Azvudine versus Paxlovid for oral treatment of COVID-19 in Chinese patients with pre-existing comorbidities
et al., Journal of Infection, doi:10.1016/j.jinf.2023.05.012, Aug 2023
Azvudine for COVID-19
48th treatment shown to reduce risk in
January 2023, now with p = 0.0000000021 from 42 studies.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
Retrospective 2,118 hospitalized COVID-19 patients in China, showing improved results with azvudine vs. paxlovid.
Standard of Care (SOC) for COVID-19 in the study country,
China, is poor with low average efficacy for approved treatments3.
Study covers paxlovid and azvudine.
|
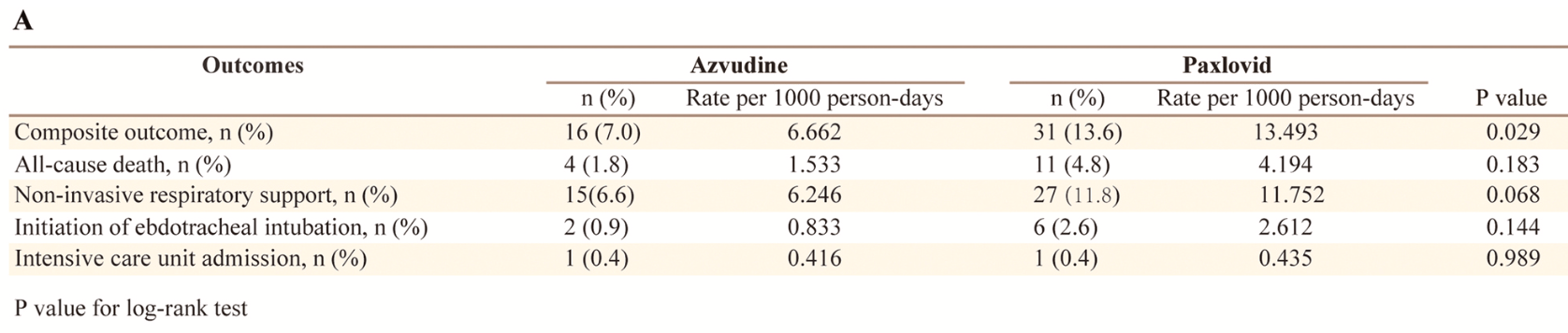
risk of death, 63.6% lower, RR 0.36, p = 0.11, treatment 4 of 228 (1.8%), control 11 of 228 (4.8%), NNT 33, propensity score matching.
|
|
risk of mechanical ventilation, 66.7% lower, RR 0.33, p = 0.28, treatment 2 of 228 (0.9%), control 6 of 228 (2.6%), NNT 57, propensity score matching.
|
|
risk of ICU admission, no change, RR 1.00, p = 1.00, treatment 1 of 228 (0.4%), control 1 of 228 (0.4%), propensity score matching.
|
|
composite outcome, 48.4% lower, RR 0.52, p = 0.03, treatment 16 of 228 (7.0%), control 31 of 228 (13.6%), NNT 15, non-invasive respiratory support, endotracheal intubation, ICU admission, all-cause death, propensity score matching.
|
|
respiratory support, 44.4% lower, RR 0.56, p = 0.07, treatment 15 of 228 (6.6%), control 27 of 228 (11.8%), NNT 19, propensity score matching.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
1.
Xiong et al., Real-world data of Azvudine-induced hepatotoxicity among hospitalized COVID-19 patients in China: a retrospective case-control study, Frontiers in Pharmacology, doi:10.3389/fphar.2025.1558054.
Dian et al., 31 Aug 2023, retrospective, China, peer-reviewed, 5 authors, study period 5 December, 2022 - 31 January, 2023, this trial compares with another treatment - results may be better when compared to placebo.
