Ribavirin is a broad-spectrum, small-molecule nucleoside analogue antiviral that inhibits hantavirus replication by depleting intracellular GTP pools via the inhibition of host inosine monophosphate dehydrogenase and by interfering directly with the viral RNA-dependent RNA polymerase.
Mar 3 2017 |
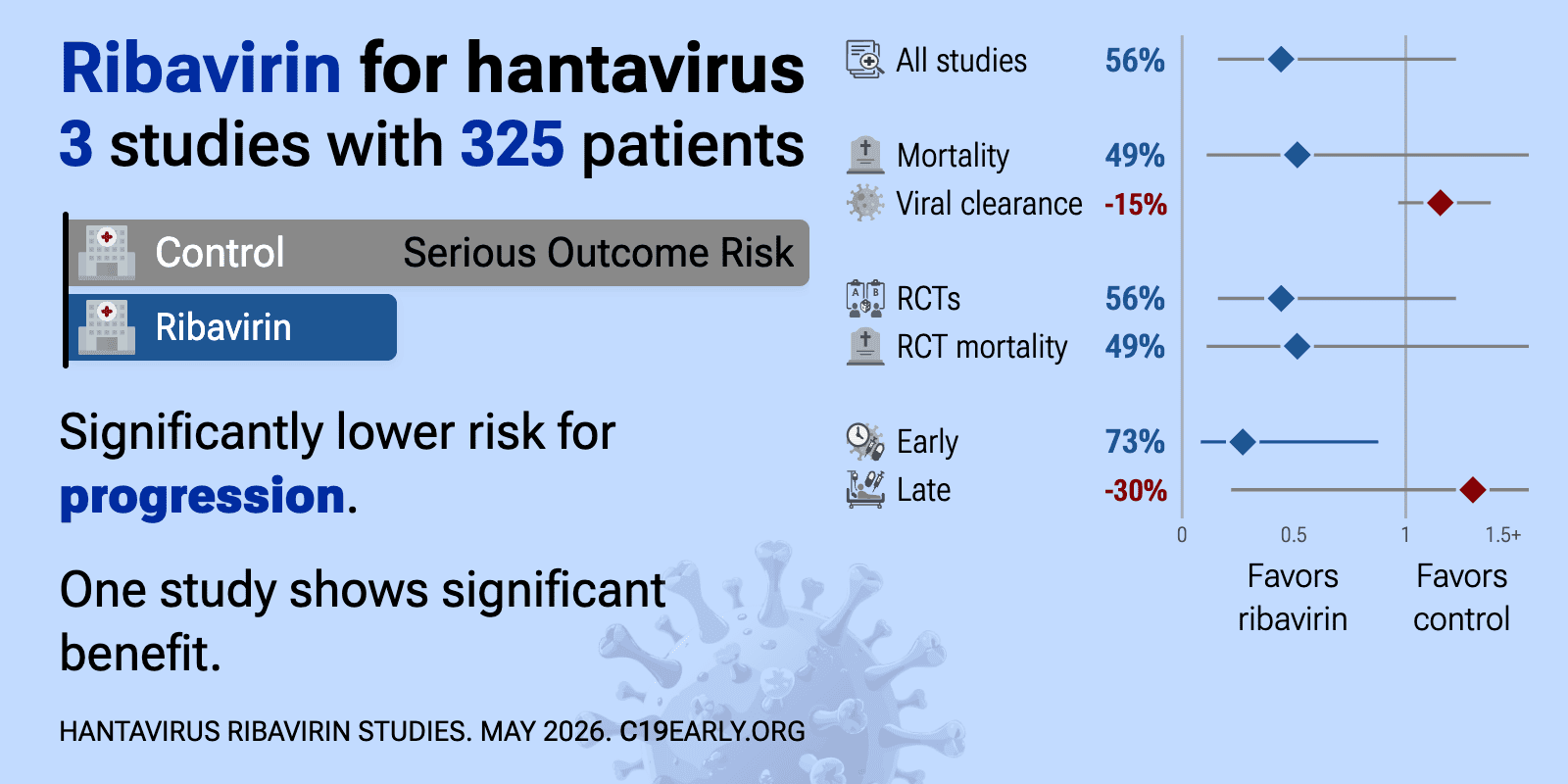
et al., Infectious Diseases, doi:10.1080/23744235.2017.1293841 | Insufficient efficacy and safety of intravenous ribavirin in treatment of haemorrhagic fever with renal syndrome caused by Puumala virus |
| 15% slower viral clearance (p=0.11). RCT 73 hospitalized patients showing no significant differences with intravenous ribavirin for treatment of haemorrhagic fever with renal syndrome (HFRS) caused by Puumala virus (PUUV). Puumala virus is relatively mild compared to other h.. | ||
Jun 4 2014 |
et al., VirusDisease, doi:10.1007/s13337-014-0219-7 | Effectiveness of the ribavirin in treatment of hantavirus infections in the Americas and Eurasia: a meta-analysis |
| Meta-analysis of 7 ribavirin studies (506 patients/animals) showing lower mortality for hemorrhagic fever with renal syndrome (HFRS), but no significant mortality benefit for hantavirus pulmonary syndrome (HPS) in humans. Benefit was seen.. | ||
Jan 31 2009 |
et al., Antiviral Research, doi:10.1016/j.antiviral.2008.09.007 | Experience with intravenous ribavirin in the treatment of hemorrhagic fever with renal syndrome in Korea |
| Retrospective 34 hospitalized patients with hemorrhagic fever with renal syndrome (HFRS) acquired in Korea, showing potential benefit with intravenous ribavirin treatment, with only 3% oliguria and 0% dialysis requirement compared to 39-6.. | ||
Nov 1 2004 |
et al., Clinical Infectious Diseases, doi:10.1086/425007 | Placebo-Controlled, Double-Blind Trial of Intravenous Ribavirin for the Treatment of Hantavirus Cardiopulmonary Syndrome in North America |
| 30% higher mortality (p=1) and 22% lower progression (p=1). RCT 36 patients (23 with confirmed hantavirus cardiopulmonary syndrome, HCPS) showing no significant differences with intravenous ribavirin treatment. Mortality and the proportion of patients surviving without extracorporeal membrane oxyg.. | ||
May 1 1999 |
et al., Antiviral Therapy, doi:10.1177/135965359900400404 | Intravenous Ribavirin for Hantavirus Pulmonary Syndrome: Safety and Tolerance during 1 Year of Open-Label Experience |
| 7% lower mortality (p=0.81). Observational study of 140 patients (30 with confirmed HPS) showing no apparent benefit with intravenous ribavirin for hantavirus pulmonary syndrome (HPS). All of the confirmed HCPS patients were enrolled late during the cardiopulmonary p.. | ||
Dec 1 1991 |
et al., Journal of Infectious Diseases, doi:10.1093/infdis/164.6.1119 | Prospective, Double-Blind, Concurrent, Placebo-Controlled Clinical Trial of Intravenous Ribavirin Therapy of Hemorrhagic Fever with Renal Syndrome |
| 74% lower mortality (p=0.04) and 73% lower progression (p=0.01). RCT 242 hospitalized patients with hemorrhagic fever with renal syndrome (HFRS) from Hantaan virus (the first hantavirus isolated) showing significantly lower mortality with intravenous ribavirin treatment. Ribavirin-treated patients had .. | ||