COVID-19 related treatment and outcomes among COVID-19 ICU patients: A retrospective cohort study
et al., Journal of Infection and Public Health, doi:10.1016/j.jiph.2021.08.030, Aug 2021
Vitamin D for COVID-19
8th treatment shown to reduce risk in
October 2020, now with p < 0.00000000001 from 137 studies, recognized in 18 countries.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
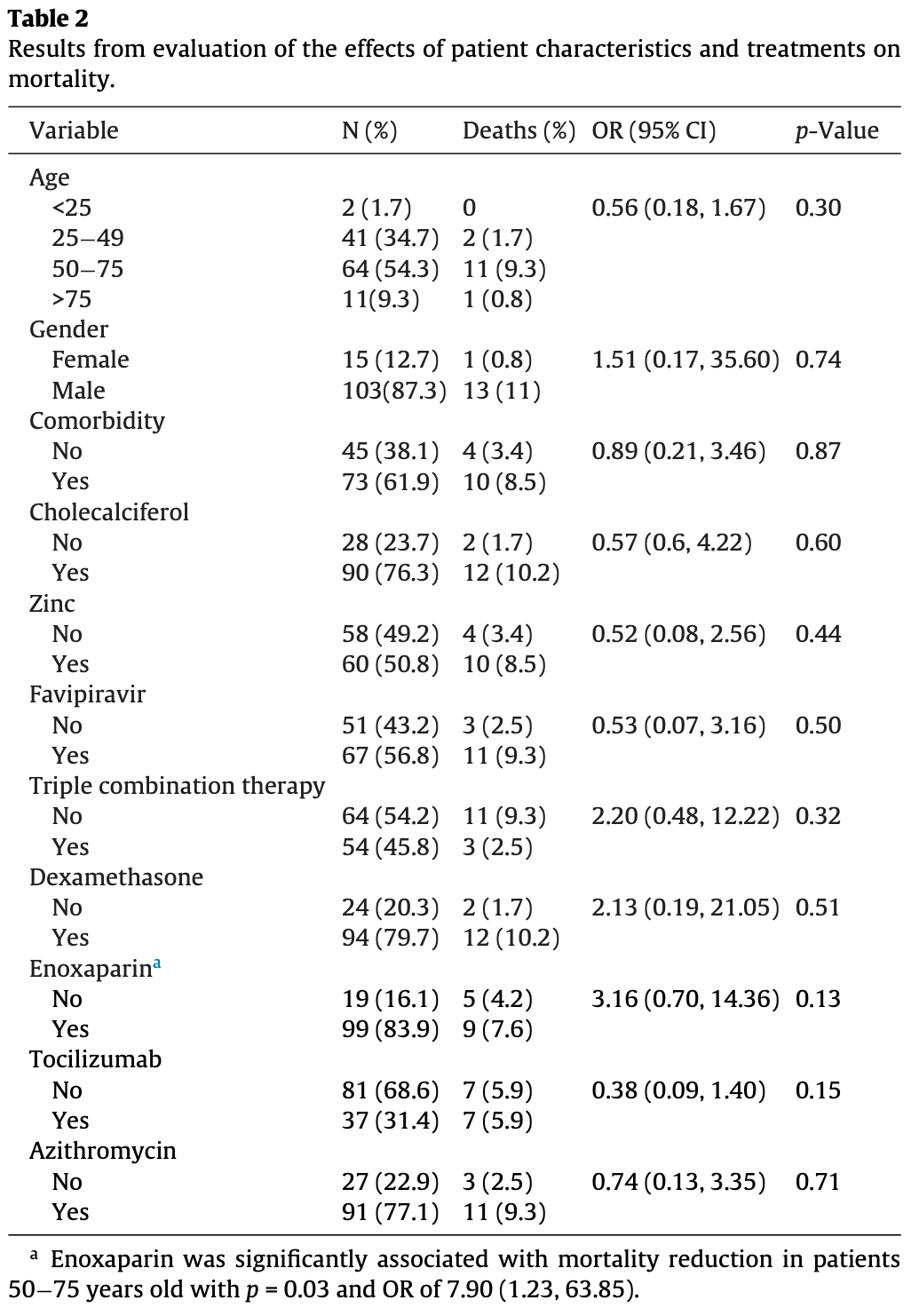
Retrospective 118 ICU patients in Saudi Arabia showing no significant differences in unadjusted results with zinc, vitamin D, and favipiravir treatment.
