Effects of Different Inhalation Therapy on Ventilator-Associated Pneumonia in Ventilated COVID-19 Patients: A Randomized Controlled Trial
et al., Microorganisms, doi:10.3390/microorganisms10061118, NCT04755972, May 2022
31st treatment shown to reduce risk in
November 2021, now with p = 0.0000000039 from 14 studies.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
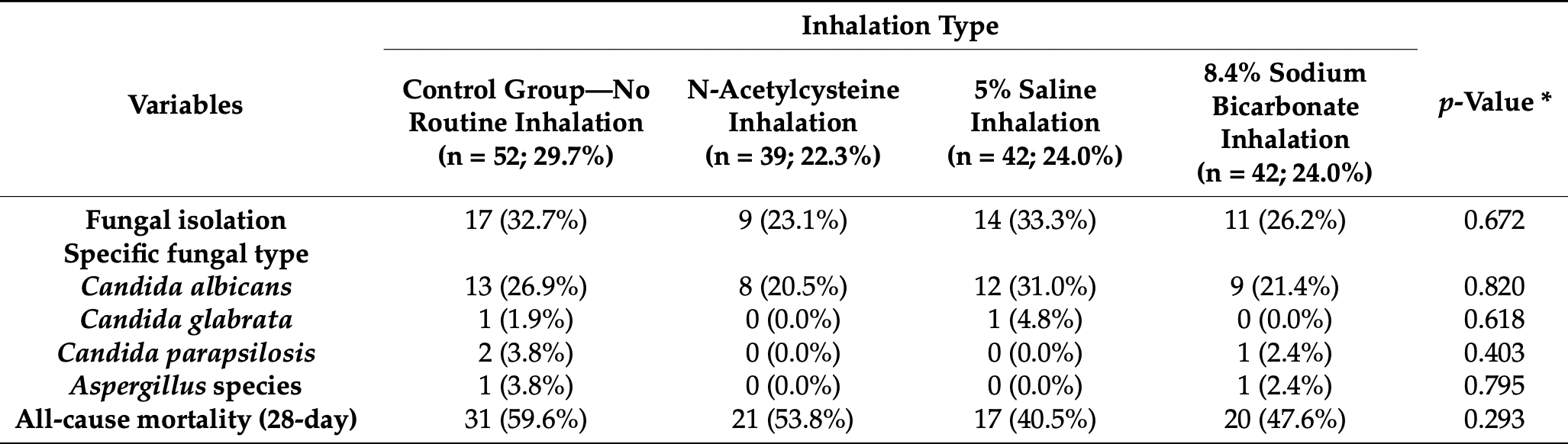
RCT mechanically ventilated patients in Croatia, 42 treated with sodium bicarbonate inhalation, and 52 control patients, showing no significant difference in mortality with treatment. Treated patients showed a lower incidence of gram-positive or MRSA-caused ventilator-associated pneumonia. ICU mortality results are from1.
Targeted administration to the respiratory tract provides treatment directly
to the typical source of initial SARS-CoV-2 infection and replication, and
allows for rapid onset of action and reduced systemic side effects (early treatment may be more beneficial).
Study covers alkalinization, NaCl, and N-acetylcysteine.
|
risk of death, 23.0% lower, RR 0.77, p = 0.13, treatment 23 of 42 (54.8%), control 37 of 52 (71.2%), NNT 6.1, ICU mortality.
|
|
risk of death, 20.1% lower, RR 0.80, p = 0.30, treatment 20 of 42 (47.6%), control 31 of 52 (59.6%), NNT 8.3, 28 day mortality.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Delić et al., 28 May 2022, Randomized Controlled Trial, Croatia, peer-reviewed, 12 authors, study period October 2020 - June 2021, trial NCT04755972 (history).
