Mortality rates of severe COVID-19-related respiratory failure with and without extracorporeal membrane oxygenation in the Middle Ruhr Region of Germany
et al., Scientific Reports, doi:10.1038/s41598-023-31944-7, Mar 2023
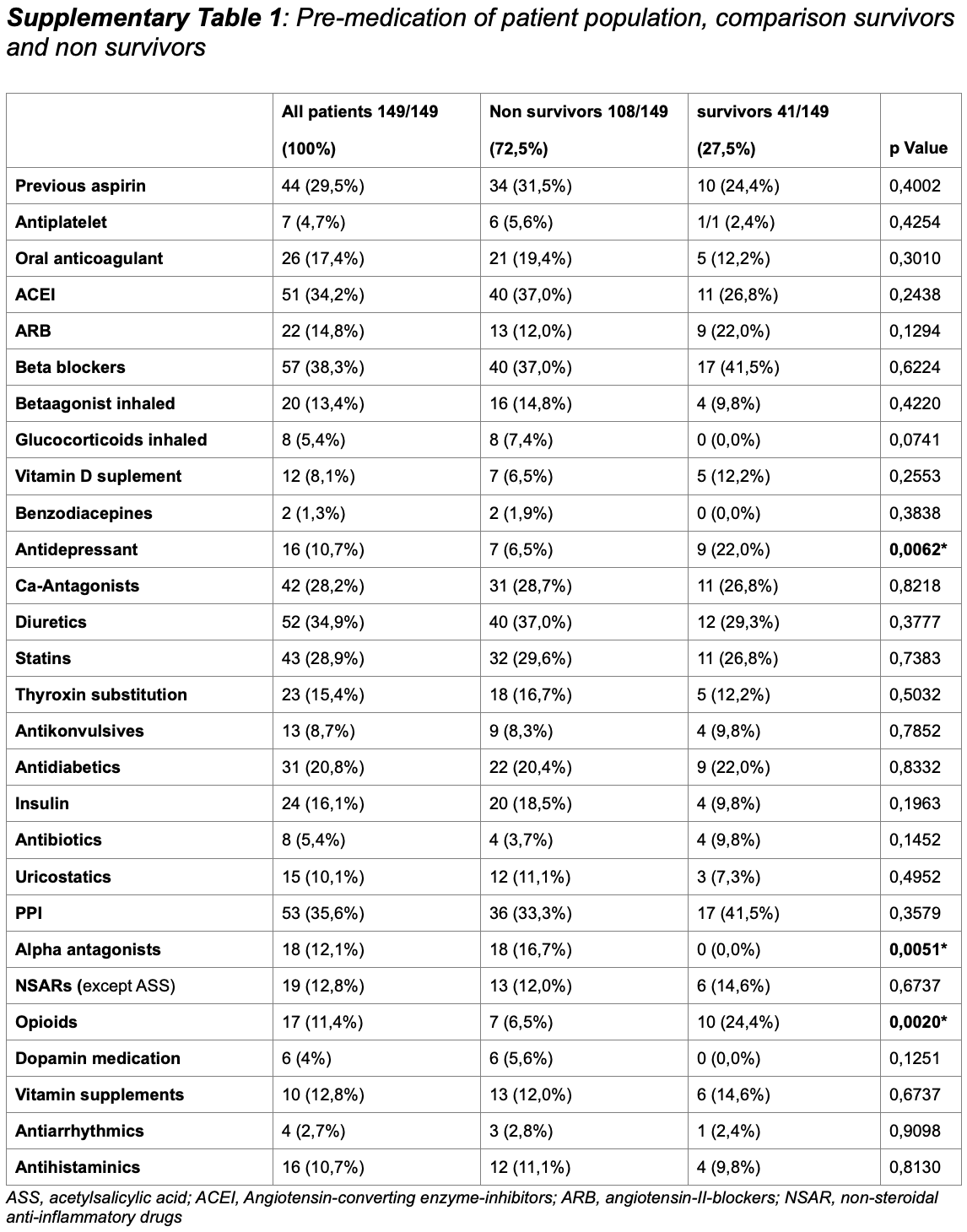
Retrospective 149 patients under invasive mechanical ventilation in Germany showing no significant difference in mortality with aspirin prophylaxis in unadjusted results.
This study is excluded in the after exclusion results of meta-analysis:
unadjusted results with no group details.
|
risk of death, 9.6% higher, RR 1.10, p = 0.43, treatment 34 of 44 (77.3%), control 74 of 105 (70.5%).
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Aweimer et al., 29 Mar 2023, retrospective, Germany, peer-reviewed, median age 67.0, 19 authors, study period 1 March, 2020 - 31 August, 2021.
Mortality rates of severe COVID-19-related respiratory failure with and without extracorporeal membrane oxygenation in the Middle Ruhr Region of Germany
Scientific Reports, doi:10.1038/s41598-023-31944-7
The use of extracorporeal membrane oxygenation (ECMO) is discussed to improve patients' outcome in severe COVID-19 with respiratory failure, but data on ECMO remains controversial. The aim of the study was to determine the characteristics of patients under invasive mechanical ventilation (IMV) with or without veno-venous ECMO support and to evaluate outcome parameters. Ventilated patients with COVID-19 with and without additional ECMO support were analyzed in a retrospective multicenter study regarding clinical characteristics, respiratory and laboratory parameters in dayto-day follow-up. Recruitment of patients was conducted during the first three COVID-19 waves at four German university hospitals of the Ruhr University Bochum, located in the Middle Ruhr Region. From March 1, 2020 to August 31, 2021, the charts of 149 patients who were ventilated for COVID-19 infection, were included (63.8% male, median age 67 years). Fifty patients (33.6%) received additional ECMO support. On average, ECMO therapy was initiated 15.6 ± 9.4 days after symptom onset, 10.6 ± 7.1 days after hospital admission, and 4.8 ± 6.4 days after the start of IMV. Male sex and higher SOFA and RESP scores were observed significantly more often in the high-volume ECMO center. Premedication with antidepressants was more often detected in survivors (22.0% vs. 6.5%; p = 0.006). ECMO patients were 14 years younger and presented a lower rate of concomitant cardiovascular
Author contributions A.A. and L.P. take responsibility for the content of the manuscript, including the data and analysis. A.A., A.M., T.R., C.D., T.B., J.F., D.B. contributed to the conception and design of the study. A.A., L.P., B.J., R.K., D.B., A.M. analyzed and interpreted the patient data. A.A., L.P. and B.J. performed the statistical analyses. A.A., A.M., I.E. and TB were major contributors in writing the manuscript. J.D., R.K., D.B., T.R., U.F., M.A., J.B., I.H. contributed to interpretation of the data and revised the manuscript. All authors read and approved the final manuscript.
Competing interests The authors declare no competing interests.
