Placebo-Controlled, Double-Blind Trial of Intravenous Ribavirin for the Treatment of Hantavirus Cardiopulmonary Syndrome in North America
et al., Clinical Infectious Diseases, doi:10.1086/425007, Nov 2004
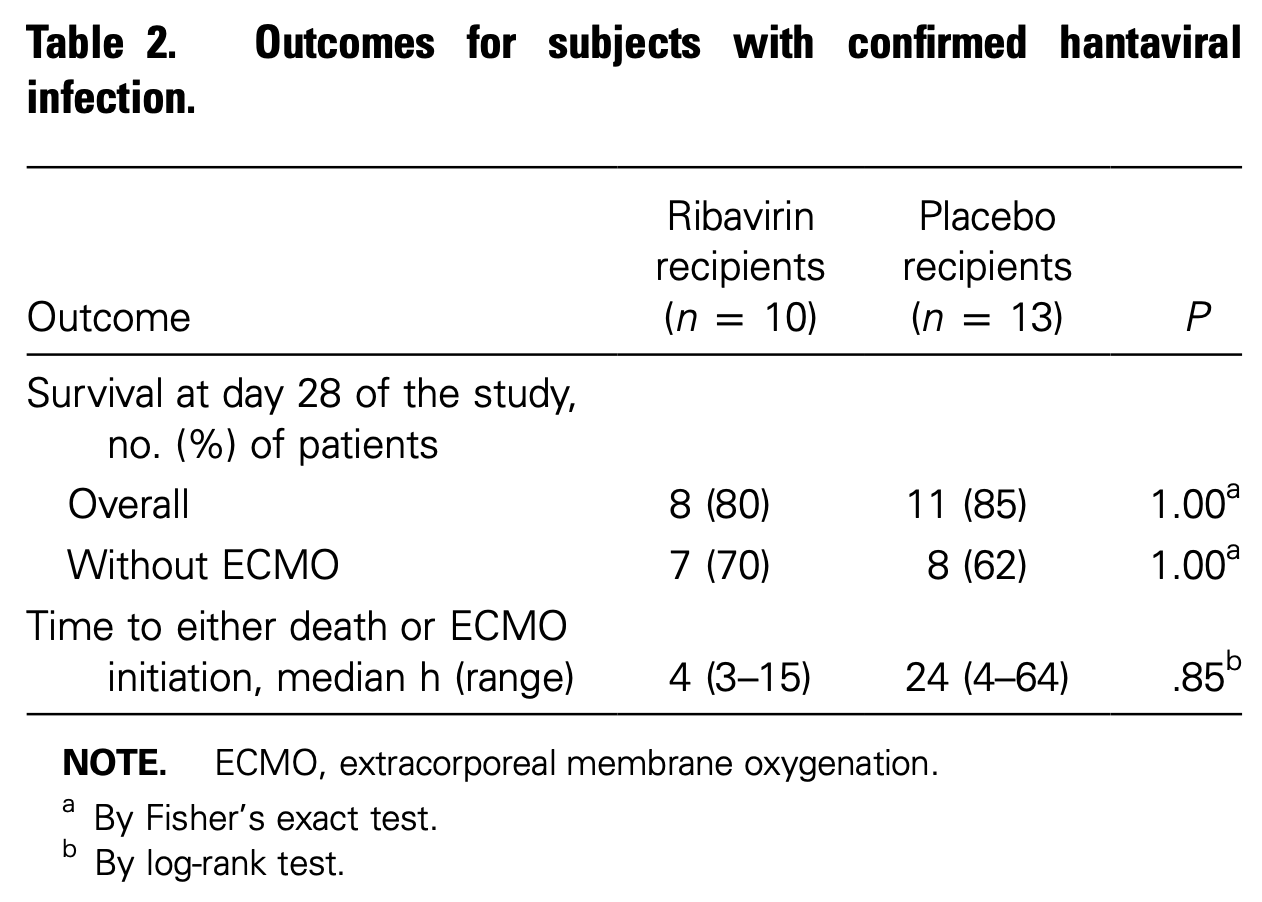
RCT 36 patients (23 with confirmed hantavirus cardiopulmonary syndrome, HCPS) showing no significant differences with intravenous ribavirin treatment. Mortality and the proportion of patients surviving without extracorporeal membrane oxygenation was similar between groups. Two ribavirin recipients and two placebo recipients died.
All patients were enrolled late in the cardiopulmonary phase. Major limitations include the very small sample size (far below the target of 130), inability to enroll any patients during the prodrome phase, and the rapid disease progression once the cardiopulmonary phase began (median time to death or ECMO initiation was only 4 hours after drug initiation in ribavirin recipients), which may have precluded any therapeutic benefit.
