The Role of Vitamin C as Adjuvant Therapy in COVID-19
et al., Cureus, doi:10.7759/cureus.11779, Nov 2020
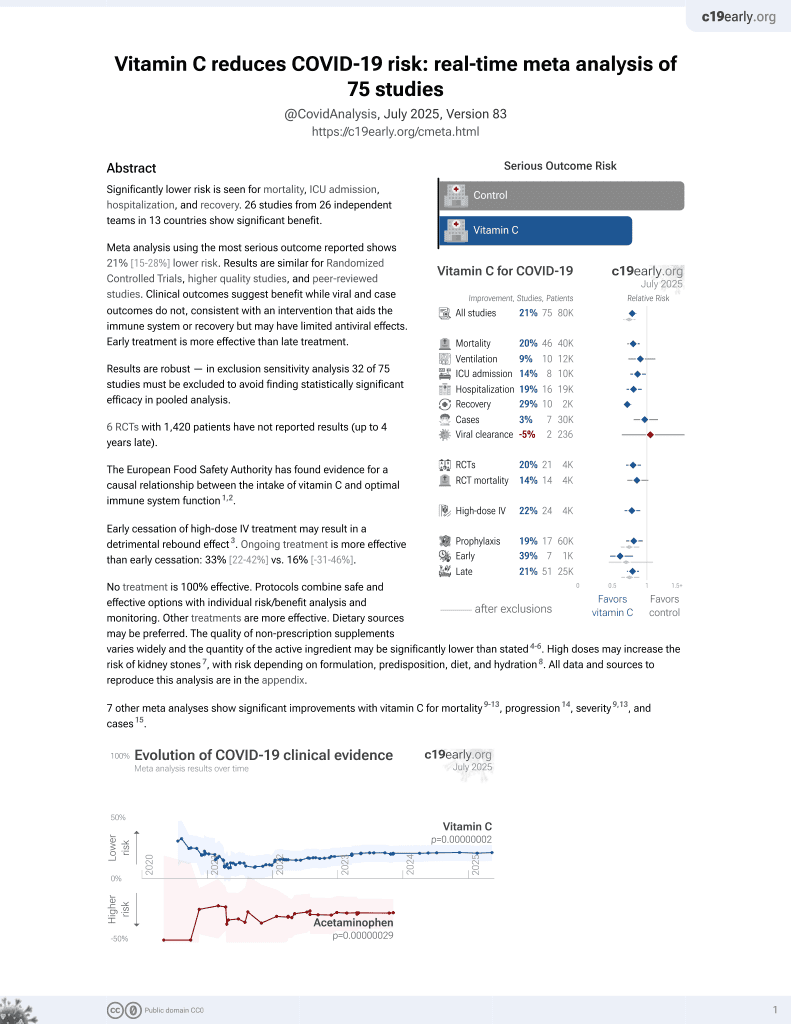
Vitamin C for COVID-19
6th treatment shown to reduce risk in
September 2020, now with p = 0.000000069 from 72 studies, recognized in 22 countries.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
RCT 150 hospitalized patients in Pakistan showing 26% faster recovery, p < 0.0001. 36% lower mortality, not statistically significant due to the small number of events. Dosage was 50 mg/kg/day of intravenous vitamin C.
This is the 2nd of 20 COVID-19 RCTs for vitamin C, which collectively show efficacy with p=0.0016.
This is the 7th of 72 COVID-19 controlled studies for vitamin C, which collectively show efficacy with p=0.000000069.
|
risk of death, 36.4% lower, RR 0.64, p = 0.45, treatment 7 of 75 (9.3%), control 11 of 75 (14.7%), NNT 19.
|
|
risk of mechanical ventilation, 20.0% lower, RR 0.80, p = 0.67, treatment 12 of 75 (16.0%), control 15 of 75 (20.0%), NNT 25.
|
|
recovery time, 26.0% lower, relative time 0.74, p < 0.001, treatment 75, control 75, days to symptom-free.
|
|
hospitalization time, 24.3% lower, relative time 0.76, p < 0.001, treatment 75, control 75, days spent in hospital.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Kumari et al., 30 Nov 2020, Randomized Controlled Trial, Pakistan, peer-reviewed, 10 authors, study period March 2020 - July 2020, dosage 50mg/kg daily.
