Beneficial effects of a mouthwash containing an antiviral phthalocyanine derivative on the length of hospital stay for COVID-19: randomised trial
et al., Scientific Reports, doi:10.1038/s41598-021-99013-5, Oct 2021
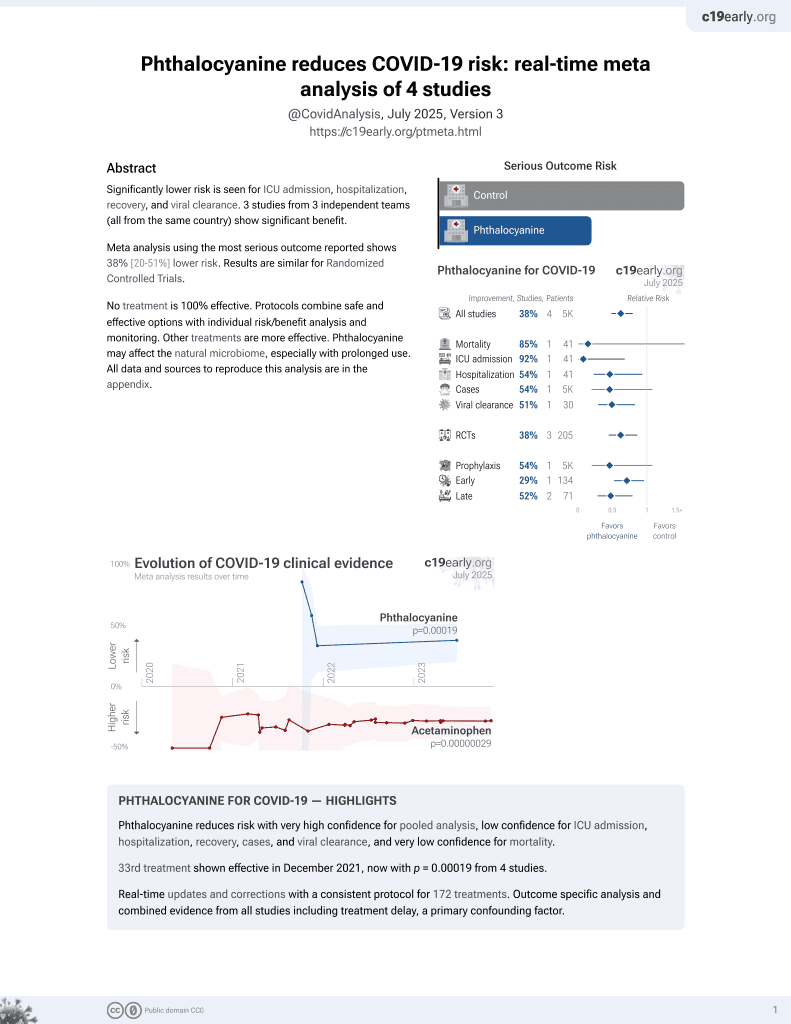
33rd treatment shown to reduce risk in
December 2021, now with p = 0.00019 from 4 studies.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
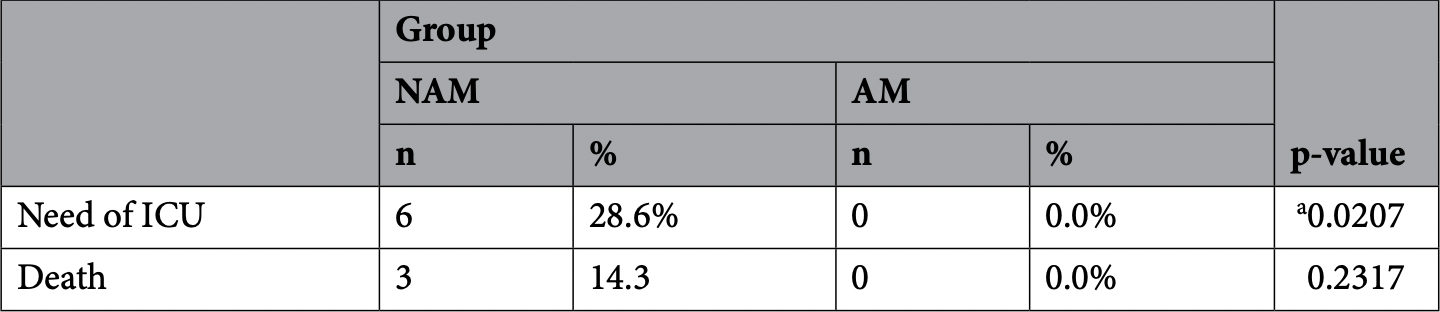
RCT 41 patients in Brazil, 20 treated with a phthalocyanine derivative mouthwash, showing shorter hosptalization and lower ICU admission with treatment. One minute gargling/rinsing 5 times per day.