Contact: ndelic@kbsplit.hr (corresponding author), tkljakgas@kbsplit.hr, lsaric@kbsplit.hr, dilic@kbsplit.hr, sdosenovic@kbsplit.hr, jdomazet@kbsplit.hr, rkovac@kbsplit.hr, sastojanovic@kbsplit.hr, bduplanc@kbsplit.hr, andrija.matetic@gmail.com, frane.runjic@gmail.com, josipa.domjanovic@gmail.com.
Effects of Different Inhalation Therapy on Ventilator-Associated Pneumonia in Ventilated COVID-19 Patients: A Randomized Controlled Trial
Microorganisms, doi:10.3390/microorganisms10061118
The effect of routine inhalation therapy on ventilator-associated pneumonia (VAP) in mechanically ventilated patients with the coronavirus disease (COVID-19) has not been well-defined. This randomized controlled trial included 175 eligible adult patients with COVID-19 who were treated with mechanical ventilation at the University Hospital of Split between October 2020 and June 2021. Patients were randomized and allocated to a control group (no routine inhalation) or one of the treatment arms (inhalation of N-acetylcysteine; 5% saline solution; or 8.4% sodium bicarbonate). The primary outcome was the incidence of VAP, while secondary outcomes included all-cause mortality. Routine inhalation therapy had no effect on the incidence of bacterial or fungal VAP nor on all-cause mortality (p > 0.05). Secondary analyses revealed a significant reduction of Gram-positive and methicillin-resistant Staphylococcus aureus (MRSA) VAP in the treatment groups. Specifically, the bicarbonate group had a statistically significantly lower incidence of Gram-positive bacterial VAP (4.8%), followed by the N-acetylcysteine group (10.3%), 5% saline group (19.0%), and control group (34.6%; p = 0.001). This difference was driven by a lower incidence of MRSA VAP in the bicarbonate group (2.4%), followed by the N-acetylcysteine group (7.7%), 5% saline group (14.3%), and control group (34.6%; p < 0.001). Longer duration of ventilator therapy was the only significant, independent predictor of any bacterial or fungal VAP in the multivariate analysis (aOR 1.14, 95% CI 1.01-1.29, p = 0.038 and aOR 1.05, 95% CI 1.01-1.10, p = 0.028, respectively). In conclusion, inhalation therapy had no effect on the overall VAP incidence or all-cause mortality. Further studies should explore the secondary findings of this study such as the reduction of Gram-positive or MRSA-caused VAP in treated patients.
Conflicts of Interest: The authors declare no conflict of interest.
References
Almand, Moore, Jaykus, Virus-Bacteria Interactions: An Emerging Topic in Human Infection, Viruses, doi:10.3390/v9030058
Bakaletz, Viral-bacterial co-infections in the respiratory tract, Curr. Opin. Microbiol, doi:10.1016/j.mib.2016.11.003
Bennett, Henderson, Donaldson, Hydrator Therapies for Chronic Bronchitis. Lessons from Cystic Fibrosis, Ann. Am. Thorac. Soc
Bergsson, Reeves, Mcnally, Chotirmall, Greene et al., LL-37 complexation with glycosaminoglycans in cystic fibrosis lungs inhibits antimicrobial activity, which can be restored by hypertonic saline, J. Immunol, doi:10.4049/jimmunol.0803959
Blonz, Kouatchet, Chudeau, Pontis, Lorber et al., Epidemiology and microbiology of ventilator-associated pneumonia in COVID-19 patients: A multicenter retrospective study in 188 patients in an un-inundated French region, Crit. Care, doi:10.1186/s13054-021-03493-w
