Prospective, Double-Blind, Concurrent, Placebo-Controlled Clinical Trial of Intravenous Ribavirin Therapy of Hemorrhagic Fever with Renal Syndrome
et al., Journal of Infectious Diseases, doi:10.1093/infdis/164.6.1119, Dec 1991
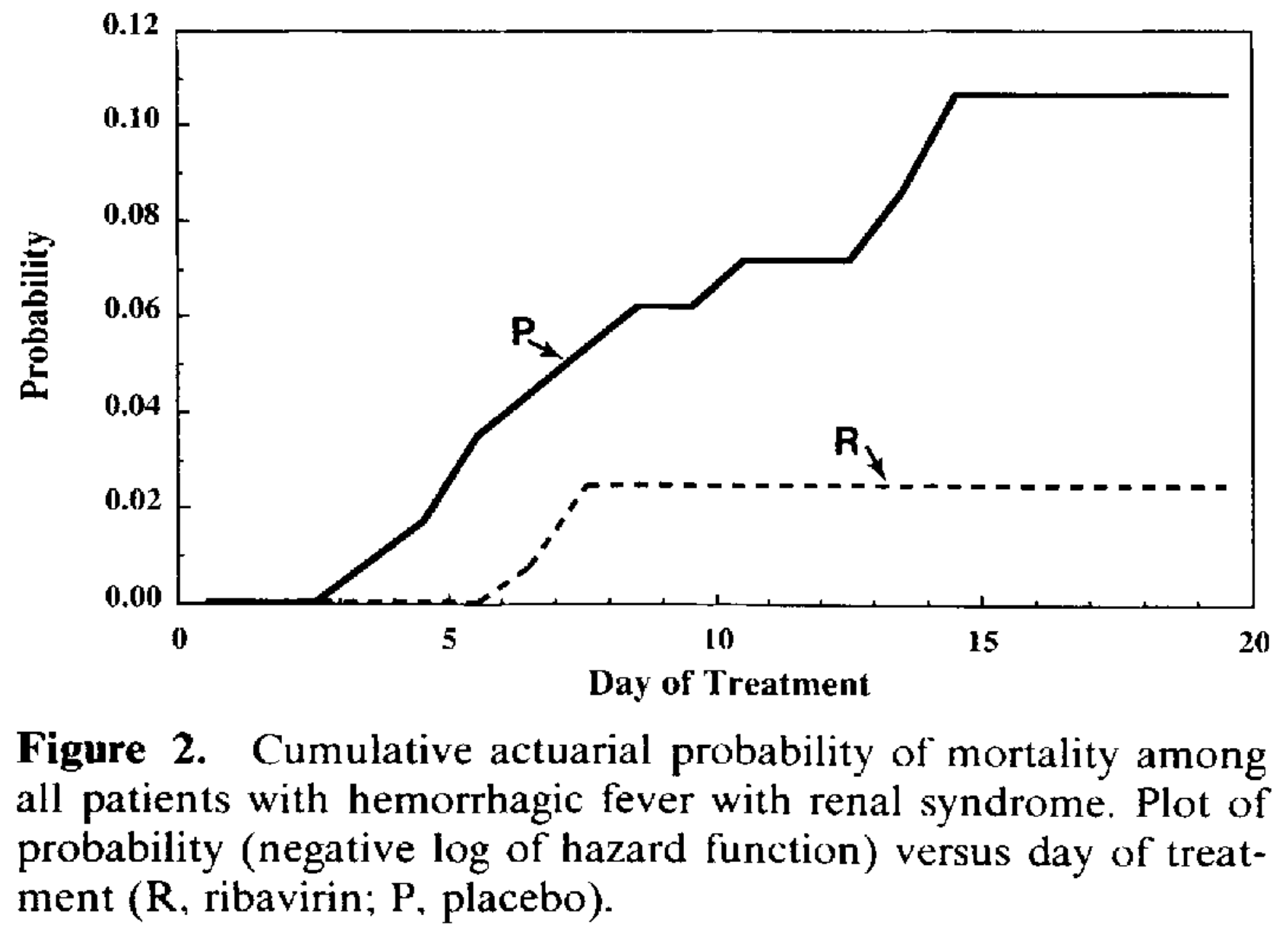
RCT 242 hospitalized patients with hemorrhagic fever with renal syndrome (HFRS) from Hantaan virus (the first hantavirus isolated) showing significantly lower mortality with intravenous ribavirin treatment. Ribavirin-treated patients had a sevenfold lower risk of death compared to placebo when adjusted for baseline variables. A notable adverse effect was a reversible hemolytic anemia during treatment, with significantly more ribavirin-treated patients reaching hematocrit below 30%, though no patients required withdrawal from the protocol. The study was conducted over two seasons in Hubei Province, China, with some baseline imbalances between treatment groups noted across the two seasons. Authors hypothesize the primary mechanism of benefit is reduction of viral replication, thereby preventing oliguria and subsequent organ damage.