This paper has several data issues:
The text states the outcome variable was coded as '1 for recovery' and concludes that enoxaparin is beneficial. However, in Table 2, the 'No' group for enoxaparin has an OR of 3.16. If an OR > 1 means higher odds of recovery, this implies that not taking enoxaparin is highly beneficial. The reference groups are inconsistently applied or the text interpretation is backwards. The reverse interpretation may apply to other results in Table 2, for example for tocilizumab: No group OR = 0.38. This would mean not receiving tocilizumab is associated with worse recovery, implying that tocilizumab is beneficial. But the authors conclude it's harmful.
The study concludes that tocilizumab and triple combination therapy significantly increase ICU stay length and should be restricted. However, tocilizumab is specifically indicated for patients experiencing severe cytokine storm (i.e., the sickest patients). Authors failed to adjust for baseline disease severity (e.g., SOFA score, mechanical ventilation status, or oxygen requirements), making the association between the drug and longer ICU stays confounded by the underlying severity of the indication.
Table 1 states 93 patients received dexamethasone and lists this as 86.7%. However, 93 out of 118 is 78.8%. Table 2 states 94 patients (79.7%) received dexamethasone. The abstract and text both claim 86.7%.
Table 1 lists patients with co-morbidities as 'Yes = 81 (68.6%), No = 37 (31.4%)'. Table 2 lists comorbidity as 'Yes = 73 (61.9%), No = 45 (38.1%)'. The totals both sum to 118, but the distribution is different.
After the primary regression model found no variables significantly associated with mortality, authors performed 'subsequent analysis' by dividing the cohort into four age groups to test all variables again. They highlight a single significant finding (enoxaparin in patients 50-75, p=0.03) without applying any correction for multiple testing.
The abstract presents the mortality benefit of enoxaparin in the 50-75 age group as a primary finding, but the methods and text reveal this was a post-hoc subsequent analysis conducted only after the main model failed to find any significant associations.
Cholecalciferol was used in this study.
Meta-analysis shows that late stage treatment with calcitriol / calcifediol (or
paricalcitol, alfacalcidol, etc.) is more effective than cholecalciferol: 66% [47‑78%] lower risk vs. 43% [32‑52%] lower risk.
Cholecalciferol requires two hydroxylation steps to become activated - first
in the liver to calcifediol, then in the kidney to calcitriol. Calcitriol,
paricalcitol, and alfacalcidol are active vitamin D analogs that do not
require conversion. This allows them to have more rapid onset of action
compared to cholecalciferol. The time delay for cholecalciferol to increase
serum calcifediol levels can be 2-3 days, and the delay for converting
calcifediol to active calcitriol can be up to 7 days.
This study is excluded in meta-analysis:
data issues pending author response.
Study covers favipiravir, zinc, and vitamin D.
|
risk of death, 66.5% higher, RR 1.66, p = 0.60, treatment 12 of 90 (13.3%), control 2 of 28 (7.1%), inverted to make RR<1 favor treatment, odds ratio converted to relative risk.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Assiri et al., 28 Aug 2021, retrospective, Saudi Arabia, peer-reviewed, 8 authors, dosage not specified.