Chang, Elhusseiny, Yeh, Sun, COVID-19 ICU and mechanical ventilation patient characteristics and outcomes-A systematic review and meta-analysis, PLoS ONE, doi:10.1371/journal.pone.0246318
Chen, Yang, Quinton, Chin, A new role for bicarbonate in mucus formation, Am. J. Physiol. Lung Cell. Mol. Physiol, doi:10.1152/ajplung.00180.2010
Cooper, Quinton, Ballard, Mucociliary transport in porcine trachea: Differential effects of inhibiting chloride and bicarbonate secretion, Am. J. Physiol. Lung Cell. Mol. Physiol, doi:10.1152/ajplung.00143.2012
Cox, Loman, Bogaert, O'grady, Co-infections: Potentially lethal and unexplored in COVID-19, Lancet Microbe, doi:10.1016/S2666-5247(20)30009-4
Donaldson, Bennett, Zeman, Knowles, Tarran et al., Mucus Clearance and Lung Function in Cystic Fibrosis with Hypertonic Saline, N. Engl. J. Med, doi:10.1056/NEJMoa043891
Everard, Hind, Ugonna, Freeman, Bradburn et al., SABRE: A multicentre randomised control trial of nebulised hypertonic saline in infants hospitalised with acute bronchiolitis, Thorax, doi:10.1136/thoraxjnl-2014-205953
Fumagalli, Panigada, Klompas, Berra, Ventilator-associated pneumonia among SARS-CoV-2 acute respiratory distress syndrome patients, Curr. Opin. Crit. Care, doi:10.1097/MCC.0000000000000908
Giacobbe, Battaglini, Enrile, Dentone, Vena et al., Incidence and Prognosis of Ventilator-Associated Pneumonia in Critically Ill Patients with COVID-19: A Multicenter Study, J. Clin. Med, doi:10.3390/jcm10040555
Gomez, Parazzi, Clinckspoor, Mauch, Pessine et al., Tolerability, and Effects of Sodium Bicarbonate Inhalation in Cystic Fibrosis, Clin. Drug Investig, doi:10.1007/s40261-019-00861-x
Helms, Tacquard, Severac, Leonard-Lorant, Ohana et al., High risk of thrombosis in patients with severe SARS-CoV-2 infection: A multicenter prospective cohort study, Intensive Care Med, doi:10.1007/s00134-020-06062-x
Icard, Rubio, The role of mucoactive agents in the mechanically ventilated patient: A review of the literature, Expert Rev. Respir. Med, doi:10.1080/17476348.2017.1359090
Ippolito, Misseri, Catalisano, Marino, Ingoglia et al., Ventilator-Associated Pneumonia in Patients with COVID-19: A Systematic Review and Meta-Analysis, Antibiotics, doi:10.3390/antibiotics10050545
King, Dasgupta, Tomkiewicz, Brown, Rheology of cystic fibrosis sputum after in vitro treatment with hypertonic saline alone and in combination with recombinant human deoxyribonuclease I, Am. J. Respir. Crit. Care Med, doi:10.1164/ajrccm.156.1.9512074
Luyt, Sahnoun, Gautier, Vidal, Burrel et al., Ventilator-associated pneumonia in patients with SARS-CoV-2-associated acute respiratory distress syndrome requiring ECMO: A retrospective cohort study, Ann. Intensive Care, doi:10.1186/s13613-020-00775-4
Maes, Higginson, Pereira-Dias, Curran, Parmar et al., Ventilator-associated pneumonia in critically ill patients with COVID-19, Crit. Care, doi:10.1186/s13054-021-03460-5
Masoompour, Anushiravani, Tafaroj Norouz, Evaluation of the Effect of Nebulized N-Acetylcysteine on Respiratory Secretions in Mechanically Ventilated Patients: Randomized Clinical Trial, Iran. J. Med. Sci
Mccullers, The co-pathogenesis of influenza viruses with bacteria in the lung, Nat. Rev. Microbiol, doi:10.1038/nrmicro3231
Melsen, Rovers, Koeman, Bonten, Estimating the attributable mortality of ventilator-associated pneumonia from randomized prevention studies, Crit. Care Med, doi:10.1097/CCM.0b013e3182281f33