References
Alshahrani, Extracorporeal membrane oxygenation for severe Middle East respiratory syndrome coronavirus, Ann. Intensive Care, doi:10.1186/s13613-017-0350-x.PMID:29330690;PMCID:PMC5768582
Arentz, Characteristics and outcomes of 21 critically Ill patients with COVID-19 in Washington State, JAMA, doi:10.1001/jama.2020.4326
Barbaro, Association of hospital-level volume of extracorporeal membrane oxygenation cases and mortality. Analysis of the extracorporeal life support organization registry, Am. J. Respir. Crit. Care Med, doi:10.1164/rccm.201409-1634OC
Barbaro, Extracorporeal life support organization. Extracorporeal membrane oxygenation for COVID-19: Evolving outcomes from the international extracorporeal life support organization registry, Lancet, doi:10.1016/S0140-6736(21)01960-7
Barbaro, Maclaren, Boonstra, Iwashyna, Slutsky et al., Extracorporeal Life Support Organization
Bercker, ECMO use in Germany: An analysis of 29,929 ECMO runs, PLoS ONE, doi:10.1371/journal.pone.0260324
Bizzarro, Infections acquired during extracorporeal membrane oxygenation in neonates, children, and adults, Pediatr. Crit. Care Med, doi:10.1097/PCC.0b013e3181e28894
Chen, Plasma CRP level is positively associated with the severity of COVID-19, Ann. Clin. Microbiol. Antimicrob, doi:10.1186/s12941-020-00362-2
Cheng, Retrospective study of critically Ill COVID-19 patients with and without extracorporeal membrane oxygenation support in Wuhan, Front. Med, doi:10.3389/fmed.2021.659793
Combes, Position paper for the organization of extracorporeal membrane oxygenation programs for acute respiratory failure in adult patients, Am. J. Respir. Crit. Care Med, doi:10.1164/rccm.201404-0630CP
Corrêa, Clinical characteristics and outcomes of COVID-19 patients admitted to the intensive care unit during the first year of the pandemic in Brazil: A single center retrospective cohort study, doi:10.31744/einstein_journal/2021AO6739
Doyle, Blood component use in critical care in patients with COVID-19 infection: A single-centre experience, Br. J. Haematol, doi:10.1111/bjh.17007
Fraaij, Severe acute respiratory infection caused by swine influenza virus in a child necessitating extracorporeal membrane oxygenation (ECMO), the Netherlands, Eurosurveill
Friedrichson, Extracorporeal membrane oxygenation in coronavirus disease 2019: A nationwide cohort analysis of 4279 runs from Germany, Eur. J. Anaesthesiol, doi:10.1097/EJA.0000000000001670
Funakoshi, Morita, Kumanogoh, Longer prehospitalization and preintubation periods in intubated non-survivors and ECMO patients with COVID-19: A systematic review and meta-analysis, Front. Med. Lausanne, doi:10.3389/fmed.2021.727101
Henriques, Multiscale Poincaré plots for visualizing the structure of heartbeat time series, BMC Med. Inform. Decis. Mak, doi:10.1186/s12911-016-0252-0.PMID:26860191;PMCID:PMC4746786
Henry, COVID-19, ECMO, and lymphopenia: A word of caution, Lancet Respir. Med, doi:10.1016/S2213-2600(20)30119-3
Hilder, Comparison of mortality prediction models in acute respiratory distress syndrome undergoing extracorporeal membrane oxygenation and development of a novel prediction score: the PREdiction of Survival on ECMO Therapy-Score (PRESET-Score), Crit. Care, doi:10.1186/s13054-017-1888-6
Hoertel, Association between antidepressant use and reduced risk of intubation or death in hospitalized patients with COVID-19: results from an observational study, Mol. Psychiatry, doi:10.1038/s41380-021-01021-4
Karagiannidis, Case characteristics, resource use, and outcomes of 10 021 patients with COVID-19 admitted to 920 German hospitals: An observational study, Lancet Respir. Med, doi:10.1016/S2213-2600(20)30316-7
Karagiannidis, Complete countrywide mortality in COVID patients receiving ECMO in Germany throughout the first three waves of the pandemic, Crit. Care, doi:10.1186/s13054-021-03831-y
Karagiannidis, Observational study of changes in utilization and outcomes in mechanical ventilation in COVID-19, PLoS ONE, doi:10.1371/journal.pone.0262315
Kelly, Petti, Noah, Covid-19, non-Covid-19 and excess mortality rates not comparable across countries, Epidemiol. Infect, doi:10.1017/S0950268821001850
Lebreton, Schmidt, Ponnaiah, Folliguet, Para et al., Paris ECMO-COVID-19 investigators. Extracorporeal membrane oxygenation network organisation and clinical outcomes during the COVID-19 pandemic in Greater Paris, France: A multicentre cohort study, Lancet Respir. Med, doi:10.1016/S2213-2600(21)00096-5
Liu, Bilirubin levels as potential indicators of disease severity in coronavirus disease patients: A retrospective cohort study, Front. Med. Lausanne, doi:10.3389/fmed.2020.598870
Mcelvaney, Characterization of the inflammatory response to severe COVID-19 illness, Am. J. Respir. Crit. Care Med
Mehta, COVID-19: Consider cytokine storm syndromes and immunosuppression, Lancet
Michelozzi, De'donato, Scortichini, Pezzotti, Stafoggia et al., Temporal dynamics in total excess mortality and COVID-19 deaths in Italian cities, BMC Public Health, doi:10.1186/s12889-020-09335-8
Nesseler, Clinical review: The liver in sepsis, Crit. Care, doi:10.1186/cc11381
Nicholson, Estimating risk of mechanical ventilation and in-hospital mortality among adult COVID-19 patients admitted to Mass General Brigham: The VICE and DICE scores, EClinicalMedicine, doi:10.1016/j.eclinm.2021.100765
Nielsen, Nørgaard, Lanzieri, Vestergaard, Moelbak, Sex-differences in COVID-19 associated excess mortality is not exceptional for the COVID-19 pandemic, Sci. Rep, doi:10.1038/s41598-021-00213-w.PMID:34675280;PMCID:PMC8531278
Niessen, Sex differences in COVID-19 mortality in the Netherlands, Infection, doi:10.1007/s15010-021-01744-0
Oliveira, Wong, Lippi, Henry, Analysis of clinical and demographic heterogeneity of patients dying from COVID-19 in Brazil versus China and Italy, Braz. J. Infect. Dis
Oskotsky, Mortality risk among patients with COVID-19 prescribed selective serotonin reuptake inhibitor antidepressants, JAMA Netw. Open, doi:10.1001/jamanetworkopen.2021.33090
Osuchowski, The COVID-19 puzzle: Deciphering pathophysiology and phenotypes of a new disease entity, Lancet Respir Med, doi:10.1016/S2213-2600(21)00218-6
Paliogiannis, Zinellu, Bilirubin levels in patients with mild and severe Covid-19: A pooled analysis, Liver Int, doi:10.1111/liv.14477
