Aspirin Is Related to Worse Clinical Outcomes of COVID-19
et al., Medicina, doi:10.3390/medicina57090931, Sep 2021
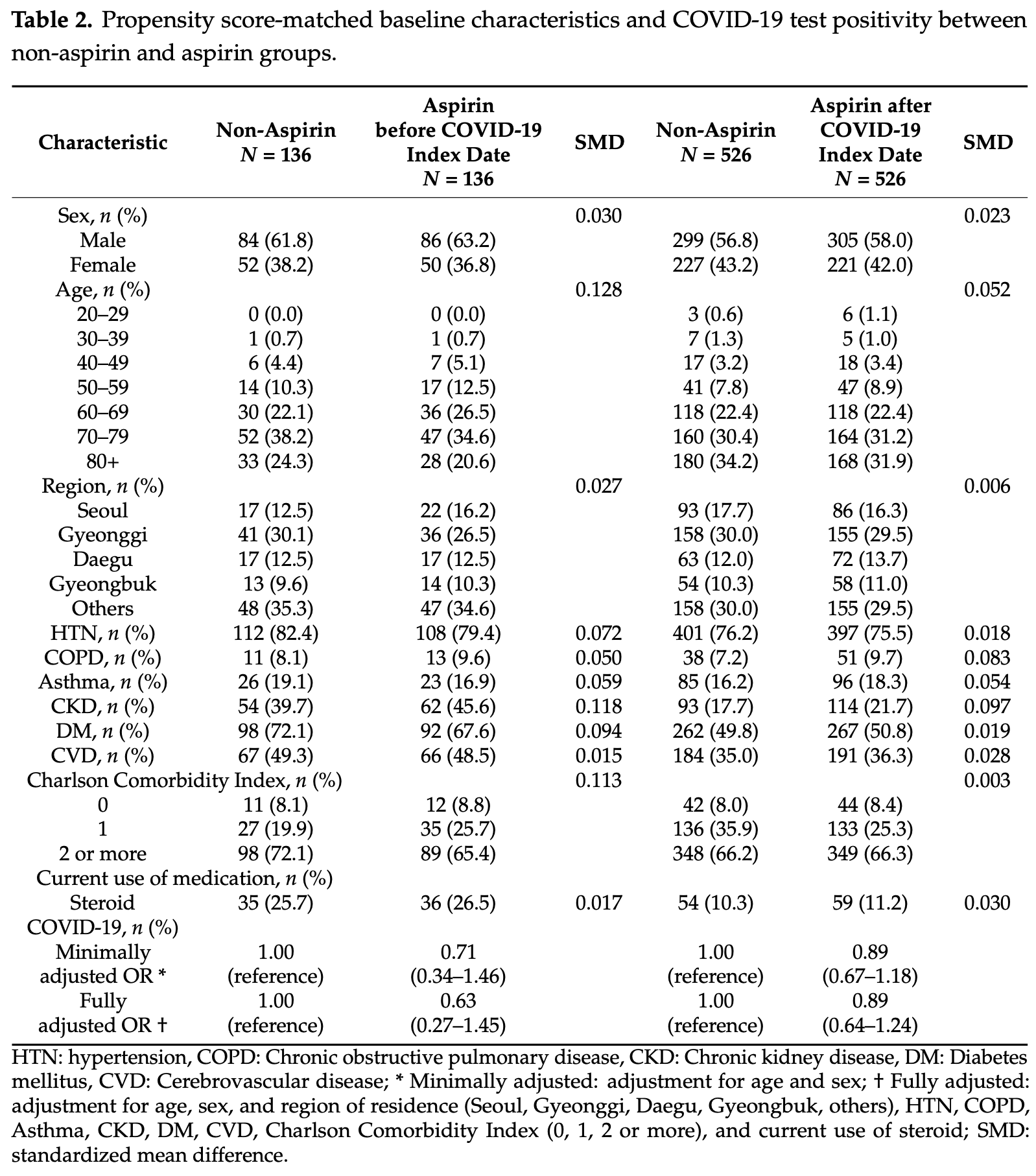
Retrospective database analysis of 22,660 patients tested for COVID-19 in South Korea. There was no significant difference in cases according to aspirin use. Aspirin use before COVID-19 was related to an increased death rate and aspirin use after COVID-19 was related to a higher risk of oxygen therapy.