Michon, Jumas-Bilak, Chiron, Lamy, Marchandin, Advances toward the elucidation of hypertonic saline effects on Pseudomonas aeruginosa from cystic fibrosis patients, PLoS ONE, doi:10.1371/journal.pone.0090164
Moretti, Van Laethem, Minini, Pierard, Malbrain, Ventilator-associated bacterial pneumonia in coronavirus 2019 disease, a retrospective monocentric cohort study, J. Infect. Chemother, doi:10.1016/j.jiac.2021.01.011
Máiz Carro, Martínez-García, Nebulized hypertonic saline in noncystic fibrosis bronchiectasis: A comprehensive review, Ther. Adv. Respir. Dis, doi:10.1177/1753466619866102
Pickens, Gao, Cuttica, Smith, Pesce et al., Bacterial Superinfection Pneumonia in Patients Mechanically Ventilated for COVID-19 Pneumonia, Am. J. Respir. Crit. Care Med, doi:10.1164/rccm.202106-1354OC
Qu, Ren, Guo, Liang, Xu et al., ffect of N-acetylcysteine inhalation on ventilator-associated pneumonia caused by biofilm in endotracheal tubes, Zhonghua Er Ke Za Zhi
Ranieri, Rubenfeld, Thompson, Ferguson, Caldwell et al., Acute Respiratory Distress Syndrome: The Berlin Definition, JAMA
Rawson, Moore, Zhu, Ranganathan, Skolimowska et al., Bacterial and Fungal Coinfection in Individuals with Coronavirus: A Rapid Review to Support COVID-19 Antimicrobial Prescribing, Clin. Infect. Dis, doi:10.1093/cid/ciaa530
Razazi, Arrestier, Haudebourg, Benelli, Carteaux et al., Correction to: Risks of ventilator-associated pneumonia and invasive pulmonary aspergillosis in patients with viral acute respiratory distress syndrome related or not to Coronavirus 19 disease, Crit. Care, doi:10.1186/s13054-021-03517-5
Robinson, Hemming, Regnis, Wong, Bailey et al., Effect of increasing doses of hypertonic saline on mucociliary clearance in patients with cystic fibrosis, Thorax, doi:10.1136/thx.52.10.900
Rogers, Mucoactive agents for airway mucus hypersecretory diseases, Respir. Care
Rouyer, Strazzulla, Youbong, Tarteret, Pitsch et al., Ventilator-Associated Pneumonia in COVID-19 Patients: A Retrospective Cohort Study, Antibiotics, doi:10.3390/antibiotics10080988
Rouzé, Martin-Loeches, Povoa, Makris, Artigas et al., Relationship between SARS-CoV-2 infection and the incidence of ventilator-associated lower respiratory tract infections: A European multicenter cohort study, Intensive Care Med, doi:10.1007/s00134-020-06323-9
Stigliani, Manniello, Zegarra-Moran, Galietta, Minicucci et al., Rheological Properties of Cystic Fibrosis Bronchial Secretion and in Vitro Drug Permeation Study: The Effect of Sodium Bicarbonate, J. Aerosol Med. Pulm. Drug Deliv, doi:10.1089/jamp.2015.1228
Van Meenen, Van Der Hoeven, Binnekade, De Borgie, Merkus et al., Effect of On-Demand vs Routine Nebulization of Acetylcysteine with Salbutamol on Ventilator-Free Days in Intensive Care Unit Patients Receiving Invasive Ventilation: A Randomized Clinical Trial, JAMA, doi:10.1001/jama.2018.0949
Wardeh, Conklin, Ko, Case reports of observed significant improvement in patients with ARDS due to COVID-19 and maximum ventilatory support after inhalation of sodium bicarbonate, J. Clin. Intensive Care Med, doi:10.29328/journal.jcicm.1001029
Wark, Mcdonald, Nebulised hypertonic saline for cystic fibrosis, Cochrane Database Syst. Rev, doi:10.1002/14651858.CD001506.pub4
Wicky, Niedermann, Timsit, Ventilator-associated pneumonia in the era of COVID-19 pandemic: How common and what is the impact?, Crit. Care, doi:10.1186/s13054-021-03571-z