COVID-19 related treatment and outcomes among COVID-19 ICU patients: A retrospective cohort study
Journal of Infection and Public Health, doi:10.1016/j.jiph.2021.08.030
Background: The COVID-19 pandemic remains an immediate and present concern, yet as of now there is still no approved therapeutic available for the treatment of COVID-19.This study aimed to investigate and report evidence concerning demographic characteristics and currently-used medications that contribute to the ultimate outcomes of COVID-19 ICU patients. Methods: A retrospective cohort study was conducted among all COVID-19 patients in the Intensive Care Unit (ICU) of Asir Central Hospital in Saudi Arabia between the 1st and 30th of June 2020. Data extracted from patients' medical records included their demographics, home medications, medications used to treat COVID-19, treatment durations, ICU stay, hospital stay, and ultimate outcome (recovery or death).Descriptive statistics and regression modelling were used to analyze and compare the results. The study was approved by the Institutional Ethics Committees at both Asir Central Hospital and King Khalid University. Results: A total of 118 patients with median age of 57 years having definite clinical and disease outcomes were included in the study. Male patients accounted for 87% of the study population, and more than 65% experienced at least one comorbidity. The mean hospital and ICU stay was 11.4 and 9.8 days, respectively. The most common drugs used were tocilizumab (31.4%), triple combination therapy (45.8%), favipiravir (56.8%), dexamethasone (86.7%), and enoxaparin (83%). Treatment with enoxaparin significantly reduced the length of ICU stay (p = 0.04) and was found to be associated with mortality reduction in patients aged 50−75 (p = 0.03), whereas the triple regimen therapy and tocilizumab significantly increased the length of ICU stay in all patients (p = 0.01, p = 0.02 respectively). Conclusion: COVID-19 tends to affect males more significantly than females. The use of enoxaparin is an important part of COVID-19 treatment, especially for those above 50 years of age, while the use of triple combination therapy and tocilizumab in COVID-19 protocols should be reevaluated and restricted to patients who have high likelihood of benefit.
Competing interests None declared.
Ethical approval Not required.
References
Ahmad, Alkharfy, Alrabiah, Alhossan, Saudi Arabia, pharmacists and COVID-19 pandemic, J Pharm Policy Pract
Alsofayan, Althunayyan, Khan, Hakawi, Assiri, Clinical characteristics of COVID-19 in Saudi Arabia: a national retrospective study, J Infect Public Health
Barry, Amri, Memish, COVID-19 in the shadows of MERS-CoV in the Kingdom of Saudi Arabia, J Epidemiol Glob Health
Bergin, Browne, Murray, Interim Guidance for the use of tocilizumab in the management of patients who have severe COVID-19 with suspected hyperinflammation
Chen, Wu, Chen, Clinical characteristics of 113 deceased patients with coronavirus disease 2019: retrospective study, BMJ
Garg, Kim, Whitaker, Hospitalization rates and characteristics of patients hospitalized with laboratory-confirmed coronavirus disease 2019 -COVID-NET, 14 states, MMWR Morb Mortal Wkly Rep
Goyal, Choi, Pinheiro, Clinical characteristics of Covid-19 in New York City, N Engl J Med
Hung, Lung, Tso, Triple combination of interferon beta-1b, lopinavir-ritonavir, and ribavirin in the treatment of patients admitted to hospital with COVID-19: an open-label, randomised, phase 2 trial, Lancet
Hussain, Farhana, Alwutayd, Efficacy of tocilizumab in Covid 19: a metanalysis of case series studies, medRxiv