Results for late treatment are listed separately1.
|
risk of death, 700.0% higher, RR 8.00, p = 0.03, treatment 6 of 15 (40.0%), control 1 of 20 (5.0%), PSM, prior aspirin use.
|
|
risk of mechanical ventilation, 433.3% higher, RR 5.33, p = 0.14, treatment 4 of 15 (26.7%), control 1 of 20 (5.0%), PSM, prior aspirin use.
|
|
risk of ICU admission, 433.3% higher, RR 5.33, p = 0.14, treatment 4 of 15 (26.7%), control 1 of 20 (5.0%), PSM, prior aspirin use.
|
|
risk of case, 33.4% lower, RR 0.67, p = 0.29, treatment 15 of 136 (11.0%), control 20 of 136 (14.7%), NNT 27, adjusted per study, odds ratio converted to relative risk, PSM, logistic regression, prior aspirin use.
|
|
risk of death, 33.7% lower, RR 0.66, p = 0.22, treatment 14 of 124 (11.3%), control 23 of 135 (17.0%), NNT 17, PSM, aspirin treatment after diagnosis.
|
|
risk of mechanical ventilation, 102.2% higher, RR 2.02, p = 0.16, treatment 13 of 124 (10.5%), control 7 of 135 (5.2%), PSM, aspirin treatment after diagnosis.
|
|
risk of ICU admission, 90.5% higher, RR 1.91, p = 0.36, treatment 7 of 124 (5.6%), control 4 of 135 (3.0%), PSM, aspirin treatment after diagnosis.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Kim et al., 4 Sep 2021, retrospective, propensity score matching, South Korea, peer-reviewed, 7 authors.
Aspirin Is Related to Worse Clinical Outcomes of COVID-19
Medicina, doi:10.3390/medicina57090931
Background and Objectives: Aspirin is used globally to reduce pain and inflammation; however, its effect in patients with coronavirus disease (COVID-19) is not fully investigated and remains controversial. We evaluated the association between aspirin and COVID-19 outcomes using nationwide data from the Korean National Health Insurance System. Materials and Methods: This was a retrospective observational cohort study that included 22,660 eligible patients who underwent COVID-19 testing in South Korea between 1 January-31 July 2020. We identified all aspirin users prescribed aspirin within two weeks before or after the index date. The primary outcome was positivity for the COVID-19 test, and secondary outcomes included conventional oxygen therapy, intensive care unit, mechanical ventilation, or death. We applied the propensity score matching method to reduce the possible bias originating from the differences in patients' baseline characteristics. Results: Of those eligible, 662 patients were prescribed aspirin. Among them, 136 patients were on aspirin within two weeks before diagnosis and 526 patients were on aspirin after diagnosis. The COVID-19 test positivity rate was not significantly different according to aspirin use. Aspirin use before COVID-19 was related to an increased death rate and aspirin use after COVID-19 was related to a higher risk of the conventional oxygen therapy. Conclusion: Aspirin use was associated with adverse effects in COVID-19 patients. Further studies for mechanisms are needed.
Author Contributions: I.K.: Project administration, data curation, formal analysis, writing the original draft. S.Y.: Data curation, formal analysis, writing original draft. M.K.: Data curation, formal analysis. H.L.: Manuscript review and editing. S.P.: Data curation. W.K.: Data curation. S.L.: Conceptualization, funding acquisition, investigation, methodology, manuscript review and editing. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement: The study protocol was approved by the Institutional Review Board (IRB) of CHA University Bundang Hospital, Korea (IRB No. CHAMC 2020-08-004) and the Health Insurance Review and Assessment Service (NHIS-2021-1-084). Informed Consent Statement: Not applicable.
