Utility of mouth rinses with povidone-iodine and hydrogen peroxide in patients with COVID-19
et al., Enfermedades Infecciosas y Microbiología Clínica, doi:10.1016/j.eimc.2021.10.005, Oct 2021
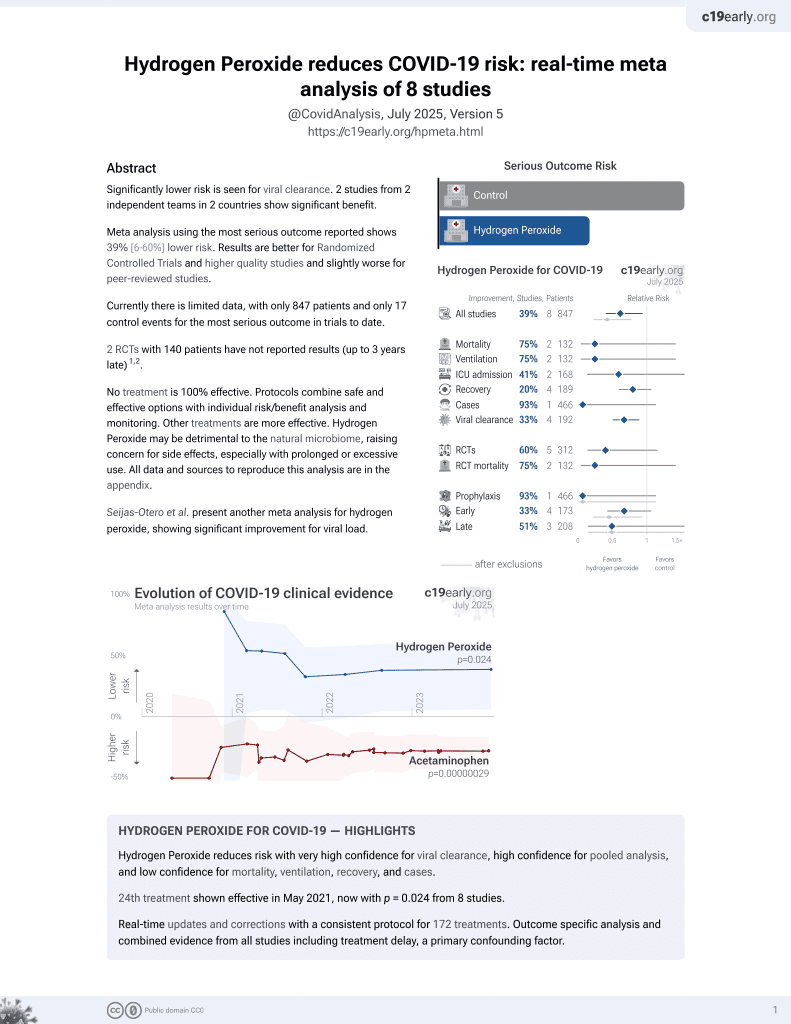
23rd treatment shown to reduce risk in
May 2021, now with p = 0.024 from 8 studies.
