In Vitro Inhibition of SARS-CoV-2 Infection by Bromhexine hydrochloride
et al., bioRxiv, doi:10.1101/2022.12.23.521817, Dec 2022
21st treatment shown to reduce risk in
April 2021, now with p = 0.00063 from 29 studies.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
In vitro study showing that bromhexine inhibits SARS-CoV-2 infection and replication in vitro by blocking the host cell protease TMPRSS2.
13 preclinical studies support the efficacy of TMPRSS2 inhibitors for COVID-19:
Study covers TMPRSS2 inhibitors and bromhexine.
1.
Haque et al., Exploring potential therapeutic candidates against COVID-19: a molecular docking study, Discover Molecules, doi:10.1007/s44345-024-00005-5.
2.
González-Paz et al., Biophysical Analysis of Potential Inhibitors of SARS-CoV-2 Cell Recognition and Their Effect on Viral Dynamics in Different Cell Types: A Computational Prediction from In Vitro Experimental Data, ACS Omega, doi:10.1021/acsomega.3c06968.
3.
Umar et al., Inhibitory potentials of ivermectin, nafamostat, and camostat on spike protein and some nonstructural proteins of SARS-CoV-2: Virtual screening approach, Jurnal Teknologi Laboratorium, doi:10.29238/teknolabjournal.v11i1.344.
4.
Unal et al., Favipiravir, umifenovir and camostat mesylate: a comparative study against SARS-CoV-2, bioRxiv, doi:10.1101/2022.01.11.475889.
5.
Sgrignani et al., Computational Identification of a Putative Allosteric Binding Pocket in TMPRSS2, Frontiers in Molecular Biosciences, doi:10.3389/fmolb.2021.666626.
6.
Yathindranath et al., Lipid Nanoparticle-Based Inhibitors for SARS-CoV-2 Host Cell Infection, International Journal of Nanomedicine, doi:10.2147/IJN.S448005.
7.
Martins et al., In Vitro Inhibition of SARS-CoV-2 Infection by Bromhexine hydrochloride, bioRxiv, doi:10.1101/2022.12.23.521817.
8.
Schultz et al., Pyrimidine inhibitors synergize with nucleoside analogues to block SARS-CoV-2, Nature, doi:10.1038/s41586-022-04482-x.
9.