Peckham, Male sex identified by global COVID-19 meta-analysis as a risk factor for death and ITU admission, Nat. Commun, doi:10.1038/s41467-020-19741-6.PMID:33298944;PMCID:PMC7726563
Reis, Dos, Moreira-Silva, Silva, Thabane et al., Effect of early treatment with fluvoxamine on risk of emergency care and hospitalisation among patients with COVID-19: The TOGETHER randomised, platform clinical trial, Lancet Glob. Health, doi:10.1016/S2214-109X(21)00448-4
Roedl, Clinical characteristics, complications and outcomes of patients with severe acute respiratory distress syndrome related to COVID-19 or influenza requiring extracorporeal membrane oxygenation-A retrospective cohort study, J. Clin. Med, doi:10.3390/jcm10225440.PMID:34830721;PMCID:PMC8619058
Schmidt, Extracorporeal membrane oxygenation for severe acute respiratory distress syndrome associated with COVID-19: A retrospective cohort study, Lancet Respir. Med, doi:10.1016/S2213-2600(20)30328-3
Schmidt, Predicting survival after extracorporeal membrane oxygenation for severe acute respiratory failure. The respiratory extracorporeal membrane oxygenation survival prediction (RESP) score, Am. J. Respir. Crit. Care Med, doi:10.1164/rccm.201311-2023OC
Stokes, Coronavirus disease 2019 case surveillance -United States, MMWR Morb. Mortal Wkly. Rep, doi:10.15585/mmwr.mm6924e2
Sukhatme, Reiersen, Vayttaden, Sukhatme, Fluvoxamine: A review of its mechanism of action and its role in COVID-19, Front. Pharmacol, doi:10.3389/fphar.2021.652688
Supady, Survival after extracorporeal membrane oxygenation in severe COVID-19 ARDS: Results from an international multicenter registry, Crit. Care, doi:10.1186/s13054-021-03486-9
Supady, Weber, Rieder, Lother, Niklaus et al., Cytokine adsorption in patients with severe COVID-19 pneumonia requiring extracorporeal membrane oxygenation (CYCOV): A single centre, open-label, randomised, controlled trial, Lancet Respir. Med, doi:10.1016/S2213-2600(21)00177-6
Valerio, Lodigiani C course of D-dimer and C-reactive protein levels in survivors and nonsurvivors with COVID-19 pneumonia: A retrospective analysis of 577 patients, Thromb. Haemost, doi:10.1055/s-0040-1721317
Williamson, Factors associated with COVID-19-related death using OpenSAFELY, Nature, doi:10.1038/s41586-020-2521-4
Www, .nature.com/scientificreports/ cohort study of the extracorporeal life support organization registry, Lancet, doi:10.1016/S0140-6736(20)32008-0
Yang, Yu, Xu, Shu, Xia et al., Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: a single-centered, retrospective, observational study, Lancet Respir. Med, doi:10.1016/S2213-2600(20)30079-5
Yitbarek, The role of C-reactive protein in predicting the severity of COVID-19 disease: A systematic review, SAGE Open Med, doi:10.1177/20503121211050755.PMID:34659766;PMCID:PMC8516378
Zhou, Yu, Du, Fan, Liu et al., Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study, Lancet, doi:10.1016/S0140-6736(20)30566-3
DOI record:
{
"DOI": "10.1038/s41598-023-31944-7",
"ISSN": [
"2045-2322"
],
"URL": "http://dx.doi.org/10.1038/s41598-023-31944-7",
"abstract": "<jats:title>Abstract</jats:title><jats:p>The use of extracorporeal membrane oxygenation (ECMO) is discussed to improve patients’ outcome in severe COVID-19 with respiratory failure, but data on ECMO remains controversial. The aim of the study was to determine the characteristics of patients under invasive mechanical ventilation (IMV) with or without veno-venous ECMO support and to evaluate outcome parameters. Ventilated patients with COVID-19 with and without additional ECMO support were analyzed in a retrospective multicenter study regarding clinical characteristics, respiratory and laboratory parameters in day-to-day follow-up. Recruitment of patients was conducted during the first three COVID-19 waves at four German university hospitals of the Ruhr University Bochum, located in the Middle Ruhr Region. From March 1, 2020 to August 31, 2021, the charts of 149 patients who were ventilated for COVID-19 infection, were included (63.8% male, median age 67 years). Fifty patients (33.6%) received additional ECMO support. On average, ECMO therapy was initiated 15.6 ± 9.4 days after symptom onset, 10.6 ± 7.1 days after hospital admission, and 4.8 ± 6.4 days after the start of IMV. Male sex and higher SOFA and RESP scores were observed significantly more often in the high-volume ECMO center. Pre-medication with antidepressants was more often detected in survivors (22.0% vs. 6.5%; <jats:italic>p</jats:italic> = 0.006). ECMO patients were 14 years younger and presented a lower rate of concomitant cardiovascular diseases (18.0% vs. 47.5%; <jats:italic>p</jats:italic> = 0.0004). Additionally, cytokine-adsorption (46.0% vs. 13.1%; <jats:italic>p</jats:italic> < 0.0001) and renal replacement therapy (76.0% vs. 43.4%; <jats:italic>p</jats:italic> = 0.0001) were carried out more frequently; in ECMO patients thrombocytes were transfused 12-fold more often related to more than fourfold higher bleeding complications. Undulating C-reactive protein (CRP) and massive increase in bilirubin levels (at terminal stage) could be observed in deceased ECMO patients. In-hospital mortality was high (Overall: 72.5%, ECMO: 80.0%, ns). Regardless of ECMO therapy half of the study population deceased within 30 days after hospital admission. Despite being younger and with less comorbidities ECMO therapy did not improve survival in severely ill COVID-19 patients. Undulating CRP levels, a massive increase of bilirubin level and a high use of cytokine-adsorption were associated with worse outcomes. In conclusion, ECMO support might be helpful in selected severe cases of COVID-19.</jats:p>",
"alternative-id": [
"31944"
],
"article-number": "5143",
"assertion": [
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "Received",
"name": "received",
"order": 1,
"value": "16 May 2022"
},
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "Accepted",
"name": "accepted",
"order": 2,
"value": "20 March 2023"
},
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "First Online",
"name": "first_online",
"order": 3,
"value": "29 March 2023"
},
{
"group": {
"label": "Competing interests",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 1,
"value": "The authors declare no competing interests."