Lower risk for viral clearance.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
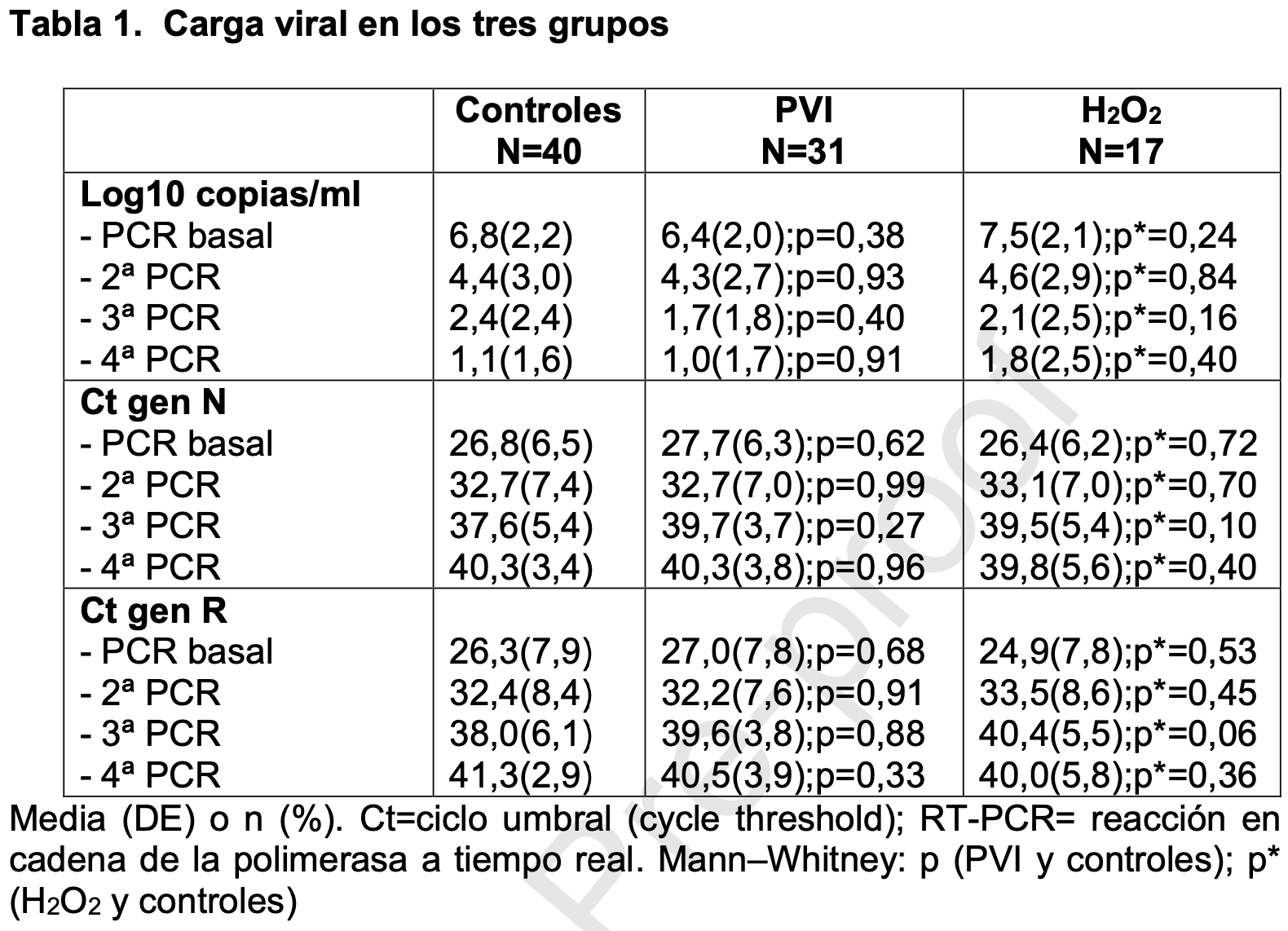
Small prospective study with 31 patients gargling povidone-iodine, 17 hydrogen peroxide, and 40 control patients, showing lower viral load mid-recovery with povidone-iodine, without reaching statistical significance. Oropharyngeal only, and only every 8 hours for two days. Results may be better with the addition of nasopharyngeal use, more frequent use, and without the two day limit.
Authors report only one of the 7 previous trials for PVP-I and COVID-19. Non-randomized study with no adjustments or group details. Some results in Figure 1 appear to be switched compared to the text and the labels in the figure. The viral clearance figures do not match the group sizes - for example authors report 62% PCR- for PVP-I at the 3rd test, however there is no number of 31 patients that rounds to 62%.
Targeted administration to the respiratory tract provides treatment directly
to the typical source of initial SARS-CoV-2 infection and replication, and
allows for rapid onset of action, higher local drug concentration, and reduced systemic side effects.
This study is excluded in the after exclusion results of meta-analysis:
unadjusted results with no group details.
