Exploratory analyses of Immunologic Features in a Randomized, Placebo-Controlled Trial of Nirmatrelvir/Ritonavir for Long COVID
et al., medRxiv, doi:10.64898/2026.02.24.26347001, PAX LC, NCT05668091, Feb 2026
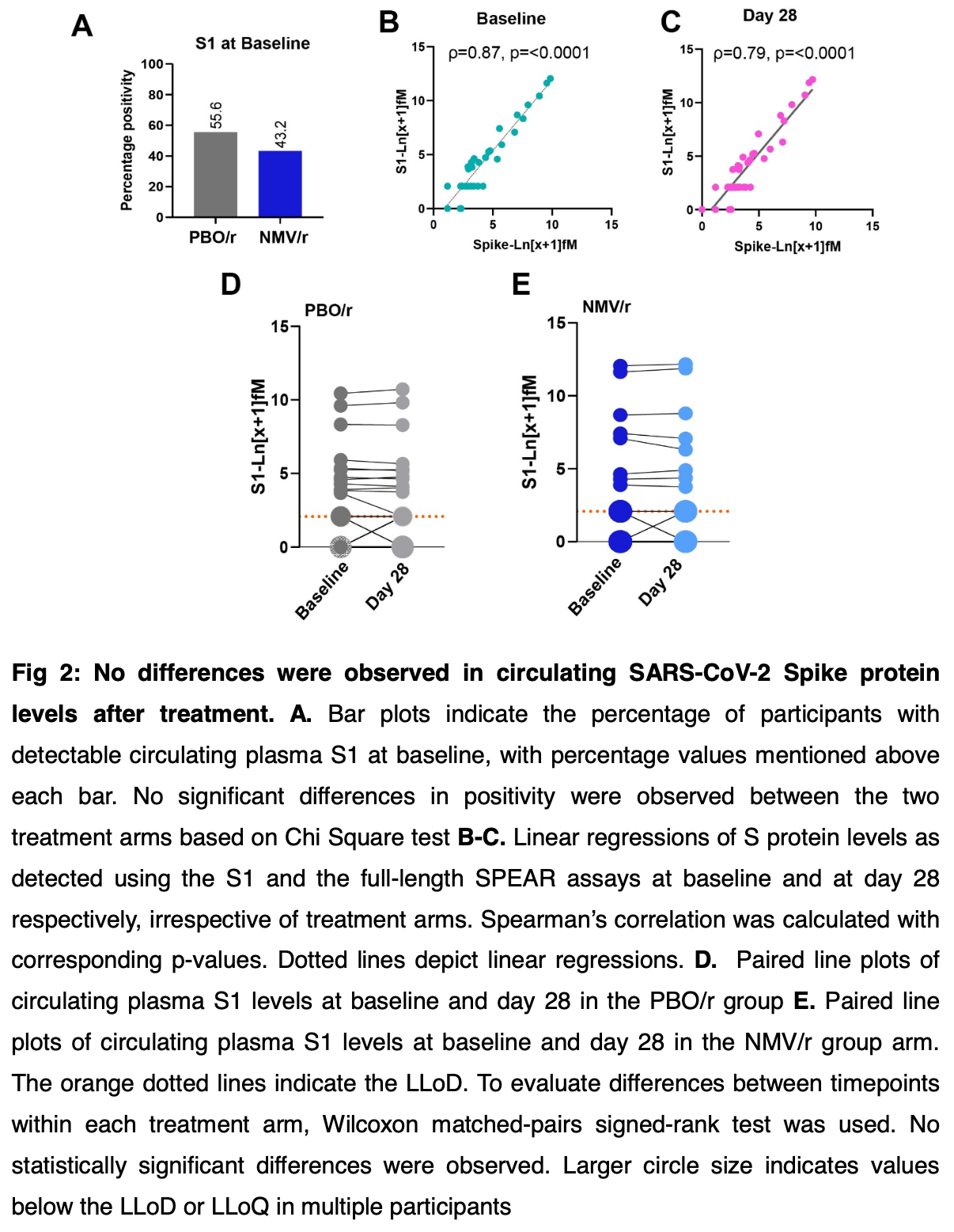
RCT 82 long COVID patients showing no significant differences with nirmatrelvir/ritonavir treatment. The study found no improvement in physical health summary scores, no changes in circulating SARS-CoV-2 spike protein levels, and no significant differences in immune cell populations or antibody responses between treatment and placebo groups after 15 days of treatment.