References
Abers, Delmonte, Ricotta, Fintzi, Fink et al., An immune-based biomarker signature is associated with mortality in COVID-19 patients, JCI Insight, doi:10.1172/jci.insight.144455
Ackermann, Verleden, Kuehnel, Haverich, Welte et al., Pulmonary vascular endothelialitis, thrombosis, and angiogenesis in Covid-19, N. Engl. J. Med
Alamdari, Afaghi, Rahimi, Tarki, Tavana et al., Mortality risk factors among hospitalized COVID-19 patients in a major referral center in Iran, Tohoku J. Exp. Med, doi:10.1620/tjem.252.73
Asghar, Aghazadeh-Habashi, Jamali, Cardiovascular effect of inflammation and nonsteroidal anti-inflammatory drugs on renin-angiotensin system in experimental arthritis, Inflammopharmacology, doi:10.1007/s10787-017-0344-1
Chow, Khanna, Kethireddy, Yamane, Levine et al., Aspirin Use Is Associated With Decreased Mechanical Ventilation, Intensive Care Unit Admission, and In-Hospital Mortality in Hospitalized Patients With Coronavirus Disease, Anesth. Analg, doi:10.1213/ANE.0000000000005292
De Moraes, Vargaftig, Lefort, Meager, Chignard, Effect of cyclo-oxygenase inhibitors and modulators of cyclic AMP formation on lipopolysaccharide-induced neutrophil infiltration in mouse lung, Br. J. Pharmacol
Du, Jiang, He, Song, Xu, Antiplatelet therapy for critically ill patients: A pairwise and Bayesian network meta-analysis, Shock. Inj. Inflamm. Sepsis Lab. Clin. Approach, doi:10.1097/SHK.0000000000001057
Guan, Ni, Hu, Liang, Ou et al., Clinical characteristics of coronavirus disease 2019 in China, N. Engl. J. Med, doi:10.1056/NEJMoa2002032
Hsia, Sarin, Oliver, Goldstein, Aspirin and thymosin increase interleukin-2 and interferon-γ production by human peripheral blood lymphocytes, Immunopharmacology, doi:10.1016/0162-3109(89)90045-3
Ikonomidis, Andreotti, Economou, Stefanadis, Toutouzas et al., Increased proinflammatory cytokines in patients with chronic stable angina and their reduction by aspirin, Circulation, doi:10.1161/01.CIR.100.8.793
Jung, Choi, You, Kim, Association of Renin-angiotensin-aldosterone System Inhibitors With Coronavirus Disease 2019 (COVID-19)-Related Outcomes in Korea: A Nationwide Population-based Cohort Study, Clin. Infect. Dis, doi:10.1093/cid/ciaa624
Kim, Shin, Kim, Analysis of Major COVID-19 Issues Using Unstructured Big Data, Knowl. Manag. Res
Klok, Kruip, Van Der Meer, Arbous, Gommers et al., Confirmation of the high cumulative incidence of thrombotic complications in critically ill ICU patients with COVID-19: An updated analysis, Thromb. Res, doi:10.1016/j.thromres.2020.04.041
Kor, Carter, Park, Festic, Banner-Goodspeed et al., Effect of aspirin on development of ARDS in at-risk patients presenting to the emergency department: The LIPS-A randomized clinical trial, JAMA, doi:10.1001/jama.2016.6330
Mehta, Mcauley, Brown, Sanchez, Tattersall et al., COVID-19: Consider cytokine storm syndromes and immunosuppression, Lancet, doi:10.1016/S0140-6736(20)30628-0
Osborne, Veigulis, Arreola, Mahajan, Roosli et al., Association of mortality and aspirin prescription for COVID-19 patients at the Veterans Health Administration, PLoS ONE, doi:10.1371/journal.pone.0246825
Panka, De Grooth, Spoelstra-De Man, Looney, Tuinman, Prevention or Treatment of Ards With Aspirin: A Review of Preclinical Models and Meta-Analysis of Clinical Studies, Shock, doi:10.1097/SHK.0000000000000745
Paranjpe, Fuster, Lala, Russak, Glicksberg et al., Association of treatment dose anticoagulation with in-hospital survival among hospitalized patients with COVID-19, J. Am. Coll. Cardiol, doi:10.1016/j.jacc.2020.05.001
Rapkiewicz, Mai, Carsons, Pittaluga, Kleiner et al., Megakaryocytes and platelet-fibrin thrombi characterize multi-organ thrombosis at autopsy in COVID-19: A case series, EClinicalMedicine, doi:10.1016/j.eclinm.2020.100434
Russell, Moss, Rigg, Van Hemelrijck, COVID-19 and treatment with NSAIDs and corticosteroids: Should we be limiting their use in the clinical setting?, Ecancermedicalscience, doi:10.3332/ecancer.2020.1023
Salah, Mehta, Meta-Analysis of the Effect of Aspirin on Mortality in COVID-19, Am. J. Cardiol