The Role of Vitamin C as Adjuvant Therapy in COVID-19
Cureus, doi:10.7759/cureus.11779
Background and objective The anti-inflammatory properties of vitamin C (VC) and the promising results it has shown in the treatment for common cold have prompted clinicians to use it as adjuvant therapy in the treatment of COVID-19. The purpose of this study was to find out the role of VC as adjunctive therapy in coronavirus disease 2019 .
Methodology This study was conducted from March to July 2020 in the COVID-19 unit of a tertiary care hospital in Karachi. In this randomized controlled trial (RCT), one group received the intervention [50 mg/kg/day of intravenous (IV) VC] along with the standard therapy, and the other group received standard therapy only. Data such as age, gender, vitals, and biochemical values as well as outcomes including the number of days required for treatment, hospital stay, need for ventilation, and mortality were compared between the two groups and recorded using a self-structured questionnaire.
Results COVID-19 patients who received IV VC became symptom-free earlier (7.1 ± 1.8 vs. 9.6 ± 2.1 days, p-value: <0.0001) and spent fewer days in the hospital (8.1 ± 1.8 vs. 10.7 ± 2.2 days, p-value: <0.0001) compared to those who received standard therapy only. However, there was no significant difference in the need for mechanical ventilation (p-value: 0.406) and mortality (p-value: 0.31) between the two groups.
Conclusion VC can significantly improve clinical symptoms in patients affected with COVID-19; however, it had no impact on mortality and the need for mechanical ventilation. More large-scale studies are required to further assess the role of VC in the treatment of COVID-19.
Additional Information Disclosures Human subjects: Consent was obtained by all participants in this study. Ghulam Muhammad Mahar Medical College IRB issued approval GMMMC/20/03-4 (electronic). This study was approved by Ghulam Muhammad Mahar Medical College IRB. Animal subjects: All authors have confirmed that this study did not involve animal subjects or tissue. Conflicts of interest: In compliance with the ICMJE uniform disclosure form, all authors declare the following: Payment/services info: All authors have declared that no financial support was received from any organization for the submitted work. Financial relationships: All authors have declared that they have no financial relationships at present or within the previous three years with any organizations that might have an interest in the submitted work. Other relationships: All authors have declared that there are no other relationships or activities that could appear to have influenced the submitted work.
References
Biancatelli, Berrill, Catravas, Marik, Quercetin and vitamin C: an experimental, synergistic therapy for the prevention and treatment of SARS-CoV-2 related disease (COVID-19), Front Immunol, doi:10.3389/fimmu.2020.01451
Cheng, Can early and high intravenous dose of vitamin C prevent and treat coronavirus disease 2019 (COVID-19)?, Med Drug Discov, doi:10.1016/j.medidd.2020.100028
Feyaerts, Luyten, Vitamin C as prophylaxis and adjunctive medical treatment for COVID-19?, Nutrition, doi:10.1016/j.nut.2020.110948
Fowler Aa 3rd, Truwit, Hite, Effect of vitamin C infusion on organ failure and biomarkers of inflammation and vascular injury in patients with sepsis and severe acute respiratory failure: the CITRIS-ALI randomized clinical trial, JAMA, doi:10.1001/jama.2019.11825
Hemilä, Chalker, Vitamin C may reduce the duration of mechanical ventilation in critically ill patients: a meta-regression analysis, J Intensive Care, doi:10.1186/s40560-020-0432-y