Bhattacharjee et al., 26 Feb 2026, Double Blind Randomized Controlled Trial, placebo-controlled, USA, preprint, 34 authors, study period March 2024 - August 2024, trial NCT05668091 (history) (PAX LC).
Contact: akiko.iwasaki@yale.edu.
Exploratory analyses of Immunologic Features in a Randomized, Placebo-Controlled Trial of Nirmatrelvir/Ritonavir for Long COVID
doi:10.64898/2026.02.24.26347001
This exploratory analysis of PAX LC, a Phase 2, 1:1 randomized, double-blind, superiority, placebo-controlled trial examined whether treatment with nirmatrelvir/ritonavir (NMV/r) versus placebo/ritonavir (PBO/r) in individuals with Long COVID could reveal immune features associated with symptom improvement. Eightytwo participants (n=45 PBO/r; n=37 NMV/r) provided blood samples at baseline (Day 0) and post-treatment (Day 28). Baseline demographic and immunological phenotypes were similar in the two groups. No significant differences were observed in major immune cell populations or organ function markers between NMV/r vs. PBO/r groups, or before vs. after the treatment. Modest hematologic changes were noted in the NMV/r arm. SARS-CoV-2-specific IgG levels remained constant, with changes in total immunoglobulin subtypes and isotypes in both arms. Both arms showed similar shifts in cytokine levels. Notably, the levels of S1 and Spike proteins in circulation remained unchanged post-treatment. Regardless of the treatment arm, participants with selfreported symptom improvement showed reductions in the level of the inflammatory chemokine RANTES. Taken together, the findings of this study demonstrate limited virological and immunological changes in response to nirmatrelvir, contributing insights into the reason for the lack of benefit of the 15-day NMV/r treatment in Long COVID.
Materials and Methods
Study design and patient characteristics The PAX LC study was a decentralized, phase 2, randomized, double-blind, placebocontrolled clinical to investigate the efficacy and safety of a 15-day regimen of orally administered nirmatrelvir/ritonavir compared with placebo/ritonavir in participants with Long COVID. The details of the study has been published elsewhere 8 . Briefly, the key inclusion criteria for participants were: being 18
Biospecimen collection Whole blood samples were collected in lithium-heparin-coated (BD 367880, BD Biosciences) and sodium-EDTA coated vacutainers (BD 367856, BD Biosciences) from participants' homes, by ExamOne phlebotomists (Quest Diagnostics) or at the Yale clinic, New Haven, CT. After collecting, biospecimens were either shipped overnight at regulated temperatures or handed over locally to researchers at Yale University in New Haven, CT. Collection tubes were de-identified upon receipt according to protocol and study identifiers were provided. Samples were processed within 48 hours of collection. Biorender 41 was used to create a graphical schematic of the CONSORT flow chart, study design, cohorts and assays.
Linear Peptide Profiling
SERA serum screening A detailed description of the SERA assay has been published 22 . For this study, plasma was incubated with a fully random 12-mer bacterial display peptide library (1 × 10 10 diversity, 10-fold oversampled) at a 1:25 dilution in a 96-well, deep well plate..