}
],
"author": [
{
"affiliation": [],
"family": "Aweimer",
"given": "Assem",
"sequence": "first"
},
{
"affiliation": [],
"family": "Petschulat",
"given": "Lea",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Jettkant",
"given": "Birger",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Köditz",
"given": "Roland",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Finkeldei",
"given": "Johannes",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Dietrich",
"given": "Johannes W.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Breuer",
"given": "Thomas",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Draese",
"given": "Christian",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Frey",
"given": "Ulrich H.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Rahmel",
"given": "Tim",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Adamzik",
"given": "Michael",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Buchwald",
"given": "Dirk",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Useini",
"given": "Dritan",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Brechmann",
"given": "Thorsten",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Hosbach",
"given": "Ingolf",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Bünger",
"given": "Jürgen",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ewers",
"given": "Aydan",
"sequence": "additional"
},
{
"affiliation": [],
"family": "El-Battrawy",
"given": "Ibrahim",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Mügge",
"given": "Andreas",
"sequence": "additional"
}
],
"container-title": "Scientific Reports",
"container-title-short": "Sci Rep",
"content-domain": {
"crossmark-restriction": false,
"domain": [
"link.springer.com"
]
},
"created": {
"date-parts": [
[
2023,
3,
29
]
],
"date-time": "2023-03-29T15:03:32Z",
"timestamp": 1680102212000
},
"deposited": {
"date-parts": [
[
2023,
3,
29
]
],
"date-time": "2023-03-29T15:06:04Z",
"timestamp": 1680102364000
},
"funder": [
{
"DOI": "10.13039/501100006254",
"doi-asserted-by": "crossref",
"name": "Ruhr-Universität Bochum"
}
],
"indexed": {
"date-parts": [
[
2023,
3,
30
]
],
"date-time": "2023-03-30T04:23:33Z",
"timestamp": 1680150213904
},
"is-referenced-by-count": 0,
"issue": "1",
"issued": {
"date-parts": [
[
2023,
3,
29
]
]
},
"journal-issue": {
"issue": "1",
"published-online": {
"date-parts": [
[
2023,
12
]
]
}
},
"language": "en",
"license": [
{
"URL": "https://creativecommons.org/licenses/by/4.0",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2023,
3,
29
]
],
"date-time": "2023-03-29T00:00:00Z",
"timestamp": 1680048000000
}
},
{
"URL": "https://creativecommons.org/licenses/by/4.0",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2023,
3,
29
]
],
"date-time": "2023-03-29T00:00:00Z",
"timestamp": 1680048000000
}
}
],
"link": [
{
"URL": "https://www.nature.com/articles/s41598-023-31944-7.pdf",
"content-type": "application/pdf",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://www.nature.com/articles/s41598-023-31944-7",
"content-type": "text/html",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://www.nature.com/articles/s41598-023-31944-7.pdf",
"content-type": "application/pdf",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "297",
"original-title": [],
"prefix": "10.1038",
"published": {
"date-parts": [
[
2023,
3,
29
]
]
},
"published-online": {
"date-parts": [
[
2023,
3,
29
]
]
},
"publisher": "Springer Science and Business Media LLC",
"reference": [
{
"DOI": "10.15585/mmwr.mm6924e2",
"author": "EK Stokes",
"doi-asserted-by": "publisher",
"first-page": "759",
"issue": "24",
"journal-title": "MMWR Morb. Mortal Wkly. Rep.",
"key": "31944_CR1",
"unstructured": "Stokes, E. K. et al. Coronavirus disease 2019 case surveillance - United States, January 22-May 30, 2020. MMWR Morb. Mortal Wkly. Rep. 69(24), 759–765. https://doi.org/10.15585/mmwr.mm6924e2 (2020).",
"volume": "69",
"year": "2020"
},
{
"DOI": "10.1016/S2213-2600(21)00218-6",
"author": "MF Osuchowski",
"doi-asserted-by": "publisher",
"first-page": "622",
"issue": "6",
"journal-title": "Lancet Respir Med.",
"key": "31944_CR2",
"unstructured": "Osuchowski, M. F. et al. The COVID-19 puzzle: Deciphering pathophysiology and phenotypes of a new disease entity. Lancet Respir Med. 9(6), 622–642. https://doi.org/10.1016/S2213-2600(21)00218-6 (2021).",
"volume": "9",
"year": "2021"
},
{
"DOI": "10.1016/j.eclinm.2021.100765",
"author": "CJ Nicholson",
"doi-asserted-by": "publisher",
"first-page": "100765",
"journal-title": "EClinicalMedicine.",
"key": "31944_CR3",
"unstructured": "Nicholson, C. J. et al. Estimating risk of mechanical ventilation and in-hospital mortality among adult COVID-19 patients admitted to Mass General Brigham: The VICE and DICE scores. EClinicalMedicine. 33, 100765. https://doi.org/10.1016/j.eclinm.2021.100765 (2021).",
"volume": "33",
"year": "2021"
},
{
"DOI": "10.1186/s13613-017-0350-x.PMID:29330690;PMCID:PMC5768582",
"author": "MS Alshahrani",
"doi-asserted-by": "publisher",
"first-page": "3",
"issue": "1",
"journal-title": "Ann. Intensive Care",
"key": "31944_CR4",
"unstructured": "Alshahrani, M. S. et al. Extracorporeal membrane oxygenation for severe Middle East respiratory syndrome coronavirus. Ann. Intensive Care 8(1), 3. https://doi.org/10.1186/s13613-017-0350-x.PMID:29330690;PMCID:PMC5768582 (2018).",
"volume": "8",
"year": "2018"
},
{
"DOI": "10.2807/1560-7917.ES.2016.21.48.30416",
"author": "PL Fraaij",
"doi-asserted-by": "publisher",
"first-page": "30416",
"journal-title": "Eurosurveill",
"key": "31944_CR5",
"unstructured": "Fraaij, P. L. et al. Severe acute respiratory infection caused by swine influenza virus in a child necessitating extracorporeal membrane oxygenation (ECMO), the Netherlands, October 2016. Eurosurveill 21, 30416 (2016).",
"volume": "21",
"year": "2016"
},
{
"DOI": "10.3390/jcm10225440.PMID:34830721;PMCID:PMC8619058",
"author": "K Roedl",
"doi-asserted-by": "publisher",
"first-page": "5440",
"issue": "22",
"journal-title": "J. Clin. Med.",
"key": "31944_CR6",
"unstructured": "Roedl, K. et al. Clinical characteristics, complications and outcomes of patients with severe acute respiratory distress syndrome related to COVID-19 or influenza requiring extracorporeal membrane oxygenation-A retrospective cohort study. J. Clin. Med. 10(22), 5440. https://doi.org/10.3390/jcm10225440.PMID:34830721;PMCID:PMC8619058 (2021).",
"volume": "10",
"year": "2021"
},
{
"DOI": "10.1016/S2213-2600(21)00096-5",
"doi-asserted-by": "publisher",
"key": "31944_CR7",
"unstructured": "Lebreton, G., Schmidt, M., Ponnaiah, M., Folliguet, T., Para, M., Guihaire, J., Lansac, E., Sage, E., Cholley, B., Mégarbane, B., Cronier, P., Zarka, J., Da Silva, D., Besset, S., Morichau-Beauchant, T., Lacombat, I., Mongardon, N., Richard, C., Duranteau, J., Cerf, C., Saiydoun, G., Sonneville, R., Chiche, J. D., Nataf, P., Longrois, D., Combes, A., Leprince, P.; Paris ECMO-COVID-19 investigators. Extracorporeal membrane oxygenation network organisation and clinical outcomes during the COVID-19 pandemic in Greater Paris, France: A multicentre cohort study. Lancet Respir. Med. 9(8), 851–862 (2021). doi: https://doi.org/10.1016/S2213-2600(21)00096-5. Erratum in: Lancet Respir. Med. 9(6), e55 (2021). Erratum in: Lancet Respir. Med. 9(7), e62 (2021)."