Hiedra, Lo, Elbashabsheh, The use of IV vitamin C for patients with COVID-19: a case series, Expert Rev Anti Infect Ther, doi:10.1080/14787210.2020.1794819
Hoang, Shaw, Fang, Han, Possible application of high-dose vitamin C in the prevention and therapy of coronavirus infection, J Glob Antimicrob Resist, doi:10.1016/j.jgar.2020.09.025
Jafarnejad, Boccardi, Hosseini, Taghizadeh, Hamedifard, A meta-analysis of randomized control trials: the impact of vitamin C supplementation on serum CRP and serum hs-CRP concentrations, Curr Pharm Des, doi:10.2174/1381612824666181017101810
Johnston, Barkyoumb, Schumacher, Vitamin C supplementation slightly improves physical activity levels and reduces cold incidence in men with marginal vitamin C status: a randomized controlled trial, Nutrients, doi:10.3390/nu6072572
Kumari, None, Cureus, doi:10.7759/cureus.117794of4
Mao, Wang, Neurologic manifestations of hospitalized patients with coronavirus disease 2019 in Wuhan, China, JAMA Neurol, doi:10.1001/jamaneurol.2020.1127
Marik, Khangoora, Rivera, Hooper, Catravas, Hydrocortisone, vitamin C, and thiamine for the treatment of severe sepsis and septic shock: a retrospective before-after study, Chest, doi:10.1016/j.chest.2016.11.036
Ran, Zhao, Wang, Wang, Zhao et al., Extra dose of vitamin C based on a daily supplementation shortens the common cold: a meta-analysis of 9 randomized controlled trials, Biomed Res Int, doi:10.1155/2018/1837634
Vorilhon, Arpajou, Roussel, Pereira, Cabaillot, Efficacy of vitamin C for the prevention and treatment of upper respiratory tract infection. A meta-analysis in children, Eur J Clin Pharmacol, doi:10.1007/s00228-018-2601-7
Zhang, Rao, Li, High-dose vitamin C infusion for the treatment of critically ill COVID-19, doi:10.21203/rs.3.rs-52778/v1
DOI record:
{
"DOI": "10.7759/cureus.11779",
"ISSN": [
"2168-8184"
],
"URL": "http://dx.doi.org/10.7759/cureus.11779",
"author": [
{
"affiliation": [],
"family": "Kumari",
"given": "Poona",
"sequence": "first"
},
{
"affiliation": [],
"family": "Dembra",
"given": "Suman",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Dembra",
"given": "Pariya",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Bhawna",
"given": "Fnu",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Gul",
"given": "Ambresha",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ali",
"given": "Basma",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Sohail",
"given": "Hamza",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Kumar",
"given": "Besham",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Memon",
"given": "Muhammad Khizar",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Rizwan",
"given": "Amber",
"sequence": "additional"
}
],
"container-title": "Cureus",
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2020,
11,
30
]
],
"date-time": "2020-11-30T09:15:55Z",
"timestamp": 1606727755000
},
"deposited": {
"date-parts": [
[
2024,
2,
9
]
],
"date-time": "2024-02-09T19:20:34Z",
"timestamp": 1707506434000
},
"indexed": {
"date-parts": [
[
2024,
3,
25
]
],
"date-time": "2024-03-25T00:54:26Z",
"timestamp": 1711328066038
},
"is-referenced-by-count": 34,
"issued": {
"date-parts": [
[
2020,
11,
30
]
]
},
"language": "en",
"link": [
{
"URL": "https://www.cureus.com/articles/45284-the-role-of-vitamin-c-as-adjuvant-therapy-in-covid-19",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "297",
"original-title": [],
"prefix": "10.7759",
"published": {
"date-parts": [
[
2020,
11,
30
]
]
},
"published-print": {
"date-parts": [
[
2020,
11,
30
]
]
},
"publisher": "Springer Science and Business Media LLC",
"reference": [
{
"key": "ref1",
"unstructured": "World Health Organization. coronavirus. (2020). Accessed: October 22, 2020: https://www.who.int/health-topics/coronavirus."
