, J Med Food Jan, doi:10.1089/jmf.2019.4445
Liang, Guan, Chen, Cancer patients in SARS-CoV-2 infection: a nationwide analysis in China, Lancet Oncol Mar, doi:10.1016/s1470-2045(20)30096-6
Martínez, Mijares, Sanctis, Effects of flavonoids and its derivatives on Immune cell responses, Recent Pat Inflamm Allergy Drug Discov, doi:10.2174/1872213x13666190426164124
Menegazzi, Campagnari, Bertoldi, Crupi, Paola et al., Protective effect of Epigallocatechin-3-Gallate (EGCG) in diseases with uncontrolled Immune activation: could such a scenario be helpful to Counteract COVID-19, Int J Mol Sci, doi:10.3390/ijms21145171
Ohishi, Goto, Monira, Isemura, Nakamura, Anti-inflammatory action of Green Tea, Antiinflamm Antiallergy Agents Med Chem, doi:10.2174/1871523015666160915154443
Ozturk, Talo, Yildirim, Baloglu, Yildirim et al., Automated detection of COVID-19 cases using deep neural networks with X-ray images, Comput Biol Med Jun, doi:10.1016/j.compbiomed.2020.103792
Pezzuto, Tammaro, Tonini, SARS-Cov-2 pneumonia and concurrent myelodysplasia complicated by Pseudomonas aeruginosa overinfection, J Virol Methods, doi:10.1016/j.jviromet.2021.114419
Sahin, Akbal-Dagistan, Culha, Antivirals and the potential benefits of orally inhaled Drug Administration in COVID-19 treatment, J Pharm Sci, doi:10.1016/j.xphs.2022.06.004
Sica, Massarotti, Myeloid suppressor cells in cancer and autoimmunity, J Autoimmun Dec, doi:10.1016/j.jaut.2017.07.010
Siddiqi, Mehra, COVID-19 illness in native and immunosuppressed states: a clinical-therapeutic staging proposal, J Heart Lung Transpl, doi:10.1016/j.healun.2020.03.012
Stratton, Tang, Lu, Pathogenesis-directed therapy of 2019 novel coronavirus disease, J Med Virol Mar, doi:10.1002/jmv.26610
Thakur, Dubey, Benitez, A systematic review and meta-analysis of geographic differences in comorbidities and associated severity and mortality among individuals with COVID-19, Sci Rep Apr, doi:10.1038/s41598-021-88130-w
Vidigal, Müsken, Becker, Effects of green tea compound epigallocatechin-3-gallate against Stenotrophomonas maltophilia infection and biofilm, PLoS ONE, doi:10.1371/journal.pone.0092876
Villar, Ferrando, Martínez, Dexamethasone treatment for the acute respiratory distress syndrome: a multicentre, randomised controlled trial, Lancet Respir Med, doi:10.1016/s2213-2600(19)30417-5
Wang, Ding, Zhao, Systems pharmacology-based drug discovery and active mechanism of natural products for coronavirus pneumonia (COVID-19): an example using flavonoids, Comput Biol Med Jan, doi:10.1016/j.compbiomed.2022.105241
Wiersinga, Rhodes, Cheng, Peacock, Prescott et al., Transmission, diagnosis, and treatment of Coronavirus Disease 2019 (COVID-19): a review, Jama, doi:10.1001/jama.2020.12839
Wu, Zhao, Yu, A new coronavirus associated with human respiratory disease in China, Nat Mar, doi:10.1038/s41586-020-2008-3
Yang, Liu, Zhou, Zhao, Zhao et al., The effect of corticosteroid treatment on patients with coronavirus infection: a systematic review and meta-analysis, J Infect Jul, doi:10.1016/j.jinf.2020.03.062
Zhao, Jia, Chen, A prospective, three-arm, randomized trial of EGCG for preventing radiation-induced esophagitis in lung cancer patients receiving radiotherapy, Radiother Oncol, doi:10.1016/j.radonc.2019.02.022
Zhao, Zhu, Xie, A phase I study of concurrent chemotherapy and thoracic radiotherapy with oral epigallocatechin-3-gallate protection in patients with locally advanced stage III non-small-cell lung cancer, Radiother Oncol, doi:10.1016/j.radonc.2013.10.014
Zhao, Zhu, Zhao, Efficacy of Epigallocatechin-3-Gallate in preventing Dermatitis in patients with breast Cancer receiving postoperative Radiotherapy: a Double-Blind, Placebo-Controlled, phase 2 Randomized Clinical Trial, JAMA Dermatol, doi:10.1001/jamadermatol.2022.1736
Zheng, Peng, Xu, Risk factors of critical & mortal COVID-19 cases: a systematic literature review and meta-analysis, J Infect Aug, doi:10.1016/j.jinf.2020.04.021
DOI record:
{
"DOI": "10.1186/s12885-024-12228-3",
"ISSN": [
"1471-2407"
],
"URL": "http://dx.doi.org/10.1186/s12885-024-12228-3",