Jin, Zhan, Peng, Chemoprophylaxis, diagnosis, treatments, and discharge management of COVID-19: an evidence-based clinical practice guideline (updated version), Mil Med Res
Jordan, Adab, Cheng, Covid-19: risk factors for severe disease and death, BMJ
Luo, Liu, Qiu, Liu, Liu et al., Tocilizumab treatment in COVID-19: a single center experience, J Med Virol
Mcmichael, Clark, Pogosjans, COVID-19 in a long-term care facility -King County, Washington, MMWR Morb Mortal Wkly Rep
Pawlowski, Venkatakrishnan, Kirkup, Enoxaparin is associated with lower rates of thrombosis, kidney injury, and mortality than Unfractionated Heparin in hospitalized COVID patients, medRxiv
Tang, Bai, Chen, Gong, Li et al., Anticoagulant treatment is associated with decreased mortality in severe coronavirus disease 2019 patients with coagulopathy, J Thromb Haemost
Tobaiqy, Qashqary, Al-Dahery, Therapeutic management of patients with COVID-19: a systematic review, Infect Prev Pract
Van Singer, Brahier, Ngai, COVID-19 risk stratification algorithms based on sTREM-1 and IL-6 in emergency department, J Allergy Clin Immunol
Venisse, Potential drug-drug interactions associated with drugs currently proposed for COVID-19 treatment in patients receiving other treatments. Fundam, Clin Pharmacol
Weiss, Murdoch, Clinical course and mortality risk of severe COVID-19, Lancet
Zhang, Zhang, Wang, Zhu, Wang et al., Analysis of clinical characteristics and laboratory findings of 95 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a retrospective analysis, Respir Res
Zhou, Yu, Du, Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study, Lancet
Zhou, Zhang, Tian, Xiong, Risk factors associated with disease progression in a cohort of patients infected with the 2019 novel coronavirus, Ann Palliat Med
DOI record:
{
"DOI": "10.1016/j.jiph.2021.08.030",
"ISSN": [
"1876-0341"
],
"URL": "http://dx.doi.org/10.1016/j.jiph.2021.08.030",
"alternative-id": [
"S1876034121002495"
],
"assertion": [
{
"label": "This article is maintained by",
"name": "publisher",
"value": "Elsevier"
},
{
"label": "Article Title",
"name": "articletitle",
"value": "COVID-19 related treatment and outcomes among COVID-19 ICU patients: A retrospective cohort study"
},
{
"label": "Journal Title",
"name": "journaltitle",
"value": "Journal of Infection and Public Health"
},
{
"label": "CrossRef DOI link to publisher maintained version",
"name": "articlelink",
"value": "https://doi.org/10.1016/j.jiph.2021.08.030"
},
{
"label": "Content Type",
"name": "content_type",
"value": "article"
},
{
"label": "Copyright",
"name": "copyright",
"value": "© 2021 The Author(s). Published by Elsevier Ltd on behalf of King Saud Bin Abdulaziz University for Health Sciences."
}
],
"author": [
{
"ORCID": "http://orcid.org/0000-0002-8721-1331",
"affiliation": [],
"authenticated-orcid": false,
"family": "Assiri",
"given": "Abdullah",
"sequence": "first"
},
{
"ORCID": "http://orcid.org/0000-0002-7967-1098",
"affiliation": [],
"authenticated-orcid": false,
"family": "Iqbal",
"given": "Mir J.",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-7057-948X",
"affiliation": [],
"authenticated-orcid": false,
"family": "Mohammed",
"given": "Atheer",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Alsaleh",
"given": "Abdulrhman",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Assiri",
"given": "Ahmed",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-8251-1853",
"affiliation": [],
"authenticated-orcid": false,
"family": "Noor",
"given": "Adeeb",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-6030-1505",