Targeted administration to the respiratory tract provides treatment directly
to the typical source of initial SARS-CoV-2 infection and replication, and
allows for rapid onset of action, higher local drug concentration, and reduced systemic side effects (early treatment may be more beneficial).
|
risk of death, 85.4% lower, RR 0.15, p = 0.23, treatment 0 of 20 (0.0%), control 3 of 21 (14.3%), NNT 7.0, relative risk is not 0 because of continuity correction due to zero events (with reciprocal of the contrasting arm).
|
|
risk of ICU admission, 92.1% lower, RR 0.08, p = 0.02, treatment 0 of 20 (0.0%), control 6 of 21 (28.6%), NNT 3.5, relative risk is not 0 because of continuity correction due to zero events (with reciprocal of the contrasting arm).
|
|
discharge, 53.7% lower, HR 0.46, p = 0.03, treatment 20, control 21, inverted to make HR<1 favor treatment, Cox proportional hazards.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
1.
Poleti et al., Use of mouthwash and dentifrice containing antimicrobial phthalocyanine derivative for the reduction of clinical symptoms of COVID-19: A randomized triple-blinded clinical trial, Journal of Evidence-Based Dental Practice, doi:10.1016/j.jebdp.2022.101777.
da Silva Santos et al., 7 Oct 2021, Double Blind Randomized Controlled Trial, Brazil, peer-reviewed, 17 authors, study period 10 August, 2020 - 4 November, 2020.
Contact: paulosss@fob.usp.br.
Beneficial effects of a mouthwash containing an antiviral phthalocyanine derivative on the length of hospital stay for COVID-19: randomised trial
Scientific Reports, doi:10.1038/s41598-021-99013-5
The risk of contamination and dissemination by SARS-CoV-2 has a strong link with nasal, oral and pharyngeal cavities. Recently, our research group observed the promising performance of an anionic phthalocyanine derivative (APD) used in a mouthwash protocol without photoexcitation; this protocol improved the general clinical condition of patients infected with SARS-CoV-2. The present two-arm study evaluated in vitro the antiviral activity and cytotoxicity of APD. Additionally, a triple-blind randomized controlled trial was conducted with 41 hospitalized patients who tested positive for COVID-19. All the included patients received World Health Organization standard care hospital treatment (non-intensive care) plus active mouthwash (experimental group AM/n = 20) or nonactive mouthwash (control group NAM/n = 21). The adjunct mouthwash intervention protocol used in both groups consisted one-minute gargling/rinsing / 5 times/day until hospital discharge. Groups were compared considering age, number of comorbidities, duration of symptoms prior admission and length of hospital stay (LOS). The associations between group and sex, age range, presence of comorbidities, admission to Intensive care unit (ICU) and death were also evaluated. The in vitro evaluation demonstrated that APD compound was highly effective for reduction of SARS-CoV-2 viral load in the 1.0 mg/mL (99.96%) to 0.125 mg/mL (92.65%) range without causing cytotoxicity. Regarding the clinical trial, the median LOS of the AM group was significantly shortened (4 days) compared with that of the NAM group (7 days) (p = 0.0314). Additionally, gargling/rinsing with APD was very helpful in reducing the severity of symptoms (no ICU care was needed) compared to not gargling/rinsing with APD (28.6% of the patients in the NAM group needed ICU care, and 50% of this ICU subgroup passed way, p = 0.0207). This study indicated that the mechanical action of the protocol involving mouthwash containing a compound with antiviral effects against SARS-CoV-2 may reduce the symptoms of the patients and the spread of infection. The use of APD in a mouthwash as an
Author contributions
Competing interests The authors declare no competing interests.