},
{
"DOI": "10.1016/S2213-2600(20)30079-5",
"doi-asserted-by": "publisher",
"key": "31944_CR8",
"unstructured": "Yang, X., Yu, Y., Xu, J., Shu, H., Xia, J., Liu, H., Wu, Y., Zhang, L., Yu, Z., Fang, M., Yu, T., Wang, Y., Pan, S., Zou, X., Yuan, S., Shang Y. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: a single-centered, retrospective, observational study. Lancet Respir. Med. 8(5), 475–481 (2020). doi: https://doi.org/10.1016/S2213-2600(20)30079-5. Erratum in: Lancet Respir. Med. 8(4), e26 (2020)."
},
{
"DOI": "10.1001/jama.2020.4326",
"author": "M Arentz",
"doi-asserted-by": "publisher",
"first-page": "1612",
"issue": "16",
"journal-title": "JAMA",
"key": "31944_CR9",
"unstructured": "Arentz, M. et al. Characteristics and outcomes of 21 critically Ill patients with COVID-19 in Washington State. JAMA 323(16), 1612–1614. https://doi.org/10.1001/jama.2020.4326 (2020).",
"volume": "323",
"year": "2020"
},
{
"DOI": "10.1016/S0140-6736(20)30566-3",
"doi-asserted-by": "publisher",
"key": "31944_CR10",
"unstructured": "Zhou, F., Yu, T., Du, R., Fan, G., Liu, Y., Liu, Z., Xiang, J., Wang, Y., Song, B., Gu, X., Guan, L., Wei, Y., Li, H., Wu, X., Xu, J., Tu, S., Zhang, Y., Chen, H., Cao, B. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 395(10229), 1054–1062 (2020). https://doi.org/10.1016/S0140-6736(20)30566-3. Epub 2020 Mar 11. Erratum in: Lancet 395(10229), 1038 (2020). Erratum in: Lancet 395(10229), 1038 (2020)."
},
{
"DOI": "10.1016/S0140-6736(21)01960-7",
"author": "RP Barbaro",
"doi-asserted-by": "publisher",
"first-page": "1230",
"issue": "10307",
"journal-title": "Lancet",
"key": "31944_CR11",
"unstructured": "Barbaro, R. P. et al. Extracorporeal life support organization. Extracorporeal membrane oxygenation for COVID-19: Evolving outcomes from the international extracorporeal life support organization registry. Lancet 398(10307), 1230–1238. https://doi.org/10.1016/S0140-6736(21)01960-7 (2021).",
"volume": "398",
"year": "2021"
},
{
"DOI": "10.1016/S0140-6736(20)32008-0",
"doi-asserted-by": "publisher",
"key": "31944_CR12",
"unstructured": "Barbaro, R. P., MacLaren, G., Boonstra, P. S., Iwashyna, T. J., Slutsky, A. S., Fan, E., Bartlett, R. H., Tonna, J. E., Hyslop, R., Fanning, J. J., Rycus, P. T., Hyer, S. J., Anders, M. M., Agerstrand, C. L., Hryniewicz, K., Diaz, R., Lorusso, R., Combes, A., Brodie, D., Extracorporeal Life Support Organization. Extracorporeal membrane oxygenation support in COVID-19: An international cohort study of the extracorporeal life support organization registry. Lancet 396(10257), 1071–1078 (2020). https://doi.org/10.1016/S0140-6736(20)32008-0. Epub 2020 Sep 25. Erratum in: Lancet 396(10257), 1070 (2020)."
},
{
"DOI": "10.1371/journal.pone.0262315",
"author": "C Karagiannidis",
"doi-asserted-by": "publisher",
"first-page": "e0262315",
"issue": "1",
"journal-title": "PLoS ONE",
"key": "31944_CR13",
"unstructured": "Karagiannidis, C. et al. Observational study of changes in utilization and outcomes in mechanical ventilation in COVID-19. PLoS ONE 17(1), e0262315. https://doi.org/10.1371/journal.pone.0262315 (2022).",
"volume": "17",
"year": "2022"
},
{
"DOI": "10.1016/S2213-2600(20)30328-3",
"author": "M Schmidt",
"doi-asserted-by": "publisher",
"first-page": "1121",
"issue": "11",
"journal-title": "Lancet Respir. Med.",
"key": "31944_CR14",
"unstructured": "Schmidt, M. et al. Extracorporeal membrane oxygenation for severe acute respiratory distress syndrome associated with COVID-19: A retrospective cohort study. Lancet Respir. Med. 8(11), 1121–1131. https://doi.org/10.1016/S2213-2600(20)30328-3 (2020).",
"volume": "8",
"year": "2020"
},
{
"DOI": "10.1016/S2213-2600(20)30316-7",
"author": "C Karagiannidis",
"doi-asserted-by": "publisher",
"first-page": "853",
"issue": "9",
"journal-title": "Lancet Respir. Med.",
"key": "31944_CR15",
"unstructured": "Karagiannidis, C. et al. Case characteristics, resource use, and outcomes of 10 021 patients with COVID-19 admitted to 920 German hospitals: An observational study. Lancet Respir. Med. 8(9), 853–862. https://doi.org/10.1016/S2213-2600(20)30316-7 (2020).",
"volume": "8",
"year": "2020"
},
{
"DOI": "10.1186/s13054-021-03831-y",
"author": "C Karagiannidis",
"doi-asserted-by": "publisher",
"first-page": "413",
"issue": "1",
"journal-title": "Crit. Care",
"key": "31944_CR16",
"unstructured": "Karagiannidis, C. et al. Complete countrywide mortality in COVID patients receiving ECMO in Germany throughout the first three waves of the pandemic. Crit. Care 25(1), 413. https://doi.org/10.1186/s13054-021-03831-y (2021).",
"volume": "25",
"year": "2021"
},
{
"DOI": "10.1017/S0950268821001850",
"author": "G Kelly",
"doi-asserted-by": "publisher",
"first-page": "e176",
"journal-title": "Epidemiol. Infect.",
"key": "31944_CR17",
"unstructured": "Kelly, G., Petti, S. & Noah, N. Covid-19, non-Covid-19 and excess mortality rates not comparable across countries. Epidemiol. Infect. 149, e176. https://doi.org/10.1017/S0950268821001850 (2021).",
"volume": "149",
"year": "2021"
},
{
"DOI": "10.1016/j.bjid.2020.05.002",
"author": "MHS Oliveira",
"doi-asserted-by": "publisher",
"first-page": "273",
"journal-title": "Braz. J. Infect. Dis.",
"key": "31944_CR18",
"unstructured": "Oliveira, M. H. S., Wong, J., Lippi, G. & Henry, B. M. Analysis of clinical and demographic heterogeneity of patients dying from COVID-19 in Brazil versus China and Italy. Braz. J. Infect. Dis. 24, 273–275 (2020).",
"volume": "24",
"year": "2020"
},
{
"DOI": "10.1186/s12889-020-09335-8",
"doi-asserted-by": "publisher",
"key": "31944_CR19",
"unstructured": "Michelozzi, P., de'Donato, F., Scortichini, M., Pezzotti, P., Stafoggia, M., De Sario, M., Costa, G., Noccioli, F., Riccardo, F., Bella, A., Demaria, M., Rossi, P., Brusaferro, S., Rezza, G., Davoli, M. Temporal dynamics in total excess mortality and COVID-19 deaths in Italian cities. BMC Public Health 20(1), 1238 (2020). https://doi.org/10.1186/s12889-020-09335-8. Erratum in: BMC Public Health 20(1), 1325 (2020)."