The, Group, Dexamethasone in hospitalized patients with Covid-19, N. Engl. J. Med
Varga, Flammer, Steiger, Haberecker, Andermatt et al., Endothelial cell infection and endotheliitis in COVID-19, Lancet, doi:10.1016/S0140-6736(20)30937-5
Wang, Li, Gu, Wang, Liu et al., Effect of antiplatelet therapy on acute respiratory distress syndrome and mortality in critically ill patients: A meta-analysis, PLoS ONE, doi:10.1371/journal.pone.0154754
Warner, Nylander, Whatling, Anti-platelet therapy: Cyclo-oxygenase inhibition and the use of aspirin with particular regard to dual anti-platelet therapy, Br. J. Clin. Pharmacol, doi:10.1111/j.1365-2125.2011.03943.x
Wu, Chen, Cai, Xia, Zhou et al., Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 pneumonia in Wuhan, China, JAMA Intern. Med, doi:10.1001/jamainternmed.2020.0994
Yuan, Chen, Li, Chen, Wang et al., Mortality and pre-hospitalization use of low-dose aspirin in COVID-19 patients with coronary artery disease, J. Cell. Mol. Med, doi:10.1111/jcmm.16198
Zhang, Kim, Lonjon, Zhu, Balance diagnostics after propensity score matching, Ann. Transl. Med, doi:10.21037/atm.2018.12.10
DOI record:
{
"DOI": "10.3390/medicina57090931",
"ISSN": [
"1648-9144"
],
"URL": "http://dx.doi.org/10.3390/medicina57090931",
"abstract": "<jats:p>Backgroundand Objectives: Aspirin is used globally to reduce pain and inflammation; however, its effect in patients with coronavirus disease (COVID-19) is not fully investigated and remains controversial. We evaluated the association between aspirin and COVID-19 outcomes using nationwide data from the Korean National Health Insurance System. Materials and Methods: This was a retrospective observational cohort study that included 22,660 eligible patients who underwent COVID-19 testing in South Korea between 1 January–31 July 2020. We identified all aspirin users prescribed aspirin within two weeks before or after the index date. The primary outcome was positivity for the COVID-19 test, and secondary outcomes included conventional oxygen therapy, intensive care unit, mechanical ventilation, or death. We applied the propensity score matching method to reduce the possible bias originating from the differences in patients’ baseline characteristics. Results: Of those eligible, 662 patients were prescribed aspirin. Among them, 136 patients were on aspirin within two weeks before diagnosis and 526 patients were on aspirin after diagnosis. The COVID-19 test positivity rate was not significantly different according to aspirin use. Aspirin use before COVID-19 was related to an increased death rate and aspirin use after COVID-19 was related to a higher risk of the conventional oxygen therapy. Conclusion: Aspirin use was associated with adverse effects in COVID-19 patients. Further studies for mechanisms are needed.</jats:p>",
"alternative-id": [
"medicina57090931"
],
"author": [
{
"ORCID": "http://orcid.org/0000-0001-6808-7390",
"affiliation": [],
"authenticated-orcid": false,
"family": "Kim",
"given": "Isaac",
"sequence": "first"
},
{
"affiliation": [],
"family": "Yoon",
"given": "Siyeong",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Kim",
"given": "Minsup",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-0957-0988",
"affiliation": [],
"authenticated-orcid": false,
"family": "Lee",
"given": "Hyunil",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Park",
"given": "Sinhyung",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Kim",
"given": "Wonsang",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Lee",
"given": "Soonchul",
"sequence": "additional"
}
],
"container-title": "Medicina",
"container-title-short": "Medicina",
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2021,
9,
6
]
],
"date-time": "2021-09-06T17:18:26Z",
"timestamp": 1630948706000
},
"deposited": {
"date-parts": [
[
2021,
9,
6
]
],
"date-time": "2021-09-06T17:26:48Z",
"timestamp": 1630949208000
},
"funder": [
{
"DOI": "10.13039/501100003725",
"award": [
"No.2020R1G1A1099728"
],
"doi-asserted-by": "publisher",
"name": "National Research Foundation of Korea"
}
],
"indexed": {
"date-parts": [
[
2024,
3,