References
Araujo, SARS-CoV-2 isolation from the first reported patients in Brazil and establishment of a coordinated task network, Mem Inst Oswaldo Cruz
Carrouel, COVID-19: A recommendation to examine the effect of mouthrinses with β-cyclodextrin combined with citrox in preventing infection and progression, J. Clin. Med
Corman, Detection of 2019 novel coronavirus (2019-nCoV) by real-time RT-PCR, Euro Surv
Da Fonseca, A phthalocyanine derivate mouthwash to gargling/rinsing as an option to reduce clinical symptoms of COVID-19: Case series, Clin. Cosmet. Investig. Dent
De Toledo, Caminha, Kallás, Sipahi, Da Silva Santos, Potential mouth rinses and nasal sprays that reduce SARS-CoV-2 viral load: What we know so far?, Clinics
Drosten, Identification of a novel coronavirus in patients with severe acute respiratory syndrome, N. Engl. J. Med
Encinar, Menendez, Potential drugs targeting early innate immune evasion of SARS-coronavirus 2 via 2'-O-methylation of viral RNA, Viruses
Gadotti, Susceptibility of the patients infected with Sars-Cov2 to oxidative stress and possible interplay with severity of the disease, Free Radic. Biol. Med
Hakansson, Orihuela, Bogaert, Bacterial-host interactions: Physiology and pathophysiology of respiratory infection, Physiol. Rev
Heugebaert, Roman, Stevens, Synthesis of isoindoles and related iso-condensed heteroaromatic pyrroles, Chem. Soc. Rev
Kawauchi, An anionic phthalocyanine decreases NRAS expression by breaking down its RNA G-quadruplex, Nat. Commun
Liu, Viral dynamics in mild and severe cases of COVID-19, Lancet Infect. Dis
Lu, Genomic characterisation and epidemiology of 2019 novel coronavirus: Implications for virus origins and receptor binding, Lancet
Magleby, Impact of SARS-CoV-2 viral load on risk of intubation and mortality among hospitalized patients with coronavirus disease 2019, Clin. Infect. Dis
Meister, Virucidal efficacy of different oral rinses against severe acute respiratory syndrome coronavirus 2, J. Infect. Dis
Moussaron, Lipophilic phthalocyanines for their potential interest in photodynamic therapy: Synthesis and photo-physical properties, Tetrahedron
Nagatake, Ahmed, Oishi, Prevention of respiratory infections by povidone-iodine gargle, Dermatology
Naqvi, Povidone-iodine solution as SARS-CoV-2 prophylaxis for procedures of the upper aerodigestive tract a theoretical framework, J. Otolaryngol. Head Neck Surg
Nyamu, Ombaka, Masika, Nganga, Antimicrobial photodynamic activity of phthalocyanine derivatives, Adv. Chem
O'donnell, Potential role of oral rinses targeting the viral lipid envelope in SARS-CoV-2 infection, Function (Oxf)
Orcina, Santos, Oral manifestation COVID-19 and the rapid resolution of symptoms post-phtalox treatment: A case series, Int. J. Odontostomat
Rees, COVID-19 length of hospital stay: A systematic review and data synthesis, BMC Med
Romeo, Iacovelli, Falconi, Targeting the SARS-CoV-2 spike glycoprotein prefusion conformation: Virtual screening and molecular dynamics simulations applied to the identification of potential fusion inhibitors, Virus Res
Sales-Medina, Discovery of clinically approved drugs capable of inhibiting SARS-CoV-2 in vitro infection using a phenotypic screening strategy and network-analysis to predict their potential to treat covid-19, BioRxiv, doi:10.1101/2020.07.09.196337
Santos, A recommendation of PHTALOX® mouthwash for preventing infection and progression of COVID-19, Acta Sci. Dent. Sci
Satomura, Prevention of upper respiratory tract infections by gargling: A randomized trial, Am. J. Prev. Med
Schuster, Kraus, Opitzb, Brütting, Eckern, Transport properties of copper phthalocyanine based organic electronic devices, Eur. Phys. J. Spec. Topics
Silveira, Efficacy of propolis as an adjunct treatment for hospitalized COVID-19 patients: A randomized, controlled clinical trial, MedRxiv, doi:10.1101/2021.01.08.20248932
Sun, Jiang, Xia, Density functional theory study of the oxygen reduction reaction on metalloporphyrins and metallophthalocyanines, J. Phys. Chem
Tashiro, Shaw, COVID-19 pandemic response in Japan: What is behind the initial flattening of the curve?, Sustainability
Teodoro, PHTALOX® antimicrobial action and cytotoxicity: In vitro study, J. Dent. Res
Thai, Factors associated with the duration of hospitalisation among COVID-19 patients in Vietnam: A survival analysis, Epidemiol. Infect