},
{
"DOI": "10.1186/s12911-016-0252-0.PMID:26860191;PMCID:PMC4746786",
"author": "TS Henriques",
"doi-asserted-by": "publisher",
"first-page": "17",
"issue": "16",
"journal-title": "BMC Med. Inform. Decis. Mak.",
"key": "31944_CR20",
"unstructured": "Henriques, T. S. et al. Multiscale Poincaré plots for visualizing the structure of heartbeat time series. BMC Med. Inform. Decis. Mak. 9(16), 17. https://doi.org/10.1186/s12911-016-0252-0.PMID:26860191;PMCID:PMC4746786 (2016).",
"volume": "9",
"year": "2016"
},
{
"DOI": "10.1164/rccm.201311-2023OC",
"author": "M Schmidt",
"doi-asserted-by": "publisher",
"first-page": "1374",
"issue": "11",
"journal-title": "Am. J. Respir. Crit. Care Med.",
"key": "31944_CR21",
"unstructured": "Schmidt, M. et al. Predicting survival after extracorporeal membrane oxygenation for severe acute respiratory failure. The respiratory extracorporeal membrane oxygenation survival prediction (RESP) score. Am. J. Respir. Crit. Care Med. 189(11), 1374–1382. https://doi.org/10.1164/rccm.201311-2023OC (2014).",
"volume": "189",
"year": "2014"
},
{
"DOI": "10.1186/s13054-017-1888-6",
"author": "M Hilder",
"doi-asserted-by": "publisher",
"first-page": "301",
"issue": "1",
"journal-title": "Crit. Care",
"key": "31944_CR22",
"unstructured": "Hilder, M. et al. Comparison of mortality prediction models in acute respiratory distress syndrome undergoing extracorporeal membrane oxygenation and development of a novel prediction score: the PREdiction of Survival on ECMO Therapy-Score (PRESET-Score). Crit. Care 21(1), 301. https://doi.org/10.1186/s13054-017-1888-6 (2017).",
"volume": "21",
"year": "2017"
},
{
"DOI": "10.1097/EJA.0000000000001670",
"author": "B Friedrichson",
"doi-asserted-by": "publisher",
"first-page": "445",
"issue": "5",
"journal-title": "Eur. J. Anaesthesiol.",
"key": "31944_CR23",
"unstructured": "Friedrichson, B. et al. Extracorporeal membrane oxygenation in coronavirus disease 2019: A nationwide cohort analysis of 4279 runs from Germany. Eur. J. Anaesthesiol. 39(5), 445–451. https://doi.org/10.1097/EJA.0000000000001670 (2022).",
"volume": "39",
"year": "2022"
},
{
"DOI": "10.1038/s41586-020-2521-4",
"author": "EJ Williamson",
"doi-asserted-by": "publisher",
"first-page": "430",
"issue": "7821",
"journal-title": "Nature",
"key": "31944_CR24",
"unstructured": "Williamson, E. J. et al. Factors associated with COVID-19-related death using OpenSAFELY. Nature 584(7821), 430–436. https://doi.org/10.1038/s41586-020-2521-4 (2020).",
"volume": "584",
"year": "2020"
},
{
"DOI": "10.3389/fmed.2021.659793",
"author": "W Cheng",
"doi-asserted-by": "publisher",
"first-page": "659793",
"journal-title": "Front. Med. (Lausanne)",
"key": "31944_CR25",
"unstructured": "Cheng, W. et al. Retrospective study of critically Ill COVID-19 patients with and without extracorporeal membrane oxygenation support in Wuhan, China. Front. Med. (Lausanne) 8, 659793. https://doi.org/10.3389/fmed.2021.659793 (2021).",
"volume": "8",
"year": "2021"
},
{
"DOI": "10.31744/einstein_journal/2021AO6739",
"author": "TD Corrêa",
"doi-asserted-by": "publisher",
"first-page": "eAO6739",
"journal-title": "Einstein (Sao Paulo)",
"key": "31944_CR26",
"unstructured": "Corrêa, T. D. et al. Clinical characteristics and outcomes of COVID-19 patients admitted to the intensive care unit during the first year of the pandemic in Brazil: A single center retrospective cohort study. Einstein (Sao Paulo) 19, eAO6739. https://doi.org/10.31744/einstein_journal/2021AO6739 (2021).",
"volume": "19",
"year": "2021"
},
{
"DOI": "10.1186/s13054-021-03486-9",
"author": "A Supady",
"doi-asserted-by": "publisher",
"first-page": "90",
"issue": "1",
"journal-title": "Crit. Care",
"key": "31944_CR27",
"unstructured": "Supady, A. et al. Survival after extracorporeal membrane oxygenation in severe COVID-19 ARDS: Results from an international multicenter registry. Crit. Care 25(1), 90. https://doi.org/10.1186/s13054-021-03486-9 (2021).",
"volume": "25",
"year": "2021"
},
{
"DOI": "10.3389/fmed.2021.727101",
"author": "K Funakoshi",
"doi-asserted-by": "publisher",
"first-page": "727101",
"issue": "8",
"journal-title": "Front. Med. Lausanne",
"key": "31944_CR28",
"unstructured": "Funakoshi, K., Morita, T. & Kumanogoh, A. Longer prehospitalization and preintubation periods in intubated non-survivors and ECMO patients with COVID-19: A systematic review and meta-analysis. Front. Med. Lausanne 15(8), 727101. https://doi.org/10.3389/fmed.2021.727101 (2021).",