"abstract": "<jats:title>Abstract</jats:title><jats:sec>\n <jats:title>Background</jats:title>\n <jats:p>The antiviral drug Nirmatrelvir was found to be a key drug in controlling the progression of pneumonia during the infectious phase of COVID-19. However, there are very few options for effective treatment for cancer patients who have viral pneumonia. Glucocorticoids is one of the effective means to control pneumonia, but there are many adverse events. EGCG is a natural low toxic compound with anti-inflammatory function. Thus, this study was designed to investigate the safety and efficacy of epigallocatechin-3-gallate (EGCG) aerosol to control COVID-19 pneumonia in cancer populations.</jats:p>\n </jats:sec><jats:sec>\n <jats:title>Methods</jats:title>\n <jats:p>The study was designed as a prospective, single-arm, open-label phase I/II trial at Shandong Cancer Hospital and Institute, between January 5, 2023 to March 31,2023 with viral pneumonia on radiographic signs after confirmed novel coronavirus infection. These patients were treated with EGCG nebulization 10 ml three times daily for at least seven days. EGCG concentrations were increased from 1760-8817umol/L to 4 levels with dose escalation following a standard Phase I design of 3–6 patients per level. Any grade adverse event caused by EGCG was considered a dose-limiting toxicity (DLT). The maximum tolerated dose (MTD) is defined as the highest dose with less than one-third of patients experiencing dose limiting toxicity (DLT) due to EGCG. The primary end points were the toxicity of EGCG and CT findings, and the former was graded by Common Terminology Criteria for Adverse Events (CTCAE) v. 5.0. The secondary end point was the laboratory parameters before and after treatment.</jats:p>\n </jats:sec><jats:sec>\n <jats:title>Result</jats:title>\n <jats:p>A total of 60 patients with high risk factors for severe COVID-19 pneumonia (factors such as old age, smoking and combined complications)were included in this phase I-II study. The 54 patients in the final analysis were pathologically confirmed to have tumor burden and completed the whole course of treatment. A patient with bucking at a level of 1760 umol/L and no acute toxicity associated with EGCG has been reported at the second or third dose gradients. At dose escalation to 8817umol/L, Grade 1 adverse events of nausea and stomach discomfort occurred in two patients, which resolved spontaneously within 1 hour. After one week of treatment, CT showed that the incidence of non-progression of pneumonia was 82% (32/39), and the improvement rate of pneumonia was 56.4% (22/39). There was no significant difference in inflammation-related laboratory parameters (white blood cell count, lymphocyte count, IL-6, ferritin, C-reactive protein and lactate dehydrogenase) before and after treatment.</jats:p>\n </jats:sec><jats:sec>\n <jats:title>Conclusion</jats:title>\n <jats:p>Aerosol inhalation of EGCG is well tolerated, and preliminary investigation in cancer population suggests that EGCG may be effective in COVID-19-induced pneumonia, which can promote the improvement of patients with moderate pneumonia or prevent them from developing into severe pneumonia.</jats:p>\n </jats:sec><jats:sec>\n <jats:title>Trial registration</jats:title>\n <jats:p>ClinicalTrials.gov Identifier: NCT05758571. Date of registration: 8 February 2023.</jats:p>\n </jats:sec>",
"alternative-id": [
"12228"
],
"article-number": "486",
"assertion": [
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "Received",
"name": "received",
"order": 1,
"value": "14 November 2023"
},
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "Accepted",
"name": "accepted",
"order": 2,
"value": "5 April 2024"
},
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "First Online",
"name": "first_online",
"order": 3,
"value": "17 April 2024"
},
{
"group": {
"label": "Declarations",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 1
},
{
"group": {
"label": "Ethics approval and consent to participate",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 2,
"value": "This study was approved by the Institutional Review and Ethical Committees at the affiliated Cancer Hospital of Shandong first Medical University and registered at ClinicalTrials.gov (NCT057-58571). Informed consent was provided by every participant."
},
{
"group": {
"label": "Consent for publication",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 3,
"value": "Not applicable."
},
{
"group": {
"label": "Competing interests",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 4,
"value": "The authors declare no competing interests."