Xu, Li, Gan, Du, Yao, Salivary glands: Potential reservoirs for COVID-19 asymptomatic infection, J. Dent. Res
Zhou, Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study, Lancet
Zhu, China novel coronavirus investigating and research team a novel coronavirus from patients with pneumonia in China, N. Engl. J. Med
DOI record:
{
"DOI": "10.1038/s41598-021-99013-5",
"ISSN": [
"2045-2322"
],
"URL": "http://dx.doi.org/10.1038/s41598-021-99013-5",
"abstract": "<jats:title>Abstract</jats:title><jats:p>The risk of contamination and dissemination by SARS-CoV-2 has a strong link with nasal, oral and pharyngeal cavities. Recently, our research group observed the promising performance of an anionic phthalocyanine derivative (APD) used in a mouthwash protocol without photoexcitation; this protocol improved the general clinical condition of patients infected with SARS-CoV-2. The present two-arm study evaluated in vitro the antiviral activity and cytotoxicity of APD. Additionally, a triple-blind randomized controlled trial was conducted with 41 hospitalized patients who tested positive for COVID-19. All the included patients received World Health Organization standard care hospital treatment (non-intensive care) plus active mouthwash (experimental group AM/n = 20) or nonactive mouthwash (control group NAM/n = 21). The adjunct mouthwash intervention protocol used in both groups consisted one-minute gargling/rinsing / 5 times/day until hospital discharge. Groups were compared considering age, number of comorbidities, duration of symptoms prior admission and length of hospital stay (LOS). The associations between group and sex, age range, presence of comorbidities, admission to Intensive care unit (ICU) and death were also evaluated. The in vitro evaluation demonstrated that APD compound was highly effective for reduction of SARS-CoV-2 viral load in the 1.0 mg/mL (99.96%) to 0.125 mg/mL (92.65%) range without causing cytotoxicity. Regarding the clinical trial, the median LOS of the AM group was significantly shortened (4 days) compared with that of the NAM group (7 days) (p = 0.0314). Additionally, gargling/rinsing with APD was very helpful in reducing the severity of symptoms (no ICU care was needed) compared to not gargling/rinsing with APD (28.6% of the patients in the NAM group needed ICU care, and 50% of this ICU subgroup passed way, p = 0.0207). This study indicated that the mechanical action of the protocol involving mouthwash containing a compound with antiviral effects against SARS-CoV-2 may reduce the symptoms of the patients and the spread of infection. The use of APD in a mouthwash as an adjuvant the hospital COVID-19 treatment presented no contraindication and reduced the hospital stay period.</jats:p><jats:p><jats:bold>Trial registration:</jats:bold> The clinical study was registered at REBEC—Brazilian Clinical Trial Register (RBR-58ftdj).</jats:p>",
"alternative-id": [
"99013"
],
"article-number": "19937",
"assertion": [
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "Received",
"name": "received",
"order": 1,
"value": "26 March 2021"
},
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "Accepted",
"name": "accepted",
"order": 2,
"value": "17 September 2021"
},
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "First Online",
"name": "first_online",
"order": 3,
"value": "7 October 2021"
},
{
"group": {
"label": "Competing interests",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 1,
"value": "The authors declare no competing interests."
}
],
"author": [
{
"affiliation": [],
"family": "da Silva Santos",
"given": "Paulo Sérgio",
"sequence": "first"
},
{
"affiliation": [],
"family": "da Fonseca Orcina",
"given": "Bernardo",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Machado",
"given": "Rafael Rahal Guaragna",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Vilhena",
"given": "Fabiano Vieira",
"sequence": "additional"
},
{
"affiliation": [],
"family": "da Costa Alves",
"given": "Lucas Marques",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Zangrando",
"given": "Mariana Schutzer Ragghianti",
"sequence": "additional"
},
{
"affiliation": [],
"family": "de Oliveira",
"given": "Rodrigo Cardoso",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Soares",
