Regeneron's REGN-COV2 antibody cocktail reduced viral levels and improved symptoms in non-hospitalized COVID-19 patients
, Press Release, Sep 2020
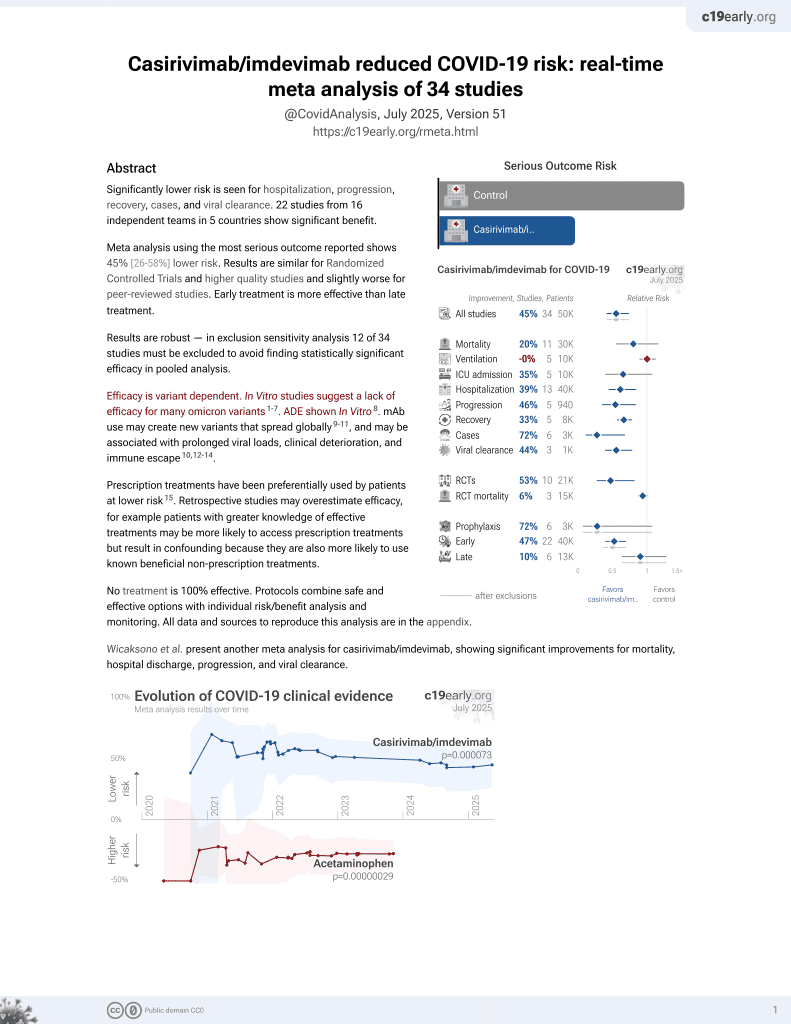
18th treatment shown to reduce risk in
March 2021, now with p = 0.000095 from 34 studies, recognized in 52 countries.
Efficacy is variant dependent.
No treatment is 100% effective. Protocols
combine treatments.
6,500+ studies for
210+ treatments. c19early.org
|
Analysis of the first 275 patients in a trial of the REGN-COV2 antibody cocktail showing reductions in viral load and the time to alleviate symptoms in non-hospitalized patients with COVID-19. Greatest improvements were seen with patients that had not mounted their own effective immune response prior to treatment.
The mean time-weighted-average change from baseline nasopharyngeal viral load through Day 7 in the seronegative (no measurable antiviral antibodies) group was a 0.60 log10 copies/mL greater reduction (p=0.03) in patients treated with high dose, and a 0.51 log10 copies/mL greater reduction (p=0.06) in patients treated with low dose, compared to placebo. In the overall population, there was a 0.51 log10 copies/mL greater reduction (p=0.0049) in patients treated with high dose, and a 0.23 log10 copies/mL greater reduction (p=0.20) in patients treated with low dose, compared to placebo.
Among seronegative patients, median time to symptom alleviation (defined as symptoms becoming mild or absent) was 13 days in placebo, 8 days in high dose (p=0.22), and 6 days in low dose (p=0.09).
Adverse reactions were similar with treatment and placebo. There were no deaths.
Efficacy is variant dependent. In Vitro research suggests a lack of efficacy for many omicron variants1-7.
Standard of Care (SOC) for COVID-19 in the study country,
the USA, is very poor with very low average efficacy for approved treatments8.
Only expensive, high-profit treatments were approved for early treatment. Low-cost treatments were excluded, reducing the probability of early treatment due to access and cost barriers, and eliminating complementary and synergistic benefits seen with many low-cost treatments.
|
recovery time, 38.0% lower, relative time 0.62, p = 0.22, treatment 92, control 91, high dose median time to recovery, group sizes estimated because they were not supplied.
|
|
recovery time, 54.0% lower, relative time 0.46, p = 0.09, treatment 92, control 91, low dose median time to recovery, group sizes estimated because they were not supplied.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
1.
Liu et al., Striking Antibody Evasion Manifested by the Omicron Variant of SARS-CoV-2, bioRxiv, doi:10.1101/2021.12.14.472719.
2.
Sheward et al., Variable loss of antibody potency against SARS-CoV-2 B.1.1.529 (Omicron), bioRxiv, doi:10.1101/2021.12.19.473354.
3.
VanBlargan et al., An infectious SARS-CoV-2 B.1.1.529 Omicron virus escapes neutralization by several therapeutic monoclonal antibodies, bioRxiv, doi:10.1101/2021.12.15.472828.
4.
Tatham et al., Lack of Ronapreve (REGN-CoV; casirivimab and imdevimab) virological efficacy against the SARS-CoV 2 Omicron variant (B.1.1.529) in K18-hACE2 mice, bioRxiv, doi:10.1101/2022.01.23.477397.
5.
Pochtovyi et al., In Vitro Efficacy of Antivirals and Monoclonal Antibodies against SARS-CoV-2 Omicron Lineages XBB.1.9.1, XBB.1.9.3, XBB.1.5, XBB.1.16, XBB.2.4, BQ.1.1.45, CH.1.1, and CL.1, Vaccines, doi:10.3390/vaccines11101533.
6.
Haars et al., Prevalence of SARS-CoV-2 Omicron Sublineages and Spike Protein Mutations Conferring Resistance against Monoclonal Antibodies in a Swedish Cohort during 2022–2023, Microorganisms, doi:10.3390/microorganisms11102417.
Regeneron et al., 29 Sep 2020, Randomized Controlled Trial, USA, preprint, 1 author.