Hempel et al., Synergistic inhibition of SARS-CoV-2 cell entry by otamixaban and covalent protease inhibitors: pre-clinical assessment of pharmacological and molecular properties, Chemical Science, doi:10.1039/D1SC01494C.
Martins et al., 26 Dec 2022, preprint, 12 authors.
Contact: rb.mj@hotmail.com.
In vitro studies are an important part of preclinical research, however results may be very different in vivo.
In Vitro Inhibition of SARS-CoV-2 Infection by Bromhexine hydrochloride
doi:10.1101/2022.12.23.521817
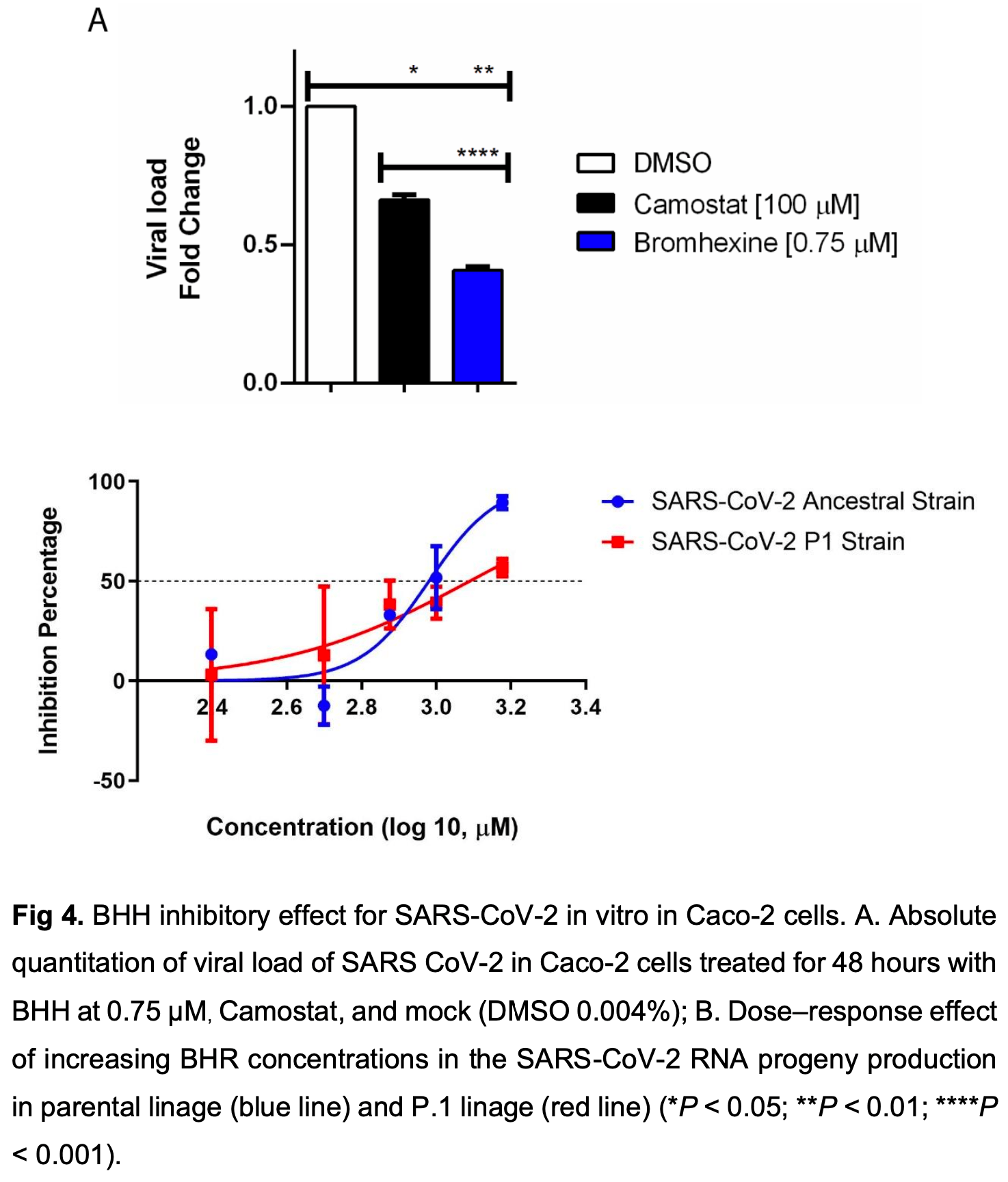
The world is enduring the SARS CoV-2 pandemic, and although extensive research has been conducted on the issue, only a few antivirals have been approved to treat patients with COVID-19. Bromhexine hydrochloride was previously identified as a potent inhibitor of TMPRSS2, an essential protease for ACE-2 virus receptor interactions. In the present study, we investigated whether bromhexine treatment could reduce SARS CoV-2 replication in vitro. To evaluate bromhexine's effectiveness against SARS COV-2 infection, viral load was measured using Caco-2 cell lines expressing TMPRSS2. Our molecular docking results indicate that bromhexine displays an affinity with the active site of TMPRSS2. The drug significantly inhibited SARS CoV-2, both parental and P1 variant strains, infection in the Caco-2 cell line, reducing about 40% of SARS-CoV-2 entrance and about 90% of viral progeny in the supernatant 48h postinfection. Furthermore, bromhexine did not exhibit any direct virucidal activity on .
Almeida, Juliano P. Souza, Marjorie Pontelli, Italo A. Castro, Thais M. Lima, and Rosa M. M. Viana designed and executed the experimental SARS-CoV-2 infection. All authors contributed to writting the manuscript. Eurico Arruda, Priscyla D. Marcato, and RBM wrote the final version of the manuscript. All authors have approved the final manuscript.
Declaration of Competing Interests The authors declare no conflict of interest.
