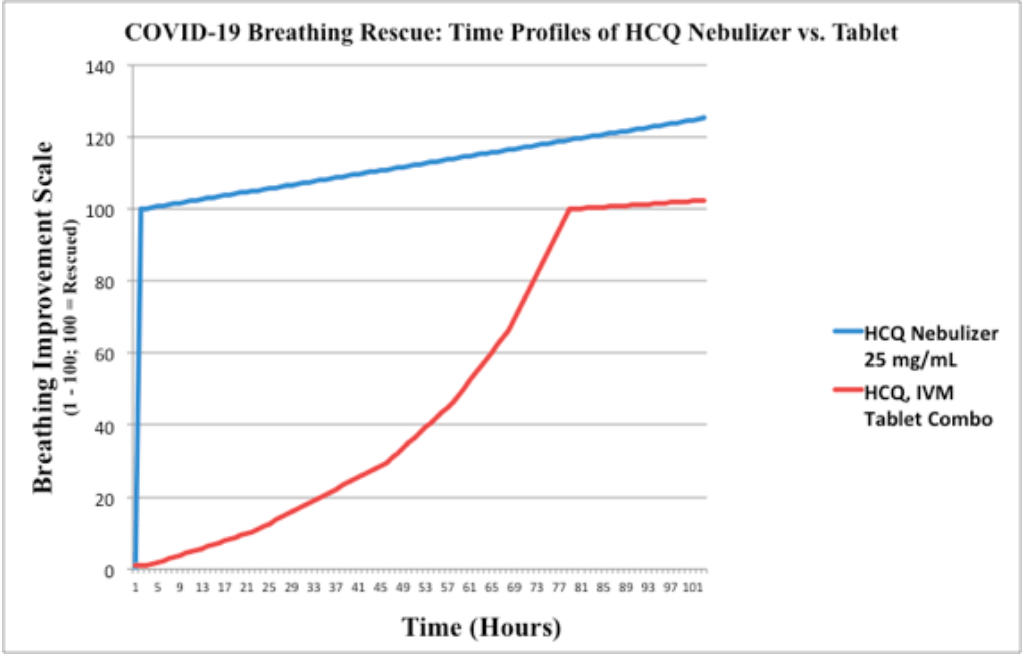
Nebulized Hydroxychloroquine for COVID-19 Treatment: 80x Improvement in Breathing
, Z., Preprint, Jan 2021
HCQ for COVID-19
1st treatment shown to reduce risk in
March 2020, now with p < 0.00000000001 from 424 studies, used in 59 countries.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
Report on the use of nebulized HCQ showing much more rapid improvement compared to tablets, with 95% of patients experiencing improved breathing within 1 hour. Author notes that the effectiveness of HCQ is time and dose dependent, with a primary issue being the time it takes for a therapeutic dosage to reach the lungs, and that treatment with tablets may take an average of 80 hours to achieve significant clinical improvement.
Author notes that it may take 3-7 days to achieve optimal alveolar concentrations with tablets, whereas nebulized HCQ administered as microdroplets directly to the lungs achieves optimal alveolar concentration in approximately one hour and is associated with faster clinical improvement, reduction in pulmonary complications, and a reduction in medical costs.
1.
Alsmadi et al., The In Vitro, In Vivo, and PBPK Evaluation of a Novel Lung-Targeted Cardiac-Safe Hydroxychloroquine Inhalation Aerogel, AAPS PharmSciTech, doi:10.1208/s12249-023-02627-3.
2.
Faísca et al., Enhanced In Vitro Antiviral Activity of Hydroxychloroquine Ionic Liquids against SARS-CoV-2, Pharmaceutics, doi:10.3390/pharmaceutics14040877.
3.
Zelenko, Z., Nebulized Hydroxychloroquine for COVID-19 Treatment: 80x Improvement in Breathing, Preprint, faculty.utrgv.edu/eleftherios.gkioulekas/zelenko/Zelenko-nebulized-hcq.pdf.
Zelenko et al., 24 Jan 2021, preprint, 1 author.