Abstract: Journal of Infection 87 (2023) e24–e27
Contents lists available at ScienceDirect
Journal of Infection
journal homepage: www.elsevier.com/locate/jinf
Letter to the Editor
Azvudine versus Paxlovid for oral treatment of
COVID-19 in Chinese patients with pre-existing
comorbidities
]]]]
]]
Dear Editor,
Chinese guidelines grant the use of Azvudine and Paxlovid in
COVID-19 patients, especially those with pre-existing comorbid
ities.1,2 Recently, Gao Y et al. reported that Paxlovid appears to be
superior to Azvudine in the virus clearance among general COVID-19
patients.3 However, a multicenter randomized controlled study de
monstrated that Paxlovid showed no significant reduction in the risk
of all-cause mortality on day 28 and the duration of virus clearance
in hospitalized adult COVID-19 patients with pre-existing co
morbidities.4 Several studies demonstrated that Azvudine could re
duce the duration of virus clearance and improve the clinical
prognosis in COVID-19 patients including those with pre-existing
comorbidities.5–8 Therefore, concerns arise about the clinical effec
tiveness of Azvudine versus Paxlovid in COVID-19 patients with preexisting comorbidities on admission.
Here, we conducted a single-center, retrospective cohort study
during the outbreak caused by the omicron from December 5,
2022 to January 31, 2023 in Xiangya Hospital of Central South
University. The study included hospitalized patients with preexisting comorbidities and confirmed diagnosis of SARS-CoV-2
infection who received Paxlovid or Azvudine. The patients with
these conditions were excluded: 1) younger than 18 years; 2) re
ceived oxygen support or mechanical ventilation on the date of
the admission; 3) not received any antiviral agents; 4) received
both Azvudine and Paxlovid. The study was approved by the in
stitutional review board of Xiangya Hospital of Central South
University, and all the patients were anonymous and no need for
individual informed consent.
The primary endpoint was a composite disease progression
outcome which was defined as any of the following events: 1) noninvasive respiratory support; 2) initiation of endotracheal intuba
tion; 3) intensive care unit admission; 4) all-cause death. The sec
ondary endpoints were each of these individual disease progression
outcomes. Patients were observed from the date of admission until
discharge, occurrence of outcome event or death, whichever came
first. We used propensity-score models conditional on baseline
characteristics, and the probability of receiving Azvudine was esti
mated in an approach of calliper matching without replacement,
with a calliper width of 0.2. The baseline covariates included age,
sex, time from symptom onset to hospitalization, COVID-19 vacci
nation status, severity of COVID-19 on admission (severe cases were
defined as having respiratory rate ≥30, or oxygen saturation ≤93%, or
PaO2/FiO2 ≤300 mmHg, or lung infiltrates > 50%), and concomitant
treatments initiated at admission (systemic steroid and antibiotics).
The standard mean differences (SMDs) were used to assess the
balance of each baseline covariates between the groups before and
after propensity-score matching which less than 0.1 indicating
covariate was balanced.9 The incidence rates of outcome events were
calculated as the number of outcome events / (sum of person ×
hospital days). Univariate Cox regression model was used to estimate
a hazard ratio (HR) with 95% confidence interval (CI) for each..
DOI record:
{
"DOI": "10.1016/j.jinf.2023.05.012",
"ISSN": [
"0163-4453"
],
"URL": "http://dx.doi.org/10.1016/j.jinf.2023.05.012",
"alternative-id": [
"S0163445323002906"
],
"assertion": [
{
"label": "This article is maintained by",
"name": "publisher",
"value": "Elsevier"
},
{
"label": "Article Title",
"name": "articletitle",
"value": "Azvudine versus Paxlovid for oral treatment of COVID-19 in Chinese patients with pre-existing comorbidities"
},
{
"label": "Journal Title",
"name": "journaltitle",
"value": "Journal of Infection"
},
{
"label": "CrossRef DOI link to publisher maintained version",
"name": "articlelink",
"value": "https://doi.org/10.1016/j.jinf.2023.05.012"
},
{
"label": "Content Type",
"name": "content_type",
"value": "simple-article"
},
{
"label": "Copyright",
"name": "copyright",
"value": "© 2023 The British Infection Association. Published by Elsevier Ltd. All rights reserved."