References
Ansarin, Tolouian, Ardalan, Taghizadieh, Varshochi et al., Effect of bromhexine on clinical outcomes and mortality in COVID-19 patients: A randomized clinical trial, Bio. Impacts
Bertram, Glowacka, Blazejewska, Soilleux, Allen et al., TMPRSS2 and TMPRSS4 facilitate trypsin-independent spread of influenza virus in Caco-2 cells, J. Virol
Borefreund, Puerner, A simple quantitative procedure using monolayer cultures for cytotoxicity assays (HTD/NR 90), J. Tissue. Cult. Methods. P
Case, Rothlauf, Chen, Liu, Zhao et al., Neutralizing Antibody and Soluble ACE2 Inhibition of a Replication-Competent VSV-SARS-CoV-2 and a Clinical Isolate of SARS-CoV-2, Cell Host Microbe. Sep
Clososki, Soldi, Da Silva, Guaratini, Lopes et al., Tenofovir Disoproxil Fumarate: New Chemical Developments and Encouraging in vitro Biological Results for SARS-CoV-2, J. Braz. Chem. Soc
Correa, Dehydrocrotonin and its β-cyclodextrin complex: Cytotoxicity in V79 fibroblasts and rat cultured hepatocytes, Eur. J. Pharmacol
Fu, Zheng, Zhou, Tang, Chen et al., Re-recognizing bromhexine hydrochloride: pharmaceutical properties and its possible role in treating pediatric COVID-19, Eur. J. Clin. Pharmacol, doi:10.1007/s00228-020-02971-4
Habtemariam, Nabavi, Ghavami, Cismaru, Berindan-Neagoe et al., Possible use of the mucolytic drug, bromhexine hydrochloride, as a prophylactic agent against SARS-CoV-2 infection based on its action on the Transmembrane Serine Protease 2, Pharmacol. Res
Harcourt, Tamin, Lu, Kamili, Sakthivel et al., Severe acute respiratory syndrome coronavirus 2 from patient with coronavirus disease
Hoffmann, Kleine-Weber, Schroeder, Krüger, Herrler et al., SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor, Cell
Hörnich, Schlagowski, Tenbusch, Kleine-Weber, Neipel et al., SARS-CoV-2 and SARS-CoV Spike-Mediated Cell-Cell Fusion Differ in Their Requirements for Receptor Expression and Proteolytic Activation, J Virol
Kim, Heinlein, Hackman, Nelson, Phenotypic analysis of mice lacking the Tmprss2-encoded protease, Mol. Cell Biol
Laskowski, Macarthur, Moss, Thornton, PROCHECKa program to check the stereochemical quality of protein structures, J. App. Cryst
Li, Sun, Zhang, Zheng, Jiang et al., Bromhexine Hydrochloride Tablets for the Treatment of Moderate COVID-19: An Open-label Randomized Controlled Pilot Study, Clin. Transl. Sci, doi:10.1111/cts.12881
Lu, Wang, Sakthivel, US CDC Real-Time Reverse Transcription PCR Panel for Detection of Severe Acute Respiratory Syndrome Coronavirus 2. Emerg, Infect. Dis, doi:10.3201/eid2608.201246
Lucas, Heinlein, Kim, Hernandez, Malik et al., The androgen-regulated protease TMPRSS2 activates a proteolytic cascade involving components of the tumor microenvironment and promotes prostate cancer metastasis, Cancer Discov
Maggio, Corsini, Repurposing the mucolytic cough suppressant and TMPRSS2 protease inhibitor bromhexine for the prevention and management of SARS-CoV-2 infection, Pharmacol. Res
Mengzhen, Jie, Xuefeng, Jie, Lizhen et al., ACE2, TMPRSS2 distribution and extrapulmonary organ injury in patients with COVID-19, Biomed Pharmacother
Mikhaylov, Lyubimtseva, Vakhrushev, Stepanov, Lebedev et al., Bromhexine Hydrochloride Prophylaxis of COVID-19 for Medical Personnel: A Randomized Open-Label Study
Miranda, Marcato, Carvalho, Silva, Ribeiro et al., Assessing the cytotoxic potential of glycoalkaloidic extract in nanoparticles against bladder cancer cells, J. Pharm. Pharmacol