Study covers hydrogen peroxide and povidone-iodine.
|
relative viral load, 12.5% better, RR 0.88, p = 0.67, treatment mean 2.1 (±2.5) n=17, control mean 2.4 (±2.4) n=40, 3rd PCR (mid-recovery).
|
|
relative viral load, 63.6% worse, RR 1.64, p = 0.16, treatment mean 1.8 (±2.5) n=31, control mean 1.1 (±1.6) n=40, 4th PCR (most patients recovered).
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Pablo-Marcos et al., 25 Oct 2021, prospective, Spain, peer-reviewed, mean age 43.0, 6 authors, study period May 2020 - November 2020.
Contact: mirvdc@humv.es.
Utilidad de los enjuagues con povidona yodada y peróxido de hidrógeno en pacientes con COVID-19
Enfermedades Infecciosas y Microbiología Clínica, doi:10.1016/j.eimc.2021.10.005
Introduction: Povidone-iodine and hydrogen peroxide could be effective in against SARS-CoV-2. Methods: A "non-interventional trial" in 88 patients (43 ± 17 yrs., 55% men) with SARS-CoV-2 in nasopharyngeal swabs (RT-PCR). 31 received mouth rinses/gargling with povidone-iodine (every 8 h, two consecutive days), 17 with mouth rinses/gargling of hydrogen peroxide, and 40 controls. Were repeated PCR in 3, 11 and 17 days. Results: After intervention the viral load (Log10 copies/ml) remained similar in povidone-iodine (4.3 ± 2.7 copies/ml), hydrogen peroxide (4.6 ± 2.9 copies/ml; p = 0.40) and controls (4.4 ± 3.0 copies/ml). The percentage of patients with a negative result in the second PCR was 27% in povidone-iodine group, 23% in hydrogen peroxide and 32% in controls; in the third PCR, 62%, 54% y 58% respectively; and in the fourth PCR, 81%, 75% y 81%. Conclusion: Our results do not support the clinical usefulness of mouth rinses/gargling with povidoneiodine or hydrogen peroxide in patients with COVID-19.
G Model We only found one other study, without a control group, in 12 hospitalised COVID-19 patients who received H 2 O 2 (1%) mouth rinses, with no change in their oropharyngeal viral load at 30 min 13 . Our study has limitations, as it is not randomised. However, we included a large number of patients with a control group and evaluated the utility of two commonly used oral antiseptics. We conclude that rinses and gargles of PVI and H 2 O 2 do not seem to have a clear utility in reducing the oropharyngeal viral load of SARS-CoV-2. We believe that the recommendation for the use of these and other oral antiseptics should be based on scientific evidence, hence more studies are needed to assess their efficacy in this pandemic.
Conflicts of interest The authors declare that they have no conflicts of interest.
References
Anderson, Sivalingam, Kang, Ananthanarayanan, Arumugam et al., Povidone-iodine demonstrates rapid in vitro virucidal activity against SARS-CoV-2, the virus causing COVID-19 disease, Infect Dis Ther, doi:10.1007/s40121-020-00316-3
Bidra, Pelletier, Westover, Frank, Brown et al., Comparison of in vitro inactivation of SARS CoV-2 with hydrogen peroxide and povidone-iodine oral antiseptic rinses, J Prosthodont, doi:10.1016/j.jhin.2020.01.022
Caruso, Del Prete, Lazzarino, Hydrogen peroxide and viral infections: a literature review with research hypothesis definition in relation to the current covid-19 pandemic, Med Hypotheses, doi:10.1016/j.micinf.201708008
Eggers, Eickmann, Zorn, Rapid and effective virucidal activity of povidone-iodine products against middle east respiratory syndrome coronavirus (MERS-CoV) and modified vaccinia virus ankara (MVA), Infect Dis Ther, doi:10.5867/medwave.2020.05.7916