Wills, Hall, Chan, Cole, Sodium chloride increases the ciliary transportability of cystic fibrosis and bronchiectasis sputum on the mucus-depleted bovine trachea, J. Clin. Investig, doi:10.1172/JCI119138
Wu, Mcgoogan, Characteristics of and Important Lessons from the Coronavirus Disease 2019 (COVID-19) Outbreak in China: Summary of a Report of 72 314 Cases from the Chinese Center for Disease Control and Prevention, JAMA, doi:10.1001/jama.2020.2648
Zhang, Mendoza-Sassi, Wainwright, Klassen, Nebulised hypertonic saline solution for acute bronchiolitis in infants, Cochrane Database Syst. Rev, doi:10.1002/14651858.CD006458.pub3
DOI record:
{
"DOI": "10.3390/microorganisms10061118",
"ISSN": [
"2076-2607"
],
"URL": "http://dx.doi.org/10.3390/microorganisms10061118",
"abstract": "<jats:p>The effect of routine inhalation therapy on ventilator-associated pneumonia (VAP) in mechanically ventilated patients with the coronavirus disease (COVID-19) has not been well-defined. This randomized controlled trial included 175 eligible adult patients with COVID-19 who were treated with mechanical ventilation at the University Hospital of Split between October 2020 and June 2021. Patients were randomized and allocated to a control group (no routine inhalation) or one of the treatment arms (inhalation of N-acetylcysteine; 5% saline solution; or 8.4% sodium bicarbonate). The primary outcome was the incidence of VAP, while secondary outcomes included all-cause mortality. Routine inhalation therapy had no effect on the incidence of bacterial or fungal VAP nor on all-cause mortality (p > 0.05). Secondary analyses revealed a significant reduction of Gram-positive and methicillin-resistant Staphylococcus aureus (MRSA) VAP in the treatment groups. Specifically, the bicarbonate group had a statistically significantly lower incidence of Gram-positive bacterial VAP (4.8%), followed by the N-acetylcysteine group (10.3%), 5% saline group (19.0%), and control group (34.6%; p = 0.001). This difference was driven by a lower incidence of MRSA VAP in the bicarbonate group (2.4%), followed by the N-acetylcysteine group (7.7%), 5% saline group (14.3%), and control group (34.6%; p < 0.001). Longer duration of ventilator therapy was the only significant, independent predictor of any bacterial or fungal VAP in the multivariate analysis (aOR 1.14, 95% CI 1.01–1.29, p = 0.038 and aOR 1.05, 95% CI 1.01–1.10, p = 0.028, respectively). In conclusion, inhalation therapy had no effect on the overall VAP incidence or all-cause mortality. Further studies should explore the secondary findings of this study such as the reduction of Gram-positive or MRSA-caused VAP in treated patients.</jats:p>",
"alternative-id": [
"microorganisms10061118"
],
"author": [
{
"affiliation": [],
"family": "Delić",
"given": "Nikola",
"sequence": "first"
},
{
"ORCID": "http://orcid.org/0000-0001-9272-6906",
"affiliation": [],
"authenticated-orcid": false,
"family": "Matetic",
"given": "Andrija",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0001-7003-8192",
"affiliation": [],
"authenticated-orcid": false,
"family": "Domjanović",
"given": "Josipa",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Kljaković-Gašpić",
"given": "Toni",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0001-5621-580X",
"affiliation": [],
"authenticated-orcid": false,
"family": "Šarić",
"given": "Lenko",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ilić",