Morris, Huey, Lindstrom, Sanner, Belew et al., Autodock4 and AutoDockTools4: automated docking with selective receptor flexiblity, J. Computational Chemistry
Pettersen, Goddard, Huang, Couch, Greenblatt et al., UCSF Chimera--a visualization system for exploratory research and analysis, J Comput Chem, doi:10.1002/jcc.20084
Reed, Muench, A Simple Method of Estimating Fifty Per Cent Endpoints, Am. J. Epidemiol
Sabino, Buss, Carvalho, Prete, Crispim et al., Resurgence of COVID-19 in Manaus, Brazil, despite high seroprevalence, Lancet
Shen, Mao, Wu, Tanaka, Zhang, TMPRSS2: A potential target for treatment of influenza virus and coronavirus infections, Biochimie
Shen, Mao, Wu, Tanaka, Zhang, TMPRSS2: A potential target for treatment of influenza virus and coronavirus infections, Biochimie
Shrimp, Kales, Simeonov, Shen, Hall, An Enzymatic TMPRSS2 Assay for Assessment of Clinical Candidates and Discovery of Inhibitors as Potential Treatment of COVID-19, ACS Pharmacol Transl Sci
Wang, Yu, Li, Wang, Yang et al., Bromhexine elevates REP2 expression to stimulate secretion from human primary conjunctiva fornix epithelial cells, FEBS Lett
Waterhouse, Bertoni, Bienert, Studer, Tauriello et al., SWISS-MODEL: homology modelling of protein structures and complexes, Nucleic Acids Res
Wiederstein, Sippl, ProSA-web: interactive web service for the recognition of errors in three-dimensional structures of proteins, Nucleic Acids Research
Wilson, Greer, Hooper, Zijlstra, Walker et al., The membrane-anchored serine protease, TMPRSS2, activates PAR-2 in prostate cancer cells, Biochem J, doi:10.1042/BJ20041066
DOI record:
{
"DOI": "10.1101/2022.12.23.521817",
"URL": "http://dx.doi.org/10.1101/2022.12.23.521817",
"abstract": "<jats:title>ABSTRACT</jats:title><jats:p>The world is enduring the SARS CoV-2 pandemic, and although extensive research has been conducted on the issue, only a few antivirals have been approved to treat patients with COVID-19. Bromhexine hydrochloride was previously identified as a potent inhibitor of TMPRSS2, an essential protease for ACE-2 virus receptor interactions. In the present study, we investigated whether bromhexine treatment could reduce SARS CoV-2 replication in vitro. To evaluate bromhexine’s effectiveness against SARS COV-2 infection, viral load was measured using Caco-2 cell lines expressing TMPRSS2. Our molecular docking results indicate that bromhexine displays an affinity with the active site of TMPRSS2. The drug significantly inhibited SARS CoV-2, both parental and P1 variant strains, infection in the Caco-2 cell line, reducing about 40% of SARS-CoV-2 entrance and about 90% of viral progeny in the supernatant 48h post-infection. Furthermore, bromhexine did not exhibit any direct virucidal activity on SARS CoV-2. In conclusion, bromhexine hydrochloride efficiently disrupts SARS CoV-2 infection in vitro and has the potential to become an effective antiviral agent in COVID-19 treatment.</jats:p>",
"accepted": {
"date-parts": [
[
2022,
12,
26
]
]
},
"author": [
{
"ORCID": "http://orcid.org/0000-0002-8902-5962",
"affiliation": [],
"authenticated-orcid": false,
"family": "Martins",
"given": "Ronaldo",
"sequence": "first"
},
{
"affiliation": [],
"family": "Ferreira",
"given": "Iasmin",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Jorge",
"given": "Daniel M. M.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Almeida",