References
Al-Aly, Long COVID science, research and policy, Nat Med, doi:10.1038/s41591-024-03173-611
Bhattacharjee, Lu, Monteiro, Tabachnikova, Wang et al., None
Booth, Extending human IgG half-life using structure-guided design, MAbs, doi:10.1080/19420862.2018.1490119
Caronia, Xi, Margolskee, Jiang, Paxlovid mouth likely is mediated by activation of the TAS2R1 bitter receptor by nirmatrelvir, Biochem Biophys Res Commun, doi:10.1016/j.bbrc.2023.10.001
Cervia-Hasler, Persistent complement dysregulation with signs of thromboinflammation in active Long Covid, Science, doi:10.1126/science.adg7942
Chatterjee, Bhattacharya, Dhama, Lee, Chakraborty, Resistance to nirmatrelvir due to mutations in the Mpro in the subvariants of SARS-CoV-2 Omicron: Another concern?, Mol Ther Nucleic Acids, doi:10.1016/j.omtn.2023.03.013
Choutka, Jansari, Hornig, Iwasaki, Unexplained post-acute infection syndromes, Nat Med, doi:10.1038/s41591-022-01810-6
Couzin-Frankel, Lessons in persistence, Science, doi:10.1126/science.adp7205
Cvancara, Postmarketing Reporting of Paxlovid-Related Dysgeusia: A Real-World Pharmacovigilance Study, Otolaryngol Head Neck Surg, doi:10.1002/ohn.278
Davis, Mccorkell, Vogel, Topol, Long COVID: major findings, mechanisms and recommendations, Nat Rev Microbiol, doi:10.1038/s41579-022-00846-2
El-Khoury, Schulz, Durant, Longitudinal Assessment of SARS-CoV-2 Antinucleocapsid and Antispike-1-RBD Antibody Testing Following PCR-Detected SARS-CoV-2 Infection, J Appl Lab Med, doi:10.1093/jalm/jfab030
Filardi, Age-dependent impairment in antibody responses elicited by a homologous CoronaVac booster dose, Sci Transl Med, doi:10.1126/scitranslmed.ade6023
Gao, Identification of soluble biomarkers that associate with distinct manifestations of long COVID, Nat Immunol, doi:10.1038/s41590-025-02135-5
Geng, Nirmatrelvir-Ritonavir and Symptoms in Adults With Postacute Sequelae of SARS-CoV-2 Infection: The STOP-PASC Randomized Clinical Trial, JAMA Intern Med, doi:10.1001/jamainternmed.2024.2007
Hammond, Oral Nirmatrelvir for High-Risk, Nonhospitalized Adults with Covid-19, N Engl J Med, doi:10.1056/NEJMoa2118542
Harmon, Deng, Breslin, Bitter Taste Receptors (T2Rs) are Sentinels that Coordinate Metabolic and Immunological Defense Responses, Curr Opin Physiol, doi:10.1016/j.cophys.2021.01.006
Haynes, Kamath, Waitz, Daugherty, Shon, Protein-Based Immunome Wide Association Studies (PIWAS) for the Discovery of Significant Disease-Associated Antigens, Front Immunol, doi:10.3389/fimmu.2021.625311
Hendrick, Pharmacovigilance of Drug-Drug Interactions with Nirmatrelvir/Ritonavir, Infect Dis Ther, doi:10.1007/s40121-024-01050-w
Iketani, Multiple pathways for SARS-CoV-2 resistance to nirmatrelvir, Nature, doi:10.1038/s41586-022-05514-2
Kamath, Antibody epitope repertoire analysis enables rapid antigen discovery and multiplex serology, Sci Rep, doi:10.1038/s41598-020-62256-9
Khalatbari, Ritonavir and Lopinavir Suppress RCE1 and CAAX Rab Proteins Sensitizing the Liver to Organelle Stress and Injury, Hepatol Commun, doi:10.1002/hep4.1515
Klein, Distinguishing features of long COVID identified through immune profiling, Nature, doi:10.1038/s41586-023-06651-y
Krumholz, The PAX LC Trial: A Decentralized, Phase 2, Randomized, Double-Blind Study of Nirmatrelvir/Ritonavir Compared with Placebo/Ritonavir for Long COVID, Am J Med, doi:10.1016/j.amjmed.2024.04.030
Liew, Large-scale phenotyping of patients with long COVID posthospitalization reveals mechanistic subtypes of disease, Nat Immunol, doi:10.1038/s41590-024-01778-0
Nelson, Krensky, Chemokines, lymphocytes and viruses: what goes around, comes around, Curr Opin Immunol, doi:10.1016/s0952-7915(98)80164-7
Nooruzzaman, Emergence of transmissible SARS-CoV-2 variants with decreased sensitivity to antivirals in immunocompromised patients with persistent infections, Nat Commun, doi:10.1038/s41467-024-51924-3