"affiliation": [],
"authenticated-orcid": false,
"family": "Nour",
"given": "Redhwan",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0003-3778-1026",
"affiliation": [],
"authenticated-orcid": false,
"family": "Khobrani",
"given": "Moteb",
"sequence": "additional"
}
],
"container-title": "Journal of Infection and Public Health",
"container-title-short": "Journal of Infection and Public Health",
"content-domain": {
"crossmark-restriction": true,
"domain": [
"clinicalkey.fr",
"clinicalkey.jp",
"clinicalkey.es",
"clinicalkey.com.au",
"clinicalkey.com",
"elsevier.com",
"sciencedirect.com"
]
},
"created": {
"date-parts": [
[
2021,
8,
28
]
],
"date-time": "2021-08-28T20:31:54Z",
"timestamp": 1630182714000
},
"deposited": {
"date-parts": [
[
2022,
5,
15
]
],
"date-time": "2022-05-15T17:09:13Z",
"timestamp": 1652634553000
},
"funder": [
{
"DOI": "10.13039/501100004686",
"award": [
"-288-40"
],
"doi-asserted-by": "publisher",
"name": "Deanship of Scientific Research, King Faisal University"
},
{
"DOI": "10.13039/501100007446",
"doi-asserted-by": "publisher",
"name": "King Khalid University"
}
],
"indexed": {
"date-parts": [
[
2024,
3,
12
]
],
"date-time": "2024-03-12T12:24:21Z",
"timestamp": 1710246261201
},
"is-referenced-by-count": 6,
"issue": "9",
"issued": {
"date-parts": [
[
2021,
9
]
]
},
"journal-issue": {
"issue": "9",
"published-print": {
"date-parts": [
[
2021,
9
]
]
}
},
"language": "en",
"license": [
{
"URL": "https://www.elsevier.com/tdm/userlicense/1.0/",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2021,
9,
1
]
],
"date-time": "2021-09-01T00:00:00Z",
"timestamp": 1630454400000
}
},
{
"URL": "http://creativecommons.org/licenses/by-nc-nd/4.0/",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2021,
8,
26
]
],
"date-time": "2021-08-26T00:00:00Z",
"timestamp": 1629936000000
}
}
],
"link": [
{
"URL": "https://api.elsevier.com/content/article/PII:S1876034121002495?httpAccept=text/xml",
"content-type": "text/xml",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://api.elsevier.com/content/article/PII:S1876034121002495?httpAccept=text/plain",
"content-type": "text/plain",
"content-version": "vor",
"intended-application": "text-mining"
}
],
"member": "78",
"original-title": [],
"page": "1274-1278",
"prefix": "10.1016",
"published": {
"date-parts": [
[
2021,
9
]
]
},
"published-print": {
"date-parts": [
[
2021,
9
]
]
},
"publisher": "Elsevier BV",
"reference": [
{
"DOI": "10.2991/jegh.k.200218.003",
"article-title": "COVID-19 in the shadows of MERS-CoV in the Kingdom of Saudi Arabia",
"author": "Barry",
"doi-asserted-by": "crossref",
"first-page": "1",
"issue": "1",
"journal-title": "J Epidemiol Glob Health",
"key": "10.1016/j.jiph.2021.08.030_bib0005",
"volume": "10",
"year": "2020"
},
{
"DOI": "10.1016/j.jiph.2020.05.026",
"article-title": "Clinical characteristics of COVID-19 in Saudi Arabia: a national retrospective study",
"author": "Alsofayan",
"doi-asserted-by": "crossref",
"first-page": "920",
"issue": "7",
"journal-title": "J Infect Public Health",
"key": "10.1016/j.jiph.2021.08.030_bib0010",
"volume": "13",
"year": "2020"
},
{
"DOI": "10.1186/s40545-020-00243-1",
"article-title": "Saudi Arabia, pharmacists and COVID-19 pandemic",
"author": "Ahmad",
"doi-asserted-by": "crossref",
"first-page": "41",
"journal-title": "J Pharm Policy Pract",
"key": "10.1016/j.jiph.2021.08.030_bib0015",
"volume": "13",
"year": "2020"
},
{
"DOI": "10.1186/s12931-020-01338-8",
"article-title": "Analysis of clinical characteristics and laboratory findings of 95 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a retrospective analysis",