"given": "Mariana Quirino Silveira",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Simão",
"given": "Andréa Name Colado",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Pietro",
"given": "Emilene Cristine Izu Nakamura",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Kuroda",
"given": "Juliana Pescinelli Garcia",
"sequence": "additional"
},
{
"affiliation": [],
"family": "de Almeida Benjamim",
"given": "Ivanilda Aparecida",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Araujo",
"given": "Danielle Bastos",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Toma",
"given": "Sérgio Hiroshi",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Flor",
"given": "Lourival",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Araki",
"given": "Koiti",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Durigon",
"given": "Edison Luiz",
"sequence": "additional"
}
],
"container-title": "Scientific Reports",
"container-title-short": "Sci Rep",
"content-domain": {
"crossmark-restriction": false,
"domain": [
"link.springer.com"
]
},
"created": {
"date-parts": [
[
2021,
10,
7
]
],
"date-time": "2021-10-07T10:25:59Z",
"timestamp": 1633602359000
},
"deposited": {
"date-parts": [
[
2021,
10,
7
]
],
"date-time": "2021-10-07T11:07:42Z",
"timestamp": 1633604862000
},
"indexed": {
"date-parts": [
[
2022,
5,
20
]
],
"date-time": "2022-05-20T05:11:35Z",
"timestamp": 1653023495119
},
"is-referenced-by-count": 5,
"issue": "1",
"issued": {
"date-parts": [
[
2021,
10,
7
]
]
},
"journal-issue": {
"issue": "1",
"published-print": {
"date-parts": [
[
2021,
12
]
]
}
},
"language": "en",
"license": [
{
"URL": "https://creativecommons.org/licenses/by/4.0",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2021,
10,
7
]
],
"date-time": "2021-10-07T00:00:00Z",
"timestamp": 1633564800000
}
},
{
"URL": "https://creativecommons.org/licenses/by/4.0",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2021,
10,
7
]
],
"date-time": "2021-10-07T00:00:00Z",
"timestamp": 1633564800000
}
}
],
"link": [
{
"URL": "https://www.nature.com/articles/s41598-021-99013-5.pdf",
"content-type": "application/pdf",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://www.nature.com/articles/s41598-021-99013-5",
"content-type": "text/html",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://www.nature.com/articles/s41598-021-99013-5.pdf",
"content-type": "application/pdf",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "297",
"original-title": [],
"prefix": "10.1038",
"published": {
"date-parts": [
[
2021,
10,
7
]
]
},
"published-online": {
"date-parts": [
[
2021,
10,
7
]
]
},
"published-print": {
"date-parts": [
[
2021,
12
]
]
},
"publisher": "Springer Science and Business Media LLC",
"reference": [
{
"author": "F Zhou",
"first-page": "10229",
"journal-title": "Lancet",
"key": "99013_CR1",
"unstructured": "Zhou, F. et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 395, 10229 (2020).",
"volume": "395",
"year": "2020"
},
{
"DOI": "10.1016/S0140-6736(20)30251-8",
"author": "R Lu",
"doi-asserted-by": "publisher",
"first-page": "10224",
"journal-title": "Lancet",
"key": "99013_CR2",
"unstructured": "Lu, R. et al. Genomic characterisation and epidemiology of 2019 novel coronavirus: Implications for virus origins and receptor binding. Lancet 395, 10224 (2020).",
"volume": "395",
"year": "2020"
},
{
"DOI": "10.1056/NEJMoa2001017",
"author": "N Zhu",
"doi-asserted-by": "publisher",
"first-page": "8",
"journal-title": "N. Engl. J. Med.",
"key": "99013_CR3",
"unstructured": "Zhu, N. et al. China novel coronavirus investigating and research team a novel coronavirus from patients with pneumonia in China, 2019. N. Engl. J. Med. 382, 8 (2020).",
"volume": "382",
"year": "2020"
},
{
"key": "99013_CR4",
"unstructured": "https://www.who.int/docs/default-source/coronaviruse/laboratory-biosafety-novel-coronavirus-version-1-1.pdf?sfvrsn=912a9847_2. Accessed 30 Jan 2021."
},
{
"DOI": "10.1093/cid/ciaa851",
"author": "R Magleby",
"doi-asserted-by": "publisher",
"first-page": "851",
"journal-title": "Clin. Infect. Dis.",
"key": "99013_CR5",
"unstructured": "Magleby, R. et al. Impact of SARS-CoV-2 viral load on risk of intubation and mortality among hospitalized patients with coronavirus disease 2019. Clin. Infect. Dis. 2, 851 (2020).",