}
],
"author": [
{
"affiliation": [],
"family": "Dian",
"given": "Yating",
"sequence": "first"
},
{
"affiliation": [],
"family": "Meng",
"given": "Yu",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Sun",
"given": "Yuming",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Deng",
"given": "Guangtong",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Zeng",
"given": "Furong",
"sequence": "additional"
}
],
"container-title": "Journal of Infection",
"container-title-short": "Journal of Infection",
"content-domain": {
"crossmark-restriction": true,
"domain": [
"clinicalkey.fr",
"clinicalkey.jp",
"clinicalkey.es",
"clinicalkey.com.au",
"clinicalkey.com",
"journalofinfection.com",
"elsevier.com",
"sciencedirect.com"
]
},
"created": {
"date-parts": [
[
2023,
5,
17
]
],
"date-time": "2023-05-17T09:07:19Z",
"timestamp": 1684314439000
},
"deposited": {
"date-parts": [
[
2023,
7,
21
]
],
"date-time": "2023-07-21T16:31:02Z",
"timestamp": 1689957062000
},
"funder": [
{
"DOI": "10.13039/501100001809",
"award": [
"82102803",
"82103183",
"82272849"
],
"doi-asserted-by": "publisher",
"name": "National Natural Science Foundation of China"
},
{
"DOI": "10.13039/501100004735",
"award": [
"2021JJ40976",
"2022JJ40767"
],
"doi-asserted-by": "publisher",
"name": "Natural Science Foundation of Hunan Province"
},
{
"DOI": "10.13039/501100011790",
"doi-asserted-by": "publisher",
"name": "Xiangya Hospital, Central South University"
}
],
"indexed": {
"date-parts": [
[
2023,
7,
22
]
],
"date-time": "2023-07-22T04:13:53Z",
"timestamp": 1689999233029
},
"is-referenced-by-count": 1,
"issue": "2",
"issued": {
"date-parts": [
[
2023,
8
]
]
},
"journal-issue": {
"issue": "2",
"published-print": {
"date-parts": [
[
2023,
8
]
]
}
},
"language": "en",
"license": [
{
"URL": "https://www.elsevier.com/tdm/userlicense/1.0/",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2023,
8,
1
]
],
"date-time": "2023-08-01T00:00:00Z",
"timestamp": 1690848000000
}
}
],
"link": [
{
"URL": "https://api.elsevier.com/content/article/PII:S0163445323002906?httpAccept=text/xml",
"content-type": "text/xml",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://api.elsevier.com/content/article/PII:S0163445323002906?httpAccept=text/plain",
"content-type": "text/plain",
"content-version": "vor",
"intended-application": "text-mining"
}
],
"member": "78",
"original-title": [],
"page": "e24-e27",
"prefix": "10.1016",
"published": {
"date-parts": [
[
2023,
8
]
]
},
"published-print": {
"date-parts": [
[
2023,
8
]
]
},
"publisher": "Elsevier BV",
"reference": [
{
"key": "10.1016/j.jinf.2023.05.012_bib1",
"unstructured": "General Office of the National Health Commission. Notice on the issuance of Diagnosis and Treatment Protocol for novel coronavirus infection (trial version 9); 2022. Accessed on: 〈http://www.gov.cn/zhengce/zhengceku/2022-03/15/content_5679257.htm〉."
},
{
"key": "10.1016/j.jinf.2023.05.012_bib2",
"unstructured": "General Office of the National Health Commission. Notice on the issuance of Diagnosis and Treatment Protocol for novel coronavirus infection (trial version 10); 2023. Accessed on: 〈http://www.gov.cn/zhengce/zhengceku/2023-01/06/content_5735343.htm〉."