24
]
],
"date-time": "2024-03-24T07:35:50Z",
"timestamp": 1711265750028
},
"is-referenced-by-count": 9,
"issue": "9",
"issued": {
"date-parts": [
[
2021,
9,
4
]
]
},
"journal-issue": {
"issue": "9",
"published-online": {
"date-parts": [
[
2021,
9
]
]
}
},
"language": "en",
"license": [
{
"URL": "https://creativecommons.org/licenses/by/4.0/",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2021,
9,
4
]
],
"date-time": "2021-09-04T00:00:00Z",
"timestamp": 1630713600000
}
}
],
"link": [
{
"URL": "https://www.mdpi.com/1648-9144/57/9/931/pdf",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "1968",
"original-title": [],
"page": "931",
"prefix": "10.3390",
"published": {
"date-parts": [
[
2021,
9,
4
]
]
},
"published-online": {
"date-parts": [
[
2021,
9,
4
]
]
},
"publisher": "MDPI AG",
"reference": [
{
"article-title": "Analysis of Major COVID-19 Issues Using Unstructured Big Data",
"author": "Kim",
"first-page": "145",
"journal-title": "Knowl. Manag. Res.",
"key": "ref1",
"volume": "22",
"year": "2021"
},
{
"key": "ref2",
"unstructured": "Coronavirus Disease (COVID-19); Weekly Epidemiologicalhttps://www.who.int/publications/m/item/weekly-epidemiological-update-on-covid-19"
},
{
"DOI": "10.1056/NEJMoa2021436",
"article-title": "Dexamethasone in hospitalized patients with Covid-19",
"doi-asserted-by": "crossref",
"first-page": "693",
"journal-title": "N. Engl. J. Med.",
"key": "ref3",
"volume": "384",
"year": "2021"
},
{
"DOI": "10.1111/j.1365-2125.2011.03943.x",
"doi-asserted-by": "publisher",
"key": "ref4"
},
{
"DOI": "10.1371/journal.pone.0154754",
"doi-asserted-by": "publisher",
"key": "ref5"
},
{
"DOI": "10.1097/SHK.0000000000001057",
"doi-asserted-by": "publisher",
"key": "ref6"
},
{
"DOI": "10.1213/ANE.0000000000005292",
"doi-asserted-by": "publisher",
"key": "ref7"
},
{
"DOI": "10.1111/jcmm.16198",
"doi-asserted-by": "publisher",
"key": "ref8"
},
{
"DOI": "10.1093/cid/ciaa624",
"doi-asserted-by": "publisher",
"key": "ref9"
},
{
"DOI": "10.21037/atm.2018.12.10",
"doi-asserted-by": "publisher",
"key": "ref10"
},
{
"DOI": "10.1016/j.jacc.2020.05.001",
"doi-asserted-by": "publisher",
"key": "ref11"
},
{
"DOI": "10.1161/01.CIR.100.8.793",
"doi-asserted-by": "publisher",
"key": "ref12"
},
{
"DOI": "10.1016/S0140-6736(20)30628-0",
"doi-asserted-by": "publisher",
"key": "ref13"
},
{
"DOI": "10.1016/j.thromres.2020.04.041",
"doi-asserted-by": "publisher",
"key": "ref14"
},
{
"DOI": "10.1016/j.eclinm.2020.100434",
"doi-asserted-by": "publisher",
"key": "ref15"
},
{
"DOI": "10.1016/S0140-6736(20)30937-5",
"doi-asserted-by": "publisher",
"key": "ref16"
},
{
"DOI": "10.1056/NEJMoa2015432",
"doi-asserted-by": "publisher",
"key": "ref17"
},
{
"DOI": "10.1097/SHK.0000000000000745",
"doi-asserted-by": "publisher",
"key": "ref18"
},
{
"DOI": "10.3332/ecancer.2020.1023",
"doi-asserted-by": "publisher",
"key": "ref19"
},
{
"DOI": "10.1007/s10787-017-0344-1",
"doi-asserted-by": "publisher",
"key": "ref20"
},
{
"DOI": "10.1620/tjem.252.73",
"doi-asserted-by": "publisher",
"key": "ref21"
},
{
"DOI": "10.1016/j.amjcard.2020.12.073",
"doi-asserted-by": "publisher",
"key": "ref22"
},
{
"DOI": "10.1001/jamainternmed.2020.0994",
"doi-asserted-by": "publisher",
"key": "ref23"
},
{
"DOI": "10.1111/j.1476-5381.1996.tb15356.x",
"doi-asserted-by": "publisher",
"key": "ref24"
},
{
"DOI": "10.1001/jama.2016.6330",
"doi-asserted-by": "publisher",
"key": "ref25"
},
{
"DOI": "10.1371/journal.pone.0246825",
"doi-asserted-by": "publisher",
"key": "ref26"
},
{
"DOI": "10.1056/NEJMoa2002032",
"doi-asserted-by": "publisher",
"key": "ref27"
},
{
"DOI": "10.1016/0162-3109(89)90045-3",
"doi-asserted-by": "publisher",
"key": "ref28"
},
{
"DOI": "10.1172/jci.insight.144455",
"doi-asserted-by": "publisher",
"key": "ref29"
}
],
"reference-count": 29,
"references-count": 29,
"relation": {},
"resource": {
"primary": {
"URL": "https://www.mdpi.com/1648-9144/57/9/931"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [
"General Medicine"
],
"subtitle": [],
"title": "Aspirin Is Related to Worse Clinical Outcomes of COVID-19",
"type": "journal-article",
"volume": "57"
}