Treatment with hydroxychloroquine vs nitazoxanide in patients with COVID-19: brief report
et al., PAMJ - Clinical Medicine, doi:10.11604/pamj-cm.2021.7.15.30981, Nov 2021
Planned RCT of HCQ vs. HCQ+nitazoxanide which was aborted due to the retracted Surgisphere paper. Authors retrospectively analyze a small set of HCQ vs. nitazoxanide patients (which were protocol deviations in the planned RCT), showing reduced hospitalization time and ICU admission with nitazoxanide.
Study covers HCQ and nitazoxanide.
|
risk of death, 68.2% lower, RR 0.32, p = 0.38, treatment 1 of 17 (5.9%), control 5 of 27 (18.5%), NNT 7.9.
|
|
risk of mechanical ventilation, 86.7% lower, RR 0.13, p = 0.15, treatment 0 of 17 (0.0%), control 4 of 27 (14.8%), NNT 6.8, relative risk is not 0 because of continuity correction due to zero events (with reciprocal of the contrasting arm).
|
|
risk of ICU admission, 59.3% lower, RR 0.41, p < 0.001, treatment 0 of 17 (0.0%), control 16 of 27 (59.3%), NNT 1.7, adjusted per study.
|
|
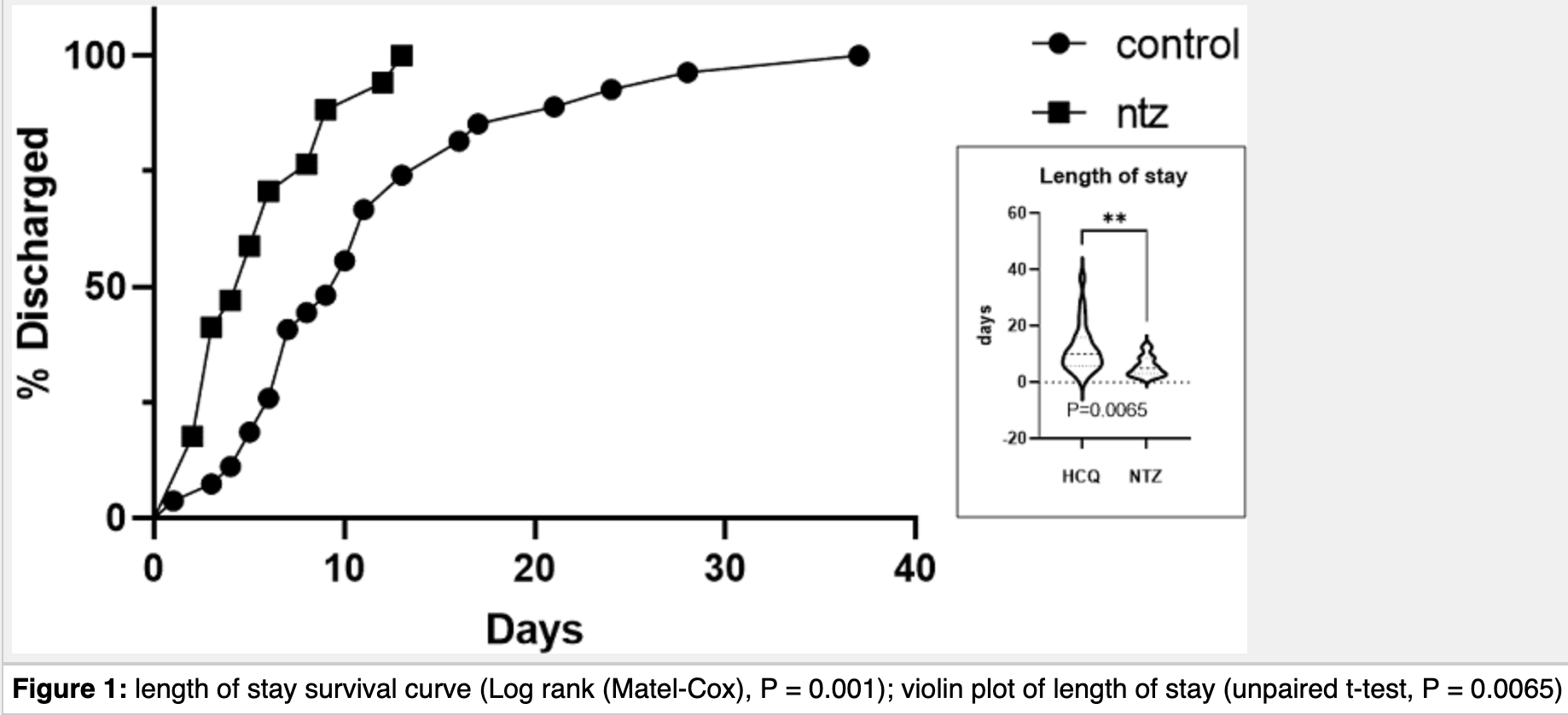
hospitalization time, 51.8% lower, relative time 0.48, p = 0.006, treatment 17, control 27.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Calderón et al., 23 Nov 2021, retrospective, Mexico, peer-reviewed, 7 authors, this trial compares with another treatment - results may be better when compared to placebo.