}
],
"author": [
{
"affiliation": [],
"family": "Yin",
"given": "Xiaoyan",
"sequence": "first"
},
{
"affiliation": [],
"family": "Zhu",
"given": "Wanqi",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Tang",
"given": "Xiaoyong",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Yang",
"given": "Guangjian",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Zhao",
"given": "Xianguang",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Zhao",
"given": "Kaikai",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Jiang",
"given": "Liyang",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Li",
"given": "Xiaolin",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Zhao",
"given": "Hong",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Wang",
"given": "Xin",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Yan",
"given": "Yuanyuan",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Xing",
"given": "Ligang",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Yu",
"given": "Jinming",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Meng",
"given": "Xiangjiao",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Zhao",
"given": "Hanxi",
"sequence": "additional"
}
],
"container-title": "BMC Cancer",
"container-title-short": "BMC Cancer",
"content-domain": {
"crossmark-restriction": false,
"domain": [
"link.springer.com"
]
},
"created": {
"date-parts": [
[
2024,
4,
17
]
],
"date-time": "2024-04-17T08:29:24Z",
"timestamp": 1713342564000
},
"deposited": {
"date-parts": [
[
2024,
4,
17
]
],
"date-time": "2024-04-17T08:30:49Z",
"timestamp": 1713342649000
},
"funder": [
{
"DOI": "10.13039/501100001809",
"award": [
"81972863",
"No.81972796"
],
"doi-asserted-by": "publisher",
"name": "National Natural Science Foundation of China"
},
{
"award": [
"ZR201911040452"
],
"name": "the foundation of Natural Science Foundation of Shandong"
},
{
"award": [
"Y-HS202102-0089"
],
"name": "CSCO-Haosen Foundation"
}
],
"indexed": {
"date-parts": [
[
2024,
4,
18
]
],
"date-time": "2024-04-18T02:22:29Z",
"timestamp": 1713406949610
},
"is-referenced-by-count": 0,
"issue": "1",
"issued": {
"date-parts": [
[
2024,
4,
17
]
]
},
"journal-issue": {
"issue": "1",
"published-online": {
"date-parts": [
[
2024,
12
]
]
}
},
"language": "en",
"license": [
{
"URL": "https://creativecommons.org/licenses/by/4.0",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2024,
4,
17
]
],
"date-time": "2024-04-17T00:00:00Z",
"timestamp": 1713312000000
}
},
{
"URL": "https://creativecommons.org/licenses/by/4.0",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2024,
4,
17
]
],
"date-time": "2024-04-17T00:00:00Z",
"timestamp": 1713312000000
}
}
],
"link": [
{
"URL": "https://link.springer.com/content/pdf/10.1186/s12885-024-12228-3.pdf",
"content-type": "application/pdf",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://link.springer.com/article/10.1186/s12885-024-12228-3/fulltext.html",
"content-type": "text/html",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://link.springer.com/content/pdf/10.1186/s12885-024-12228-3.pdf",
"content-type": "application/pdf",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "297",
"original-title": [],
"prefix": "10.1186",
"published": {
"date-parts": [
[
2024,
4,
17
]
]
},
"published-online": {
"date-parts": [
[
2024,
4,
17
]
]
},
"publisher": "Springer Science and Business Media LLC",
"reference": [
{
"DOI": "10.1001/jama.2020.12839",
"author": "WJ Wiersinga",
"doi-asserted-by": "publisher",
"first-page": "782",
"issue": "8",
"journal-title": "Jama Aug",
"key": "12228_CR1",
"unstructured": "Wiersinga WJ, Rhodes A, Cheng AC, Peacock SJ, Prescott HC, Pathophysiology. Transmission, diagnosis, and treatment of Coronavirus Disease 2019 (COVID-19): a review. Jama Aug. 2020;25(8):782–93. https://doi.org/10.1001/jama.2020.12839.",
"volume": "25",
"year": "2020"
},
{
"DOI": "10.1038/s41586-020-2008-3",
"author": "F Wu",
"doi-asserted-by": "publisher",
"first-page": "265",
"issue": "7798",
"journal-title": "Nat Mar",
"key": "12228_CR2",
"unstructured": "Wu F, Zhao S, Yu B, et al. A new coronavirus associated with human respiratory disease in China. Nat Mar. 2020;579(7798):265–9. https://doi.org/10.1038/s41586-020-2008-3.",
"volume": "579",
"year": "2020"
},
{
"DOI": "10.1093/cid/ciaa1012",
"author": "L Kim",
"doi-asserted-by": "publisher",
"first-page": "e206",
"issue": "9",
"journal-title": "Clin Infect Dis May",