"given": "Darko",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Došenović",
"given": "Svjetlana",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Domazet",
"given": "Josipa",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Kovač",
"given": "Ruben",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Runjić",
"given": "Frane",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Stipić",
"given": "Sanda Stojanović",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Duplančić",
"given": "Božidar",
"sequence": "additional"
}
],
"container-title": "Microorganisms",
"container-title-short": "Microorganisms",
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2022,
5,
30
]
],
"date-time": "2022-05-30T03:43:03Z",
"timestamp": 1653882183000
},
"deposited": {
"date-parts": [
[
2022,
5,
30
]
],
"date-time": "2022-05-30T04:00:51Z",
"timestamp": 1653883251000
},
"indexed": {
"date-parts": [
[
2022,
5,
30
]
],
"date-time": "2022-05-30T04:42:53Z",
"timestamp": 1653885773518
},
"is-referenced-by-count": 0,
"issue": "6",
"issued": {
"date-parts": [
[
2022,
5,
28
]
]
},
"journal-issue": {
"issue": "6",
"published-online": {
"date-parts": [
[
2022,
6
]
]
}
},
"language": "en",
"license": [
{
"URL": "https://creativecommons.org/licenses/by/4.0/",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2022,
5,
28
]
],
"date-time": "2022-05-28T00:00:00Z",
"timestamp": 1653696000000
}
}
],
"link": [
{
"URL": "https://www.mdpi.com/2076-2607/10/6/1118/pdf",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "1968",
"original-title": [],
"page": "1118",
"prefix": "10.3390",
"published": {
"date-parts": [
[
2022,
5,
28
]
]
},
"published-online": {
"date-parts": [
[
2022,
5,
28
]
]
},
"publisher": "MDPI AG",
"reference": [
{
"DOI": "10.1001/jama.2020.2648",
"doi-asserted-by": "publisher",
"key": "ref1"
},
{
"key": "ref2"
},
{
"DOI": "10.3390/antibiotics10050545",
"doi-asserted-by": "publisher",
"key": "ref3"
},
{
"DOI": "10.1097/CCM.0b013e3182281f33",
"doi-asserted-by": "publisher",
"key": "ref4"
},
{
"DOI": "10.1097/MCC.0000000000000908",
"doi-asserted-by": "publisher",
"key": "ref5"
},
{
"DOI": "10.1080/17476348.2017.1359090",
"doi-asserted-by": "publisher",
"key": "ref6"
},
{
"DOI": "10.1007/s40261-019-00861-x",
"doi-asserted-by": "publisher",
"key": "ref7"
},
{
"article-title": "Evaluation of the Effect of Nebulized N-Acetylcysteine on Respiratory Secretions in Mechanically Ventilated Patients: Randomized Clinical Trial",
"author": "Masoompour",
"first-page": "309",
"journal-title": "Iran. J. Med. Sci.",
"key": "ref8",
"volume": "40",
"year": "2015"
},
{
"DOI": "10.1002/14651858.CD001506.pub4",
"doi-asserted-by": "publisher",
"key": "ref9"
},
{
"article-title": "Acute Respiratory Distress Syndrome: The Berlin Definition",
"author": "Ranieri",
"first-page": "2526",
"journal-title": "JAMA",
"key": "ref10",
"volume": "307",
"year": "2012"
},
{
"DOI": "10.1186/s13054-021-03460-5",
"doi-asserted-by": "publisher",
"key": "ref11"
},
{
"article-title": "Hydrator Therapies for Chronic Bronchitis. Lessons from Cystic Fibrosis",
"author": "Bennett",
"first-page": "S186",
"journal-title": "Ann. Am. Thorac. Soc.",
"key": "ref12",
"volume": "13",
"year": "2016"
},
{
"DOI": "10.1056/NEJMoa043891",
"doi-asserted-by": "publisher",
"key": "ref13"
},
{
"DOI": "10.1136/thx.52.10.900",
"doi-asserted-by": "publisher",
"key": "ref14"
},
{