Owen, An oral SARS-CoV-2 M(pro) inhibitor clinical candidate for the treatment of COVID-19, Science, doi:10.1126/science.abl4784
Peluso, Chronic viral coinfections differentially affect the likelihood of developing long COVID, J Clin Invest, doi:10.1172/JCI163669
Peluso, Deeks, Mechanisms of long COVID and the path toward therapeutics, Cell, doi:10.1016/j.cell.2024.07.054
Peluso, Plasma-based antigen persistence in the post-acute phase of COVID-19, Lancet Infect Dis, doi:10.1016/S1473-3099(24)00211-1
Proal, SARS-CoV-2 reservoir in post-acute sequelae of COVID-19 (PASC), Nat Immunol, doi:10.1038/s41590-023-01601-2
Proal, Targeting the SARS-CoV-2 reservoir in long COVID, Lancet Infect Dis, doi:10.1016/S1473-3099(24)00769-2
Rodriguez, None
Sawano, Nirmatrelvir-ritonavir versus placebo-ritonavir in individuals with long COVID in the USA (PAX LC): a double-blind, randomised, placebocontrolled, phase 2, decentralised trial, Lancet Infect Dis, doi:10.1016/S1473-3099(25)00073-8
Su, Multiple early factors anticipate post-acute COVID-19 sequelae, Cell, doi:10.1016/j.cell.2022.01.014
Swank, Measurement of circulating viral antigens post-SARS-CoV-2 infection in a multicohort study, Clin Microbiol Infect, doi:10.1016/j.cmi.2024.09.001
Swank, Persistent Circulating Severe Acute Respiratory Syndrome Coronavirus 2 Spike Is Associated With Post-acute Coronavirus Disease 2019 Sequelae, Clin Infect Dis, doi:10.1093/cid/ciac722
Toussi, Pharmacokinetics of Oral Nirmatrelvir/Ritonavir, a Protease Inhibitor for Treatment of COVID-19, in Subjects With Renal Impairment, Clin Pharmacol Ther, doi:10.1002/cpt.2688
Weinstein, Extended nirmatrelvir-ritonavir treatment durations for immunocompromised patients with COVID-19 (EPIC-IC): a placebo-controlled, randomised, double-blind, phase 2 trial, Lancet Infect Dis, doi:10.1016/S1473-3099(25)00221-X
Woodruff, Chronic inflammation, neutrophil activity, and autoreactivity splits long COVID, Nat Commun, doi:10.1038/s41467-023-40012-7
Yin, Long COVID manifests with T cell dysregulation, inflammation and an uncoordinated adaptive immune response to SARS-CoV-2, Nat Immunol, doi:10.1038/s41590-023-01724-6
DOI record:
{
"DOI": "10.64898/2026.02.24.26347001",
"URL": "http://dx.doi.org/10.64898/2026.02.24.26347001",
"abstract": "<jats:p>This exploratory analysis of PAX LC, a Phase 2, 1:1 randomized, double-blind, superiority, placebo-controlled trial examined whether treatment with nirmatrelvir/ritonavir (NMV/r) versus placebo/ritonavir (PBO/r) in individuals with Long COVID could reveal immune features associated with symptom improvement. Eighty-two participants (n=45 PBO/r; n=37 NMV/r) provided blood samples at baseline (Day 0) and post-treatment (Day 28). Baseline demographic and immunological phenotypes were similar in the two groups. No significant differences were observed in major immune cell populations or organ function markers between NMV/r vs. PBO/r groups, or before vs. after the treatment. Modest hematologic changes were noted in the NMV/r arm. SARS-CoV-2-specific IgG levels remained constant, with changes in total immunoglobulin subtypes and isotypes in both arms. Both arms showed similar shifts in cytokine levels. Notably, the levels of S1 and Spike proteins in circulation remained unchanged post-treatment. Regardless of the treatment arm, participants with self-reported symptom improvement showed reductions in the level of the inflammatory chemokine RANTES. Taken together, the findings of this study demonstrate limited virological and immunological changes in response to nirmatrelvir, contributing insights into the reason for the lack of benefit of the 15-day NMV/r treatment in Long COVID.</jats:p>",
"accepted": {
"date-parts": [
[
2026,
2,
26
]
]
},
"author": [
{
"ORCID": "https://orcid.org/0000-0002-0801-1543",
"affiliation": [
{
"name": "Yale School of Medicine;"
}
],
"authenticated-orcid": false,
"family": "Bhattacharjee",