"key": "12228_CR3",
"unstructured": "Kim L, Garg S, O’Halloran A, et al. Risk factors for Intensive Care Unit Admission and In-hospital mortality among hospitalized adults identified through the US Coronavirus Disease 2019 (COVID-19)-Associated Hospitalization Surveillance Network (COVID-NET). Clin Infect Dis May. 2021;4(9):e206–14. https://doi.org/10.1093/cid/ciaa1012.",
"volume": "4",
"year": "2021"
},
{
"DOI": "10.1016/j.jinf.2020.04.021",
"author": "Z Zheng",
"doi-asserted-by": "publisher",
"first-page": "e16",
"issue": "2",
"journal-title": "J Infect Aug",
"key": "12228_CR4",
"unstructured": "Zheng Z, Peng F, Xu B, et al. Risk factors of critical & mortal COVID-19 cases: a systematic literature review and meta-analysis. J Infect Aug. 2020;81(2):e16–25. https://doi.org/10.1016/j.jinf.2020.04.021.",
"volume": "81",
"year": "2020"
},
{
"DOI": "10.2188/jea.JE20220321",
"author": "S Gallus",
"doi-asserted-by": "publisher",
"first-page": "367",
"issue": "7",
"journal-title": "J Epidemiol Jul",
"key": "12228_CR5",
"unstructured": "Gallus S, Bosetti C, Gorini G, et al. The Association of Tobacco Smoking, second-hand smoke, and Novel Tobacco products with COVID-19 severity and mortality in Italy: results from the COSMO-IT Study. J Epidemiol Jul. 2023;5(7):367–71. https://doi.org/10.2188/jea.JE20220321.",
"volume": "5",
"year": "2023"
},
{
"DOI": "10.1038/s41598-021-88130-w",
"author": "B Thakur",
"doi-asserted-by": "publisher",
"first-page": "8562",
"issue": "1",
"journal-title": "Sci Rep Apr",
"key": "12228_CR6",
"unstructured": "Thakur B, Dubey P, Benitez J, et al. A systematic review and meta-analysis of geographic differences in comorbidities and associated severity and mortality among individuals with COVID-19. Sci Rep Apr. 2021;20(1):8562. https://doi.org/10.1038/s41598-021-88130-w.",
"volume": "20",
"year": "2021"
},
{
"DOI": "10.1056/NEJMoa2118542",
"author": "J Hammond",
"doi-asserted-by": "publisher",
"first-page": "1397",
"issue": "15",
"journal-title": "N Engl J Med",
"key": "12228_CR7",
"unstructured": "Hammond J, Leister-Tebbe H, Gardner A, et al. Oral nirmatrelvir for High-Risk, nonhospitalized adults with Covid-19. N Engl J Med. Apr 2022;14(15):1397–408. https://doi.org/10.1056/NEJMoa2118542.",
"volume": "14",
"year": "2022"
},
{
"DOI": "10.1016/j.jaut.2017.07.010",
"author": "A Sica",
"doi-asserted-by": "publisher",
"first-page": "117",
"journal-title": "J Autoimmun Dec",
"key": "12228_CR8",
"unstructured": "Sica A, Massarotti M. Myeloid suppressor cells in cancer and autoimmunity. J Autoimmun Dec. 2017;85:117–25. https://doi.org/10.1016/j.jaut.2017.07.010.",
"volume": "85",
"year": "2017"
},
{
"DOI": "10.1016/s1470-2045(20)30096-6",
"author": "W Liang",
"doi-asserted-by": "publisher",
"first-page": "335",
"issue": "3",
"journal-title": "Lancet Oncol Mar",
"key": "12228_CR9",
"unstructured": "Liang W, Guan W, Chen R, et al. Cancer patients in SARS-CoV-2 infection: a nationwide analysis in China. Lancet Oncol Mar. 2020;21(3):335–7. https://doi.org/10.1016/s1470-2045(20)30096-6.",
"volume": "21",
"year": "2020"
},
{
"DOI": "10.1158/2159-8290.Cd-20-0422",
"author": "M Dai",
"doi-asserted-by": "publisher",
"first-page": "783",
"issue": "6",
"journal-title": "Cancer Discov Jun",
"key": "12228_CR10",
"unstructured": "Dai M, Liu D, Liu M, et al. Patients with Cancer Appear more vulnerable to SARS-CoV-2: a Multicenter Study during the COVID-19 outbreak. Cancer Discov Jun. 2020;10(6):783–91. https://doi.org/10.1158/2159-8290.Cd-20-0422.",
"volume": "10",
"year": "2020"
},
{
"DOI": "10.1164/rccm.202008-3265OC",
"author": "DA Dorward",
"doi-asserted-by": "publisher",
"first-page": "192",
"issue": "2",
"journal-title": "Am J Respir Crit Care Med Jan",
"key": "12228_CR11",
"unstructured": "Dorward DA, Russell CD, Um IH, et al. Tissue-specific immunopathology in fatal COVID-19. Am J Respir Crit Care Med Jan. 2021;15(2):192–201. https://doi.org/10.1164/rccm.202008-3265OC.",
"volume": "15",
"year": "2021"
},
{
"DOI": "10.3390/ijms21145171",
"doi-asserted-by": "publisher",
"key": "12228_CR12",
"unstructured": "Menegazzi M, Campagnari R, Bertoldi M, Crupi R, Di Paola R, Cuzzocrea S. Protective effect of Epigallocatechin-3-Gallate (EGCG) in diseases with uncontrolled Immune activation: could such a scenario be helpful to Counteract COVID-19? Int J Mol Sci Jul. 2020;21(14). https://doi.org/10.3390/ijms21145171."