},
{
"DOI": "10.3390/nu6072572",
"article-title": "Vitamin C supplementation slightly improves physical activity levels and reduces cold incidence in men with marginal vitamin C status: a randomized controlled trial",
"author": "Johnston CS",
"doi-asserted-by": "publisher",
"journal-title": "Nutrients",
"key": "ref2",
"unstructured": "Johnston CS, Barkyoumb GM, Schumacher SS. Vitamin C supplementation slightly improves physical activity levels and reduces cold incidence in men with marginal vitamin C status: a randomized controlled trial. Nutrients. 2014, 6:2572-2583. 10.3390/nu6072572",
"volume": "6",
"year": "2014"
},
{
"DOI": "10.2174/1381612824666181017101810",
"article-title": "A meta-analysis of randomized control trials: the impact of vitamin C supplementation on serum CRP and serum hs-CRP concentrations",
"author": "Jafarnejad S",
"doi-asserted-by": "publisher",
"journal-title": "Curr Pharm Des",
"key": "ref3",
"unstructured": "Jafarnejad S, Boccardi V, Hosseini B, Taghizadeh M, Hamedifard Z. A meta-analysis of randomized control trials: the impact of vitamin C supplementation on serum CRP and serum hs-CRP concentrations. Curr Pharm Des. 2018, 24:3520-3528. 10.2174/1381612824666181017101810",
"volume": "24",
"year": "2018"
},
{
"DOI": "10.1080/14787210.2020.1794819",
"article-title": "The use of IV vitamin C for patients with COVID-19: a case series",
"author": "Hiedra R",
"doi-asserted-by": "publisher",
"journal-title": "Expert Rev Anti Infect Ther",
"key": "ref4",
"unstructured": "Hiedra R, Lo KB, Elbashabsheh M, et al.. The use of IV vitamin C for patients with COVID-19: a case series. Expert Rev Anti Infect Ther. 2020, 18:1259-1261. 10.1080/14787210.2020.1794819",
"volume": "18",
"year": "2020"
},
{
"DOI": "10.1016/j.jgar.2020.09.025",
"article-title": "Possible application of high-dose vitamin C in the prevention and therapy of coronavirus infection",
"author": "Hoang BX",
"doi-asserted-by": "publisher",
"journal-title": "J Glob Antimicrob Resist",
"key": "ref5",
"unstructured": "Hoang BX, Shaw G, Fang W, Han B. Possible application of high-dose vitamin C in the prevention and therapy of coronavirus infection. J Glob Antimicrob Resist. 2020, 23:256-262. 10.1016/j.jgar.2020.09.025",
"volume": "23",
"year": "2020"
},
{
"key": "ref6",
"unstructured": "NHSRC. clinical management guideline for COVID-19 infections. (2020). Accessed: Nov 29, 2020: http://www.nhsrc.gov.pk/SiteImage/Misc/files/20200704%20Clinical%20Management%20Guidelines%20for%20COVID-19%20infecti...."
},
{
"DOI": "10.1001/jamaneurol.2020.1127",
"article-title": "Neurologic manifestations of hospitalized patients with coronavirus disease 2019 in Wuhan, China",
"author": "Mao L",
"doi-asserted-by": "publisher",
"journal-title": "JAMA Neurol",
"key": "ref7",
"unstructured": "Mao L, Jin H, Wang M, et al.. Neurologic manifestations of hospitalized patients with coronavirus disease 2019 in Wuhan, China. JAMA Neurol. 2020, 77:683-690. 10.1001/jamaneurol.2020.1127",
"volume": "77",
"year": "2020"
},
{
"DOI": "10.1016/j.nut.2020.110948",
"article-title": "Vitamin C as prophylaxis and adjunctive medical treatment for COVID-19?",
"author": "Feyaerts AF",
"doi-asserted-by": "publisher",
"journal-title": "Nutrition",
"key": "ref8",
"unstructured": "Feyaerts AF, Luyten W. Vitamin C as prophylaxis and adjunctive medical treatment for COVID-19?. Nutrition. 2020, 79:110948. 10.1016/j.nut.2020.110948",
"volume": "79",
"year": "2020"
},
{
"DOI": "10.3389/fimmu.2020.01451",
"article-title": "Quercetin and vitamin C: an experimental, synergistic therapy for the prevention and treatment of SARS-CoV-2 related disease (COVID-19)",
"author": "Colunga Biancatelli RML",
"doi-asserted-by": "publisher",
"journal-title": "Front Immunol",
"key": "ref9",
"unstructured": "Colunga Biancatelli RML, Berrill M, Catravas JD, Marik PE. Quercetin and vitamin C: an experimental, synergistic therapy for the prevention and treatment of SARS-CoV-2 related disease (COVID-19). Front Immunol. 2020, 11:1451. 10.3389/fimmu.2020.01451",
"volume": "11",
"year": "2020"
},
{
"DOI": "10.1016/j.medidd.2020.100028",
"article-title": "Can early and high intravenous dose of vitamin C prevent and treat coronavirus disease 2019 (COVID-19)?",