Fini, Oral saliva and COVID-19, Oral Oncol, doi:10.1016/j.oraloncology.2020.104821
Guenezan, Garcia, Strasters, Jousselin, Lévêque et al., Povidone iodine mouthwash, gargle, and nasal spray to reduce nasopharyngeal viral load in patients with COVID-19: a randomized clinical trial, JAMA Otolaryngol Head Neck Surg, doi:10.1111/jopr.13220
Kampf, Todt, Pfaender, Steinmann, Persistence of coronaviruses on inanimate surfaces and their inactivation with biocidal agents, J Hosp Infect, doi:10.1007/s40121-020-00316-3
Kariwa, Fujii, Takashima, Inactivation of SARS coronavirus by means of povidone-iodine, physical conditions, and chemical reagents, Jpn J Vet Res
Liu, Chen, Liu, Chen, Liu et al., The role of oxidative stress in influenza virus infection, Microbes Infect, doi:10.1007/s00784-020-03549-1
Liu, Liao, Qian, Yuan, Wang et al., Community transmission of severe acute respiratory syndrome coronavirus 2, Shenzhen, China, 2020, Emerg Infect Dis, doi:10.3201/eid2606.200239
Maximilian, Gottsauner, Schmidt, Scholz, Buchalla et al., A prospective clinical pilot study on the effects of a hydrogen peroxide mouthrinse on the intraoral viral load of SARS-CoV-2, Clin Oral Investig, doi:10.1007/s00784-020-03549-1
Meselson, Droplets and aerosols in the transmission of SARS-CoV-2, N Engl J Med
Ortega, Rech, Haje, Gallo, Pérez-Sayáns et al., Do hydrogen peroxide mouthwashes have a virucidal effect? A systematic review, J Hosp Infect, doi:10.1016/j.mehy.2020.109910
Sriwilaijaroen, Wilairat, Hiramatsu, Takahashi, Suzuki et al., Mechanisms of the action of povidone-iodine against human and avian influenza A viruses: its effects on hemagglutination and sialidase activities, Virol J, doi:10.1001/jamaoto.2020.5490
Vargas-Buratovic, Verdugo-Paiva, Véliz-Paiva, López-Tagle, Ahumada-Salinas et al., Dental recommendations in the COVID-19 pandemic: a narrative review, Medwave, doi:10.1002/alr.22735
Yan, Bleier, Prophylactic and therapeutic topical povidone-iodine in coronavirus disease 2019 (COVID-19): What is the evidence?, Int Forum Allergy Rhinol, doi:10.1016/j.oraloncology.2020.104821
DOI record:
{
"DOI": "10.1016/j.eimc.2021.10.005",
"ISSN": [
"0213-005X"
],
"URL": "http://dx.doi.org/10.1016/j.eimc.2021.10.005",
"alternative-id": [
"S0213005X21002950"
],
"author": [
{
"affiliation": [],
"family": "Pablo-Marcos",
"given": "Daniel",
"sequence": "first"
},
{
"affiliation": [],
"family": "Abascal",
"given": "Beatriz",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Lloret",
"given": "Lara",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Gutiérrez Cuadra",
"given": "Manuel",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Velasco",
"given": "Nieves",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Valero",
"given": "Carmen",
"sequence": "additional"
}
],
"container-title": [
"Enfermedades Infecciosas y Microbiología Clínica"
],
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2021,
10,
25
]
],
"date-time": "2021-10-25T13:30:28Z",
"timestamp": 1635168628000
},
"deposited": {
"date-parts": [
[
2021,
11,
23
]
],
"date-time": "2021-11-23T22:44:03Z",
"timestamp": 1637707443000
},
"funder": [
{
"DOI": "10.13039/501100003339",
"doi-asserted-by": "publisher",
"name": "CSIC"
}
],
"indexed": {
"date-parts": [
[
2021,
11,
24
]
],
"date-time": "2021-11-24T06:46:40Z",
"timestamp": 1637736400566