"volume": "2",
"year": "2020"
},
{
"key": "99013_CR6",
"unstructured": "https://www.who.int/publications/i/item/clinical-management-of-covid-19. Accessed: 30 Jan 2021."
},
{
"DOI": "10.3390/su12135250",
"author": "A Tashiro",
"doi-asserted-by": "publisher",
"first-page": "5250",
"journal-title": "Sustainability.",
"key": "99013_CR7",
"unstructured": "Tashiro, A. & Shaw, R. COVID-19 pandemic response in Japan: What is behind the initial flattening of the curve?. Sustainability. 12, 5250 (2020).",
"volume": "12",
"year": "2020"
},
{
"author": "VB O'Donnell",
"first-page": "1",
"journal-title": "Function (Oxf).",
"key": "99013_CR8",
"unstructured": "O’Donnell, V. B. et al. Potential role of oral rinses targeting the viral lipid envelope in SARS-CoV-2 infection. Function (Oxf). 1, 1 (2020).",
"volume": "1",
"year": "2020"
},
{
"DOI": "10.1177/0022034519889050",
"author": "J Xu",
"doi-asserted-by": "publisher",
"first-page": "8",
"journal-title": "J. Dent. Res.",
"key": "99013_CR9",
"unstructured": "Xu, J., Li, Y., Gan, F., Du, Y. & Yao, Y. Salivary glands: Potential reservoirs for COVID-19 asymptomatic infection. J. Dent. Res. 99, 8 (2020).",
"volume": "99",
"year": "2020"
},
{
"DOI": "10.1186/s40463-020-00474-x",
"author": "SHS Naqvi",
"doi-asserted-by": "publisher",
"first-page": "1",
"journal-title": "J. Otolaryngol. Head Neck Surg.",
"key": "99013_CR10",
"unstructured": "Naqvi, S. H. S. et al. Povidone-iodine solution as SARS-CoV-2 prophylaxis for procedures of the upper aerodigestive tract a theoretical framework. J. Otolaryngol. Head Neck Surg. 49, 1 (2020).",
"volume": "49",
"year": "2020"
},
{
"DOI": "10.3390/jcm9041126",
"author": "F Carrouel",
"doi-asserted-by": "publisher",
"first-page": "4",
"journal-title": "J. Clin. Med.",
"key": "99013_CR11",
"unstructured": "Carrouel, F. et al. COVID-19: A recommendation to examine the effect of mouthrinses with β-cyclodextrin combined with citrox in preventing infection and progression. J. Clin. Med. 9, 4 (2020).",
"volume": "9",
"year": "2020"
},
{
"DOI": "10.1093/infdis/jiaa471",
"author": "TL Meister",
"doi-asserted-by": "publisher",
"first-page": "8",
"journal-title": "J. Infect. Dis.",
"key": "99013_CR12",
"unstructured": "Meister, T. L. et al. Virucidal efficacy of different oral rinses against severe acute respiratory syndrome coronavirus 2. J. Infect. Dis. 222, 8 (2020).",
"volume": "222",
"year": "2020"
},
{
"author": "PSS Santos",
"first-page": "12",
"journal-title": "Acta Sci. Dent. Sci.",
"key": "99013_CR13",
"unstructured": "Santos, P. S. S. et al. A recommendation of PHTALOX® mouthwash for preventing infection and progression of COVID-19. Acta Sci. Dent. Sci. 4, 12 (2020).",
"volume": "4",
"year": "2020"
},
{
"DOI": "10.2147/CCIDE.S295423",
"author": "OB da Fonseca",
"doi-asserted-by": "publisher",
"first-page": "47",
"journal-title": "Clin. Cosmet. Investig. Dent.",
"key": "99013_CR14",
"unstructured": "da Fonseca, O. B. et al. A phthalocyanine derivate mouthwash to gargling/rinsing as an option to reduce clinical symptoms of COVID-19: Case series. Clin. Cosmet. Investig. Dent. 13, 47–50 (2021).",
"volume": "13",
"year": "2021"
},
{
"DOI": "10.4067/S0718-381X2021000100067",
"author": "BF Orcina",
"doi-asserted-by": "publisher",
"first-page": "67",
"issue": "1",
"journal-title": "Int. J. Odontostomat.",
"key": "99013_CR15",
"unstructured": "Orcina, B. F. & Santos, P. S. S. Oral manifestation COVID-19 and the rapid resolution of symptoms post-phtalox treatment: A case series. Int. J. Odontostomat. 15(1), 67–70 (2021).",
"volume": "15",
"year": "2021"
},
{
"key": "99013_CR16",
"unstructured": "https://vihema.gov.vn. Accessed 30 Jan 2021."
},
{
"key": "99013_CR17",
"unstructured": "https://www.jica.go.jp/COVID-19/en/index.html. Accessed 30 Jan 2021."
},
{
"DOI": "10.1140/epjst/e2010-01214-3",
"author": "C Schuster",
"doi-asserted-by": "publisher",
"first-page": "117",
"journal-title": "Eur. Phys. J. Spec. Topics.",
"key": "99013_CR18",
"unstructured": "Schuster, C., Kraus, M., Opitzb, A., Brütting, W. & Eckern, U. Transport properties of copper phthalocyanine based organic electronic devices. Eur. Phys. J. Spec. Topics. 180, 117–134 (2010).",
"volume": "180",
"year": "2010"
},
{
"author": "SN Nyamu",
"first-page": "2598062",
"journal-title": "Adv. Chem.",