},
{
"DOI": "10.1016/j.jinf.2023.03.023",
"article-title": "Antiviral effect of azvudine and nirmatrelvir-ritonavir among hospitalized patients with COVID-19",
"author": "Gao",
"doi-asserted-by": "crossref",
"first-page": "e158",
"issue": "6",
"journal-title": "J Infect",
"key": "10.1016/j.jinf.2023.05.012_bib3",
"volume": "86",
"year": "2023"
},
{
"article-title": "Efficacy and safety of Paxlovid in severe adult patients with SARS-Cov-2 infection: a multicenter randomized controlled study",
"author": "Liu",
"journal-title": "Lancet Reg Health West Pac",
"key": "10.1016/j.jinf.2023.05.012_bib4",
"volume": "33",
"year": "2023"
},
{
"DOI": "10.1038/s41392-021-00835-6",
"article-title": "Azvudine is a thymus-homing anti-SARS-CoV-2 drug effective in treating COVID-19 patients",
"author": "Zhang",
"doi-asserted-by": "crossref",
"first-page": "414",
"issue": "1",
"journal-title": "Signal Transduct Target Ther",
"key": "10.1016/j.jinf.2023.05.012_bib5",
"volume": "6",
"year": "2021"
},
{
"DOI": "10.1002/advs.202001435",
"article-title": "A randomized, open-label, controlled clinical trial of azvudine tablets in the treatment of mild and common COVID-19, a pilot study",
"author": "Ren",
"doi-asserted-by": "crossref",
"issue": "19",
"journal-title": "Adv Sci",
"key": "10.1016/j.jinf.2023.05.012_bib6",
"volume": "7",
"year": "2020"
},
{
"article-title": "Oral azvudine (FNC) tablets in patients infected with SARS-CoV-2 omicron variant: a retrospective cohort study",
"author": "Chen",
"journal-title": "medRxiv",
"key": "10.1016/j.jinf.2023.05.012_bib7",
"year": "2023"
},
{
"article-title": "Real-world effectiveness of Azvudine in hospitalized patients with COVID-19: a retrospective cohort study",
"author": "Shen",
"journal-title": "medRxiv",
"key": "10.1016/j.jinf.2023.05.012_bib8",
"year": "2023"
},
{
"DOI": "10.1016/S1473-3099(22)00507-2",
"article-title": "Real-world effectiveness of early molnupiravir or nirmatrelvir-ritonavir in hospitalised patients with COVID-19 without supplemental oxygen requirement on admission during Hong Kong’s omicron BA.2 wave: a retrospective cohort study",
"author": "Wong",
"doi-asserted-by": "crossref",
"first-page": "1681",
"issue": "12",
"journal-title": "Lancet Infect Dis",
"key": "10.1016/j.jinf.2023.05.012_bib9",
"volume": "22",
"year": "2022"
},
{
"DOI": "10.1056/NEJMoa2204919",
"article-title": "Nirmatrelvir use and severe covid-19 outcomes during the omicron surge",
"author": "Arbel",
"doi-asserted-by": "crossref",
"first-page": "790",
"issue": "9",
"journal-title": "N Engl J Med",
"key": "10.1016/j.jinf.2023.05.012_bib10",
"volume": "387",
"year": "2022"
}
],
"reference-count": 10,
"references-count": 10,
"relation": {},
"resource": {
"primary": {
"URL": "https://linkinghub.elsevier.com/retrieve/pii/S0163445323002906"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [
"Infectious Diseases",
"Microbiology (medical)"
],
"subtitle": [],
"title": "Azvudine versus Paxlovid for oral treatment of COVID-19 in Chinese patients with pre-existing comorbidities",
"type": "journal-article",
"update-policy": "http://dx.doi.org/10.1016/elsevier_cm_policy",
"volume": "87"
}