The mean duration of fever at the time of enrollment was 3.7 days during the 1985-1986 season, and 4.3 days during the 1986-1987 season. About 90% of patients were enrolled prior to the oliguric phase.
Authors noted that early treatment - defined as on or before day 4 of illness - was more effective.
Using the unadjusted raw counts to estimate mortality may be more reliable - the adjusted result relies on an unstable logistic regression model driven by only 13 total deaths, resulting in a very wide confidence interval that makes the exact magnitude of the reduction statistically fragile. The adjusted model used baseline Total Serum Protein and AST. Researchers used a mean-imputation strategy for missing baseline data, therefore the specific covariate values fed into this already fragile model may have been artificially smoothed out.
All three ribavirin-treated patients who died were oliguric on presentation.
|
risk of death, 73.7% lower, RR 0.26, p = 0.04, treatment 3 of 122 (2.5%), control 10 of 107 (9.3%), NNT 15, p-value adjusted for treatment site and stratification by seasons.
|
|
risk of death, 73.7% lower, RR 0.26, p = 0.04, treatment 3 of 122 (2.5%), control 10 of 107 (9.3%), NNT 15, unadjusted.
|
|
risk of death, 84.5% lower, RR 0.16, p = 0.03, treatment 3 of 122 (2.5%), control 10 of 107 (9.3%), adjusted per study, inverted to make RR<1 favor treatment, odds ratio converted to relative risk.
|
|
oliguric phase, 73.0% lower, OR 0.27, p = 0.01, treatment 122, control 107, inverted to make OR<1 favor treatment, RR approximated with OR.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Huggins et al., 1 Dec 1991, Double Blind Randomized Controlled Trial, placebo-controlled, China, peer-reviewed, 15 authors, HFRS, Hantaan.
Ribavirin is a broad-spectrum, small-molecule nucleoside analogue antiviral that inhibits hantavirus replication by depleting intracellular GTP pools via the inhibition of host inosine monophosphate dehydrogenase and by interfering directly with the viral RNA-dependent RNA polymerase.
Abstract: ## Prospective, Double-Blind, Concurrent, Placebo-Controlled Clinical Trial of Intravenous Ribavirin Therapy of Hemorrhagic Fever with Renal Syndrome
John W. Huggins, Chin Min Hsiang, Thomas M. Cosgriff,* Mei Ying Guang, Joseph I. Smith, Zhen Ou Wu, James W. LeDuc, Zhi Ming Zheng, James M. Meegan,* Qi Nan Wang, Dwayne D. Oland, Xi En Gui,
Paul H. Gibbs, Guang Hui Yuan, and Tian Ming Zhang
A prospective, randomized, double-blind, concurrent, placebo-controlled clinical trial ofintra venous ribavirin (loading dose of 33 mg/kg, 16 mg/kg every 6 h for 4 days, and 8 rug/kg every 8 h for 3 days) was conducted in 242 patients with serologically confirmed hemorrhagic fever with renal syndrome (HFRS) in the People'sRepublic of China. Mortality was significantly reduced (sevenfold decrease in risk) among ribavirin-treated patients, when comparisons were adjusted for baseline risk estimators of mortality (P = .01; two-tailed). HFRS typically consists of five consecutive but frequently overlapping clinical phases. Only occurrence of oliguric phase and hemorrhage was associated with severity of clinical disease in the placebo group. Ribavirin ther apy also resulted in a significant reduction in the risk ofentering the oliguric phase and experienc ing hemorrhage, The only ribavirin-related side effect was a well-recognized, fully reversible anemia after completion of therapy.