"key": "99013_CR19",
"unstructured": "Nyamu, S. N., Ombaka, L., Masika, E. & Nganga, M. Antimicrobial photodynamic activity of phthalocyanine derivatives. Adv. Chem. 2, 2598062 (2018).",
"volume": "2",
"year": "2018"
},
{
"DOI": "10.1016/j.tet.2013.09.035",
"author": "A Moussaron",
"doi-asserted-by": "publisher",
"first-page": "10116",
"issue": "47",
"journal-title": "Tetrahedron",
"key": "99013_CR20",
"unstructured": "Moussaron, A. et al. Lipophilic phthalocyanines for their potential interest in photodynamic therapy: Synthesis and photo-physical properties. Tetrahedron 69(47), 10116–10122 (2013).",
"volume": "69",
"year": "2013"
},
{
"DOI": "10.1039/c2cs35093a",
"author": "TS Heugebaert",
"doi-asserted-by": "publisher",
"first-page": "5626",
"issue": "17",
"journal-title": "Chem. Soc. Rev.",
"key": "99013_CR21",
"unstructured": "Heugebaert, T. S., Roman, B. I. & Stevens, C. V. Synthesis of isoindoles and related iso-condensed heteroaromatic pyrroles. Chem. Soc. Rev. 41(17), 5626–5640 (2012).",
"volume": "41",
"year": "2012"
},
{
"DOI": "10.3390/v12050525",
"author": "JA Encinar",
"doi-asserted-by": "publisher",
"first-page": "525",
"issue": "5",
"journal-title": "Viruses",
"key": "99013_CR22",
"unstructured": "Encinar, J. A. & Menendez, J. A. Potential drugs targeting early innate immune evasion of SARS-coronavirus 2 via 2’-O-methylation of viral RNA. Viruses 12(5), 525 (2020).",
"volume": "12",
"year": "2020"
},
{
"DOI": "10.1016/j.virusres.2020.198068",
"author": "A Romeo",
"doi-asserted-by": "publisher",
"first-page": "198068",
"journal-title": "Virus Res.",
"key": "99013_CR23",
"unstructured": "Romeo, A., Iacovelli, F. & Falconi, M. Targeting the SARS-CoV-2 spike glycoprotein prefusion conformation: Virtual screening and molecular dynamics simulations applied to the identification of potential fusion inhibitors. Virus Res. 286, 198068 (2020).",
"volume": "286",
"year": "2020"
},
{
"DOI": "10.1590/0074-02760200342",
"author": "DB Araujo",
"doi-asserted-by": "publisher",
"first-page": "e200342",
"journal-title": "Mem Inst Oswaldo Cruz.",
"key": "99013_CR24",
"unstructured": "Araujo, D. B. et al. SARS-CoV-2 isolation from the first reported patients in Brazil and establishment of a coordinated task network. Mem Inst Oswaldo Cruz. 115, e200342 (2020).",
"volume": "115",
"year": "2020"
},
{
"author": "VM Corman",
"first-page": "200045",
"issue": "3",
"journal-title": "Euro Surv.",
"key": "99013_CR25",
"unstructured": "Corman, V. M. et al. Detection of 2019 novel coronavirus (2019-nCoV) by real-time RT-PCR. Euro Surv. 25(3), 200045 (2020).",
"volume": "25",
"year": "2020"
},
{
"DOI": "10.1056/NEJMoa030747",
"author": "C Drosten",
"doi-asserted-by": "publisher",
"first-page": "1967",
"issue": "20",
"journal-title": "N. Engl. J. Med.",
"key": "99013_CR26",
"unstructured": "Drosten, C. et al. Identification of a novel coronavirus in patients with severe acute respiratory syndrome. N. Engl. J. Med. 348(20), 1967–1976 (2003).",
"volume": "348",
"year": "2003"
},
{
"DOI": "10.1101/2020.07.09.196337",
"author": "DF Sales-Medina",
"doi-asserted-by": "publisher",
"journal-title": "BioRxiv.",
"key": "99013_CR27",
"unstructured": "Sales-Medina, D. F. et al. Discovery of clinically approved drugs capable of inhibiting SARS-CoV-2 in vitro infection using a phenotypic screening strategy and network-analysis to predict their potential to treat covid-19. BioRxiv. https://doi.org/10.1101/2020.07.09.196337 (2020).",
"year": "2020"
},
{
"DOI": "10.1177/0022034520911037",
"author": "GR Teodoro",
"doi-asserted-by": "publisher",
"first-page": "0839",
"journal-title": "J. Dent. Res.",
"key": "99013_CR28",
"unstructured": "Teodoro, G. R. et al. PHTALOX® antimicrobial action and cytotoxicity: In vitro study. J. Dent. Res. 99, 0839 (2020).",
"volume": "99",
"year": "2020"
},
{
"DOI": "10.1186/s12916-020-01726-3",
"author": "EM Rees",
"doi-asserted-by": "publisher",
"first-page": "270",
"issue": "1",
"journal-title": "BMC Med.",
"key": "99013_CR29",
"unstructured": "Rees, E. M. et al. COVID-19 length of hospital stay: A systematic review and data synthesis. BMC Med. 18(1), 270 (2020).",
"volume": "18",
"year": "2020"
},
{
"DOI": "10.1101/2021.01.08.20248932",
"author": "MAD Silveira",
"doi-asserted-by": "publisher",
"journal-title": "MedRxiv.",
"key": "99013_CR30",