"author": "Zhang",
"doi-asserted-by": "crossref",
"first-page": "74",
"issue": "1",
"journal-title": "Respir Res",
"key": "10.1016/j.jiph.2021.08.030_bib0020",
"volume": "21",
"year": "2020"
},
{
"DOI": "10.1111/jth.14817",
"article-title": "Anticoagulant treatment is associated with decreased mortality in severe coronavirus disease 2019 patients with coagulopathy",
"author": "Tang",
"doi-asserted-by": "crossref",
"first-page": "1094",
"issue": "5",
"journal-title": "J Thromb Haemost",
"key": "10.1016/j.jiph.2021.08.030_bib0025",
"volume": "18",
"year": "2020"
},
{
"DOI": "10.1136/bmj.m1091",
"article-title": "Clinical characteristics of 113 deceased patients with coronavirus disease 2019: retrospective study",
"author": "Chen",
"doi-asserted-by": "crossref",
"first-page": "m1091",
"journal-title": "BMJ",
"key": "10.1016/j.jiph.2021.08.030_bib0030",
"volume": "368",
"year": "2020"
},
{
"DOI": "10.1016/S0140-6736(20)30566-3",
"article-title": "Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study",
"author": "Zhou",
"doi-asserted-by": "crossref",
"first-page": "1054",
"issue": "10229",
"journal-title": "Lancet",
"key": "10.1016/j.jiph.2021.08.030_bib0035",
"volume": "395",
"year": "2020"
},
{
"DOI": "10.21037/apm.2020.03.26",
"article-title": "Risk factors associated with disease progression in a cohort of patients infected with the 2019 novel coronavirus",
"author": "Zhou",
"doi-asserted-by": "crossref",
"first-page": "428",
"issue": "2",
"journal-title": "Ann Palliat Med",
"key": "10.1016/j.jiph.2021.08.030_bib0040",
"volume": "9",
"year": "2020"
},
{
"DOI": "10.1111/fcp.12588",
"article-title": "Potential drug-drug interactions associated with drugs currently proposed for COVID-19 treatment in patients receiving other treatments",
"author": "Venisse",
"doi-asserted-by": "crossref",
"journal-title": "Fundam Clin Pharmacol",
"key": "10.1016/j.jiph.2021.08.030_bib0045",
"year": "2020"
},
{
"DOI": "10.1016/j.infpip.2020.100061",
"article-title": "Therapeutic management of patients with COVID-19: a systematic review",
"author": "Tobaiqy",
"doi-asserted-by": "crossref",
"first-page": "100061",
"issue": "3",
"journal-title": "Infect Prev Pract",
"key": "10.1016/j.jiph.2021.08.030_bib0050",
"volume": "2",
"year": "2020"
},
{
"author": "IBM Corp. Released",
"key": "10.1016/j.jiph.2021.08.030_bib0055",
"series-title": "IBM SPSS statistics for windows, version 27.0",
"year": "2020"
},
{
"key": "10.1016/j.jiph.2021.08.030_bib0060",
"unstructured": "GraphPad Prism. https://www.graphpad.com/guides/prism/latest/user-guide/citing_graphpad_prism.htm. [Accessed 23 October 2020]."
},
{
"DOI": "10.15585/mmwr.mm6912e1",
"article-title": "COVID-19 in a long-term care facility — King County, Washington, February 27–March 9, 2020",
"author": "McMichael",
"doi-asserted-by": "crossref",
"first-page": "339",
"issue": "12",
"journal-title": "MMWR Morb Mortal Wkly Rep",
"key": "10.1016/j.jiph.2021.08.030_bib0065",
"volume": "69",
"year": "2020"
},
{
"DOI": "10.15585/mmwr.mm6915e3",
"article-title": "Hospitalization rates and characteristics of patients hospitalized with laboratory-confirmed coronavirus disease 2019 — COVID-NET, 14 states, March 1–30, 2020",
"author": "Garg",
"doi-asserted-by": "crossref",
"first-page": "458",
"issue": "15",
"journal-title": "MMWR Morb Mortal Wkly Rep",
"key": "10.1016/j.jiph.2021.08.030_bib0070",
"volume": "69",
"year": "2020"
},
{
"author": "Centers for Disease Control and Prevention",
"key": "10.1016/j.jiph.2021.08.030_bib0075",