"DOI": "10.1164/ajrccm.156.1.9512074",
"doi-asserted-by": "publisher",
"key": "ref15"
},
{
"DOI": "10.1172/JCI119138",
"doi-asserted-by": "publisher",
"key": "ref16"
},
{
"DOI": "10.4049/jimmunol.0803959",
"doi-asserted-by": "publisher",
"key": "ref17"
},
{
"DOI": "10.1371/journal.pone.0090164",
"doi-asserted-by": "publisher",
"key": "ref18"
},
{
"DOI": "10.1177/1753466619866102",
"doi-asserted-by": "publisher",
"key": "ref19"
},
{
"DOI": "10.1136/thoraxjnl-2014-205953",
"doi-asserted-by": "publisher",
"key": "ref20"
},
{
"article-title": "Mucoactive agents for airway mucus hypersecretory diseases",
"author": "Rogers",
"first-page": "1176",
"journal-title": "Respir. Care",
"key": "ref21",
"volume": "52",
"year": "2007"
},
{
"DOI": "10.1152/ajplung.00180.2010",
"doi-asserted-by": "publisher",
"key": "ref22"
},
{
"DOI": "10.29328/journal.jcicm.1001029",
"doi-asserted-by": "publisher",
"key": "ref23"
},
{
"DOI": "10.1186/s13054-021-03517-5",
"doi-asserted-by": "publisher",
"key": "ref24"
},
{
"DOI": "10.1186/s13613-020-00775-4",
"doi-asserted-by": "publisher",
"key": "ref25"
},
{
"DOI": "10.1007/s00134-020-06323-9",
"doi-asserted-by": "publisher",
"key": "ref26"
},
{
"DOI": "10.1371/journal.pone.0246318",
"doi-asserted-by": "publisher",
"key": "ref27"
},
{
"DOI": "10.1186/s13054-021-03571-z",
"doi-asserted-by": "publisher",
"key": "ref28"
},
{
"DOI": "10.1007/s00134-020-06062-x",
"doi-asserted-by": "publisher",
"key": "ref29"
},
{
"DOI": "10.1016/j.jiac.2021.01.011",
"doi-asserted-by": "publisher",
"key": "ref30"
},
{
"DOI": "10.3390/jcm10040555",
"doi-asserted-by": "publisher",
"key": "ref31"
},
{
"DOI": "10.3390/v9030058",
"doi-asserted-by": "publisher",
"key": "ref32"
},
{
"DOI": "10.1038/nrmicro3231",
"doi-asserted-by": "publisher",
"key": "ref33"
},
{
"DOI": "10.1016/j.mib.2016.11.003",
"doi-asserted-by": "publisher",
"key": "ref34"
},
{
"DOI": "10.1016/S2666-5247(20)30009-4",
"doi-asserted-by": "publisher",
"key": "ref35"
},
{
"DOI": "10.1093/cid/ciaa530",
"doi-asserted-by": "publisher",
"key": "ref36"
},
{
"DOI": "10.1002/14651858.CD006458.pub3",
"doi-asserted-by": "publisher",
"key": "ref37"
},
{
"article-title": "ffect of N-acetylcysteine inhalation on ventilator-associated pneumonia caused by biofilm in endotracheal tubes",
"author": "Qu",
"first-page": "278",
"journal-title": "Zhonghua Er Ke Za Zhi",
"key": "ref38",
"volume": "54",
"year": "2016"
},
{
"DOI": "10.1001/jama.2018.0949",
"doi-asserted-by": "publisher",
"key": "ref39"
},
{
"DOI": "10.1152/ajplung.00143.2012",
"doi-asserted-by": "publisher",
"key": "ref40"
},
{
"DOI": "10.1089/jamp.2015.1228",
"doi-asserted-by": "publisher",
"key": "ref41"
},
{
"DOI": "10.1186/s13054-021-03493-w",
"doi-asserted-by": "publisher",
"key": "ref42"
},
{
"DOI": "10.3390/antibiotics10080988",
"doi-asserted-by": "publisher",
"key": "ref43"
},
{
"DOI": "10.1164/rccm.202106-1354OC",
"doi-asserted-by": "publisher",
"key": "ref44"
}
],
"reference-count": 44,
"references-count": 44,
"relation": {},
"resource": {
"primary": {
"URL": "https://www.mdpi.com/2076-2607/10/6/1118"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [
"Virology",
"Microbiology (medical)",
"Microbiology"
],
"subtitle": [],
"title": "Effects of Different Inhalation Therapy on Ventilator-Associated Pneumonia in Ventilated COVID-19 Patients: A Randomized Controlled Trial",
"type": "journal-article",
"volume": "10"
}
delic