"given": "Leticia",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Souza",
"given": "Juliano P.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Pontelli",
"given": "Marjorie",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0001-7885-7547",
"affiliation": [],
"authenticated-orcid": false,
"family": "Castro",
"given": "Italo A.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Lima",
"given": "Thais M.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Viana",
"given": "Rosa M. M.",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-7856-7512",
"affiliation": [],
"authenticated-orcid": false,
"family": "Zamboni",
"given": "Dario",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Marcato",
"given": "Priscyla D.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Arruda",
"given": "Eurico",
"sequence": "additional"
}
],
"container-title": [],
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2022,
12,
27
]
],
"date-time": "2022-12-27T02:35:12Z",
"timestamp": 1672108512000
},
"deposited": {
"date-parts": [
[
2022,
12,
28
]
],
"date-time": "2022-12-28T18:50:38Z",
"timestamp": 1672253438000
},
"group-title": "Pharmacology and Toxicology",
"indexed": {
"date-parts": [
[
2022,
12,
29
]
],
"date-time": "2022-12-29T05:55:39Z",
"timestamp": 1672293339711
},
"institution": [
{
"name": "bioRxiv"
}
],
"is-referenced-by-count": 0,
"issued": {
"date-parts": [
[
2022,
12,
26
]
]
},
"link": [
{
"URL": "https://syndication.highwire.org/content/doi/10.1101/2022.12.23.521817",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "246",
"original-title": [],
"posted": {
"date-parts": [
[
2022,
12,
26
]
]
},
"prefix": "10.1101",
"published": {
"date-parts": [
[
2022,
12,
26
]
]
},
"publisher": "Cold Spring Harbor Laboratory",
"reference": [
{
"article-title": "Effect of bromhexine on clinical outcomes and mortality in COVID-19 patients: A randomized clinical trial",
"first-page": "209",
"issue": "4",
"journal-title": "Bio. Impacts",
"key": "2022122810501320000_2022.12.23.521817v1.1",
"volume": "10",
"year": "2020"
},
{
"DOI": "10.1128/JVI.00239-10",
"doi-asserted-by": "publisher",
"key": "2022122810501320000_2022.12.23.521817v1.2"
},
{
"DOI": "10.1007/BF01666038",
"doi-asserted-by": "crossref",
"key": "2022122810501320000_2022.12.23.521817v1.3",
"unstructured": "Borefreund, E. , Puerner, J.A. , (1984). A simple quantitative procedure using monolayer cultures for cytotoxicity assays (HTD/NR 90). J. Tissue. Cult. Methods. P 7–9."
},
{
"DOI": "10.1016/j.chom.2020.06.021",
"article-title": "Neutralizing Antibody and Soluble ACE2 Inhibition of a Replication-Competent VSV-SARS-CoV-2 and a Clinical Isolate of SARS-CoV-2",
"doi-asserted-by": "crossref",
"first-page": "475",
"issue": "3",
"journal-title": "Cell Host Microbe",
"key": "2022122810501320000_2022.12.23.521817v1.4",
"volume": "28",
"year": "2020"
},
{
"DOI": "10.21577/0103-5053.20200106",
"doi-asserted-by": "crossref",
"key": "2022122810501320000_2022.12.23.521817v1.5",
"unstructured": "Clososki, G.C. , Soldi, R.A. , da Silva, R.M. , Guaratini, T. , Lopes, J.N.C. , Pereira, P.R.R. , Lopes, J.L.C. , dos Santos, T. , Martins, R.B. , Costa, C.S. , de Carvalho, A.N. , da Silva, L.L.P. , Arruda, E. , Lopes, N.P. , (2020). Tenofovir Disoproxil Fumarate: New Chemical Developments and Encouraging in vitro Biological Results for SARS-CoV-2. J. Braz. Chem. Soc. vol.31, no.8."