},
{
"DOI": "10.1016/j.compbiomed.2020.103792",
"author": "T Ozturk",
"doi-asserted-by": "publisher",
"first-page": "103792",
"journal-title": "Comput Biol Med Jun",
"key": "12228_CR13",
"unstructured": "Ozturk T, Talo M, Yildirim EA, Baloglu UB, Yildirim O, Rajendra Acharya U. Automated detection of COVID-19 cases using deep neural networks with X-ray images. Comput Biol Med Jun. 2020;121:103792. https://doi.org/10.1016/j.compbiomed.2020.103792.",
"volume": "121",
"year": "2020"
},
{
"DOI": "10.1016/0027-5107(85)90109-5",
"author": "T Kada",
"doi-asserted-by": "publisher",
"first-page": "127",
"issue": "1–2",
"journal-title": "Mutat Res Jun-Jul",
"key": "12228_CR14",
"unstructured": "Kada T, Kaneko K, Matsuzaki S, Matsuzaki T, Hara Y. Detection and chemical identification of natural bio-antimutagens. A case of the green tea factor. Mutat Res Jun-Jul. 1985;150(1–2):127–32. https://doi.org/10.1016/0027-5107(85)90109-5.",
"volume": "150",
"year": "1985"
},
{
"DOI": "10.1080/13543776.2016.1203419",
"author": "L Chakrawarti",
"doi-asserted-by": "publisher",
"first-page": "907",
"issue": "8",
"journal-title": "Expert Opin Ther Pat Aug",
"key": "12228_CR15",
"unstructured": "Chakrawarti L, Agrawal R, Dang S, Gupta S, Gabrani R. Therapeutic effects of EGCG: a patent review. Expert Opin Ther Pat Aug. 2016;26(8):907–16. https://doi.org/10.1080/13543776.2016.1203419.",
"volume": "26",
"year": "2016"
},
{
"DOI": "10.1016/j.radonc.2019.02.022",
"author": "H Zhao",
"doi-asserted-by": "publisher",
"first-page": "186",
"journal-title": "Radiother Oncol Aug",
"key": "12228_CR16",
"unstructured": "Zhao H, Jia L, Chen G, et al. A prospective, three-arm, randomized trial of EGCG for preventing radiation-induced esophagitis in lung cancer patients receiving radiotherapy. Radiother Oncol Aug. 2019;137:186–91. https://doi.org/10.1016/j.radonc.2019.02.022.",
"volume": "137",
"year": "2019"
},
{
"DOI": "10.1016/j.radonc.2013.10.014",
"author": "H Zhao",
"doi-asserted-by": "publisher",
"first-page": "132",
"issue": "1",
"journal-title": "Radiother Oncol Jan",
"key": "12228_CR17",
"unstructured": "Zhao H, Zhu W, Xie P, et al. A phase I study of concurrent chemotherapy and thoracic radiotherapy with oral epigallocatechin-3-gallate protection in patients with locally advanced stage III non-small-cell lung cancer. Radiother Oncol Jan. 2014;110(1):132–6. https://doi.org/10.1016/j.radonc.2013.10.014.",
"volume": "110",
"year": "2014"
},
{
"DOI": "10.1001/jamadermatol.2022.1736",
"author": "H Zhao",
"doi-asserted-by": "publisher",
"first-page": "779",
"issue": "7",
"journal-title": "JAMA Dermatol Jul",
"key": "12228_CR18",
"unstructured": "Zhao H, Zhu W, Zhao X, et al. Efficacy of Epigallocatechin-3-Gallate in preventing Dermatitis in patients with breast Cancer receiving postoperative Radiotherapy: a Double-Blind, Placebo-Controlled, phase 2 Randomized Clinical Trial. JAMA Dermatol Jul. 2022;1(7):779–86. https://doi.org/10.1001/jamadermatol.2022.1736.",
"volume": "1",
"year": "2022"
},
{
"DOI": "10.1089/jmf.2019.4445",
"author": "X Li",
"doi-asserted-by": "publisher",
"first-page": "43",
"issue": "1",
"journal-title": "J Med Food Jan",
"key": "12228_CR19",
"unstructured": "Li X, Xing L, Zhang Y, et al. Phase II trial of Epigallocatechin-3-Gallate in Acute Radiation-Induced Esophagitis for Esophagus Cancer. J Med Food Jan. 2020;23(1):43–9. https://doi.org/10.1089/jmf.2019.4445.",
"volume": "23",
"year": "2020"
},
{
"DOI": "10.1016/j.yrtph.2018.03.019",
"author": "J Hu",
"doi-asserted-by": "publisher",
"first-page": "412",
"journal-title": "Regul Toxicol Pharmacol Jun",
"key": "12228_CR20",
"unstructured": "Hu J, Webster D, Cao J, Shao A. The safety of green tea and green tea extract consumption in adults - results of a systematic review. Regul Toxicol Pharmacol Jun. 2018;95:412–33. https://doi.org/10.1016/j.yrtph.2018.03.019.",
"volume": "95",
"year": "2018"
},
{
"DOI": "10.1016/j.ejps.2010.06.010",
"author": "A Dube",
"doi-asserted-by": "publisher",
"first-page": "219",
"issue": "2",
"journal-title": "Eur J Pharm Sci Oct",
"key": "12228_CR21",
"unstructured": "Dube A, Nicolazzo JA, Larson I. Chitosan nanoparticles enhance the intestinal absorption of the green tea catechins (+)-catechin and (-)-epigallocatechin gallate. Eur J Pharm Sci Oct. 2010;9(2):219–25. https://doi.org/10.1016/j.ejps.2010.06.010.",
"volume": "9",
"year": "2010"
},
{
"DOI": "10.3390/molecules23092346",
"doi-asserted-by": "publisher",
"key": "12228_CR22",
"unstructured": "Cai ZY, Li XM, Liang JP, et al. Bioavailability of tea catechins and its improvement. Molecules Sep. 2018;13(9). https://doi.org/10.3390/molecules23092346."