Treatment with hydroxychloroquine vs nitazoxanide in patients with COVID-19: brief report
doi:10.11604/pamj-cm.2021.7.15.30981
Introduction: coronavirus pandemic has led to 1.8 million deaths worldwide as of May 1 st , 2021. Nitazoxanide (NTZ) is currently being studied in several ambulatory clinical trials to control viral infections including SARS-CoV-2. This study evaluates the effect of treatment with NTZ compared to Hydroxychloroquine (HCQ) in hospitalized patients. Methods: in this clinical study performed from May to August 2020, two groups of COVID-19 patients were compared: A) a control group treated with HCQ 200 mg PO twice a day for seven days and B) an experimental group treated with NTZ 500 mg PO every 6 hours for seven days. Student's t-test between the two groups and Relative Risk (RR) with a 95% confidence interval (CI), were calculated. In all cases a P<0.05 was considered statistically significant. Results: twenty-seven ( 27 ) patients (females 17, males 10) treated with HCQ and 17 patients (females 8, males 9) administered NTZ were included in the study (mean age 44.6 ± 14.4 years old). At the moment of hospital admission, the following variables were statistically significant: BMI (P=0.0225) and leucocyte counts (P=0.069). In the follow-up period, the statistically significant variables were hospital length of stay (P=0.003257) and Intensive Care Units (ICU) admission with a lower RR in the group taking NTZ vs HCQ of 0.4074 (95% CI: 0.2451 to 0.5927, attributable risk (P1 -P2): 0.5926, P≤0.0001, NNT: 1.688). Conclusion: in comparison to HCQ, NTZ significantly reduces the risk to be hospitalized in the ICU and this approach could be replicated easily in any hospital.
Competing interests The authors declare no competing interests.
Authors' contributions
References
Albani, Sepe, Fusina, Prezioso, Baronio et al., Thromboprophylaxis with enoxaparin is associated with a lower death rate in patients hospitalized with SARS-CoV-2 infection. A cohort study, EClinical Medicine, doi:10.1016/j.eclinm.2020.100562
Anastasiou, Eleftheriadou, Tentolouris, Tsilingiris, Tentolouris, In Vitro Data of Current Therapies for SARS-CoV-2, Curr Med Chem, doi:10.2174/0929867327666200513075430
Annie, Sirbu, Frazier, Broce, Lucas, Hydroxychloroquine in hospitalized COVID-19 patients: real world experience assessing mortality, Pharmacotherapy, doi:10.1002/phar.2467
Beigel, Tomashek, Dodd, Mehta, Zingman et al., Remdesivir for the treatment of Covid-19 -Final Report, N Engl J Med, doi:10.1056/NEJMoa2007764
Blum, Cimerman, Hunter, Tierno, Lacerda et al., Nitazoxanide superiority to placebo to treat moderate COVID-19 -a pilot prove of concept randomized double-blind clinical trial, E Clinical Medicine, doi:10.1016/j.eclinm.2021.100981
Cadegiani, Goren, Wambier, Mccoy, Early COVID-19 therapy with azithromycin plus nitazoxanide, ivermectin or hydroxychloroquine in outpatient settings significantly improved COVID-19 outcomes compared to known outcomes in untreated patients, New Microbes New Infect, doi:10.1016/j.nmni.2021.100915
Dhama, Sharun, Tiwari, Maryam, Yashpal et al., COVID-19, an emerging coronavirus infection: advances and prospects in designing and developing vaccines, immunotherapeutics, and therapeutics, Hum Vaccines Immunother
Group, Horby, Mafham, Linsell, Effect of Hydroxychloroquine in Hospitalized patients with Covid-19, N Engl J Med, doi:10.1056/NEJMoa2022926
Mendieta Zerón, Calderón, Coria, Nitazoxanide as prophylactic use to reduce the intensity of COVID-19 outbreaks in health personnel, World Acad Sci J, doi:10.3892/wasj.2021.94
Pepperrell, Pilkington, Owen, Wang, Hill, Review of safety and minimum pricing of nitazoxanide for potential treatment of COVID-19, J Virus Erad, doi:10.1016/S2055-6640(20)30017-0
Rocco, Silva, Cruz, Junior, Tierno et al., Early use of nitazoxanide in mild Covid-19 disease: randomized, placebo-controlled trial. medRxiv, Eur Respir J, doi:10.1183/13993003.03725-2020
Rossignol, Bardin, Oaks, Bostick, Vora et al., Early treatment with nitazoxanide prevents worsening of mild and moderate COVID-19 and subsequent hospitalization, medRxiv, doi:10.1101/2021.04.19.21255441
Vijayvargiya, Garrigos, Almeida, Gurram, Stevens et al., Treatment considerations for COVID-19: a critical review of the evidence (or Lack Thereof), Mayo Clin Proc, doi:10.1016/j.mayocp.2020.04.027
Yavuz, Ünal, Antiviral treatment of COVID-19, Turk J Med Sci, doi:10.3906/sag-2004-145