},
{
"DOI": "10.1016/j.ejphar.2005.01.016",
"article-title": "Dehydrocrotonin and its β-cyclodextrin complex: Cytotoxicity in V79 fibroblasts and rat cultured hepatocytes",
"doi-asserted-by": "crossref",
"first-page": "17",
"issue": "1-2",
"journal-title": "Eur. J. Pharmacol",
"key": "2022122810501320000_2022.12.23.521817v1.6",
"volume": "510",
"year": "2005"
},
{
"DOI": "10.1007/s00228-020-02971-4",
"doi-asserted-by": "publisher",
"key": "2022122810501320000_2022.12.23.521817v1.7"
},
{
"DOI": "10.1016/j.phrs.2020.104853",
"article-title": "Possible use of the mucolytic drug, bromhexine hydrochloride, as a prophylactic agent against SARS-CoV-2 infection based on its action on the Transmembrane Serine Protease 2",
"doi-asserted-by": "crossref",
"first-page": "104853",
"journal-title": "Pharmacol. Res",
"key": "2022122810501320000_2022.12.23.521817v1.8",
"volume": "157",
"year": "2020"
},
{
"DOI": "10.3201/eid2606.200516",
"article-title": "Severe acute respiratory syndrome coronavirus 2 from patient with coronavirus disease, United States",
"doi-asserted-by": "crossref",
"first-page": "1266",
"journal-title": "Emerg. Infect. Dis",
"key": "2022122810501320000_2022.12.23.521817v1.9",
"volume": "26",
"year": "2020"
},
{
"DOI": "10.1016/j.cell.2020.02.052",
"doi-asserted-by": "publisher",
"key": "2022122810501320000_2022.12.23.521817v1.10"
},
{
"article-title": "SARS-CoV-2 and SARS-CoV Spike-Mediated Cell-Cell Fusion Differ in Their Requirements for Receptor Expression and Proteolytic Activation",
"first-page": "e00002",
"issue": "9",
"journal-title": "J Virol",
"key": "2022122810501320000_2022.12.23.521817v1.11",
"volume": "95",
"year": "2021"
},
{
"DOI": "10.1128/MCB.26.3.965-975.2006",
"doi-asserted-by": "publisher",
"key": "2022122810501320000_2022.12.23.521817v1.12"
},
{
"DOI": "10.1107/S0021889892009944",
"doi-asserted-by": "publisher",
"key": "2022122810501320000_2022.12.23.521817v1.13"
},
{
"DOI": "10.1111/cts.12881",
"doi-asserted-by": "publisher",
"key": "2022122810501320000_2022.12.23.521817v1.14"
},
{
"DOI": "10.3201/eid2608.201246",
"doi-asserted-by": "publisher",
"key": "2022122810501320000_2022.12.23.521817v1.15"
},
{
"DOI": "10.1158/2159-8290.CD-13-1010",
"doi-asserted-by": "publisher",
"key": "2022122810501320000_2022.12.23.521817v1.16"
},
{
"DOI": "10.1016/j.phrs.2020.104837",
"article-title": "Repurposing the mucolytic cough suppressant and TMPRSS2 protease inhibitor bromhexine for the prevention and management of SARS-CoV-2 infection",
"doi-asserted-by": "crossref",
"first-page": "104837",
"journal-title": "Pharmacol. Res",
"key": "2022122810501320000_2022.12.23.521817v1.17",
"volume": "157",
"year": "2020"
},
{
"DOI": "10.1016/j.biopha.2020.110678",
"article-title": "ACE2, TMPRSS2 distribution and extrapulmonary organ injury in patients with COVID-19",
"doi-asserted-by": "crossref",
"first-page": "110678",
"journal-title": "Biomed Pharmacother",
"key": "2022122810501320000_2022.12.23.521817v1.18",
"volume": "131",
"year": "2020"
},
{
"DOI": "10.1101/2021.03.03.21252855",
"doi-asserted-by": "crossref",
"key": "2022122810501320000_2022.12.23.521817v1.19",
"unstructured": "Mikhaylov E. N. , Lyubimtseva T. A. , Vakhrushev A. D. , Stepanov D. , Lebedev D. S. , Vasilieva E. Y. , Konradi A. O. , Evgeny V. Shlyakhto E. V. , (2022). Bromhexine Hydrochloride Prophylaxis of COVID-19 for Medical Personnel: A Randomized Open-Label Study. Interdisciplinary Perspectives on Infectious Diseases, Article ID 4693121."