},
{
"DOI": "10.1371/journal.pone.0092876",
"author": "PG Vidigal",
"doi-asserted-by": "publisher",
"first-page": "e92876",
"issue": "4",
"journal-title": "PLoS ONE",
"key": "12228_CR23",
"unstructured": "Vidigal PG, Müsken M, Becker KA, et al. Effects of green tea compound epigallocatechin-3-gallate against Stenotrophomonas maltophilia infection and biofilm. PLoS ONE. 2014;9(4):e92876. https://doi.org/10.1371/journal.pone.0092876.",
"volume": "9",
"year": "2014"
},
{
"DOI": "10.1016/j.xphs.2022.06.004",
"author": "G Sahin",
"doi-asserted-by": "publisher",
"first-page": "2652",
"issue": "10",
"journal-title": "J Pharm Sci Oct",
"key": "12228_CR24",
"unstructured": "Sahin G, Akbal-Dagistan O, Culha M, et al. Antivirals and the potential benefits of orally inhaled Drug Administration in COVID-19 treatment. J Pharm Sci Oct. 2022;111(10):2652–61. https://doi.org/10.1016/j.xphs.2022.06.004.",
"volume": "111",
"year": "2022"
},
{
"DOI": "10.3390/covid1010002",
"author": "S Bettuzzi",
"doi-asserted-by": "publisher",
"first-page": "2",
"issue": "1",
"journal-title": "COVID",
"key": "12228_CR25",
"unstructured": "Bettuzzi S, Gabba L, Cataldo S. Efficacy of a polyphenolic, standardized Green Tea Extract for the treatment of COVID-19 syndrome: a proof-of-Principle Study. COVID. 2021;1(1):2–12.",
"volume": "1",
"year": "2021"
},
{
"DOI": "10.1038/s41579-022-00713-0",
"author": "MM Lamers",
"doi-asserted-by": "publisher",
"first-page": "270",
"issue": "5",
"journal-title": "Nat Rev Microbiol May",
"key": "12228_CR26",
"unstructured": "Lamers MM, Haagmans BL. SARS-CoV-2 pathogenesis. Nat Rev Microbiol May. 2022;20(5):270–84. https://doi.org/10.1038/s41579-022-00713-0.",
"volume": "20",
"year": "2022"
},
{
"DOI": "10.1016/j.healun.2020.03.012",
"doi-asserted-by": "publisher",
"key": "12228_CR27",
"unstructured": "Siddiqi HK, Mehra MR. COVID-19 illness in native and immunosuppressed states: a clinical-therapeutic staging proposal. J Heart Lung Transpl. May 2020;39(5):405–7. https://doi.org/10.1016/j.healun.2020.03.012."