},
"is-referenced-by-count": 0,
"issn-type": [
{
"type": "print",
"value": "0213-005X"
}
],
"issued": {
"date-parts": [
[
2021,
10
]
]
},
"language": "es",
"license": [
{
"URL": "https://www.elsevier.com/tdm/userlicense/1.0/",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2021,
10,
1
]
],
"date-time": "2021-10-01T00:00:00Z",
"timestamp": 1633046400000
}
}
],
"link": [
{
"URL": "https://api.elsevier.com/content/article/PII:S0213005X21002950?httpAccept=text/xml",
"content-type": "text/xml",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://api.elsevier.com/content/article/PII:S0213005X21002950?httpAccept=text/plain",
"content-type": "text/plain",
"content-version": "vor",
"intended-application": "text-mining"
}
],
"member": "78",
"original-title": [],
"prefix": "10.1016",
"published": {
"date-parts": [
[
2021,
10
]
]
},
"published-print": {
"date-parts": [
[
2021,
10
]
]
},
"publisher": "Elsevier BV",
"reference": [
{
"DOI": "10.1056/NEJMc2009324",
"article-title": "Droplets and aerosols in the transmission of SARS-CoV-2",
"author": "Meselson",
"doi-asserted-by": "crossref",
"first-page": "2063",
"journal-title": "N Engl J Med",
"key": "10.1016/j.eimc.2021.10.005_bib0085",
"volume": "382",
"year": "2020"
},
{
"DOI": "10.3201/eid2606.200239",
"article-title": "Community transmission of severe acute respiratory syndrome coronavirus 2, Shenzhen, China, 2020",
"author": "Liu",
"doi-asserted-by": "crossref",
"first-page": "1320",
"journal-title": "Emerg Infect Dis",
"key": "10.1016/j.eimc.2021.10.005_bib0090",
"volume": "26",
"year": "2020"
},
{
"DOI": "10.1016/j.oraloncology.2020.104821",
"article-title": "Oral saliva and COVID-19",
"author": "Baghizadeh Fini",
"doi-asserted-by": "crossref",
"first-page": "104821",
"journal-title": "Oral Oncol",
"key": "10.1016/j.eimc.2021.10.005_bib0095",
"volume": "108",
"year": "2020"
},
{
"DOI": "10.1002/alr.22735",
"article-title": "Prophylactic and therapeutic topical povidone-iodine in coronavirus disease 2019 (COVID-19): What is the evidence?",
"author": "Yan",
"doi-asserted-by": "crossref",
"first-page": "1271",
"journal-title": "Int Forum Allergy Rhinol",
"key": "10.1016/j.eimc.2021.10.005_bib0100",
"volume": "10",
"year": "2020"
},
{
"DOI": "10.5867/medwave.2020.05.7916",
"article-title": "Dental recommendations in the COVID-19 pandemic: A narrative review",
"author": "Vargas-Buratovic",
"doi-asserted-by": "crossref",
"first-page": "e7916",
"journal-title": "Medwave",
"key": "10.1016/j.eimc.2021.10.005_bib0105",
"volume": "20",
"year": "2020"
},
{
"DOI": "10.1007/s40121-015-0091-9",
"article-title": "Rapid and effective virucidal activity of povidone-iodine products against middle east respiratory syndrome coronavirus (MERS-CoV) and modified vaccinia virus Ankara (MVA)",
"author": "Eggers",
"doi-asserted-by": "crossref",
"first-page": "491",
"journal-title": "Infect Dis Ther",
"key": "10.1016/j.eimc.2021.10.005_bib0110",
"volume": "4",
"year": "2015"
},
{
"article-title": "Inactivation of SARS coronavirus by means of povidone-iodine, physical conditions, and chemical reagents",
"author": "Kariwa",
"first-page": "105",
"journal-title": "Jpn J Vet Res",
"key": "10.1016/j.eimc.2021.10.005_bib0115",
"volume": "52",
"year": "2004"
},
{
"DOI": "10.1007/s40121-020-00316-3",
"article-title": "Povidone-iodine demonstrates rapid in vitro virucidal activity against SARS-CoV-2. The virus causing COVID-19 disease",