"unstructured": "Silveira, M. A. D. et al. Efficacy of propolis as an adjunct treatment for hospitalized COVID-19 patients: A randomized, controlled clinical trial. MedRxiv. https://doi.org/10.1101/2021.01.08.20248932 (2021).",
"year": "2021"
},
{
"DOI": "10.1152/physrev.00040.2016",
"author": "AP Hakansson",
"doi-asserted-by": "publisher",
"first-page": "781",
"issue": "2",
"journal-title": "Physiol. Rev.",
"key": "99013_CR31",
"unstructured": "Hakansson, A. P., Orihuela, C. J. & Bogaert, D. Bacterial-host interactions: Physiology and pathophysiology of respiratory infection. Physiol. Rev. 98(2), 781–811 (2018).",
"volume": "98",
"year": "2018"
},
{
"DOI": "10.1016/j.amepre.2005.06.013",
"author": "K Satomura",
"doi-asserted-by": "publisher",
"first-page": "302",
"issue": "4",
"journal-title": "Am. J. Prev. Med.",
"key": "99013_CR32",
"unstructured": "Satomura, K. et al. Prevention of upper respiratory tract infections by gargling: A randomized trial. Am. J. Prev. Med. 29(4), 302–307 (2005).",
"volume": "29",
"year": "2005"
},
{
"DOI": "10.1159/000057722",
"author": "T Nagatake",
"doi-asserted-by": "publisher",
"first-page": "32",
"issue": "Suppl 1",
"journal-title": "Dermatology",
"key": "99013_CR33",
"unstructured": "Nagatake, T., Ahmed, K. & Oishi, K. Prevention of respiratory infections by povidone-iodine gargle. Dermatology 204(Suppl 1), 32–36 (2002).",
"volume": "204",
"year": "2002"
},
{
"DOI": "10.1016/S1473-3099(20)30232-2",
"author": "Y Liu",
"doi-asserted-by": "publisher",
"first-page": "656",
"issue": "6",
"journal-title": "Lancet Infect. Dis.",
"key": "99013_CR34",
"unstructured": "Liu, Y. et al. Viral dynamics in mild and severe cases of COVID-19. Lancet Infect. Dis. 20(6), 656–657 (2020).",
"volume": "20",
"year": "2020"
},
{
"DOI": "10.6061/clinics/2020/e2328",
"author": "T-A de Toledo",
"doi-asserted-by": "publisher",
"first-page": "e2328",
"journal-title": "Clinics",
"key": "99013_CR35",
"unstructured": "de Toledo, T.-A., Caminha, R. D. G., Kallás, M. S., Sipahi, A. M. & da Silva Santos, P. S. Potential mouth rinses and nasal sprays that reduce SARS-CoV-2 viral load: What we know so far?. Clinics 75, e2328 (2020).",
"volume": "75",
"year": "2020"
},
{
"DOI": "10.1038/s41467-018-04771-y",
"author": "K Kawauchi",
"doi-asserted-by": "publisher",
"first-page": "2271",
"issue": "1",
"journal-title": "Nat. Commun.",
"key": "99013_CR36",
"unstructured": "Kawauchi, K. et al. An anionic phthalocyanine decreases NRAS expression by breaking down its RNA G-quadruplex. Nat. Commun. 9(1), 2271 (2018).",
"volume": "9",
"year": "2018"
},
{
"author": "S Sun",
"first-page": "9511",
"issue": "19",
"journal-title": "J. Phys. Chem.",
"key": "99013_CR37",
"unstructured": "Sun, S., Jiang, N. & Xia, D. Density functional theory study of the oxygen reduction reaction on metalloporphyrins and metallophthalocyanines. J. Phys. Chem. 115(19), 9511–9517 (2011).",
"volume": "115",
"year": "2011"
},
{
"DOI": "10.1016/j.freeradbiomed.2021.01.044",
"author": "AC Gadotti",
"doi-asserted-by": "publisher",
"first-page": "184",
"journal-title": "Free Radic. Biol. Med.",
"key": "99013_CR38",
"unstructured": "Gadotti, A. C. et al. Susceptibility of the patients infected with Sars-Cov2 to oxidative stress and possible interplay with severity of the disease. Free Radic. Biol. Med. 165, 184–190 (2021).",
"volume": "165",
"year": "2021"
},
{
"DOI": "10.1017/S0950268820001259",
"author": "PQ Thai",
"doi-asserted-by": "publisher",
"first-page": "e114",
"journal-title": "Epidemiol. Infect.",
"key": "99013_CR39",
"unstructured": "Thai, P. Q. et al. Factors associated with the duration of hospitalisation among COVID-19 patients in Vietnam: A survival analysis. Epidemiol. Infect. 148, e114 (2020).",
"volume": "148",
"year": "2020"
}
],
"reference-count": 39,
"references-count": 39,
"relation": {},
"resource": {
"primary": {
"URL": "https://www.nature.com/articles/s41598-021-99013-5"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [
"Multidisciplinary"
],
"subtitle": [],
"title": "Beneficial effects of a mouthwash containing an antiviral phthalocyanine derivative on the length of hospital stay for COVID-19: randomised trial",
"type": "journal-article",
"update-policy": "http://dx.doi.org/10.1007/springer_crossmark_policy",
"volume": "11"
}