Authors hypothesize that ribavirin is likely ineffective in the cardiopulmonary stage due to the rapid rate of disease progression compared to hemorrhagic fever with renal syndrome, where ribavirin showed benefit.
|
risk of death, 30.0% higher, RR 1.30, p = 1.00, treatment 2 of 10 (20.0%), control 2 of 13 (15.4%).
|
|
ECMO/death, 22.0% lower, RR 0.78, p = 1.00, treatment 3 of 10 (30.0%), control 5 of 13 (38.5%), NNT 12.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Mertz et al., 1 Nov 2004, Double Blind Randomized Controlled Trial, placebo-controlled, Mexico, peer-reviewed, 15 authors, HCPS, Sin Nombre.
Contact: gmertz@salud.unm.edu.
Ribavirin is a broad-spectrum, small-molecule nucleoside analogue antiviral that inhibits hantavirus replication by depleting intracellular GTP pools via the inhibition of host inosine monophosphate dehydrogenase and by interfering directly with the viral RNA-dependent RNA polymerase.
Abstract: ## Placebo-Controlled, Double-Blind T rial of Intravenous Ribavirin for the T reatment of Hantavirus Cardiopulmonary Syndrome in North America
Gregory J. Mertz, 1 Lil Miedzinski, 6 Diane Goade, 1 Andrew T. Pavia, 2 Brian Hjelle, 1 Christine O. Hansbarger, 1 Howard Levy, 1,a Frederick T. Koster, 1 Kenneth Baum, 3 Adeline Lindemulder, 6 Wenquan Wang, 4 Laura Riser, 4 Humberto Fernandez, 5 and Richard J. Whitley, 4 for the Collaborative Antiviral Study Group
1 University of New Mexico, Albuquerque; 2 University of Utah, Salt Lake City; 3 University of Colorado, Denver; 4 University of Alabama at Birmingham; 5 ICN Pharmaceuticals, Costa Mesa, California; and 6 University of Alberta, Edmonton, Alberta, Canada
Background. Ribavirin is active in vitro against hantaviruses, but the findings of an open trial of the use of intravenous ribavirin for the treatment of hantavirus cardiopulmonary syndrome (HCPS) were inconclusive.
Methods. Subjects with suspected HCPS in the prodrome or cardiopulmonary phase but without shock were eligible for randomization to receive either intravenous ribavirin (33 mg/kg [ ⩽ 2 g], followed by 16 mg/kg [ ⩽ 1 g] given every 6 h for 4 days and by 8 mg/kg [ ⩽ .5 g] given every 8 h for 3 days) or placebo (administered for 7 days or until the initial Sin Nombre virus antibody test result was confirmed to be negative). The primary outcome was survival at day 28 of the study without the need for extracorporeal membrane oxygenation (ECMO).
Results. Thirty-six subjects were enrolled in the trial from March 1996 through July 2001, at which point the study was terminated prematurely because of both the slow rate of accrual of subjects and the findings of a futility analysis. Of the 36 subjects enrolled, 23 (all of whom were enrolled during the cardiopulmonary stage of HCPS) had HCPS confirmed by serologic testing. The severity of illness at entry into the study was similar among the 10 subjects with HCPS who received ribavirin and the 13 subjects with HCPS who received placebo. The proportion of subjects who survived and who did not require ECMO was similar among ribavirin recipients and placebo recipients (70% vs. 62%, respectively); 2 ribavirin recipients and 2 placebo recipients died, including 3 of 7 subjects treated with ECMO. The frequency of adverse events, including anemia, was similar between treatment groups.
Conclusions. The rate of accrual of subjects in the present study was inadequate to clearly assess the safety or efficacy of ribavirin in the treatment of HCPS. However, ribavirin was well tolerated, and the lack of trends supporting the use of intravenous ribavirin suggests that it is probably ineffective in the treatment of HCPS in the cardiopulmonary stage.
Since the recognition of hantavirus cardiopulmonary syndrome (HCPS) in 1993, HCPS has been identified throughout most of North and South America and in Panama. Known also as 'hantavirus pulmonary syn-
Received 23 April 2004; accepted 16 June 2004; electronically published 11 October 2004.
a Present affiliation: Now also affiliated with Acute Care-US Medical Division, Eli Lilly, Indianapolis, Indiana.
Investigators and institutions who participated in the study are listed at the end of the text.
Reprints or correspondence: Dr. Gregory J. Mertz, Internal Medicine, MSC10 5550, 1 University of New Mexico, Albuquerque, New Mexico 87131-0001 (gmertz@salud.unm.edu).
drome' (HPS), we prefer the term 'hantavirus cardiopulmonary syndrome' because almost..