"author": "Anderson",
"doi-asserted-by": "crossref",
"first-page": "669",
"journal-title": "Infect Dis Ther",
"key": "10.1016/j.eimc.2021.10.005_bib0120",
"volume": "9",
"year": "2020"
},
{
"DOI": "10.1016/j.jhin.2020.01.022",
"article-title": "Persistence of coronaviruses on inanimate surfaces and their inactivation with biocidal agents",
"author": "Kampf",
"doi-asserted-by": "crossref",
"first-page": "246",
"journal-title": "J Hosp Infect",
"key": "10.1016/j.eimc.2021.10.005_bib0125",
"volume": "104",
"year": "2020"
},
{
"DOI": "10.1111/jopr.13220",
"article-title": "Comparison of in vitro inactivation of SARS CoV-2 with hydrogen peroxide and povidone-iodine oral antiseptic rinses",
"author": "Bidra",
"doi-asserted-by": "crossref",
"first-page": "599",
"journal-title": "J Prosthodont",
"key": "10.1016/j.eimc.2021.10.005_bib0130",
"volume": "29",
"year": "2020"
},
{
"article-title": "Povidone iodine mouthwash gargle, and nasal spray to reduce nasopharyngeal viral load in patients with COVID-19: A randomized clinical trial",
"author": "Guenezan",
"first-page": "e205490",
"journal-title": "JAMA Otolaryngol Head Neck Surg",
"key": "10.1016/j.eimc.2021.10.005_bib0135",
"volume": "4",
"year": "2021"
},
{
"DOI": "10.1186/1743-422X-6-124",
"article-title": "Mechanisms of the action of povidone-iodine against human and avian influenza A viruses: Its effects on hemagglutination and sialidase activities",
"author": "Sriwilaijaroen",
"doi-asserted-by": "crossref",
"first-page": "124",
"journal-title": "Virol J",
"key": "10.1016/j.eimc.2021.10.005_bib0140",
"volume": "6",
"year": "2009"
},
{
"DOI": "10.1007/s00784-020-03549-1",
"article-title": "A prospective clinical pilot study on the effects of a hydrogen peroxide mouthrinse on the intraoral viral load of SARS-CoV-2",
"author": "Maximilian",
"doi-asserted-by": "crossref",
"first-page": "3707",
"journal-title": "Clin Oral Investig",
"key": "10.1016/j.eimc.2021.10.005_bib0145",
"volume": "24",
"year": "2020"
},
{
"DOI": "10.1016/j.micinf.2017.08.008",
"article-title": "The role of oxidative stress in influenza virus infection",
"author": "Liu",
"doi-asserted-by": "crossref",
"first-page": "580",
"journal-title": "Microbes Infect",
"key": "10.1016/j.eimc.2021.10.005_bib0150",
"volume": "19",
"year": "2017"
},
{
"DOI": "10.1016/j.mehy.2020.109910",
"article-title": "Hydrogen peroxide and viral infections: A literature review with research hypothesis definition in relation to the current covid-19 pandemic",
"author": "Caruso",
"doi-asserted-by": "crossref",
"first-page": "109910",
"journal-title": "Med Hypotheses",
"key": "10.1016/j.eimc.2021.10.005_bib0155",
"volume": "144",
"year": "2020"
},
{
"DOI": "10.1016/j.jhin.2020.10.003",
"article-title": "Do hydrogen peroxide mouthwashes have a virucidal effect? A systematic review",
"author": "Ortega",
"doi-asserted-by": "crossref",
"first-page": "657",
"journal-title": "J Hosp Infect",
"key": "10.1016/j.eimc.2021.10.005_bib0160",
"volume": "106",
"year": "2020"
}
],
"reference-count": 16,
"references-count": 16,
"relation": {},
"score": 1,
"short-container-title": [
"Enfermedades Infecciosas y Microbiología Clínica"
],
"short-title": [],
"source": "Crossref",
"subject": [
"Microbiology (medical)"
],
"subtitle": [],
"title": [
"Utilidad de los enjuagues con povidona yodada y peróxido de hidrógeno en pacientes con COVID-19"
],
"type": "journal-article"
}
pablomarcos