"given": "Bornali",
"sequence": "first"
},
{
"ORCID": "https://orcid.org/0000-0003-2789-1758",
"affiliation": [
{
"name": "Yale School of Medicine;"
}
],
"authenticated-orcid": false,
"family": "Sawano",
"given": "Mitsuaki",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Center for Infection and Immunity, Yale School of Medicine, New Haven, CT, USA;"
}
],
"family": "Hooper",
"given": "William B.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Center for Infection and Immunity, Yale School of Medicine, New Haven, CT, USA; \tDepartment of Biostatistics, Yale School of Public Health, New Haven, CT, USA;"
}
],
"family": "Wang",
"given": "Kexin",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Immunobiology, Yale University School of Medicine, New Haven, CT, USA;"
}
],
"family": "Tabachnikova",
"given": "Alexandra",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Immunobiology, Yale University School of Medicine, New Haven, CT, USA;"
}
],
"family": "Monteiro",
"given": "Valter Silva",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Immunobiology, Yale University School of Medicine, New Haven, CT, USA;"
}
],
"family": "Lu",
"given": "Peiwen",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Center for Infection and Immunity, Yale School of Medicine, New Haven, CT, USA;"
}
],
"family": "Baevova",
"given": "Pavlina",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Center for Infection and Immunity, Yale School of Medicine, New Haven, CT, USA;"
}
],
"family": "Rodrigues",
"given": "Gisele C.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Center for Infection and Immunity, Yale School of Medicine, New Haven, CT, USA;"
}
],
"family": "Fisher",
"given": "Victoria L.",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0002-4557-9437",
"affiliation": [
{
"name": "Yale School of Medicine;"
}
],
"authenticated-orcid": false,
"family": "Caraballo",
"given": "Cesar",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0001-9467-6199",
"affiliation": [
{
"name": "Yale School of Medicine;"
}
],
"authenticated-orcid": false,
"family": "Khera",
"given": "Rohan",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Yale-New Haven Hospital;"
}
],
"family": "Li",
"given": "Shu-Xia",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Yale University;"
}
],
"family": "Herrin",
"given": "Jeph",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Yale Center for Clinical Investigation, Yale School of Medicine, New Haven, CT, USA;"
}
],
"family": "Christian",
"given": "Dany",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Section of Cardiovascular Medicine, Department of Internal Medicine, Yale School of Medicine, New Haven, CT, USA; Center for Outcomes Research and Evaluation, Yale New Haven Hospital, New Haven, CT, USA;"
}
],
"family": "Coppi",
"given": "Andreas",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Section of Cardiovascular Medicine, Department of Internal Medicine, Yale School of Medicine, New Haven, CT, USA; Center for Outcomes Research and Evaluation, Yale New Haven Hospital, New Haven, CT, USA;"
}
],
"family": "Warner",
"given": "Frederick",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Yale Center for Clinical Investigation, Yale School of Medicine, New Haven, CT, USA;"
}
],
"family": "Holub",
"given": "Julie",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Yale Center for Clinical Investigation, Yale School of Medicine, New Haven, CT, USA;"
}
],
"family": "Henriquez",
"given": "Yashira",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Section of Cardiovascular Medicine, Department of Internal Medicine, Yale School of Medicine, New Haven, CT, USA; Center for Outcomes Research and Evaluation, Yale New Haven Hospital, New Haven, CT, USA;"
}
],
"family": "Johnson",
"given": "Maria A.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Yale Center for Clinical Investigation, Yale School of Medicine, New Haven, CT, USA;"