},
{
"DOI": "10.1126/scitranslmed.abj7790",
"author": "F D’Agnillo",
"doi-asserted-by": "publisher",
"first-page": "eabj7790",
"issue": "620",
"journal-title": "Sci Transl Med Nov",
"key": "12228_CR28",
"unstructured": "D’Agnillo F, Walters KA, Xiao Y, et al. Lung epithelial and endothelial damage, loss of tissue repair, inhibition of fibrinolysis, and cellular senescence in fatal COVID-19. Sci Transl Med Nov. 2021;17(620):eabj7790. https://doi.org/10.1126/scitranslmed.abj7790.",
"volume": "17",
"year": "2021"
},
{
"DOI": "10.1002/jmv.26610",
"author": "CW Stratton",
"doi-asserted-by": "publisher",
"first-page": "1320",
"issue": "3",
"journal-title": "J Med Virol Mar",
"key": "12228_CR29",
"unstructured": "Stratton CW, Tang YW, Lu H. Pathogenesis-directed therapy of 2019 novel coronavirus disease. J Med Virol Mar. 2021;93(3):1320–42. https://doi.org/10.1002/jmv.26610.",
"volume": "93",
"year": "2021"
},
{
"DOI": "10.1056/NEJMcp2009575",
"author": "DA Berlin",
"doi-asserted-by": "publisher",
"first-page": "2451",
"issue": "25",
"journal-title": "N Engl J Med Dec",
"key": "12228_CR30",
"unstructured": "Berlin DA, Gulick RM, Martinez FJ. Severe Covid-19. N Engl J Med Dec. 2020;17(25):2451–60. https://doi.org/10.1056/NEJMcp2009575.",
"volume": "17",
"year": "2020"
},
{
"DOI": "10.1002/jmv.26209",
"author": "I Andrianopoulos",
"doi-asserted-by": "publisher",
"first-page": "120",
"issue": "1",
"journal-title": "J Med Virol Jan",
"key": "12228_CR31",
"unstructured": "Andrianopoulos I, Papathanasiou A, Papathanakos G, Chaidos A, Koulouras V. Tocilizumab’s efficacy in patients with Coronavirus Disease 2019 (COVID-19) is determined by the presence of cytokine storm. J Med Virol Jan. 2021;93(1):120–1. https://doi.org/10.1002/jmv.26209.",
"volume": "93",
"year": "2021"
},
{
"DOI": "10.1016/s0140-6736(20)30920-x",
"author": "H Li",
"doi-asserted-by": "publisher",
"first-page": "1517",
"issue": "10235",
"journal-title": "Lancet May",
"key": "12228_CR32",
"unstructured": "Li H, Liu L, Zhang D, et al. SARS-CoV-2 and viral sepsis: observations and hypotheses. Lancet May. 2020;9(10235):1517–20. https://doi.org/10.1016/s0140-6736(20)30920-x.",
"volume": "9",
"year": "2020"
},
{
"DOI": "10.1016/s2213-2600(19)30417-5",
"doi-asserted-by": "publisher",
"key": "12228_CR33",
"unstructured": "Villar J, Ferrando C, Martínez D, et al. Dexamethasone treatment for the acute respiratory distress syndrome: a multicentre, randomised controlled trial. Lancet Respir Med. Mar 2020;8(3):267–76. https://doi.org/10.1016/s2213-2600(19)30417-5."
},
{
"DOI": "10.1016/j.jinf.2020.03.062",
"author": "Z Yang",
"doi-asserted-by": "publisher",
"first-page": "e13",
"issue": "1",
"journal-title": "J Infect Jul",
"key": "12228_CR34",
"unstructured": "Yang Z, Liu J, Zhou Y, Zhao X, Zhao Q, Liu J. The effect of corticosteroid treatment on patients with coronavirus infection: a systematic review and meta-analysis. J Infect Jul. 2020;81(1):e13–20. https://doi.org/10.1016/j.jinf.2020.03.062.",
"volume": "81",
"year": "2020"
},
{
"DOI": "10.1016/j.jviromet.2021.114419",
"author": "A Pezzuto",
"doi-asserted-by": "publisher",
"first-page": "114419",
"journal-title": "J Virol Methods Feb",
"key": "12228_CR35",
"unstructured": "Pezzuto A, Tammaro A, Tonini G, et al. SARS-Cov-2 pneumonia and concurrent myelodysplasia complicated by Pseudomonas aeruginosa over-infection. J Virol Methods Feb. 2022;300:114419. https://doi.org/10.1016/j.jviromet.2021.114419.",
"volume": "300",
"year": "2022"
},
{
"DOI": "10.1016/j.compbiomed.2022.105241",
"author": "B Wang",
"doi-asserted-by": "publisher",
"first-page": "105241",
"journal-title": "Comput Biol Med Jan",
"key": "12228_CR36",
"unstructured": "Wang B, Ding Y, Zhao P, et al. Systems pharmacology-based drug discovery and active mechanism of natural products for coronavirus pneumonia (COVID-19): an example using flavonoids. Comput Biol Med Jan. 2022;26:143:105241. https://doi.org/10.1016/j.compbiomed.2022.105241.",
"volume": "26",
"year": "2022"
},
{
"DOI": "10.1016/j.ejphar.2020.173107",
"author": "T Gharibi",
"doi-asserted-by": "publisher",
"first-page": "173107",
"journal-title": "Eur J Pharmacol Jul",