"series-title": "Coronavirus disease 2019 (COVID-19): cases in U.S.",
"year": "2020"
},
{
"DOI": "10.1056/NEJMc2010419",
"article-title": "Clinical characteristics of Covid-19 in New York City",
"author": "Goyal",
"doi-asserted-by": "crossref",
"first-page": "2372",
"issue": "24",
"journal-title": "N Engl J Med",
"key": "10.1016/j.jiph.2021.08.030_bib0080",
"volume": "382",
"year": "2020"
},
{
"DOI": "10.1016/S0140-6736(20)30633-4",
"article-title": "Clinical course and mortality risk of severe COVID-19",
"author": "Weiss",
"doi-asserted-by": "crossref",
"first-page": "1014",
"issue": "10229",
"journal-title": "Lancet",
"key": "10.1016/j.jiph.2021.08.030_bib0085",
"volume": "395",
"year": "2020"
},
{
"DOI": "10.1136/bmj.m1198",
"article-title": "Covid-19: risk factors for severe disease and death",
"author": "Jordan",
"doi-asserted-by": "crossref",
"first-page": "m1198",
"journal-title": "BMJ",
"key": "10.1016/j.jiph.2021.08.030_bib0090",
"volume": "368",
"year": "2020"
},
{
"article-title": "COVID-19 risk stratification algorithms based on sTREM-1 and IL-6 in emergency department",
"author": "Van Singer",
"journal-title": "J Allergy Clin Immunol",
"key": "10.1016/j.jiph.2021.08.030_bib0095",
"year": "2020"
},
{
"article-title": "Enoxaparin is associated with lower rates of thrombosis, kidney injury, and mortality than Unfractionated Heparin in hospitalized COVID patients",
"author": "Pawlowski",
"journal-title": "medRxiv",
"key": "10.1016/j.jiph.2021.08.030_bib0100",
"year": "2020"
},
{
"article-title": "Chemoprophylaxis, diagnosis, treatments, and discharge management of COVID-19: an evidence-based clinical practice guideline (updated version)",
"author": "Jin",
"first-page": "41",
"issue": "1",
"journal-title": "Mil Med Res",
"key": "10.1016/j.jiph.2021.08.030_bib0105",
"volume": "7",
"year": "2020"
},
{
"DOI": "10.1002/jmv.25801",
"article-title": "Tocilizumab treatment in COVID-19: a single center experience",
"author": "Luo",
"doi-asserted-by": "crossref",
"first-page": "814",
"issue": "7",
"journal-title": "J Med Virol",
"key": "10.1016/j.jiph.2021.08.030_bib0110",
"volume": "92",
"year": "2020"
},
{
"article-title": "Efficacy of tocilizumab in Covid 19: a metanalysis of case series studies",
"author": "Mohamed Hussain",
"journal-title": "medRxiv",
"key": "10.1016/j.jiph.2021.08.030_bib0115",
"year": "2020"
},
{
"key": "10.1016/j.jiph.2021.08.030_bib0120",
"unstructured": "Bergin C, Browne P, Murray P, et al. Interim Guidance for the use of tocilizumab in the management of patients who have severe COVID-19 with suspected hyperinflammation [v3. 0]."
},
{
"DOI": "10.1016/S0140-6736(20)31042-4",
"article-title": "Triple combination of interferon beta-1b, lopinavir-ritonavir, and ribavirin in the treatment of patients admitted to hospital with COVID-19: an open-label, randomised, phase 2 trial",
"author": "Hung",
"doi-asserted-by": "crossref",
"first-page": "1695",
"issue": "10238",
"journal-title": "Lancet",
"key": "10.1016/j.jiph.2021.08.030_bib0125",
"volume": "395",
"year": "2020"
}
],
"reference-count": 25,
"references-count": 25,
"relation": {},
"resource": {
"primary": {
"URL": "https://linkinghub.elsevier.com/retrieve/pii/S1876034121002495"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [
"Infectious Diseases",
"Public Health, Environmental and Occupational Health",
"General Medicine"
],
"subtitle": [],
"title": "COVID-19 related treatment and outcomes among COVID-19 ICU patients: A retrospective cohort study",
"type": "journal-article",
"update-policy": "http://dx.doi.org/10.1016/elsevier_cm_policy",
"volume": "14"
}