}
],
"family": "Goddard",
"given": "Theresa B.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Yale Center for Clinical Investigation, Yale School of Medicine, New Haven, CT, USA;"
}
],
"family": "Rocco",
"given": "Erica",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Yale Center for Clinical Investigation, Yale School of Medicine, New Haven, CT, USA;"
}
],
"family": "Hummel",
"given": "Amy C.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Center for Outcomes Research and Evaluation, Yale New Haven Hospital, New Haven, CT, USA;"
}
],
"family": "Mouslmani",
"given": "Mohammad AL",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Trusted Medical PLLC, Middletown, CT, USA;"
}
],
"family": "Carr",
"given": "Kevin D.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Pfizer, Inc., Cambridge, MA, New York, NY, and Boulder, CO, USA;"
}
],
"family": "Charnas",
"given": "Lawrence",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Pfizer, Inc., Cambridge, MA, New York, NY, and Boulder, CO, USA;"
}
],
"family": "Jesus",
"given": "Magdia De",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Pfizer, Inc., Cambridge, MA, New York, NY, and Boulder, CO, USA;"
}
],
"family": "Nepert",
"given": "Dale",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Pfizer, Inc., Cambridge, MA, New York, NY, and Boulder, CO, USA;"
}
],
"family": "Abreu",
"given": "Paula",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Law Offices of Barry J. Gammons, Nashville, TN, USA;"
}
],
"family": "Ziegler",
"given": "Frank W.",
"sequence": "additional",
"suffix": "III"
},
{
"affiliation": [
{
"name": "UMKC: University of Missouri Kansas City;"
}
],
"family": "Spertus",
"given": "John",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Center for Infection and Immunity, Yale School of Medicine, New Haven, CT, USA; Department of Biostatistics, Yale School of Public Health, New Haven, CT, USA;"
}
],
"family": "Guan",
"given": "Leying",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0003-2046-127X",
"affiliation": [
{
"name": "Yale University;"
}
],
"authenticated-orcid": false,
"family": "Krumholz",
"given": "Harlan",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0002-7824-9856",
"affiliation": [
{
"name": "Yale University School of Medicine"
}
],
"authenticated-orcid": false,
"family": "Iwasaki",
"given": "Akiko",
"sequence": "additional"
}
],
"container-title": [],
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2026,
2,
26
]
],
"date-time": "2026-02-26T16:45:12Z",
"timestamp": 1772124312000
},
"deposited": {
"date-parts": [
[
2026,
2,
26
]
],
"date-time": "2026-02-26T16:45:12Z",
"timestamp": 1772124312000
},
"group-title": "Public and Global Health",
"indexed": {
"date-parts": [
[
2026,
2,
26
]
],
"date-time": "2026-02-26T17:31:55Z",
"timestamp": 1772127115992,
"version": "3.50.1"
},
"institution": [
{
"name": "medRxiv"
}
],
"is-referenced-by-count": 0,
"issued": {
"date-parts": [
[
2026,
2,
26
]
]
},
"license": [
{
"URL": "http://creativecommons.org/licenses/by/4.0/",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2026,
2,
26
]
],
"date-time": "2026-02-26T00:00:00Z",
"timestamp": 1772064000000
}
}
],
"link": [
{
"URL": "https://syndication.highwire.org/content/doi/10.64898/2026.02.24.26347001",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "54368",
"original-title": [],
"posted": {
"date-parts": [
[
2026,
2,
26
]
]
},
"prefix": "10.64898",
"published": {
"date-parts": [
[
2026,
2,
26
]
]
},
"publisher": "openRxiv",
"reference-count": 0,
"references-count": 0,
"relation": {},
"resource": {
"primary": {
"URL": "http://medrxiv.org/lookup/doi/10.64898/2026.02.24.26347001"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"subtype": "preprint",
"title": "Exploratory analyses of Immunologic Features in a Randomized, Placebo-Controlled Trial of Nirmatrelvir/Ritonavir for Long COVID",
"type": "posted-content"
}