"volume": "15",
"year": "2021"
},
{
"DOI": "10.1164/rccm.201409-1634OC",
"author": "RP Barbaro",
"doi-asserted-by": "publisher",
"first-page": "894",
"issue": "8",
"journal-title": "Am. J. Respir. Crit. Care Med.",
"key": "31944_CR29",
"unstructured": "Barbaro, R. P. et al. Association of hospital-level volume of extracorporeal membrane oxygenation cases and mortality. Analysis of the extracorporeal life support organization registry. Am. J. Respir. Crit. Care Med. 191(8), 894–901. https://doi.org/10.1164/rccm.201409-1634OC (2015).",
"volume": "191",
"year": "2015"
},
{
"DOI": "10.1164/rccm.201404-0630CP",
"author": "A Combes",
"doi-asserted-by": "publisher",
"first-page": "488",
"issue": "5",
"journal-title": "Am. J. Respir. Crit. Care Med.",
"key": "31944_CR30",
"unstructured": "Combes, A. et al. Position paper for the organization of extracorporeal membrane oxygenation programs for acute respiratory failure in adult patients. Am. J. Respir. Crit. Care Med. 190(5), 488–496. https://doi.org/10.1164/rccm.201404-0630CP (2014).",
"volume": "190",
"year": "2014"
},
{
"DOI": "10.1371/journal.pone.0260324",
"author": "S Bercker",
"doi-asserted-by": "publisher",
"first-page": "e0260324",
"issue": "12",
"journal-title": "PLoS ONE",
"key": "31944_CR31",
"unstructured": "Bercker, S. et al. ECMO use in Germany: An analysis of 29,929 ECMO runs. PLoS ONE 16(12), e0260324. https://doi.org/10.1371/journal.pone.0260324 (2021).",
"volume": "16",
"year": "2021"
},
{
"DOI": "10.1038/s41467-020-19741-6.PMID:33298944;PMCID:PMC7726563",
"author": "H Peckham",
"doi-asserted-by": "publisher",
"first-page": "6317",
"issue": "1",
"journal-title": "Nat. Commun.",
"key": "31944_CR32",
"unstructured": "Peckham, H. et al. Male sex identified by global COVID-19 meta-analysis as a risk factor for death and ITU admission. Nat. Commun. 11(1), 6317. https://doi.org/10.1038/s41467-020-19741-6.PMID:33298944;PMCID:PMC7726563 (2020).",
"volume": "11",
"year": "2020"
},
{
"DOI": "10.1038/s41598-021-00213-w.PMID:34675280;PMCID:PMC8531278",
"author": "J Nielsen",
"doi-asserted-by": "publisher",
"first-page": "20815",
"issue": "1",
"journal-title": "Sci. Rep.",
"key": "31944_CR33",
"unstructured": "Nielsen, J., Nørgaard, S. K., Lanzieri, G., Vestergaard, L. S. & Moelbak, K. Sex-differences in COVID-19 associated excess mortality is not exceptional for the COVID-19 pandemic. Sci. Rep. 11(1), 20815. https://doi.org/10.1038/s41598-021-00213-w.PMID:34675280;PMCID:PMC8531278 (2021).",
"volume": "11",
"year": "2021"
},
{
"DOI": "10.1007/s15010-021-01744-0",
"author": "A Niessen",
"doi-asserted-by": "publisher",
"journal-title": "Infection",
"key": "31944_CR34",
"unstructured": "Niessen, A. et al. Sex differences in COVID-19 mortality in the Netherlands. Infection https://doi.org/10.1007/s15010-021-01744-0 (2022).",
"year": "2022"
},
{
"DOI": "10.1164/rccm.202005-1583OC",
"author": "OJ McElvaney",
"doi-asserted-by": "publisher",
"first-page": "812",
"journal-title": "Am. J. Respir. Crit. Care Med.",
"key": "31944_CR35",
"unstructured": "McElvaney, O. J. et al. Characterization of the inflammatory response to severe COVID-19 illness. Am. J. Respir. Crit. Care Med. 202, 812–821 (2020).",
"volume": "202",
"year": "2020"
},
{
"DOI": "10.1016/S0140-6736(20)30628-0",
"author": "P Mehta",
"doi-asserted-by": "publisher",
"first-page": "1033",
"journal-title": "Lancet",
"key": "31944_CR36",
"unstructured": "Mehta, P. et al. COVID-19: Consider cytokine storm syndromes and immunosuppression. Lancet 395, 1033–1034 (2020).",
"volume": "395",
"year": "2020"
},
{
"DOI": "10.1016/S2213-2600(21)00177-6",
"doi-asserted-by": "publisher",
"key": "31944_CR37",
"unstructured": "Supady, A., Weber, E., Rieder, M., Lother, A., Niklaus, T., Zahn, T., Frech, F., Müller, S., Kuhl, M., Benk, C., Maier, S., Trummer, G., Flügler, A., Krüger, K., Sekandarzad, A., Stachon, P., Zotzmann, V., Bode, C., Biever, P. M., Staudacher, D., Wengenmayer, T., Graf, E., Duerschmied D. Cytokine adsorption in patients with severe COVID-19 pneumonia requiring extracorporeal membrane oxygenation (CYCOV): A single centre, open-label, randomised, controlled trial. Lancet Respir. Med. 9(7), 755–762 (2021). https://doi.org/10.1016/S2213-2600(21)00177-6. Erratum in: Lancet Respir. Med. (2021)."