"author": "Cheng RZ",
"doi-asserted-by": "publisher",
"journal-title": "Med Drug Discov",
"key": "ref10",
"unstructured": "Cheng RZ. Can early and high intravenous dose of vitamin C prevent and treat coronavirus disease 2019 (COVID-19)?. Med Drug Discov. 2020, 5:100028. 10.1016/j.medidd.2020.100028",
"volume": "5",
"year": "2020"
},
{
"DOI": "10.1001/jama.2019.11825",
"article-title": "Effect of vitamin C infusion on organ failure and biomarkers of inflammation and vascular injury in patients with sepsis and severe acute respiratory failure: the CITRIS-ALI randomized clinical trial",
"author": "Fowler AA 3rd",
"doi-asserted-by": "publisher",
"journal-title": "JAMA",
"key": "ref11",
"unstructured": "Fowler AA 3rd, Truwit JD, Hite RD, et al.. Effect of vitamin C infusion on organ failure and biomarkers of inflammation and vascular injury in patients with sepsis and severe acute respiratory failure: the CITRIS-ALI randomized clinical trial. JAMA. 2019, 322:1261-1270. 10.1001/jama.2019.11825",
"volume": "322",
"year": "2019"
},
{
"DOI": "10.1155/2018/1837634",
"article-title": "Extra dose of vitamin C based on a daily supplementation shortens the common cold: a meta-analysis of 9 randomized controlled trials",
"author": "Ran L",
"doi-asserted-by": "publisher",
"journal-title": "Biomed Res Int",
"key": "ref12",
"unstructured": "Ran L, Zhao W, Wang J, Wang H, Zhao Y, Tseng Y, Bu H. Extra dose of vitamin C based on a daily supplementation shortens the common cold: a meta-analysis of 9 randomized controlled trials. Biomed Res Int. 2018, 2018:1837634. 10.1155/2018/1837634",
"volume": "2018",
"year": "2018"
},
{
"DOI": "10.1007/s00228-018-2601-7",
"article-title": "Efficacy of vitamin C for the prevention and treatment of upper respiratory tract infection. A meta-analysis in children",
"author": "Vorilhon P",
"doi-asserted-by": "publisher",
"journal-title": "Eur J Clin Pharmacol",
"key": "ref13",
"unstructured": "Vorilhon P, Arpajou B, Vaillant Roussel H, Merlin É, Pereira B, Cabaillot A. Efficacy of vitamin C for the prevention and treatment of upper respiratory tract infection. A meta-analysis in children. Eur J Clin Pharmacol. 2019, 75:303-311. 10.1007/s00228-018-2601-7",
"volume": "75",
"year": "2019"
},
{
"DOI": "10.1186/s40560-020-0432-y",
"article-title": "Vitamin C may reduce the duration of mechanical ventilation in critically ill patients: a meta-regression analysis",
"author": "Hemilä H",
"doi-asserted-by": "publisher",
"journal-title": "J Intensive Care",
"key": "ref14",
"unstructured": "Hemilä H, Chalker E. Vitamin C may reduce the duration of mechanical ventilation in critically ill patients: a meta-regression analysis. J Intensive Care. 2020, 8:15. 10.1186/s40560-020-0432-y",
"volume": "8",
"year": "2020"
},
{
"DOI": "10.1016/j.chest.2016.11.036",
"article-title": "Hydrocortisone, vitamin C, and thiamine for the treatment of severe sepsis and septic shock: a retrospective before-after study",
"author": "Marik PE",
"doi-asserted-by": "publisher",
"journal-title": "Chest",
"key": "ref15",
"unstructured": "Marik PE, Khangoora V, Rivera R, Hooper MH, Catravas J. Hydrocortisone, vitamin C, and thiamine for the treatment of severe sepsis and septic shock: a retrospective before-after study. Chest. 2017, 151:1229-1238. 10.1016/j.chest.2016.11.036",
"volume": "151",
"year": "2017"
},
{
"DOI": "10.21203/rs.3.rs-52778/v1",
"article-title": "High-dose vitamin C infusion for the treatment of critically ill COVID-19 [PREPRINT]",
"author": "Zhang J",
"doi-asserted-by": "publisher",
"journal-title": "Res Square",
"key": "ref16",
"unstructured": "Zhang J, Rao X, Li Y, et al.. High-dose vitamin C infusion for the treatment of critically ill COVID-19 [PREPRINT]. Res Square. 2020, 10.21203/rs.3.rs-52778/v1",
"year": "2020"
}
],
"reference-count": 16,
"references-count": 16,
"relation": {},
"resource": {
"primary": {
"URL": "https://www.cureus.com/articles/45284-the-role-of-vitamin-c-as-adjuvant-therapy-in-covid-19"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [
"Aerospace Engineering"
],
"subtitle": [],
"title": "The Role of Vitamin C as Adjuvant Therapy in COVID-19",
"type": "journal-article"
}