DOI record:
{
"DOI": "10.1086/425007",
"ISSN": [
"1058-4838",
"1537-6591"
],
"URL": "http://dx.doi.org/10.1086/425007",
"author": [
{
"affiliation": [],
"family": "Mertz",
"given": "G. J.",
"sequence": "first"
},
{
"affiliation": [],
"family": "Miedzinski",
"given": "L.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Goade",
"given": "D.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Pavia",
"given": "A. T.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Hjelle",
"given": "B.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Hansbarger",
"given": "C. O.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Levy",
"given": "H.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Koster",
"given": "F. T.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Baum",
"given": "K.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Lindemulder",
"given": "A.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Wang",
"given": "W.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Riser",
"given": "L.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Fernandez",
"given": "H.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Whitley",
"given": "R. J.",
"sequence": "additional"
},
{
"affiliation": [],
"name": "Collaborative Antiviral Study Group",
"sequence": "additional"
}
],
"container-title": "Clinical Infectious Diseases",
"container-title-short": "Clinical Infectious Diseases",
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2004,
10,
15
]
],
"date-time": "2004-10-15T20:51:13Z",
"timestamp": 1097873473000
},
"deposited": {
"date-parts": [
[
2023,
4,
29
]
],
"date-time": "2023-04-29T15:43:36Z",
"timestamp": 1682783016000
},
"indexed": {
"date-parts": [
[
2026,
1,
9
]
],
"date-time": "2026-01-09T14:45:35Z",
"timestamp": 1767969935593,
"version": "3.49.0"
},
"is-referenced-by-count": 144,
"issue": "9",
"issued": {
"date-parts": [
[
2004,
11,
1
]
]
},
"journal-issue": {
"issue": "9",
"published-print": {
"date-parts": [
[
2004,
11,
1
]
]
}
},
"language": "en",
"link": [
{
"URL": "http://academic.oup.com/cid/article-pdf/39/9/1307/5864268/39-9-1307.pdf",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "286",
"original-title": [],
"page": "1307-1313",
"prefix": "10.1093",
"published": {
"date-parts": [
[
2004,
11,
1
]
]
},
"published-print": {
"date-parts": [
[
2004,
11,
1
]
]
},
"publisher": "Oxford University Press (OUP)",
"reference": [
{
"DOI": "10.1097/00003246-199602000-00012",
"ISSN": "https://id.crossref.org/issn/0090-3493",
"author": "Hallin",
"doi-asserted-by": "publisher",
"first-page": "252",
"issn-type": "print",
"issue": "2",
"journal-title": "Critical care medicine",
"key": "2_16197130",
"volume": "24",
"year": "1996"
},
{
"DOI": "10.1093/infdis/164.6.1119",
"ISSN": "https://id.crossref.org/issn/0022-1899",
"doi-asserted-by": "publisher",
"first-page": "1119",
"issn-type": "print",
"issue": "6",
"journal-title": "Journal of Infectious Diseases",
"key": "3_38726651",
"volume": "164",
"year": "1991"
},
{
"first-page": "229",
"journal-title": "ANTIVIRAL CHEMOTHERAPY NEW DIRECTIONS FOR CLINICAL APPLICATION AND RESEARCH",
"key": "4_38270148",
"volume": "3",
"year": "1993"
},
{
"DOI": "10.1177/135965359900400404",
"ISSN": "https://id.crossref.org/issn/1359-6535",
"author": "Chapman",
"doi-asserted-by": "crossref",
"first-page": "211",
"issn-type": "print",
"issue": "4",
"journal-title": "Antiviral therapy",
"key": "5_6516376",
"volume": "4",
"year": "1999"
},
{
"DOI": "10.1086/315467",
"ISSN": "https://id.crossref.org/issn/0022-1899",
"doi-asserted-by": "publisher",
"first-page": "1569",
"issn-type": "print",
"issue": "5",
"journal-title": "Journal of Infectious Diseases",
"key": "6_38579526",
"volume": "181",
"year": "2000"
},
{
"DOI": "10.1086/315657",
"ISSN": "https://id.crossref.org/issn/0022-1899",
"doi-asserted-by": "publisher",
"first-page": "43",
"issn-type": "print",
"issue": "1",
"journal-title": "Journal of Infectious Diseases",
"key": "8_38580385",
"volume": "182",
"year": "2000"
},
{
"DOI": "10.1128/JVI.77.18.9894-9905.2003",
"ISSN": "https://id.crossref.org/issn/0022-538X",
"author": "Custer",
"doi-asserted-by": "publisher",
"first-page": "9894",
"issn-type": "print",
"issue": "18",
"journal-title": "Journal of Virology",
"key": "9_17786012",
"volume": "77",
"year": "2003"
}
],
"reference-count": 7,
"references-count": 7,
"relation": {},
"resource": {
"primary": {
"URL": "https://academic.oup.com/cid/article-lookup/doi/10.1086/425007"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "Placebo-Controlled, Double-Blind Trial of Intravenous Ribavirin for the Treatment of Hantavirus Cardiopulmonary Syndrome in North America",
"type": "journal-article",
"volume": "39"
}