},
{
"DOI": "10.1001/jamanetworkopen.2021.33090",
"author": "T Oskotsky",
"doi-asserted-by": "publisher",
"first-page": "e2133090",
"issue": "11",
"journal-title": "JAMA Netw. Open",
"key": "31944_CR38",
"unstructured": "Oskotsky, T. et al. Mortality risk among patients with COVID-19 prescribed selective serotonin reuptake inhibitor antidepressants. JAMA Netw. Open 4(11), e2133090. https://doi.org/10.1001/jamanetworkopen.2021.33090 (2021).",
"volume": "4",
"year": "2021"
},
{
"DOI": "10.1038/s41380-021-01021-4",
"author": "N Hoertel",
"doi-asserted-by": "publisher",
"first-page": "5199",
"issue": "9",
"journal-title": "Mol. Psychiatry",
"key": "31944_CR39",
"unstructured": "Hoertel, N. et al. Association between antidepressant use and reduced risk of intubation or death in hospitalized patients with COVID-19: results from an observational study. Mol. Psychiatry 26(9), 5199–5212. https://doi.org/10.1038/s41380-021-01021-4 (2021).",
"volume": "26",
"year": "2021"
},
{
"DOI": "10.3389/fphar.2021.652688",
"author": "VP Sukhatme",
"doi-asserted-by": "publisher",
"first-page": "652688",
"issue": "12",
"journal-title": "Front. Pharmacol.",
"key": "31944_CR40",
"unstructured": "Sukhatme, V. P., Reiersen, A. M., Vayttaden, S. J. & Sukhatme, V. V. Fluvoxamine: A review of its mechanism of action and its role in COVID-19. Front. Pharmacol. 20(12), 652688. https://doi.org/10.3389/fphar.2021.652688 (2021).",
"volume": "20",
"year": "2021"
},
{
"DOI": "10.1016/S2214-109X(21)00448-4",
"doi-asserted-by": "publisher",
"key": "31944_CR41",
"unstructured": "Reis, G., Dos Santos Moreira-Silva, E. A., Silva, D. C. M., Thabane, L., Milagres, A. C., Ferreira, T. S., Dos Santos, C. V. Q., de Souza Campos, V. H., Nogueira, A. M. R., de Almeida, A. P. F. G., Callegari, E. D., de Figueiredo Neto, A. D., Savassi, L. C. M., Simplicio, M. I. C., Ribeiro, L. B., Oliveira, R., Harari, O., Forrest, J. I., Ruton, H., Sprague, S., McKay, P., Glushchenko, A. V., Rayner, C. R., Lenze, E. J., Reiersen, A. M., Guyatt, G. H., Mills, E. J., TOGETHER investigators. Effect of early treatment with fluvoxamine on risk of emergency care and hospitalisation among patients with COVID-19: The TOGETHER randomised, platform clinical trial. Lancet Glob. Health 10(1), e42–e51 (2022). https://doi.org/10.1016/S2214-109X(21)00448-4. Erratum in: Lancet Glob. Health. 10(4), e481 (2022)"
},
{
"DOI": "10.1186/s12941-020-00362-2",
"author": "W Chen",
"doi-asserted-by": "publisher",
"first-page": "18",
"issue": "1",
"journal-title": "Ann. Clin. Microbiol. Antimicrob.",
"key": "31944_CR42",
"unstructured": "Chen, W. et al. Plasma CRP level is positively associated with the severity of COVID-19. Ann. Clin. Microbiol. Antimicrob. 19(1), 18. https://doi.org/10.1186/s12941-020-00362-2 (2020).",
"volume": "19",
"year": "2020"
},
{
"DOI": "10.1055/s-0040-1721317",
"author": "L Valerio",
"doi-asserted-by": "publisher",
"first-page": "98",
"issue": "1",
"journal-title": "Thromb. Haemost.",
"key": "31944_CR43",
"unstructured": "Valerio, L. et al. Lodigiani C course of D-dimer and C-reactive protein levels in survivors and nonsurvivors with COVID-19 pneumonia: A retrospective analysis of 577 patients. Thromb. Haemost. 121(1), 98–101. https://doi.org/10.1055/s-0040-1721317 (2021).",
"volume": "121",
"year": "2021"
},
{
"DOI": "10.1177/20503121211050755.PMID:34659766;PMCID:PMC8516378",
"author": "GY Yitbarek",
"doi-asserted-by": "publisher",
"first-page": "205031212110507",
"issue": "9",
"journal-title": "SAGE Open Med.",
"key": "31944_CR44",
"unstructured": "Yitbarek, G. Y. et al. The role of C-reactive protein in predicting the severity of COVID-19 disease: A systematic review. SAGE Open Med. 11(9), 20503121211050756. https://doi.org/10.1177/20503121211050755.PMID:34659766;PMCID:PMC8516378 (2021).",
"volume": "11",
"year": "2021"
},
{
"DOI": "10.3389/fmed.2020.598870",
"author": "Z Liu",
"doi-asserted-by": "publisher",
"first-page": "598870",
"issue": "7",
"journal-title": "Front. Med. Lausanne",
"key": "31944_CR45",
"unstructured": "Liu, Z. et al. Bilirubin levels as potential indicators of disease severity in coronavirus disease patients: A retrospective cohort study. Front. Med. Lausanne 9(7), 598870. https://doi.org/10.3389/fmed.2020.598870 (2020).",
"volume": "9",
"year": "2020"
},
{
"DOI": "10.1111/liv.14477",
"author": "P Paliogiannis",
"doi-asserted-by": "publisher",
"first-page": "1787",
"issue": "7",
"journal-title": "Liver Int.",
"key": "31944_CR46",
"unstructured": "Paliogiannis, P. & Zinellu, A. Bilirubin levels in patients with mild and severe Covid-19: A pooled analysis. Liver Int. 40(7), 1787–1788. https://doi.org/10.1111/liv.14477 (2020).",
"volume": "40",
"year": "2020"
},
{
"DOI": "10.1186/cc11381",
"author": "N Nesseler",
"doi-asserted-by": "publisher",
"first-page": "235",
"issue": "5",
"journal-title": "Crit. Care.",
"key": "31944_CR47",
"unstructured": "Nesseler, N. et al. Clinical review: The liver in sepsis. Crit. Care. 16(5), 235. https://doi.org/10.1186/cc11381 (2012).",
"volume": "16",
"year": "2012"
},
{
"DOI": "10.1016/S2213-2600(20)30119-3",
"author": "BM Henry",
"doi-asserted-by": "publisher",
"first-page": "e24",
"issue": "4",
"journal-title": "Lancet Respir. Med.",
"key": "31944_CR48",
"unstructured": "Henry, B. M. COVID-19, ECMO, and lymphopenia: A word of caution. Lancet Respir. Med. 8(4), e24. https://doi.org/10.1016/S2213-2600(20)30119-3 (2020).",
"volume": "8",
"year": "2020"
},
{
"DOI": "10.1097/PCC.0b013e3181e28894",
"author": "MJ Bizzarro",
"doi-asserted-by": "publisher",
"first-page": "277",
"issue": "3",
"journal-title": "Pediatr. Crit. Care Med.",
"key": "31944_CR49",
"unstructured": "Bizzarro, M. J. et al. Infections acquired during extracorporeal membrane oxygenation in neonates, children, and adults. Pediatr. Crit. Care Med. 12(3), 277–281. https://doi.org/10.1097/PCC.0b013e3181e28894 (2011).",
"volume": "12",
"year": "2011"
},
{
"DOI": "10.1111/bjh.17007",
"author": "AJ Doyle",
"doi-asserted-by": "publisher",
"first-page": "382",
"issue": "3",
"journal-title": "Br. J. Haematol.",
"key": "31944_CR50",
"unstructured": "Doyle, A. J. et al. Blood component use in critical care in patients with COVID-19 infection: A single-centre experience. Br. J. Haematol. 191(3), 382–385. https://doi.org/10.1111/bjh.17007 (2020).",
"volume": "191",
"year": "2020"
}
],
"reference-count": 50,
"references-count": 50,
"relation": {},
"resource": {
"primary": {
"URL": "https://www.nature.com/articles/s41598-023-31944-7"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [
"Multidisciplinary"
],
"subtitle": [],
"title": "Mortality rates of severe COVID-19-related respiratory failure with and without extracorporeal membrane oxygenation in the Middle Ruhr Region of Germany",
"type": "journal-article",
"update-policy": "http://dx.doi.org/10.1007/springer_crossmark_policy",
"volume": "13"
}
aweimer