Hemorrhagic fever with renal syndrome (HFRS) has been recognized as a clinical entity in Asia since at least 1913. A similar, but milder, form of the disease was first reported in Scandinavia in the 1930s. The characteristic clinical associa tion includes fever, hemorrhage, and renal insufficiency. Its clinical, pathophysiologic, and pathologic features were sum marized in 1954 [1]. HFRS occurs as a seasonal disease in China, with tens to hundreds of thousands of cases recog nized annually in endemic regions. Mortality in Asian forms of HFRS is -- 5%-1 0% in centers where modern medical re sources are available but may be higher elsewhere.
Received 27 July 1990; revised 23 August 1991.
Written informed consent was given by participants. For the protection of human subjects, investigators adhered to US law 45CFR46 and the laws of the People's Republic of China. The clinical protocol and amendments (including the informed consent form) were reviewed and approved by the following scientific and institutional use review boards: USAMRIID. US Army Surgeon General's, Hubei Medical University, Hubei Ministry of Health, and the Chinese National Ministry of Health Drug Control Board.
The views of the authors do not purport to reflect the positions of either the US Department of the Army or the Department of Defense.
The use of tradenames is for identification only and does not constitute endorsement by the US Department of the Army.
Financial support: Institutional funds and US Army Medical Research and Development Command (DAMD17-86-G-6002 to Hubei Medical Uni versity and participating hospitals).
Reprints or correspondence: Dr. John W. Huggins, Department ofAnti viral Studies. Virology Division. US Army Medical Research Institute of Infectious Diseases, Fort Detrick, Frederick. MD 21702-50 II.
* Present addresses: Department of Oncology, Fitzsimmons Army Center, Denver, Colorado (T.M.C.); World Health Organization. Geneva, Switzerland (J.M.M.).
Ribavirin is an antiviral chemotherapeutic drug that in hibits replication of several hemorrhagic fever viruses. It has..
DOI record:
{
"DOI": "10.1093/infdis/164.6.1119",
"ISSN": [
"0022-1899",
"1537-6613"
],
"URL": "http://dx.doi.org/10.1093/infdis/164.6.1119",
"author": [
{
"affiliation": [],
"family": "Huggins",
"given": "J. W.",
"sequence": "first"
},
{
"affiliation": [],
"family": "Hsiang",
"given": "C. M.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Cosgriff",
"given": "T. M.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Guang",
"given": "M. Y.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Smith",
"given": "J. I.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Wu",
"given": "Z. O.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "LeDuc",
"given": "J. W.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Zheng",
"given": "Z. M.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Meegan",
"given": "J. M.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Wang",
"given": "Q. N.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Oland",
"given": "D. D.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Gui",
"given": "X. E.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Gibbs",
"given": "P. H.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Yuan",
"given": "G. H.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Zhang",
"given": "T. M.",
"sequence": "additional"
}
],
"container-title": "Journal of Infectious Diseases",
"container-title-short": "Journal of Infectious Diseases",
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2011,
3,
18
]
],
"date-time": "2011-03-18T14:22:25Z",
"timestamp": 1300458145000
},
"deposited": {
"date-parts": [
[
2017,
1,
25
]
],
"date-time": "2017-01-25T15:35:48Z",
"timestamp": 1485358548000
},
"indexed": {
"date-parts": [
[
2026,
4,
11
]
],
"date-time": "2026-04-11T14:24:51Z",
"timestamp": 1775917491218,
"version": "3.50.1"
},
"is-referenced-by-count": 342,
"issue": "6",
"issued": {
"date-parts": [
[
1991,
12,
1
]
]
},
"journal-issue": {
"issue": "6",
"published-print": {
"date-parts": [
[
1991,
12,
1
]
]
}
},
"language": "en",
"link": [
{
"URL": "https://academic.oup.com/jid/article/164/6/1119/825527/Prospective-DoubleBlind-Concurrent",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "286",
"original-title": [],
"page": "1119-1127",
"prefix": "10.1093",
"published": {
"date-parts": [
[
1991,
12,
1
]
]
},
"published-print": {
"date-parts": [
[
1991,
12,
1
]
]
},
"publisher": "Oxford University Press (OUP)",
"reference-count": 0,
"references-count": 0,
"relation": {},
"resource": {
"primary": {
"URL": "https://academic.oup.com/jid/article-lookup/doi/10.1093/infdis/164.6.1119"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "Prospective, Double-Blind, Concurrent, Placebo-Controlled Clinical Trial of Intravenous Ribavirin Therapy of Hemorrhagic Fever with Renal Syndrome",
"type": "journal-article",
"volume": "164"
}