},
{
"DOI": "10.1111/jphp.13145",
"article-title": "Assessing the cytotoxic potential of glycoalkaloidic extract in nanoparticles against bladder cancer cells",
"doi-asserted-by": "crossref",
"first-page": "1520",
"issue": "10",
"journal-title": "J. Pharm. Pharmacol",
"key": "2022122810501320000_2022.12.23.521817v1.20",
"volume": "71",
"year": "2019"
},
{
"article-title": "Autodock4 and AutoDockTools4: automated docking with selective receptor flexiblity",
"first-page": "2785",
"journal-title": "J. Computational Chemistry",
"key": "2022122810501320000_2022.12.23.521817v1.21",
"volume": "16",
"year": "2009"
},
{
"DOI": "10.1002/jcc.20084",
"doi-asserted-by": "publisher",
"key": "2022122810501320000_2022.12.23.521817v1.22"
},
{
"DOI": "10.1093/oxfordjournals.aje.a118408",
"doi-asserted-by": "publisher",
"key": "2022122810501320000_2022.12.23.521817v1.23"
},
{
"DOI": "10.1016/S0140-6736(21)00183-5",
"article-title": "Resurgence of COVID-19 in Manaus, Brazil, despite high seroprevalence",
"doi-asserted-by": "crossref",
"first-page": "452",
"issue": "10273",
"journal-title": "Lancet",
"key": "2022122810501320000_2022.12.23.521817v1.24",
"volume": "397",
"year": "2021"
},
{
"DOI": "10.1016/j.biochi.2017.07.016",
"doi-asserted-by": "publisher",
"key": "2022122810501320000_2022.12.23.521817v1.25"
},
{
"DOI": "10.1016/j.biochi.2017.07.016",
"doi-asserted-by": "publisher",
"key": "2022122810501320000_2022.12.23.521817v1.26"
},
{
"DOI": "10.1021/acsptsci.0c00106",
"article-title": "An Enzymatic TMPRSS2 Assay for Assessment of Clinical Candidates and Discovery of Inhibitors as Potential Treatment of COVID-19",
"doi-asserted-by": "crossref",
"first-page": "997",
"issue": "5",
"journal-title": "ACS Pharmacol Transl Sci",
"key": "2022122810501320000_2022.12.23.521817v1.27",
"volume": "3",
"year": "2020"
},
{
"DOI": "10.1093/nar/gky1049",
"doi-asserted-by": "publisher",
"key": "2022122810501320000_2022.12.23.521817v1.28"
},
{
"DOI": "10.1002/1873-3468.13560",
"article-title": "Bromhexine elevates REP2 expression to stimulate secretion from human primary conjunctiva fornix epithelial cells",
"doi-asserted-by": "crossref",
"first-page": "153",
"issue": "1",
"journal-title": "FEBS Lett",
"key": "2022122810501320000_2022.12.23.521817v1.29",
"volume": "594",
"year": "2020"
},
{
"DOI": "10.1093/nar/gky427",
"doi-asserted-by": "publisher",
"key": "2022122810501320000_2022.12.23.521817v1.30"
},
{
"article-title": "ProSA-web: interactive web service for the recognition of errors in three-dimensional structures of proteins",
"first-page": "W407",
"issue": "1",
"journal-title": "Nucleic Acids Research",
"key": "2022122810501320000_2022.12.23.521817v1.31",
"volume": "35-2",
"year": "2007"
},
{
"DOI": "10.1042/BJ20041066",
"doi-asserted-by": "publisher",
"key": "2022122810501320000_2022.12.23.521817v1.32"
},
{
"DOI": "10.1016/j.bpj.2011.10.024",
"doi-asserted-by": "publisher",
"key": "2022122810501320000_2022.12.23.521817v1.33"
},
{
"article-title": "Determination of bromhexine in plasma by gas chromatography-electron capture detection and pharmacokinetic studies",
"first-page": "543",
"issue": "6",
"journal-title": "Se. Pu",
"key": "2022122810501320000_2022.12.23.521817v1.34",
"volume": "18",
"year": "2000"
},
{
"article-title": "TMPRSS2 Contributes to Virus Spread and Immunopathology in the Airways of Murine Models after Coronavirus Infection",
"first-page": "e01815",
"issue": "6",
"journal-title": "J Virol",
"key": "2022122810501320000_2022.12.23.521817v1.35",
"volume": "93",
"year": "2019"
},
{
"article-title": "HPLC-MS assay for the determination of bromhexine in human plasma and application to bioequivalence studies",
"first-page": "114",
"journal-title": "Yao Xue Yu Lin Chuang Yan Jiu",
"key": "2022122810501320000_2022.12.23.521817v1.36",
"volume": "16",
"year": "2008"
}
],
"reference-count": 36,
"references-count": 36,
"relation": {},
"resource": {
"primary": {
"URL": "http://biorxiv.org/lookup/doi/10.1101/2022.12.23.521817"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subtitle": [],
"subtype": "preprint",
"title": "In Vitro Inhibition of SARS-CoV-2 Infection by Bromhexine hydrochloride",
"type": "posted-content"
}