"key": "12228_CR37",
"unstructured": "Gharibi T, Babaloo Z, Hosseini A, et al. Targeting STAT3 in cancer and autoimmune diseases. Eur J Pharmacol Jul. 2020;5:878:173107. https://doi.org/10.1016/j.ejphar.2020.173107.",
"volume": "5",
"year": "2020"
},
{
"DOI": "10.4049/jimmunol.173.9.5794",
"author": "O Aktas",
"doi-asserted-by": "publisher",
"first-page": "5794",
"issue": "9",
"journal-title": "J Immunol Nov",
"key": "12228_CR38",
"unstructured": "Aktas O, Prozorovski T, Smorodchenko A, et al. Green tea epigallocatechin-3-gallate mediates T cellular NF-kappa B inhibition and exerts neuroprotection in autoimmune encephalomyelitis. J Immunol Nov. 2004;1(9):5794–800. https://doi.org/10.4049/jimmunol.173.9.5794.",
"volume": "1",
"year": "2004"
},
{
"DOI": "10.2174/1871523015666160915154443",
"author": "T Ohishi",
"doi-asserted-by": "publisher",
"first-page": "74",
"issue": "2",
"journal-title": "Antiinflamm Antiallergy Agents Med Chem",
"key": "12228_CR39",
"unstructured": "Ohishi T, Goto S, Monira P, Isemura M, Nakamura Y. Anti-inflammatory action of Green Tea. Antiinflamm Antiallergy Agents Med Chem. 2016;15(2):74–90. https://doi.org/10.2174/1871523015666160915154443.",
"volume": "15",
"year": "2016"
},
{
"DOI": "10.2174/1872213x13666190426164124",
"author": "G Martínez",
"doi-asserted-by": "publisher",
"first-page": "84",
"issue": "2",
"journal-title": "Recent Pat Inflamm Allergy Drug Discov",
"key": "12228_CR40",
"unstructured": "Martínez G, Mijares MR, De Sanctis JB. Effects of flavonoids and its derivatives on Immune cell responses. Recent Pat Inflamm Allergy Drug Discov. 2019;13(2):84–104. https://doi.org/10.2174/1872213x13666190426164124.",
"volume": "13",
"year": "2019"
},
{
"DOI": "10.1093/carcin/bgp127",
"author": "F Colotta",
"doi-asserted-by": "publisher",
"first-page": "1073",
"issue": "7",
"journal-title": "Carcinog Jul",
"key": "12228_CR41",
"unstructured": "Colotta F, Allavena P, Sica A, Garlanda C, Mantovani A. Cancer-related inflammation, the seventh hallmark of cancer: links to genetic instability. Carcinog Jul. 2009;30(7):1073–81. https://doi.org/10.1093/carcin/bgp127.",
"volume": "30",
"year": "2009"
},
{
"DOI": "10.1038/nrc3611",
"author": "E Elinav",
"doi-asserted-by": "publisher",
"first-page": "759",
"issue": "11",
"journal-title": "Nat Rev Cancer Nov",
"key": "12228_CR42",
"unstructured": "Elinav E, Nowarski R, Thaiss CA, Hu B, Jin C, Flavell RA. Inflammation-induced cancer: crosstalk between tumours, immune cells and microorganisms. Nat Rev Cancer Nov. 2013;13(11):759–71. https://doi.org/10.1038/nrc3611.",
"volume": "13",
"year": "2013"
},
{
"DOI": "10.1016/j.annonc.2020.03.286",
"author": "F Cortiula",
"doi-asserted-by": "publisher",
"first-page": "553",
"issue": "5",
"journal-title": "Ann Oncol May",
"key": "12228_CR43",
"unstructured": "Cortiula F, Pettke A, Bartoletti M, Puglisi F, Helleday T. Managing COVID-19 in the oncology clinic and avoiding the distraction effect. Ann Oncol May. 2020;31(5):553–5. https://doi.org/10.1016/j.annonc.2020.03.286.",
"volume": "31",
"year": "2020"
},
{
"DOI": "10.1038/s41392-021-00603-6",
"author": "H Bian",
"doi-asserted-by": "publisher",
"first-page": "194",
"issue": "1",
"journal-title": "Signal Transduct Target Ther May",
"key": "12228_CR44",
"unstructured": "Bian H, Zheng ZH, Wei D, et al. Safety and efficacy of meplazumab in healthy volunteers and COVID-19 patients: a randomized phase 1 and an exploratory phase 2 trial. Signal Transduct Target Ther May. 2021;17(1):194. https://doi.org/10.1038/s41392-021-00603-6.",
"volume": "17",
"year": "2021"
}
],
"reference-count": 44,
"references-count": 44,
"relation": {},
"resource": {
"primary": {
"URL": "https://bmccancer.biomedcentral.com/articles/10.1186/s12885-024-12228-3"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [
"Cancer Research",
"Genetics",
"Oncology"
],
"subtitle": [],
"title": "Phase I/II clinical trial of efficacy and safety of EGCG oxygen nebulization inhalation in the treatment of COVID-19 pneumonia patients with cancer",
"type": "journal-article",
"update-policy": "http://dx.doi.org/10.1007/springer_crossmark_policy",
"volume": "24"
}